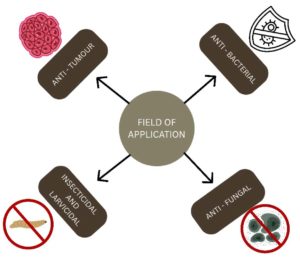
The Xenorhabdus genus encompasses of the bacteria that forms symbiotic association with the Entomopathogenic nematodes of Steinernema genus. The Xenorhabdus spp. exhibits dual relationship of pathogenic relationship with insects and mutualistic relationship with the nematodes. The Xenorhabdus spp. isolation is performed using Galleria mellonella as a bait. Xenorhabdus spp. produces secondary metabolites that have various inhibitory activities, from insecticidal to anti-tumour activity, with mechanisms ranging from membrane disruption to inhibition of RNA, protein synthesis, and immune suppression. These activities have been employed in various fields such as agriculture field for pesticides, mosquito control for larvicide, in medicine for antibiotics and particularly in cancer therapy. The compounds like rhabduscin is used in insecticides, lecithinase in larvicide, Xenorhabdicin as antibiotic and rhabdopeptides in anti-tumour activity. In general, Xenorhabdus is an important but not thoroughly investigated resource for creating sustainable biocontrol agents and new therapeutic compounds.
Entomopathogenic Nematodes, Biocontrol Agents, Anti-microbial Activity, Insecticidal Activity, Anti-tumour Activity, Therapeutic Compounds
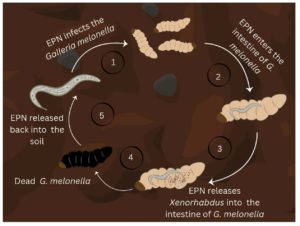
Xenorhabdus genus was first described by Thomas and Poinar. In their study, nine isolates were taken from the lumen of nematodes of the genera Neoaplectana and Heterorhabditis.1 Xenorhabdus spp. exist in a symbiotic relationship that can be pathogenic or mutually beneficial. The life cycle of the bacteria starts and ends with the colonisation in the intestines Xenorhabdus spp. start their life cycle by colonising the nematodes (Figure 1). The nematodes upon entering the insect, release the bacteria into the haemolymph. Xenorhabdus spp. multiplies and produces toxins that kill the insects. The nematodes multiply by consuming nutrients from the insect cadaver, which is converted by the bacteria. The Xenorhabdus spp. exhibits mutualism with the nematodes by providing the specific nutrients for their growth and exhibits pathogenicity towards insects.2
Figure 1. Life cycle and infection process of the Entomopathogenic nematode and the symbiotic Xenorhabus in the Galleria mellonella
Entomopathogenic bacteria
Entomopathogenic nematodes are a group of insect-pathogenic that mostly belong to the genera Steinernema and Heterorhabditis. Entomopathogenic nematodes exist in a mutualistic relationship with bacterial genera Xenorhabdus and Photorhabdus.3 One species of nematodes associates with only one species of bacteria, whereas one species of bacteria can associate with many species of nematodes. The Entomopathogenic nematodes enter the insect host by cuticle penetration through the openings of the host, reaching the gut they convert the host tissue into nutrients for their reproduction.4 Entomopathogenic nematodes are mostly free-living soil organisms, so isolated from the soil using the Galleria mellonella,5 Galleria mellonella Linnaeus, 1758 and Achroia grisella Fabricius, 1794 are the key pests of honeybee colonies worldwide. They cause wax comb damages which lead to colony perishing and absconding. To control these pests, their genetic background and evolution should be considered. In this study, genetic diversity and demographic history of 2 species of wax moth were explored based on mitochondrial genes (COI) as a bait. The larvae that are infected by Steinernema appear black in colour, whereas those infected with Heterorhabditis appear brick red in colour. This pigment is associated with the symbiotic bacteria. The nematodes are collected using the white trap method followed by surface sterilization and molecular characterization.6
Galleria mellonella
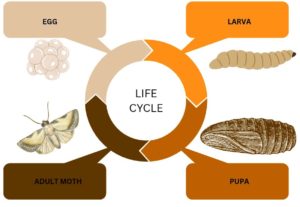
Galleria mellonella is commonly known as the Greater Wax Moth (GWM). This GWM refers to a collection of moths that infiltrate and overrun the bee hives and their products. Egg, larva, pupa, and adult moth are the different life stages of the G. mellonella (Figure 2). The life cycle takes around weeks to months based on the biotic and abiotic factors. The GWM is nocturnal and is active at the onset of the scotophase, that is, between 18:00-24:00 hours. During this phase, the mating of the male and female moth occurs; the female flies towards the male when it is attracted by the acoustic sound from the male. After mating, the female flies back to the hive and lays around 50 to 100 eggs in the cleft and cranny to increase the survival rate of the egg and larvae. The larvae feed on the honey, pollen, and brood resulting in the destruction of the hive structure. Prior to pupation, the larval stage lasts from 28 days to 6 months. The pupa stage takes around 1-9 weeks, and the life cycle continues.7
Xenorhabdus isolation
The G. mellonella larvae are reared artificially and are used as bait to isolate the Xenorhabdus. Soil samples from different locations were collected, and the large debris was removed. The soil samples are moistened, to this the G. mellonella is placed and the container is closed with the cloth to provide a dark condition with proper aeration. The temperature of this setup is maintained at around 25-28 °C. This setup is checked regularly for 7 days to identify any infection in the larvae.8
The growth temperature of Xenorhabdus lies in the range between 32 °C and 42 °C. The bacteria can be isolated through an indirect or direct method. The direct method involves the isolation of the bacteria from the infective nematodes, whereas the indirect method, involves the isolation from the G. mellonella haemocoel9 isolated from at least 27 species of Steinernema nematodes and collected in 32 countries, using three complementary approaches: 16S rRNA gene sequencing, molecular typing and phenotypic characterization. The 16S rRNA gene sequences of the Xenorhabdus strains were highly conserved (similarity coefficient >95%. The G. mellonella is infected by the Steinernema nematode, and the dead cadaver appears black in colour. The cadaver is washed with ethanol, and it is placed in a sterile petri plate. The haemolymph is collected from the 3rd segment of the insect cadaver, and it is streaked in NBTA after 4 days of incubation in the dark. The morphological identification provides a primary confirmation of the species. The Xenorhabdus species appears as dark blue colonies.10 These colonies are further cultured in the LB media under shaking conditions to obtain the secondary metabolites for 78 hours at 30 °C. The supernatant is collected for further analysis of the secondary metabolites.11
Secondary metabolites
The secondary metabolites produced by Xenorhabdus bacteria have multiple activities, like anti-bacterial, anti-fungal, insecticide, protozoal, and also against cancer cells (Figure 3). The antibiotic production in the X. nematophila is regulated by the Lrp gene, but this gene activity is not the same in all other species of Xenorhabdus. The isolated secondary metabolite includes depsipeptides, xenocoumacins, rhabdopeptides, bicornitun, etc (Table 1).12 Xenorhabdus khoisanae J194 with South African origin was cultured in non-aerated and aerated broth; it was observed that there was a rapid increase in biomass in non-aerated broth compared with to aerated. The sequence encoding novel peptides rhabdin and xenopep was confirmed using whole-genome sequencing. Rhabdin is effective against Gram-positive bacteria such as S. aureus ATCC 25923, S. aureus Xen 31, L. monocytogenes and Gram-negative bacteria such as K. pneumoniae, P. aeruginosa, and E. coli. Xenopep was effective only against Gram-positive bacteria such as S. aureus ATCC 25923, L. monocytogenes, and S. epidermidis.13
Table (1):
Secondary metabolites produced by various Xenorhabdus spp. against a range of pathogens and their mode of action
| Secondary Metabolite | Xenorhabdus spp. | Activity | Mode of Action | Ref. |
|---|---|---|---|---|
| Xenoamicin | X. doucetiae, X. mauleonii | Anti-protozoal, weak cytotoxic | Hydrophobic peptide interacts with cell membranes; target site unknown |
12
12
12
|
| Taxlllaid A–G (Lipodepsipeptides) | X. indica | Anti-protozoal; cytotoxic to HeLa cells | Fatty acid chain insertion disrupts protozoan membrane integrity | |
| Xenematides A–D | X. nematophila, X. bovienii | Antibacterial, weak insecticidal | Cyclic depsipeptide damages bacterial membranes; insect toxicity via hemocyte inhibition | |
| Xenobactin | Xenorhabdus sp. PB30.3 | Antibacterial (M. luteus) | Likely membrane-targeting due to hydrophobic nature | |
| Szentiamide | X. szentirmaii | Anti-P. falciparum, T. brucei, T. cruzi, weak cytotoxic | Unknown precise target; interacts with protozoan membranes | |
| Xenocoumacin 1 | X. nematophila, X. kozodoii | Broad-spectrum antibacterial, antifungal, anti-ulcer | Inhibits bacterial cell wall synthesis and fungal growth; damages gastric pathogens | |
| Xenocoumacin 2 | Same as above | Reduced antibacterial, no antifungal, anti-ulcer | Modified from Xcn1 to avoid self-toxicity; retains anti-ulcer effect | |
| Fabclavines | X. budapestensis, X. szentirmaii | Antibacterial, antifungal, anti-parasitic | PKS–NRPS hybrid; disrupts microbial membranes and possibly metabolic enzymes | |
| Pristinamycin IIA | X. nematophila | Antibacterial (Gram-positive, MRSA) | Inhibits bacterial protein synthesis by binding 50S ribosomal subunit | |
| Xenortides A–D | X. nematophila | Weak anti-protozoal | Peptidic amides; tryptamide variants more active; possible membrane interaction | |
| Rhabdopeptides | X. nematophila, X. cabanillasii | Anti-protozoal, weak cytotoxic | Linear peptides disrupt protozoan membrane and affect hemocytes | |
| Bicornitun A–C | X. budapestensis | Antibacterial, antifungal, plant pathogen cytotoxic | Arginine-rich peptides inhibit colony formation and mycelial growth | |
| PAX Peptides | X. nematophila | Antibacterial, antifungal | Lysine-rich cyclolipopeptides disrupt fungal cell membranes | |
| Cabanillasin | X. cabanillasii | Antifungal (human pathogens) | Inhibits fungal cell wall formation and growth | |
| Nemaucin | X. cabanillasii | Antibacterial (MRSA) | Structurally similar to fabclavine; disrupts bacterial cell membrane | |
| Xenorhabdins | X. bovienii | Antibacterial, antifungal, insecticidal, anticancer | Inhibits RNA synthesis; heterobicyclic dithiolopyrrolone core | |
| Xenorxides | X. bovienii | Similar to xenorhabdins | Oxidized sulfur moiety variant; same RNA synthesis inhibition | |
| Indole derivatives | X. nematophila, X. bovienii | Antibacterial, antifungal, immune suppression | Inhibits RNA synthesis; suppresses insect immune eicosanoid pathway | |
| Nematophin | X. nematophila | Antibacterial (MRSA) | α-carbonyl acyl group essential; inhibits bacterial growth via protein target interference | |
| GP-19 & EP-20 (Unnamed peptides) | X. budapestensis | Broad-spectrum antibacterial, antifungal | GP-19 disrupts membranes; EP-20 inhibits nucleic acid and protein synthesis | |
| Benzylideneacetone | X. nematophila | Antibacterial (Gram-negative plant pathogens) | Inhibits phospholipase A2, suppressing insect immune response | |
| Rhabduscin | X. nematophila | Insecticidal | Inhibits phenoloxidase in melanization pathway | |
| Xenocin | X. nematophila | Antibacterial (low-iron conditions) | Bacteriocin binds toxin receptors under iron depletion | |
| Xenorhabdicin | X. nematophila, X. bovienii, X. beddingii | Bactericidal (against related bacteria) | Phage-tail-like bacteriocin punctures target bacterial membranes |
Applications
Anti-bacterial activity of secondary metabolites from Xenorhabdus spp.
EPNs from 230 soil samples were isolated and it was identified as the genus Steinernema and Heterorhabditis. From these isolates 6 Xenorhabdus and 8 Photorhabdus was isolated, and the antibacterial activity was evaluated against the 2 MRSA (Methicillin-resistant Staphylococcus aureus) strains, 2 MDR (Multidrug-resistant) strains, 1 XDR (Extensively drug-resistant) strain, and E. faecalis ATCC 51299 by the disk diffusion method. The results stated that the Xenorhabdus had no inhibitory effect against any antibiotic-resistant strain. In Photorhabdus, the P. luminescens subspecies was able to inhibit a broad range of antibiotic-resistant strains. 1 Xenorhabdus and 3 Photorhabdus strains were tested against 5 antibiotic-resistant strains, and the result indicated the MIC and MBC range between 7.81-0.98 mg/ml, exhibiting moderate to strong activity. Time-kill study was performed against S. aureus strain PB36 (MRSA), and it expressed that Photorhabdus reduced the S. aureus strain PB36 (MRSA) 105 to 103 CFU/ml within 30 minutes; also, there was no bacterial regrowth, exhibiting the strong bactericidal activity (Figure 4). On the contrary, the S. aureus strain PB36 treated with Xenorhabdus continued to grow, referring to no bactericidal activity.14 10 EPN were isolated from different agricultural sites, and it was found that 2 symbiotic bacteria evinced antibacterial activity. The primary characterisation was performed based on the triphenyl tetrazolium and bromothymol blue dye absorption from NBTA plates. It revealed that the colonies, which was blue in colour, were Xenorhabdus and brown were Photorhabdus species. Further molecular characterisation was performed using 16S rRNA sequencing. The antibacterial compound was isolated by the solvent extraction method using ethyl acetate, followed by HPLC. The extract from the Xenorhabdus and Photorhabdus exhibited antibacterial activity against 6 bacterial pathogens (E. coli, S. aureus, B. subtilus, P. mirabilis, E. faecalis, and P. stutzeri). At increasing concentration of the Xenorhabdus and Photorhabdus strain, displayed increasing zone of inhibition. Compared to Photorhabdus, Xenorhabdus evinced strong antibacterial activity.15
Figure 4. Antimicrobial compounds derived from Xenorhabdus and Photorhabdus spp. against E. coli, S. aureus, B. subtilis, P. mirabilis, E. faecalis, P. stutzeri, S. aureus (MRSA), MDR/XDR A. baumannii, ESBL/CRE E. coli, MDR P. aeruginosa, K. pneumoniae, E. faecalis (VRE)
Anti-fungal activity of Xenorhabdus spp.
Two Photorhabdus and three Xenorhabdus species were used to study the antifungal activity against Cryphonectria parasitica16 Fusarium oxysporum17 Rhizoctonia solani18 and Sclerotinia sclerotiorum.19 The Xenorhabdus strain X. szentirmaii has strong anti-fungal activity against C. parasitica compared to other strains (Table 2). In the case of F. oxysporum all the bacterial strains had an anti-fungal activity; the X. szentirmaii at a concentration of 5% had the highest inhibition even after 15 days of incubation, which was absent in the case of other strains. Whereas the other Xenorhabdus strains had no effect against F. oxysporum. The R. solani and S. sclerotiorum were inhibited only by the X. szentirmaii at the end of 5th and 10th day of inhibition and the other strains had no inhibitory effect. A dose of 40% of X. szentirmaii supernatant inhibited the R. solani and 20% dose inhibited the S. sclerotiorum. The antifungal activity of X. szentirmaii is due to the fabclavine compounds.20
Table (2): Antifungal compounds derived from various bacterial strains
Bacterial Strain |
Target Pathogens |
Antifungal Compounds |
Mode of Action |
Results |
Ref. |
|---|---|---|---|---|---|
Xenorhabdus szentirmaii, Xenorhabdus budapestensis |
Fusarium oxysporum f. sp. lycopersici, Fusarium solani, Alternaria alternata, Verticillium dahliae |
Secondary metabolites (unspecified; tested in powder and liquid forms) |
Growth inhibition via bioactive metabolites; |
Powder formulations retained activity longer and showed higher inhibition rates than liquid metabolites for most tested fungi |
26 |
Xenorhabdus indica strain AB |
Fusarium oxysporum f. sp. cucumerinum |
Methyl anthranilate, Decanenitrile, Aniline, Furfuryl alcohol |
Disruption of cell wall and membrane integrity, altered permeability, upregulation of redox and cell wall integrity genes |
Methyl anthranilate & decanenitrile gave 100% inhibition at 50 µL/plate |
21 |
Xenorhabdus szentirmaii |
Cryphonectria parasitica, Fusarium oxysporum, Rhizoctonia solani, Sclerotinia sclerotiorum |
Fabclavines (PKS–NRPS hybrid) |
Broad-spectrum, heat-stable; disrupts fungal growth |
Most effective strain tested; strong inhibition against all, especially C. parasitica |
20 |
Xenorhabdus bovienii |
Fusarium solani NK-NH1 |
Lipids, organic acids, organoheterocyclic compounds; VOCs (2-pentanone, 1-hexen-3-one derivatives) |
VOCs alter gene expression: increase in ribosome biogenesis & oxidative phosphorylation; decrease cell cycle, meiosis, DNA replication, autophagy |
Strong inhibition of F. solani; potential for protecting cultural heritage artifacts |
22 |
Xenorhabdus nematophila YL001 |
Sclerotinia sclerotiorum |
Xenocoumacin 1 (Xcn1) |
Increases membrane permeability, reduces EPS, inhibits PG & cellulase activity, damages mitochondria; no effect on oxalic acid |
EC50 = 2.86 µg/mL; high preventive & curative efficacy in oilseed rape and broad bean |
23 |
X. nematophila, Photorhabdus laumondii subsp. laumondii |
Botrytis cinerea, Armillaria mellea |
Not specified (cell-free supernatants & unfiltered ferments) |
Growth inhibition depends on culture medium & fermentation time; unfiltered ferments more effective |
Up to 74.3% inhibition of B. cinerea (TSB/NB media); strong suppression of A. mellea; selective antibacterial effect on B. thuringiensis ABTS-351 |
25 |
The Xenorhabdus indica strain AB releases a volatile organic compound that has an anti-fungal activity against F. oxysporum. The VOCs inhibit the growth by 60% through spore germination inhibition. A total of six VOCs was isolated, among which four had anti-fungal activity against F. oxysporum. The four VOCs are methyl anthranilate, decanenitrile, aniline, and furfuryl alcohol. The methyl anthranilate and decanenitrile inhibit the growth by hindering the mycelial growth. A 50 µL/plate quantity of methyl anthranilate has the property of inhibiting the growth and reproduction of F. oxysporum through a membrane-breaking mechanism. Aniline reduces the spore germination rate by 60% with a dose of 50 µL.21
The Xenorhabdus bovienii supernatant had a strong inhibition against the Fusarium solani NK-NH1 strain with just 2 days of inhibition. Ven diagram analysis was made between the control and the treatment group to identify the unique metabolite responsible for the inhibitory action. Around 52.75% is the lipids and lipid like compounds. The VOCs (2-pentanone, 3-(phenylmethyl), and 1-hexen-3-one, 5-) produced by the Xenorhabdus bovienii contributed to the anti-fungal activity.22
10 μg/mL of Xcn1 isolated from X. nematophila YL001 exhibited a strong inhibition against Sclerotinia sclerotiorum, R. solani, B. cinerea, and E. turcicum. The samples treated with Xcn1 underwent morphological changes like coiled and disfigured hyphae with minimal distribution. S. sclerotiorum makes the medium acidic by producing oxalic acid during growth. This acidification process occurred in the Xcn1-treated group, but with some time delay. This process did not render with the inhibitory effect of the Xcn1.23
Xenorhabdus nematophila was cultured in different media such as LB, TSB, and NB. There was a significant difference in the inhibitory effect of the X. nematophila. The metabolites extracted after 3 days from LB media had a strong inhibition against Botrytis cinerea24 compared to the other TSB and NB.25
Insecticidal and larvicidal activity of Xenorhabdus spp.
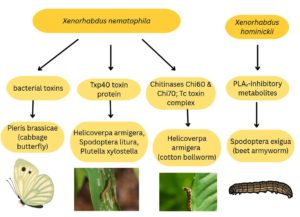
Xenorhabdus nematophila and Xenorhabdus innexi insecticidal activity against the Manduca sexta27 was evaluated. Xenorhabdus nematophila exhibited insecticidal activity by producing several metabolites like rhabduscin that reduces the activation of the phenoloxidase (PO) system by 30%. This PO system is a crucial part of the insect’s innate immune system. Xenorhabdus innexi has no activity against M. sexta. Xenorhabdus innexi is non-toxic against most of the insects that were tested, as it does not possess virulent genes to secrete the toxins. The X. innexi, along with its nematode S. scapterisci, possesses some toxicity, but when it is tested along there was no insecticidal activity. This is due to the absence of genes for Tc toxins and other virulent characteristics.28
Xenorhabdus hominickii supernatant was extracted using 4 different organic solvents, such as hexane, ethyl acetate, chloroform, and butanol. The butanol extract exhibited strong inhibition against haemocyte PLA2 of Spodoptera exigua,29 which has a crucial role in the insect’s immune system (Figure 5). From the butanol extract, fifteen fractions were evaluated, of which two fractions and eight subfractions expressed strong inhibition. From the leaf-dipping method for evaluating the feed toxicity, it was inferred that 3-ethoxy-4-methoxyphenol (EMP) and phthalimide (PM) from the eight compounds indicated strong insecticidal activity.30 Steinernema carpocapsae mutualist with Xenorhabdus nematophila expressed high toxicity against larvae of P. brassicae31 Pieris brassicae Linnaeus (Lepidoptera: Pieridae. Xenorhabdus nematophila HB310 strain releases protein toxin complex (Tc), which has insecticide properties.32
Xenorhabdus sp. isolated from Corcyra cephalonica,33 which was infected by an entomopathogenic bacterium. The Xenorhabdus sp. occurs in two forms or two phases. Phase I is referred to as the primary form, which absorbs bromophenol blue and appears as dark blue rod-shaped colonies. Phase II is also referred to as the secondary form. MacConkey agar medium appears in red, confirming the presence of this form after 1 week of time. These two forms have the ability to produce lecithinase, but the primary form produces a higher concentration compared to the secondary form. The larvicidal activity of the lecithinase was studied against Culex quinquefasciatus34 larvae. The lecithinase from the Xenorhabdus sp. exhibited high activity at 24 °C, with a pH of 7. The activity of the lecithinase was inversely correlated with the pH. The haemolytic property of the lecithinase was studied through a blood agar plate. The results confirmed that lecithinase concentration is positively correlated with the larvicidal activity against the larvae.35
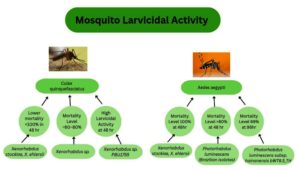
Two and four isolated Xenorhabdus and Photorhabdus species were chosen for larvicidal activity testing against Aedes aegypti36 and C. quinquefasciatus larvae (Figure 6). Photorhabdus exhibited strong larvicidal activity against them after 24 hours of exposure.37
Figure 6. Larvicidal activity of Xenorhabdus and Photorhabdus sp. against Aedes aegypti and Culex quinquefasciatus
The cell suspension and cell-free supernatants of the Photorhabdus and Xenorhabdus were tested for the larvicidal activity against Culex pipiens. It was concluded that the cell-free supernatant showed a high mortality rate compared to the cell suspension against the development stages of Culex pipiens.38 Xenorhabdus bovienii KCS-4S cell-free suspension exhibited an increasing mortality rate of 95% with a gradual increase in the exposure time of 72 hours against the first two larval development stages. This is the only strain that also expressed a mortality rate over 50% against the pupae stage. The time taken to kill 50% of the population (LT50) was higher in the case of cell suspension than in the cell-free supernatant.39
Anti-tumour
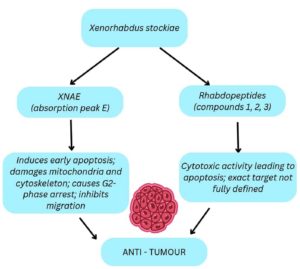
HN_xs01 strain isolated from nematodes obtained from the foot of Yuelu Mountain using Galleria mellonella as bait was identified as 99% similar to X. stockiae. The anti-tumour activity was verified using B16 melanoma cells; the cells that were treated with the HN_xs01 exhibited signs of apoptosis that include morphological changes like shrinkage and detachment, unlike the control cells, which were elongated and attached. Three compounds were purified from the crude extract of the strain, and the molecular formula was determined using NMR spectra, and these structures belonged to rhabdopeptides. The difference between the three compounds (1,2,3) is due to the number of residues of the amino acid N-methyl-valine. The cytotoxicity assay on the A431 cells and K562 cells revealed that compound 3 has strong cytotoxic activity compared to the other two compounds. The anti-tumour targeting activity of the strain was tested using C57BL/C mice bearing the B16 tumour. Different concentrations of HN_xs01 were administered intravenously. At high concentrations, the mice died immediately; lowering the concentration lowered the mortality of the mice, and the safe concentration was identified in the range of 1 × 106 CFU/ 100 μL and 5 × 107 CFU/100 μL. Comparison of HN_xs01 with the probiotic E. coli Nissle 1917 was performed to study the tumour-specific colonisation, and it was observed that the HN_xs01 strain colonised only in the tumour-specific area, whereas in the case of Nissle 1917 was detected in the other organs of the mice, like the liver, kidney, and spleen, than in the tumour region. It was also observed that after the injection of the mice with the HN_xs01 strain, the weight decreased, and it regained its normal weight after 3 days. It was observed that the HN_xs01 mimicked the pulmonary metastasis of B16 tumours and controlled them more effectively.40 The purified supernatant fractions obtained from the UHPLC peak E and F was analysed for the anti-tumour activity on the HUVEC, B16, 4T1, and HeLa cells. It was observed that the peak E fraction had a strong inhibitory effect and was less toxic to the non-tumour cells, and it was further termed as XNAE (Figure 7). This XNAE was used in the CCK-8 reduction assay to assess the in vitro inhibitory effect on the HUVEC, B16, and HeLa cells. There was no effect on the HUVEC, whereas notable cell migration inhibition in B16 and HeLa cells. The ability of the XNAE to induce apoptosis and arrest the cell cycle was tested on the B16 cell. The result suggested that there was some extent of apoptotic activity on the B16 cells, and the cell cycle was arrested in the G1 phase, inhibiting the proliferation.11
Xenorhabdus sp. are symbiotic bacteria found in entomopathogenic nematodes. They are a valuable but underexplored source of bioactive compounds that can be used in agriculture, medicine, and public health. Their diverse secondary metabolites include depsipeptides, xenocoumacins, fabclavines, and volatile organic compounds. These metabolites have broad-spectrum activities, such as antibacterial, antifungal, insecticidal, antitumor, and mosquito larvicidal effects. Research shows that these compounds work through different mechanisms. They can disrupt cell membranes, inhibit RNA or protein synthesis, suppress immune responses, and interfere with the development of pests and pathogens. Notably, some strains can target tumours while minimizing off-target effects, which emphasizes their potential in cancer treatment.
In agriculture, products derived from Xenorhabdus could act as eco-friendly biocontrol agents against resistant microbial pathogens and harmful insect pests. This may reduce the reliance on chemical pesticides. Their effectiveness against disease-carrying insects like A. aegypti and Culex sp. further highlights their importance in vector control programs. However, variations in activity between strains, differences in metabolite yield, and possible toxicity issues require more optimization. This includes genetic engineering, improving fermentation processes, and conducting thorough safety assessments. Overall, Xenorhabdus presents a promising opportunity for developing sustainable, bio-based solutions in pest management, pharmaceutical development, and controlling disease vectors.
ACKNOWLEDGMENTS
The authors are thankful to the Department of Biotechnology, Karunya Institute of Technology and Sciences, for the continued support and for providing facilities during the preparation of this review article.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
None.
DATA AVAILABILITY
All datasets generated or analysed during this study are included in the manuscript.
ETHICS STATEMENT
This article does not contain any studies on human participants or animals performed by any of the authors.
- Thomas GM, Poinar GO. Xenorhabdus gen. nov., a Genus of Entomopathogenic, Nematophilic Bacteria of the Family Enterobacteriaceae. Int J Syst Bacteriol. 1979;29(4):352-360.
Crossref - Goodrich-Blair H, Clarke DJ. Mutualism and pathogenesis in Xenorhabdus and Photorhabdus: two roads to the same destination. Mol Microbiol. 2007;64(2):260-268.
Crossref - Tarasco E, Fanelli E, Salvemini C, et al. Entomopathogenic nematodes and their symbiotic bacteria: from genes to field uses. Front Insect Sci. 2023;3:1195254.
Crossref - Shapiro-Ilan D, Hazir S, Glazer I. Basic and applied research. In: Lacey LA, ed. Microbial Control of Insect and Mite Pests: From Theory to Practice. Elsevier; 2017:91-105.
Crossref - Sopaladawan PN. Genetic Diversity and Demographic History of Wax Moths, Galleria mellonella Linnaeus, 1758 and Achroia grisella Fabricius, 1794 (Lepidoptera: Pyralidae). Trends Sci. 2024;22(1):8946.
Crossref - Mathew J, Vani C, Titus J. Isolation and Mass Production of Entomopathogenic Nematode Steinernema saimkayai using Semi Solid Dog Feed Medium. J Pure Appl Microbiol. 2020;14(1):499-507.
Crossref - Kwadha CA, Ong’amo GO, Ndegwa PN, Raina SK, Fombong AT. The Biology and Control of the Greater Wax Moth, Galleria mellonella. Insects. 2017;8(2):61.
Crossref - Jissin M, Vani C. Biogenic larvicidal formulation of metabolites from Steinernema saimkayi symbiont Xenorhabdus stockiae KUT6 against dengue vector Aedes aegypti. Trop Biomed. 2020;37(3):791-802.
Crossref - Tailliez P, Pages S, Ginibre N, Boemare N. New insight into diversity in the genus Xenorhabdus, including the description of ten novel species. Int J Syst Evol Microbiol. 2006;56(12):2805-2818.
Crossref - Fukruksa C, Yimthin T, Suwannaroj M, et al. Isolation and identification of Xenorhabdus and Photorhabdus bacteria associated with entomopathogenic nematodes and their larvicidal activity against Aedes aegypti. Parasit Vectors. 2017;10(1).
Crossref - Huang X, Tang Q, Liu S, et al. Discovery of an antitumor compound from xenorhabdus stockiae HN_xs01. World J Microbiol Biotechnol. 2024;40(3).
Crossref - Dreyer J, Malan AP, Dicks LMT. Bacteria of the Genus Xenorhabdus, a Novel Source of Bioactive Compounds. Front Microbiol. 2018;9:3177.
Crossref - Booysen E, Rautenbach M, Stander MA, Dicks LMT. Profiling the Production of Antimicrobial Secondary Metabolites by Xenorhabdus khoisanae J194 Under Different Culturing Conditions. Front Chem. 2021;9:626653.
Crossref - Muangpat P, Suwannaroj M, Yimthin T, et al. Antibacterial activity of Xenorhabdus and Photorhabdus isolated from entomopathogenic nematodes against antibiotic-resistant bacteria. PloS One. 2020;15(6):e0234129.
Crossref - Chandrakasan G, Garcia-Trejo JF, Feregrino-Perez AA, Aguirre-Becerra H, Garcia ER, Nieto-Ramirez MI. Preliminary Screening on Antibacterial Crude Secondary Metabolites Extracted from Bacterial Symbionts and Identification of Functional Bioactive Compounds by FTIR, HPLC and Gas Chromatography–Mass Spectrometry. Molecules. 2024;29(12):2914.
Crossref - Scientific Opinion on the pest categorisation of Cryphonectria parasitica (Murrill) Barr. EFSA J. 2014;12(10):3859.
Crossref - de Oliveira LDJMG, Rodrigues AAC, Candido e Silva EKCE. Morphological and phylogenetic characterization of Fusarium Link. Aust J Crop Sci. 2021;(15(12):2021):1406-1415.
Crossref - Akber MA, Mubeen M, Sohail MA, et al. Global distribution, traditional and modern detection, diagnostic, and management approaches of Rhizoctonia solani associated with legume crops. Front Microbiol. 2023;13:1091288.
Crossref - Thomas C, Wilken PM, Coetzee MPA, Visagie CM. Advancing the taxonomy of Sclerotinia (Helotiales, Sclerotiniaceae): a review and recommendations for an important plant-pathogenic genus. IMA Fungus. 2026;17:e175737.
Crossref - Cimen H, Touray M, Gulsen SH, et al. Antifungal activity of different Xenorhabdus and Photorhabdus species against various fungal phytopathogens and identification of the antifungal compounds from X. szentirmaii. Appl Microbiol Biotechnol. 2021;105(13):5517-5528.
Crossref - Baiome BA, Ye X, Yuan Z, Gaafar YZA, Melak S, Cao H. Identification of Volatile Organic Compounds Produced by Xenorhabdus indica Strain AB and Investigation of Their Antifungal Activities. Appl Environ Microbiol. 2022;88(13).
Crossref - Wang Y, Zhang F, Wang C, et al. Antifungal Substances Produced by Xenorhabdus bovienii and Its Inhibition Mechanism against Fusarium solani. Int J Mol Sci. 2022;23(16):9040.
Crossref - Zhang S, Han Y, Wang L, et al. Antifungal Activity and Mechanism of Xenocoumacin 1, a Natural Product from Xenorhabdus nematophila against Sclerotinia sclerotiorum. J Fungi. 2024;10(3):175.
Crossref - Garfinkel AR. The History of Botrytis Taxonomy, the Rise of Phylogenetics, and Implications for Species Recognition. Phytopathology. 2021;111(3):437-454.
Crossref - Vicente-Diez I, Duenas-Hernani J, Campos-Herrera R. Antifungal activity of Xenorhabdus and Photorhabdus against aerial and soilborne grapevine pathogens: varying efficiencies and non-target effects. Biol Control. 2025;205:105759.
Crossref - Gulcu B. Comparison of Powder and Liquid Forms of Antifungal Metabolites Produced by Xenorhabdus szentirmaii, the Symbionts of Entomopathogenic Nematodes, against Gray Mold Botrytis cinerea. J Agric Sci Technol. 2022;24(2):457-464.
- Miao Z, Cao X, Jiang H. Digestion-related proteins in the tobacco hornworm, Manduca sexta. Insect Biochem Mol Biol. 2020;126:103457.
Crossref - Kim IH, Aryal SK, Aghai DT, et al. The insect pathogenic bacterium Xenorhabdus innexi has attenuated virulence in multiple insect model hosts yet encodes a potent mosquitocidal toxin. BMC Genomics. 2017;18(1).
Crossref - Candano RN, Panis WND, Navasero MV, Navasero MM. Host Plants of Spodoptera exigua (Hübner) (Lepidoptera: Noctuidae) In The Philippines and Inventory of World Records. Philipp Entomol. 2020;34(1):1-20
- Mollah MdMI, Dekebo A, Kim Y. Immunosuppressive Activities of Novel PLA2 Inhibitors from Xenorhabdus hominickii, an Entomopathogenic Bacterium. Insects. 2020;11(8):505.
Crossref - Tomar P, Thakur N, Sidhu AK, et al. The Isolation, Identification, and Insecticidal Activities of Indigenous Entomopathogenic Nematodes (Steinernema carpocapsae) and Their Symbiotic Bacteria (Xenorhabdus nematophila) against the Larvae of Pieris brassicae. Horticulturae. 2023;9(8):874.
Crossref - Liu J, Bai H, Song P, et al. Insecticidal Activity of Chitinases from Xenorhabdus nematophila HB310 and Its Relationship with the Toxin Complex. Toxins. 2022;14(9):646.
Crossref - Roh SJ, Jeon JH, Kim DS, Byun BK. The complete mitochondrial genome of unique snout moth, Cathayia obliquella (Pyralidae: Galleriinae) with its phylogenetic implications and a redescription of external morphology. J Asia-Pac Biodivers. 2020;13(4):613-624.
Crossref - Garcia-Escobar GC, Gonzalez JJT, AguirreObando OA. Assessing Phylogeographic Patterns and Genetic Diversity in Culex quinquefasciatus (Diptera: Culicidae) via mtDNA Sequences from Public Databases. Zool Stud. 2024;63:e58.
Crossref - Sheetal BP, Geetha P, Vaidehi D, Bharathi D. Mosquitocidal efficacy of lecithinase derived from entomopathogenic bacteria Xenorhabdus sp. strain PBU1755 against filarial vector Culex quinquefasciatus. Biocatal Agric Biotechnol. 2019;17:492-498.
Crossref - Soares IMN, Polonio JC, Zequi JAC, Golias HC. Molecular techniques for the taxonomy of Aedes Meigen, 1818 (Culicidae: Aedini): A review of studies from 2010 to 2021. Acta Trop. 2022;236:106694.
Crossref - Thanwisai A, Muangpat P, Meesil W, et al. Entomopathogenic Nematodes and Their Symbiotic Bacteria from the National Parks of Thailand and Larvicidal Property of Symbiotic Bacteria against Aedes aegypti and Culex quinquefasciatus. Biology. 2022;11(11):1658.
Crossref - Aardema ML, vonHoldt BM, Fritz ML, Davis SR. Global evaluation of taxonomic relationships and admixture within the Culex pipiens complex of mosquitoes. Parasit Vectors. 2020;13(1):8.
Crossref - Yüksel E, Yıldırım A, İmren M, Canhilal R, Dababat AA. Xenorhabdus and Photorhabdus Bacteria as Potential Candidates for the Control of Culex pipiens L. (Diptera: Culicidae), the Principal Vector of West Nile Virus and Lymphatic Filariasis. Pathogens. 2023;12(9):1095.
Crossref - Zhang C, Chen H, Hüttel S, et al. A novel tumor-targeting strain of Xenorhabdus stockiae exhibits potent biological activities. Front Bioeng Biotechnol. 2022;10.
Crossref
© The Author(s) 2026. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.