ISSN: 0973-7510
E-ISSN: 2581-690X
Genome editing through efficient and versatile bacterial immunity based CRISPR Cas9 system has revolutionized the molecular biology. Every day new achievements in basic and applied research using CRISPR are coming in. Here, we review the some of the important advancements in methods to increase its efficiency in site-specific genome editing and achievements in crop improvement researches. It is expected from this review that it will provide recent updates on the CRISPR-Cas9 system and will help users involved in applied researches in plant biology to make them exploit the full potential of this genome editing tool.
CRISPR/Cas9, Genome editing, CRIS Paint, DSB, Transgene-free, mushroom.
After the discovery of restriction enzymes in the 1970s, molecular biologists gained the ability to manipulate genetic material and that lead to the development of rDNA technology. This was a remarkable beginning of the new era that enabled scientists to study genes, manipulate them and utilize the generated information for developments in medical and agricultural fields. Later on, genome editing tools were developed which revolutionised the field of molecular biology. These tools were sequence specific and efficient in making changes in genetic materials in vivo. Applications of these genome editing tools helped in understanding the function of unknown genes and regulatory elements. These sequence-specific genome editing nucleases bring out DSB (double strand break) at specific desired location in the genome. These DSBs in the DNA are then repaired either through NHEJ (non-homologous end joining) or through HDR (homology-directed repair) 1. HDR pathway uses template strand for repair so less chance of error is there whereas, NHEJ which does not use any template strand for repair may lead to insertion-deletions (InDels) in the repaired sequence. These genome editing tools includes meganucleases 2, zinc finger nucleases (ZFNs) 3,4, transcription activator-like effector nucleases (TALENs) 5,6, and most recently the RNA-guided clustered regularly interspaced short palindromic repeats (CRISPR) Cas9 nuclease (Cas stands for CRISPR-associated) 7,8. Among these genome editing tools meganucleases, ZFNs and TALENs recognize specific DNA sequence via protein-DNA interactions whereas CRISPR-Cas9 nuclease recognizes by RNA-DNA interactions 9. A general comparison of all these nucleases has been summarized in Table 1. All these preceding techniques to CRISPR faces one or more limitations in genome editing that reduces their utilization in genome editing. Meganucleases were lack of clear correspondence between its protein residue and their target DNA sequence. ZFN protein domains shows context dependent binding preference due to cross talk between adjacent modules when assembled into a larger array 10. TALENs also suffered the context dependent specificity and also render construction of novel TALE array labour intensive and costly affair 11,12. The newly discovered CRISPR-Cas9 nuclease is guided by short guide RNA that pair via Watson-Crick base pairing with its target DNA sequence and it overcomes the challenges of previously used nucleases. The ease of cas-9 targeting, efficient site specificity and ability for multiple editing made the CRISPR-Cas9 system more popular and helped in opening a wide range of practical applications in the medical and crop improvement research field 13-16. Although, there are several reviews and publications about the basic research on CRISPR/Cas9 but a few are there which includes applied researches about the CRISPR system especially on agriculturally important crop plants. Here, in this review we will focus on general feature of CRISPR, on its recent developments and applications regarding to the model and field crop plants.
Table (1):
A general Comparison among the sequence specific genome editing tools
particulars |
zfns |
talens |
crispr/cas9 |
|---|---|---|---|
target specificity determined by |
zinc finger proteins |
transcription activator-like effector protein |
crrna or sgrna |
type of interaction |
protein-dna |
protein-dna |
rna-dna |
nucleases used |
foki |
foki |
cas9 |
specificity determining length of target site |
18-36 bp |
30-40bp |
20bp |
target site |
rich in g content |
start with t and end with a |
end with pam sequences like ngg or nag |
Historical Developments in CRISPR/Cas9 system
CRISPR-Cas system is basically an immunity system present in bacteria. It was first discovered in 1987 by a group of scientists working on iap enzyme involved in isozyme conversion of alkaline phosphatase in E.coli. They curiously reported a set of 29 nucleotide repeats downstream of the iap gene 17. Later on, it was found that these repeats elements are present in more than 40% of sequenced bacteria and 90% of archea 18. By 2005, on the systematic analysis of the spacer sequences separating the individual direct repeats showed their extrachromosomal and phage-associated origins 19,20. It was hypothesized that this is the cause of immunity in the bacteria which leads to failure of those phages to infect bacterial cell which carries spacers corresponding to phage genome 19. After that, a rapid increase in the research to know this mechanism leads to a revelation of many details about the CRISPR system 21-24. By 2011 types and components of CRISPR were known and efforts to apply it for genome editing began 25. From 2011 to 2016 several successful genome editing has been done in bacteria 26-28, plants 11,13,14 and animals 29-31.
CRISPR/Cas9 system: types and components
CRISPR system is of three types, type I, II and III. Type II contain only one endonuclease, cas9 that makes it simple and easy to use 32. That is why type II is highly used for genome editing experiments. In vivo in the bacterial cell, the CRISPR-cas9 system consists of crRNA (CRISPR RNA), tracrRNA (Trans CRISPR RNA) and Cas9 nuclease protein (Fig.1). The crRNA contains targeted complementary sequence, tracrRNA gives stability to crRNA and Cas9 act as nuclease which carries active sites to cleave target DNA sequence 32,33. By fusing crRNA and transcrRNA a single guide RNA (SgRNA) can be constructed that facilitates DNA cleavage by Cas9 in vitro 32. For CRISPR Cas9 system to recognize 20 bp long target sequence, a PAM (Protospacer Adjacent Motif) sequence is present at 3’ end of the target sequence. The consensus nucleotide sequence of PAM is NGG, where N stands for any nucleotide among A, T, G or C 34. CRISPR Cas9 complex scans target genome for PAM motif, bind to it and then initiate upstream pairing between the target sequence and crRNA or SgRNA. If sequences found complementary it triggers the cas9 nuclease activity to cut the DNA strands 35,36.
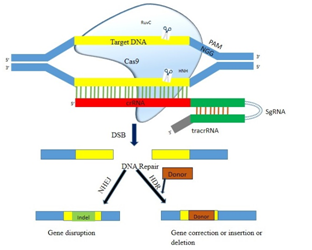
Fig. 1. mechanism of action of rna guided crispr-cas9 in creation of double strand breaks (dsb) in the target dna sequence and the possible pathways of dna repair.(hdr: homology directed repair, nhej: non homologous end joining)
Application on plants
Shortly after the discovery of the mechanism of the CRISPR-cas9 system, its ability to edit genome was started by scientists in many model plants and animals. It has successfully been used in many model plants like in Nicotiana tabaccum 37, N. benthamiana 38 and Arabidopsis 37. It has also been exploited in crops like as in maize 39, wheat 40, rice 37, sorghum 37, tomato 41 and sweet orange 42. Through genome editing mutations are created in the genome with varying efficiencies from 1.1% to as high as 84.8% in the first generation 13. These mutations are stably heritable across several generations with high percentage. Transgene-free progeny carrying only desired mutation can be isolated from later generations after the transgene (SgRNA or Cas9 gene) were segregate out 43, 44. Improvement of rice blast resistance by engineering a CRISPR/Cas9 SSN (C-ERF922) targeting the transcription factor gene OsERF922 in rice were reported 45. In T0 generation mutants having insertion or deletion in the target site obtained. All these mutations transmitted to subsequent generations. In the T1 and T2 generations rice plants harboring desired mutations at the target site but not having the transferred transgene through segregations were obtained. In T2 generation homozygous mutants on comparison with wild type, it was found that lesions for blast resistance reduced significantly and other desirable agronomic traits remained same. Moving a step ahead in genome editing through CRISPR, it is now being used for multiplex gene editing in the single experiment. For multiplexing two or more SgRNA are simultaneously expressed. It has successfully accomplished in rice 9, tomato 46 and Arabidopsis 47. By the simultaneous application of two SgRNA, there was a large and significant deletion made of up to 245 kb in the chromosome in rice protoplasts 48. Simultaneous mutagenesis of seven FT-like genes by CRISPR in rice was also a great achievement 49. Such exciting success stories show that CRISPR is a powerful and efficient tool to carry out life sciences researches. Gene knock-in or targeted gene replacement of a defected gene with an externally supplied desired gene is also highly desirable for plant biologists 14. It has been accomplished in rice 50 , soybean 51 , Maize 52 and tomato 53. In most of the cases, a reporter gene is used for HDR-mediated knock-in. However, a report has been published where npt II selectable marker has been successfully inserted into the endogenous ALS gene in Arabidopsis 54. CRISPR/Cas system was coupled with VIGS (Virus induces gene silencing) technique in tobacco 55. The ease of working with CRISPR and its flexibility has enabled it to be suitable for crops with multiple genome or polyploids. For example, in three homeoallelic genes that were responsible for powdery mildew are edited and resistant genotypes were produced using single SgRNA 40. In rice, single gene OsERF922 was targeted at several sites and it was found that it has not only increased the frequency of mutants but also increased the per cent homozygous mutants 45. Yinong Yang, a plant pathologist at Pennsylvania State University (Penn State) in University Park, engineered the common white button mushroom (Agaricus bisporus) fungus to resist browning 56. It is achieved by targeting the family of genes that encodes polyphenol oxidase (PPO), an enzyme that causes browning. By deleting a few of base pairs in the mushroom’s genome, he knocked out one of six PPO genes, reducing the enzyme’s activity by 30% 56. In a recent study, by transiently expressing CRISPR/Cas9 DNA (TECCDNA), or through transient expression of the in vitro transcripts of Cas9-coding sequence and guide RNA (TECCRNA), scientists have demonstrated a simple and efficient genome-editing approach to regenerate mutant plants from wheat callus cells. Homozygous mutant plants with no detectable transgenes are identified in T0 populations. They found that these genome-editing methods are highly efficient for both hexaploid and tetraploid wheat and should be applicable to other plant species 57.
Important considerations and recent advances about CRISPR/Cas9 system
Genome editing efforts in plants using CRISPR Cas9 faces several problems, especially of off-target activity. Many factors like cas9/SgRNA constructs or delivery methods also affect its efficiency of causing mutation. A concentration ratio between Cas9 and SgRNA plays the important role. Improper concentration ratio may lead to off-target cleavage 58. In Arabidopsis when 1:1 (Cas9:SgRNA) ratio was used, optimal mutagenesis reported 47. Cas9 is a bacterial protein so codon optimization is needed while it is used for gene expression in eukaryotic plant cells 14. There are several studies which show that codon optimization of Cas9 could lead to efficient genome editing 59-61. The specificity of CRISPR Cas9 is determined by the complementarity between SgRNA and the target DNA. Hence a perfect match between the ‘seed sequence’ (the last 8-12 bases of guide sequence) and the region near to PAM sequence in target DNA is very crucial and important 32,62,63. To avoid the off-target activity due to mismatch some bioinformatics tools may be used to design suitable SgRNA using genome information of that organism 64,65. Promoters also play important role in expressing Cas9 protein inside the cell. So optimal promoters are to be used. For example, in dicots, 35S CaMV is preferred for Cas9 and U6 promoter for SgRNA expression. Whereas in the case of monocots both 35S and Ubi work well for Cas9 expression but the different promoter is to be used for SgRNA in different species such as OsU3 for rice and TaU6 for wheat 13,66. Cas9 specificity was increase 100-1500 fold by the use of a pair of Cas9 nickase variants, which were directed to target on opposite strands up to 100 bp apart. The pair of nicks in each strand lead to DSB. Where the potential off-target sites were unlikely to be sufficiently close to each other to induce more than individual nick which is generally perfectly repaired 67,68. With the development of dCas9 (dead Cas9) in which Cas9 nuclease activity is mutated and is attached with FokI. Two dCass9 having their specific target site 15-20 bp apart on opposite strands are used to cleave DNA. By using this modified dCas9, accuracy is increased by 5000 fold. The type of delivery method used for transformation is also equally important for the high efficiency of targeted genome editing generally for plant cells Agrobacterium-mediated transformation used. Other direct transfer methods: particle bombardment and PEG also has an attractive advantage for HDR-mediated genome editing because they can feed cells with sufficient amounts of DNA repair templates to boost the HDR efficacy 14. In the recent report group of scientists co-transformed both Cas9 protein and in-vitro SgRNA into plant protoplast and a single mutated protoplast could turn into a fertile homozygous mutant plant via protoplast regeneration without any foreign DNA integration 69. The specificity and the efficiency of CRISPR-Cas9 system also affected by the location and the context of the target, epigenetic factors like histone modifications. However, in vitro and in vivo assays have shown that it efficiently cleaves methylated DNA. Chromatin immunoprecipitation followed by sequencing (ChIP-seq) has been used to identify DNA sites bound genome-wide by dCas9 in human and mouse cells. Analyses of off-target binding sites detected by ChIP-seq have shown that very few of these are cleaved or mutagenized by catalytically active Cas9, consistent with the proposed mechanism that more extensive pairing of the gRNA mediates a conformational change that enables Cas9 cleavage 70,71. O’Geen and colleagues applied targeted sequence capture 72, which allowed to survey 1200 genomic loci simultaneously including potential off-target sites identified by ChIP-seq and by computational prediction. A high frequency of indels was observed at both target sites and one off-target site, while no cleavage activity could be detected at other ChIP-bound regions. Their results confirm the high specificity of CRISPR endonucleases and demonstrate that sequence capture can be used as a high-throughput genome-wide approach to identify off-target activity. When an extra amino acid is fused at N-terminus of Cas9 lead to variation in its cleavage efficiency. These results help in explaining the distinct editing efficiencies by different Cas9 variants differed only at the N-terminus 14,73. To study the gene function site-specific insertion of heterologous genetic material into genomes is important and in a recent study it has been done using the CRISPR-Cas9 system. A modular system entitled CRISPaint (CRISPR-assisted insertion tagging) that allows precise and efficient integration of large heterologous DNA cassettes into eukaryotic genomes was developed. CRISPaint makes use of the CRISPR/Cas9 system to introduce a double-strand break (DSB) at a user-defined genomic location. A universal donor DNA, optionally provided as minicircle DNA, is cleaved simultaneously to be integrated at the genomic DSB 74.
The crisper cas9 system is efficient, highly specific and simple system. All these features make this a promising genome editing tool for plants and animals. This tool will have a large impact on crop breeding. As in recent examples of bringing powdery mildew resistance in wheat 40, enhancing blast resistance in rice 45 and several other examples indicates that CRISPR Cas9 will be an efficient tool for applied research in plant breeding practices for crop improvement. Nowadays CRISPR is being used not only for genome editing purposes but also for other gene expression regulation and epigenetic modifications. After completion of genome editing in the plant transgene (Cas9 or SgRNA) free plants can be obtained in next few segregating generations. This overcomes the traditional limitations of genetically modified crops which suffers much because of the presence of transgenes. The white button mushroom edited with CRISPRCas9 get a pass from the USDA agency. The agency’s Animal and Plant Health Inspection Service (APHIS) has reported that these organisms do not qualify as something that the agency must regulate 56. Although several general techniques and modified techniques of using CRISPR-Cas system have been developed but still few problems like off-target mutations, epigenetic effects on genome editing, effects of nearby genes and dependence on delivery methods exists and need to be solved with great efforts in the coming future.
- Capecchi, M. R. Altering the genome by homologous recombination. Science, 1989; 244(4910): 1288-1292.
- Smith, J., Grizot, S., Arnould, S., Duclert, A., Epinat, J.C., Chames, P., Prieto, J., Redondo, P., Blanco, F.J., Bravo, J., Montoya, G. A combinatorial approach to create artificial homing endonucleases cleaving chosen sequences. Nucleic Acids Res., 2006; 34(22): e149.
- Urnov, F.D., Miller, J.C., Lee, Y.L., Beausejour, C.M., Rock, J.M., Augustus, S., Jamieson, A.C., Porteus, M.H., Gregory, P.D., Holmes, M.C. Highly efficient endogenous human gene correction using designed zinc-finger nucleases. Nature, 2005; 435(7042): 646-651.
- Miller, J.C., Holmes, M.C., Wang, J., Guschin, D.Y., Lee, Y.L., Rupniewski, I., Beausejour, C.M., Waite, A.J., Wang, N.S., Kim, K.A., Gregory, P.D. An improved zinc-finger nuclease architecture for highly specific genome editing. Nat. Biotechnol., 2007; 25(7): 778-785.
- Miller, J.C., Tan, S., Qiao, G., Barlow, K.A., Wang, J., Xia, D.F., Meng, X., Paschon, D.E., Leung, E., Hinkley, S.J., Dulay, G.P. A TALE nuclease architecture for efficient genome editing. Nat. Biotechnol., 2011; 29(2): 143-148.
- Boch, J., Scholze, H., Schornack, S., Landgraf, A., Hahn, S., Kay, S., Lahaye, T., Nickstadt, A., Bonas, U. Breaking the code of DNA binding specificity of TAL-type III effectors. Science, 2009; 326(5959): 1509-1512.
- Kim, H., Kim, J.S. A guide to genome engineering with programmable nucleases. Nature Reviews Genetics, 2014; 15(5): 321-334.
- Gaj, T., Gersbach, C.A., Barbas, C.F. ZFN, TALEN, and CRISPR/Cas-based methods for genome engineering. Trends in biotechnology, 2013; 31(7): 397-405.
- Zhang, H., Zhang, J., Wei, P., Zhang, B., Gou, F., Feng, Z., Mao, Y., Yang, L., Zhang, H., Xu, N. The CRISPR/Cas9 system produces specific and homozygous targeted gene editing in rice in one generation. Plant Biotechnol. J., 2014; 12(6): 797-807.
- Maeder, M.L., Thibodeau-Beganny, S., Osiak, A., Wright, D.A., Anthony, R.M., Eichtinger, M., Jiang, T., Foley, J.E., Winfrey, R.J., Townsend, J.A. Rapid ‘‘open-source’’ engineering of customized zinc-finger nucleases for highly efficient gene modification. Mol. Cell, 2008; 31: 294–301.
- Hsu, P.D., Lander, E.S., Zhang, F. Development and applications of CRISPR-Cas9 for genome engineering. Cell, 2014; 157(6): 1262-1278.
- Juillerat, A., Dubois, G., Valton, J., Thomas, S., Stella, S., Marechal, A., Langevin, S., Benomari, N., Bertonati, C., Silva, G.H., Daboussi, F. Comprehensive analysis of the specificity of transcription activator-like effector nucleases. Nucleic Acids Res., 2014; 42(8): 5390-5402.
- Song, G., Jia, M., Chen, K., Kong, X., Khattak, K., Xie, C., Li, A., Mao, L. CRISPR/Cas9: A powerful tool for crop genome editing. The crop journal, 2016; 4(2): 75-82.
- Zhang, D., Li, Z., Li, J.F. Targeted gene manipulation in plants using the CRISPR/Cas technology. Journal of Genetics and Genomics, 2016a; 43(5): 251-262.
- Schiml, S., Puchta, H. Revolutionizing plant biology: multiple ways of genome engineering by CRISPR/Cas. BMC Plant Methods, 2016; 12(1): 8.
- Paul III, J.W., Qi, Y. CRISPR/Cas9 for plant genome editing: accomplishments, problems and prospects. Plant cell reports, 2016; 35: 1417.
- Ishino, Y., Shinagawa, H., Makino, K., Amemura, M., Nakata, A. Nucleotide sequence of the iap gene, responsible for alkaline phosphatase isozyme conversion in Escherichia coli, and identification of the gene product. J. Bacteriol., 1987; 169(12): 5429-5433.
- Mojica, F.J., Diez-Villasenor, C., Soria, E., Juez, G. Biological significance of a family of regularly spaced repeats in the genomes of Archaea, Bacteria and mitochondria. Mol. Microbiol., 2000; 36(1): 244-246.
- Mojica, F.J., Garcia-Martinez, J., Soria, E. Intervening sequences of regularly spaced prokaryotic repeats derive from foreign genetic elements. J. Mol. Evol., 2005; 60(2): 174-182.
- Bolotin, A., Quinquis, B., Sorokin, A., Ehrlich, S.D. Clustered regularly interspaced short palindrome repeats (CRISPRs) have spacers of extrachromosomal origin. Microbiol., 2005; 151(8): 2551-2561.
- Barrangou, R., Fremaux, C., Deveau, H., Richards, M., Boyaval, P., Moineau, S., Romero, D.A., Horvath, P. CRISPR provides acquired resistance against viruses in prokaryotes. Science, 2007; 315(5819): 1709-1712.
- Brouns, S.J., Jore, M.M., Lundgren, M., Westra, E.R., Slijkhuis, R.J., Snijders, A.P., Dickman, M.J., Makarova, K.S., Koonin, E.V., Vander Oost, J. Small CRISPR RNAs guide antiviral defense in prokaryotes. Science, 2008; 321(5891): 960-964.
- Marraffini, L.A., Sontheimer, E.J. CRISPR interference limits horizontal gene transfer in staphylococci by targeting DNA. Science, 2008; 322(5909): 1843-1845.
- Hale, C.R., Zhao, P., Olson, S., Duff, M.O., Graveley, B.R., Wells, L., Terns, R.M., Terns, M.P. RNA-guided RNA cleavage by a CRISPR RNA-Cas protein complex. Cell, 2009; 139(5): 945-956.
- Deltcheva, E., Chylinski, K., Sharma, C.M., Gonzales, K., Chao, Y., Pirzada, Z.A., Eckert, M.R., Vogel, J., Charpentier, E. CRISPR RNA maturation by trans-encoded small RNA and host factor RNase III. Nature, 2011; 471(7340): 602-607.
- Sapranauskas, R., Gasiunas, G., Fremaux, C., Barrangou, R., Horvath, P., Siksnys, V. The Streptococcus thermophilus CRISPR/Cas system provides immunity in Escherichia coli. Nucleic Acids Res., 2011; 39(21): 9275-9282.
- Ran, F.A., Hsu, P.D., Lin, C.Y., Gootenberg, J.S., Konermann, S., Trevino, A.E., Scott, D.A., Inoue, A., Matoba, S., Zhang, Y., Zhang, F. Double nicking by RNA-guided CRISPR Cas9 for enhanced genome editing specificity. Cell, 2013; 154(6): 1380-1389.
- Sorek, R., Lawrence, C.M., Wiedenheft, B. CRISPR-mediated adaptive immune systems in bacteria and archaea. Annu. Rev. Biochem., 2013; 82: 237-266.
- Niu, Y., Shen, B., Cui, Y., Chen, Y., Wang, J., Wang, L., Kang, Y., Zhao, X., Si, W., Li, W. Generation of gene-modified cynomolgus monkey via Cas9/RNA-mediated gene targeting in one-cell embryos. Cell, 2014; 156: 836–843.
- Horii, T., Tamura, D., Morita, S., Kimura, M., Hatada, I. Generation of an ICF syndrome model by efficient genome editing of human induced pluripotent stem cells using the CRISPR system. International journal of molecular sciences, 2013; 14(10): 19774-19781.
- Ding, Q., Regan, S.N., Xia, Y., Oostrom, L.A., Cowan, C.A., Musunuru, K. Enhanced efficiency of huma pluripotent stem cell genome editing through replacing TALENs with CRISPRs. Cell Stem Cell, 2013; 12(4): 393-394.
- Jinek, M., Chylinski, K., Fonfara, I., Hauer, M., Doudna, J.A., Charpentier, E. A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science, 2012; 337(6096): 816-821.
- Gasiunas, G., Barrangou, R., Horvath, P., Siksnys, V. Cas9-crRNA ribonucleoprotein complex mediates specific DNA cleavage for adaptive immunity in bacteria. Proc. Natl. Acad. Sci. USA, 2012; 109(39): E2579-E2586.
- Shah, S.A., Erdmann, S., Mojica, F.J., Garrett, R.A. Protospacer recognition motifs: mixed identities and functional diversity. RNA Biol., 2013; 10(5): 891-899.
- Sternberg, S.H., Redding, S., Jinek, M., Greene, E.C., Doudna, J.A. DNA interrogation by the CRISPR RNA-guided endonuclease Cas9. Nature, 2014; 507(7490): 62-67.
- Nishimasu, H., Ran, F.A., Hsu, P.D., Konermann, S., Shehata, S.I., Dohmae, N., Ishitani, R., Zhang, F., Nureki, O. Crystal structure of Cas9 in complex with guide RNA and target DNA. Cell, 2014; 156(5): 935-941.
- Jiang, W., Zhou, H., Bi, H., Fromm, M., Yang, B., Weeks, D.P. Demonstration of CRISPR/Cas9/sgRNA-mediated targeted gene modification in Arabidopsis, tobacco, sorghum and rice. Nucleic Acids Res., 2013; 41(20): e188.
- Nekrasov, V., Staskawicz, B., Weigel, D., Jones, J.D., Kamoun, S. Targeted mutagenesis in the model plant Nicotiana benthamiana using Cas9 RNA-guided endonuclease. Nat. Biotechnol., 2013; 31(8): 691-693.
- Liang, Z., Zhang, K., Chen, K., Gao, C. Targeted mutagenesis in Zea mays using TALENs and the CRISPR/Cas system. J. Genet. Genomics, 2014; 41(2): 63-68.
- Wang, Y., Cheng, X., Shan, Q., Zhang, Y., Liu, J., Gao, C., Qiu, J.L. Simultaneous editing of three homoeoalleles in hexaploid bread wheat confers heritable resistance to powdery mildew. Nat. Biotechnol., 2014; 32(9): 947-951
- Pan, C., Ye, L., Qin, L., Liu, X., He, Y., Wang, J., Lu, G. CRISPR/Cas9-mediated efficient and heritable targeted mutagenesis in tomato plants in the first and later generations. Nat. sci. rep., 2016; 6: 24765.
- Jia, H., Wang, N. Targeted genome editing of sweet orange using Cas9/sgRNA. PLOS ONE, 2014; 9(4): e93806.
- Xu, R.F., Li, H., Qin, R.Y., Li, J., Qiu, C.H., Yang, Y.C., Ma, H., Li, L., Wei, P.C., Yang, J.B. Generation of inheritable and “transgene clean” targeted genome-modified rice in later generations using the CRISPR/Cas9 system. Sci. Rep. 2015; 5: 11491.
- Lawrenson, T., Shorinola, O., Stacey, N., Li, C., Ostergaard, L., Patron, N., Uauy, C., Harwood, W. Induction of targeted, heritable mutations in barley andBrassica oleraceausing RNA-guided Cas9 nuclease. Genome Biol. 2015; 16: 258.
- Wang, F., Wang, C., Liu, P., Lei, C., Hao, W., Gao, Y., Liu, Y.G., Zhao, K. Enhanced Rice Blast Resistance by CRISPR/Cas9-Targeted Mutagenesis of the ERF Transcription Facto Gene OsERF922. PLoS ONE, 2016; 11(4): e0154027.
- Brooks, C., Nekrasov, V., Lippman, Z., VanEck, J. Efficient gene editing in tomato in the first generation using the CRISPR/Cas9 system. Plant Physiol., 2014; 166(3): 1292-1297.
- Li, J.F., Norville, J.E., Aach, J., McCormack, M., Zhang, D., Bush, J., Church, G.M., Sheen, J. Multiplex and homologous recombination-mediated genome editing in Arabidopsisand Nicotiana benthamiana using guide RNA and Cas9. Nat. Biotechnol., 2013; 31(8): 688-691.
- Zhou, H., Liu, B., Weeks, D.P., Spalding, M.H., Yang, B. Large chromosomal deletions and heritable small genetic changes induced by CRISPR/Cas9 in rice. Nucleic Acids Res., 2014; 42(17): 10903-10914.
- Ma, X., Zhang, Q., Zhu, Q., Liu, W., Chen, Y., Qiu, R., Wang, B., Yang, Z., Li, H., Lin, Y., Xie, Y., Shen, R., Chen, S., Wang, Z., Chen, Y., Guo, J.,Chen, L., Zhao, X., Dong, Z., Liu, Y.G. A robust CRISPR/Cas9 system for convenient, high-efficiency multiplex genome editing in monocot and dicot plants. Mol. Plant, 2015; 8(8): 1274-1284.
- Endo, M., Mikami, M., Toki, S. Bi-allelic gene targeting in rice. Plant Physiol., 2016; 170(2): 667-677.
- Li, Z., Liu, Z.B., Xing, A., Moon, B.P., Koellhoffer, J.P., Huang, L., Ward, R.T., Clifton, E., Falco, S.C., Cigan, A.M. Cas9-guide RNA directed genome editing in soybean. Plant Physiol., 2015; 169(2): 960-970.
- Svitashev, S., Young, J.K., Schwartz, C., Gao, H., Falco, S.C., Cigan, A.M. Targeted mutagenesis, precise genome editing, and site-specific gene nsertion in maize using Cas9 and guide RNA. Plant Physiol., 2015; 169(2): 931-945.
- Cermak, T., Baltes, N.J., Cegan, R., Zhang, Y., Voytas, D.F. High-frequency, precise modification of the tomato genome. Genome Biol., 2015; 16(1): 232.
- Schiml, S., Fauser, F., Puchta, H. The CRISPR/Cas system can be used as nuclease for in planta gene targeting and as paired nickases for directed mutagenesis in Arabidopsis resulting in heritable progeny. Plant J., 2014; 80: 1139-1150
- Ali, Z., Abul-faraj, A., Li, L., Ghosh, N., Piatek, M., Mahjoub, A., Aouida, M., Piatek, A., Baltes, N.J., Voytas, D.F., Dinesh-Kumar, S., Mahfouz, M.M. Efficient virus-mediated genome editing in plants using the CRISPR/Cas9 system. Mol. Plant, 2015; 8(8): 1288-1291.
- Waltz E. Gene-edited CRISPR mushroom escapes US regulation. Nat. Biotechnol. News. 2016; 532: 293.
- Zhang, Y., Liang, Z., Zong, Y., Wang, Y., Liu, J., Chen, K., Qiu, J., Gao, C. Efficient and transgene-free genome editing in wheat through transient expression of CRISPR/Cas9 DNA or RNA. Nature Communications, 2016b; 7: 12617.
- Pattanayak, V., Lin, S., Guilinger, J.P., Ma, E., Doudna, J.A., and Liu, D.R. High-throughput profiling of off-target DNA cleavage reveals RNAprogrammed Cas9 nuclease specificity. Nat. Biotechnol., 2013; 31: 839–843.
- Xing, H.L., Dong, L., Wang, Z.P., Zhang, H.Y., Han, C.Y., Liu, B., Wang, X.C., Chen, Q.J. A CRISPR/Cas9 toolkit for multiplex genome editing in plants. BMC Plant Biol. 2014; 14: 327.
- Xu, R.F., Li, H., Qin, R., Wang, L., Li, L., Wei, P., Yang, J. Gene targeting using the Agrobacterium tumefaciens-mediated CRISPR-Cas system in rice. Rice, 2014; 7(1): 5.
- Mikami, M., Toki, S., Endo, M. Comparison of CRISPR/Cas9 expression constructs for efficient targeted mutagenesis in rice. Plant Mol. Biol. 2015; 88: 561e57.
- Cong, L., Ran, F.A., Cox, D., Lin, S., Barretto, R., Habib, N., Hsu, P.D., Wu, X., Jiang, W., Marraffini, L.A., Zhang, F. Multiplex genome engineering using CRISPR/Cas systems. Science, 2013; 339(6121): 819-823.
- Qi, L.S., Larson M.H., Gilbert, L.A., Doudna, J.A., Weissman, J.S., Arkin, A.P., Lim, W.A. Repurposing CRISPR as an RNA-guided platform for sequence-specific control of gene expression. Cell, 2013; 152(5): 1173-1183.
- Xiao, A., Cheng, Z.C., Kong, L., Zhu, Z.Y., Lin, S., Gao, G., Zhang, B. CasOT: a genome-wide Cas9/gRNA off-target searching tool. Bioinformatics, 2014; 30(8): 1180-1182.
- Yan, M., Zhou, S.R., Xue, H.W. CRISPR primer designer: design primers for knockout and chromosome imaging CRISPR-Cas system. J. Integr. Plant Biol., 2015; 57(7): 613-617.
- Shan, Q.W., Wang, Y.P., Li, J., Gao, C.X. Genome editing in rice and wheat using the CRISPR/Cas9 system. Nat. Protoc., 2014; 9(10): 2395-2410.
- Shen, B., Zhang, W., Zhang, J., Zhou, J., Wang, J., Chen, L., Wang, L., Hodgkins, A., Iyer, V., Huang, X., Skarnes, W.C. Efficient genome modification by CRISPR-Cas9 nickase with minimal off-target effects. Nat. methods, 2014; 11(4): 399-402.
- Belhaj, K., Garcia, A., Kamoun, S., Patron, N. Editing plant genomes with CRISPR/Cas9. Current opinion biotechnol., 2015; 32:76-84.
- Woo, J.W., Kim, J., Kwon, S.I., Corvalan, C., Cho, S.W., Kim, H., Kim, S.G., Kim, S.T., Choe, S., Kim, J.S. DNA-free genome editing in plants with preassembled CRISPR-Cas9 ribonucleoproteins. Nat. Biotechnol. 2015; 33: 1162e1164.
- Wu, X., Scott, D.A., Kriz, A.J., Chiu, A.C., Hsu, P.D., Dadon, D.B., Cheng, A.W., Trevino, A.E., Konermann, S., Chen, S., Jaenisch, R. Genome-wide binding of the CRISPR endonuclease Cas9 in mammalian cells. Nature biotechnology, 2014; 32(7): 670-676.
- Cencic, R., Miura, H., Malina, A., Robert, F., Ethier, S., Schmeing, T.M., Dostie, J., Pelletier, J. Protospacer adjacent motif (PAM)-distal sequences engage CRISPR Cas9 DNA target cleavage. PLoS One, 2014; 9(10): e109213.
- O’Geen, H., Henry, I.M., Bhakta, M.S., Meckler, J.F., Segal, D.J. A genome-wide analysis of Cas9 binding specificity using ChIP-seq and targeted sequence capture. Nucleic Acids Res., 2015; 43(6): 3389-3404.
- Johnson, R.A., Gurevich, V., Filler, S., Samach, A., Levy, A.A. Comparative assessments of CRISPR-Cas nucleases’ cleavage efficiency in planta. Plant Mol. Biol. 2015; 87: 143e156.
- Schmid-Burgk, J.L., Honing, K., Ebert, T.S., Hornung, V. CRISPaint allows modular base-specific gene tagging using a ligase-4-dependent mechanism. Nature Communications, 2016; 7.
© The Author(s) 2016. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


