ISSN: 0973-7510
E-ISSN: 2581-690X
Mycetoma is a slowly progressive, chronic granulomatous infection of skin and subcutaneous tissue caused by traumatic inoculation of either fungi (eumycotic) or bacteria (actinomycotic). The disease is characterized by triad of tumefaction, discharging sinuses and grains. Here, we report a case of eumycetoma in an infant presenting with multiple discharging sinuses in lower limb. Aspirate and biopsy tissue from the sinuses were processed. The discharge revealed presence of white grains. Potassium hydroxide (KOH) mount revealed presence of hyaline septate hyphae. Histopathological examination showed granulation tissue and fungal hyphae. Repeated culture on sabouraud’s dextrose agar (SDA) with chloramphenicol showed growth of Aspergillus flavus on each occasion. Antifungal susceptibility testing was done following Clinical Laboratory Standard Institute (CLSI) M38-A2 protocol and showed high minimum inhibitory concentration (MIC) for fluconazole, caspofungin, anidulofungin and micafungin. The patient responded to itraconazole. Hence, exact categorization of lesion is essential for effective therapy and better prognosis.
Aspergillus flavus, Eumycetoma, White grain mycetoma
Mycetoma is a chronic slowly progressive localized subcutaneous infection caused by actinomycetes or fungi and characterized by formation of micro abscesses discharging grains containing aggregates of the causative agents which are discharged onto the skin surface through multiple sinuses.1 The granules in mycetoma are mostly black, yellow and white.2
In India, actinomycotic mycetoma is prevalent in south India, southeast Rajasthan and Chandigarh; while eumycetoma is mainly reported from north India and central Rajasthan.3 Here, we report an unusual rapidly progressive eumycetoma in an infant.
Case history
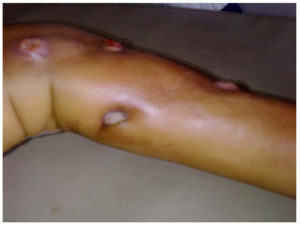
A nine month old male baby, presented with multiple non-healing discharging sinuses (Figure 1) in the left lower limb of three month duration. The lesion started with a small swelling in the left lower limb. Within one month, it had grown in size with development of small ulcers turning in to multiple discharging sinuses. The patient had low-grade intermittent fever. The parents of the patient could not recollect any history of trauma. A provisional diagnosis of osteomyelitis was made.
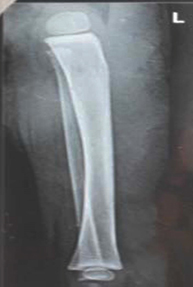
Before investigating the case, a written informed consent was obtained from the parents of the patient. Examination revealed swelling of affected lower limb with multiple discharging sinuses covered with slough with deep induration involving an area of 3.5 X 2.5 inches. The sinuses were fixed to deeper structures discharging purulent fluid with white granules. There was no regional lymphadenopathy. Laboratory investigation revealed low hemoglobin (7.6 g/dl), normocytic, normochromic anemia and a high total leucocyte count (23.32 × 10³/µl). Mantoux and Venereal Disease Research Laboratory (VDRL) tests were negative. Chest X-ray was within normal limit. X-ray (left leg) showed soft tissue swelling with obliteration of fascial planes (Figure 2). Radiologically there was no evidence of osteomyelitis.
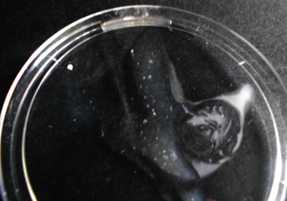
After thorough cleaning of the lesions, a pasteur pipette was introduced in to the sinuses. The aspirated material was rinsed in sterile saline and allowed to settle. A few white, irregular granules of variable size were observed in the aspirate and discharges. (Figure 3) The granules were crushed and subjected to microscopy and culture.4 Biopsy tissue from the sinus was also collected.
Gram stain, potassium hydroxide (KOH) mount, Ziehl-Neelsen stain and modified Ziehl-Neelsen stain (1% H2SO4) were done. The specimens were inoculated in to blood agar and MacConkey agar for bacterial culture and incubated at 37oC for 3days. The granules were also inoculated onto two sets of Sabouraud’s dextrose agar with chloramphenicol (0.05 mg/ml) (SDA-C) and incubated at 30°C and 37°C respectively. These were also inoculated onto two sets of brain heart infusion agar with 5% sheep blood and were incubated at 37°C for 14 days under aerobic and anaerobic conditions. The specimens were also processed to exclude tubercular etiology by standard methods.
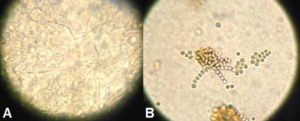
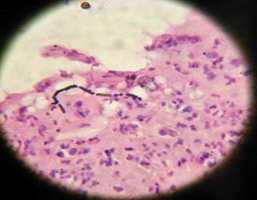
The microscopic examination of KOH mount of the aspirate and biopsy tissue showed septate hyaline hyphae and spores. (Figure 4A & B) Gram staining was performed to distinguish between actinomycetoma and eumycetoma which showed gram negative septate hyphae. (Figure 5) Histopathological examination of the hematoxylin-eosin stained tissue revealed nonspecific granulomatous inflammation with a central focus of acute inflammatory reaction surrounding grains in the subcutaneous tissue which was surrounded by histiocytes and a mixed inflammatory infiltrate comprising lymphocytes, plasma cells, eosinophils, macrophages and fibrosis along with thick club shaped structures and septate fungal hyphae.
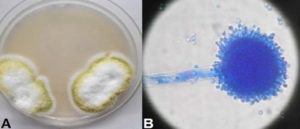
Yellow to green velvety colonies appeared on SDA-C plates after 4 days. Microscopic morphology of the colony showed conidiophores with single phialides covering approximately the entire vesicle which was compatible with that of Aspergillus flavus.. (Figure 6A & B). Repeatedly three different samples were tested and on each occasion Aspergillus flavus was isolated. Bacteriological cultures were sterile.
Fig. 6. A-Aspergillus flavus colony on Sabouraud’s dextrose agar with chloramphenicol B-Lactophenol cotton blue mount from colony (×400)
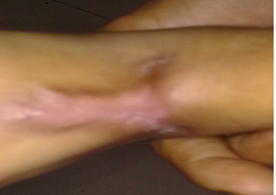
The antifungal susceptibility test done at Department of Medical Microbiology, Postgraduate Institute of Medical Education and Research, Chandigarh by CLSI M38-A2 protocol showed the MIC of amphotericin B, fluconazole, voriconazole, itraconazole, posaconazole, caspofungin, anidulofungin and micafungin to be 0.5µg/ml, 32 µg/ml, 0.5 µg/ml, 0.06 µg/ml, 0.06 µg/ml, 32 µg/ml, 16 µg/ml and 16 µg/ml respectively.5 Surgical curettage and debridement were done along with treatment with itraconazole (5mg/kg/day) for 8 weeks. The patient responded to treatment with regression of swelling after two months without any discharge. No relapse was seen on subsequent follow up for 6 months. (Figure 7).
The reliable epidemiological information regarding eumycetoma is limited even though it was first reported from India in the middle of the 19th century. Eumycetoma usually affects adult males involving limbs and other exposed parts of the body. Children are the least affected group. The case presented here was an infant who presented with a rapidly progressive course of the disease which is an unusual presentation. There are very few reports on eumycetoma in children from India. (Table 1)6-9
Table (1):
Summary of the case reports on eumycetoma in children from India
Ref No. |
Author & year |
Geographical region |
Age of patient |
Site |
Etiological agent |
Treatment |
Outcome |
|---|---|---|---|---|---|---|---|
9 |
Capoor et al. (2007) |
Uttar Pradesh, India |
8 years |
Foot |
Exophialajeanselmei |
Ketoconazole and surgical curettage |
Cured |
10 |
Joshi et al. (2014) |
Karnataka, India |
2 years |
Submendibular region |
Agent not identified |
Ketoconazole |
Not known |
11 |
Purkayastha et al. (2015) |
Karnatala, India |
30 week |
Right ventricle of heart |
Candida albicans |
Amphotericin B + Fluconazole |
Died |
12 |
Tendolkar et al. (2016) |
Uttar Pradesh, India |
8 years |
Forearm |
Madurellamycetomatis |
Voriconazole + surgical debridement |
Relapsed after six months and refused for further treatment |
Aspergillus species are uncommon cause of eumycetoma. Many antifungals showed higher MIC values against this isolate. Also, the development of the lesion was very fast which might lead to early involvement of bones if differentiation of the lesions was not done accurately for appropriate therapy.
The differential diagnosis for multiple discharging sinuses in children includes osteomyelitis and tuberculosis. Staphylococcal infection and mucormycosis, both are rapidly progressive in nature, and later is associated with necrosis of skin and subcutaneous tissue leading to eschar formation. Other diseases presenting as discharging sinuses in children are actinomycosis, botryomycosis, nocardiosis, and sporotrichosis. Sinuses of actinomycosis and nocardiosis usually discharge granules; botryomycosis is associated with subcutaneous nodules and large verrucous lesions; and in sporotrichosis, ascending chain of nodules develops along the skin lymphatic channels. Eumycotic and actinomycotic mycetoma are characterized by multiple punched out lytic lesions and both osteolytic and osteosclerotic lesions in bones respectively.10 In this regard, yaws and syphilis should also be considered.[6] Differentiation between actinomycetoma and eumycetoma is important because of their differences in response to treatment. The colour of grains helps a great deal to make a presumptive diagnosis so that proper treatment of mycetoma can be started in time.
In conclusion, it is utmost necessary that, the clinicians become aware of the disease to suspect and investigate for fungal etiology, especially in the absence of response to antibacterial therapy.
ACKNOWLEDGMENTS
I express my sincere gratitude to Dr. Arunaloke Chakrabarti, Professor & Head, Dr. Shivaprakash M Rudramurthy, Professor, Mrs. Sunita Gupta, Senior Medical Technologist, Department of Medical Microbiology, Postgraduate Institute of Medical Education and Research (PGIMER), Chandigarh, for doing antifungal susceptibility test of the isolate.
- Rippon JW. Mycetoma. In: Medical Mycology. 3rd ed. The pathogenic fungi and pathogenic actinomycetes. WB Saunders: London; 1988. p.80-118.
- Klokke AH, Swami DG, Angul R, Verghese A. The casual agent of mycetoma in South India. Trans Royal Soc Trop Med Hyg. 1968;15:17-22.
- Chakrabarti A, Singh K. Mycetoma in Chandigarh and surrounding areas. Indian J Med Microbiol. 1998;16:64-5.
- Mahgoub ES. Agents of Mycetoma. In: Principles and Practice of Infectious Disease (Mandell LG, Bennett JE, Douglas R, ed). 3rd ed. Churchill Livingstone: New York; 1990, p 1977.
- Clinical Laboratory Standard Institute. Reference method for broth dilution antifungal susceptibility testing of filamentous fungi; Approved standard-Second edition. Clinical Laboratory Standard Institute. 2008;Wayne PA.
- Capoor MR, Khanna G, Nair D, Hasan A, Deb RM, Aggarwal P. Eumycetoma pedis due to Exophiala jeanselmei. Indian J Med Microbiol. 2007; 25(2):155-7.
- Joshi A, Acharya S, Anehosur VS, Tayaar AS, Gopalkrishnan K. Oral eumycetoma of infancy: A rare presentation and a brief review. Journal of Cranio-Maxillo-Facial Surgery. 2014; 42:35-40.
- Purkayastha J, Lewis LE, Ramesh Bhat Y, Morakhia Jwalit V, Ranjan Shetty K, Muhammad Najih L. Fungal endocarditis with right ventricular candidal mycetoma in a premature neonate. Journal of Microbiology and Infectious Diseases. 2015; 5(3): 142-145.
- Tendolkar U, Binoti S, Baveja S, Mehta N, Samaddar A, Banshelkikar S, Das SS. Unusual Presentation of Madurella Mycetomatis Mycetoma in a Paediatric Patient in India. MOJ Clin Med Case Rep. 2016; 4(5):00103.
- Iffat H, Abid K. Mycetoma Revisited. N Dermatol Online. 2011; 2(3):147-150.
© The Author(s) 2017. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.