ISSN: 0973-7510
E-ISSN: 2581-690X
Wound infections is one of the major problems worldwide. Millions of people around the world require several medical treatments for wound infections. The extensive use of antibiotics to treat wound infection leads to emerging new microbial strains that are resistant to many antibiotics. There is a growing concern on the emergence and re-emergence of drug-resistant pathogens such as multi-resistant bacterial strains. Hence, the development of new antimicrobial compounds or the modification of those that already exist to improve antibacterial activity is a high research priority. Metallic nanoparticles (NPs) are considered as new alternative treatment for wound infection with superior antibacterial activity. In this study, new formulation of titanium oxide (TiO2) NPs with different sizes were synthesized and characterized. Genotoxicity, mutagenicity and antibacterial activities of TiO2 NPs against the causative agents of wound infection were investigated. Antibacterial activity of TiO2 NPs was conducted against three ATCC® bacterial strains: methicillin-resistant Staphylococcus aureus (MRSA), Escherichia coli and Pseudomonas aeruginosa. The results clearly illustrate a superior antibacterial activity of all newly formulated TiO2 NPs against the most causative agents of wound infection. Most of our TiO2 NPs showed non-genotoxic and non-mutagenic results at the maximum concentrations. Findings of this study will enhance the future of the therapeutic strategies against the resistant pathogenic strains that cause wound infections.
TiO2 nanoparticles, antibacterial activity, multi-drug-resistance pathogens, MRSA, E. coli., P. aeruginosa, genotoxicity
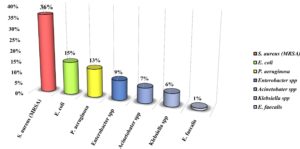
The emergence of antimicrobial resistance pathogenic strains considered as one of the major concerns World-Wide (Bassetti et al., 2015). Wound infection accounted as a common life-threatening global health problem resulting in 300,000 death every year (Song et al., 2016). Recent studies confirmed that chronic wound infections affect about 6.5 million people in U.S. alone (Kline and Bowdish, 2016). Delayed wound healing occurs due to several factors such as age, chronic diseases and infection with the pathogenic microorganisms (Morton and Phillips, 2016). These factors increase the spread of infection to the surrounding tissue and longer patients hospitalization (Gainza et al., 2015, Insan et al., 2015). Wounds are vulnerable to be infected with different microorganisms (Singh et al., 2014, Kaushik et al., 2019, Khansa et al., 2019). Thus, the inappropriate use of antibiotics such as β-lactams, vancomycin, daptomycin and rifampicin leads to the development and dissemination of multi-drug-resistant (MDR) bacteria (Chudobova et al., 2015, Friaes et al., 2015). The most common MDR bacterial species colonize wounds are methicillin-resistance S. aureus (MRSA) followed by E. coli, P. aeruginosa, Enterobacter species, Acinetobacter species, Klebsiella species and Enterococcus species (Fig. 1) (Gupta et al., 2015, Serra et al., 2015, Dhar and Han, 2020). Chowdhury et al., reported that the prevalence of bacteria isolated from infected wounds were 80% Gram negative (mainly E. coli and Pseudomonas) and 20% were Gram positive (MRSA) (Chowdhury et al., 2016, Atef et al., 2019).
Researchers are seeking out for alternative treatment scenarios to overcome the antibiotics resistance crisis since MDR pathogens take over 25,000 lives in the European Union and 23,000 lives in the USA every year (Baym et al., 2016). Nowadays, metallic NPs have been studied as highly promising alternative approach to treat wound infection (Huh and Kwon, 2011, Pachaiappan et al., 2020). These NPs have a potential broad spectrum antimicrobial activity and able to inhibit a wide range of MDR bacteria, including MRSA, P. aeruginosa and E. coli (Yah and Simate, 2015, Mihai et al., 2019). The antimicrobial activity of NPs are driven by several factors such as size, surface charge, shape and concentration (Sportelli et al., 2016). Titanium oxide (TiO2) NPs have been widely used as photocatalysts among all photocatalytic compounds (Ravishankar Rai and Jamuna Bai, 2011, Naskar and Kim, 2020). TiO2 NPs are self-cleaning, non-toxic, chemically stable highly photo-reactive and have broad-spectrum antibiotic capability (Priyanka et al., 2016, Bui et al., 2017).
In this study, we investigated the antibacterial activity of newly formulated and synthesized TiO2 NPs against the most common MDR pathogenic strains that cause wound infections. The antibacterial activity of TiO2 NPs has been investigated against the tested MDR strains at dose response manner versus several exposure time to determine their best inhibitory effect at a specific time and concentration. Introducing titanium oxide (TiO2) NPs as antibacterial agents are pushing for a novel step in the development and improvement of revolutionary therapeutic strategies.
Synthesis and Preparation of TiO2 Nanoparticles
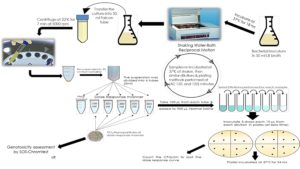
TiO2 NPs with different sizes were prepared at the nanotechnology centre, King Abdulaziz University. The synthesis of TiO2 monocrystalline structures with diameter of 3~8 nm was achieved by hydrothermal and solvothermal conditions by microwave-assisted green synthesis. The electromagnetic energy of microwave has a frequency range between (0.3-300) GHz that equivalent to meV energy from (1.24×10-6) to (1.24×10-3) (Mirzaei and Neri, 2016, Yadav et al., 2020). All samples were synthesized using 3.38 mM of Titanium (IV) Isopropoxide (Ti [OCH (CH3)2]4) that dissolved in different ratio of deionized water and ethanol. The mixtures were adjusted to 50 ml in 100 ml Teflon vessel by adding the solvent then sealed it. The synthesis was conducted at 170 ℃ in microwave oven for 90 minutes then cooled down to room temperature. TiO2 nanopowders were dried in desiccator after centrifugation and washing for three times with deionized water.
Characterization of TiO2 NPs
X-Ray Diffraction (XRD) analysis was used to determine the particles size and nature. XRD of TiO2 NPs measurements was performed by using Rigaku, Ultima-IV, which equipped with Cu-Ka radiation (λ=1.54060 nm) and operated at 40 kV and 40 mA at room temperature. The XRD spectra were measured at step size 0.05°C and 2θ angular region lied between 10°C to 80°C. In addition, the morphological features of the synthesized TiO2 NPs were examined by field emission scanning electron microscope Jeol, Japan (FESEM- JSM-7600F).
Growth Characterization of Multi-Drug-Resistance Pathogens
Methicillin-resistant Staphylococcus aureus (MRSA) (ATCC® 43300MINIPACK™), Pseudomonas aeruginosa (ATCC® 27853™) and Escherichia coli (ATCC® 25922™) were purchased from the American Type Culture Collection (ATCC) org. (Manassas, USA). The bacterial strains were characterized by monitoring the optical density (OD) and colony forming units (CFUs) of the bacterial cells over time. Luria-Bertani (LB) agar and broth were purchased from Micromaster Laboratories Pvt. Ltd. (Maharashtra, India) and prepared according to manufacture instructions. A full loop of the overnight second sub-culture colonies of each strain were inoculated in Erlenmeyer flask containing 50 ml LB broth. The inoculum was incubated in a shaker incubator (GFL Shaking Water Bath 1083 from UNIQUE Medical Laboratory Equipment Trading & Services, Sharjah, UAE) at 37°C and 150 rpm for 18 hours. The bacterial growth was monitored by measuring the optical density at wavelength 600 nm (OD600) using a spectrophotometer (GENESYS™ 20 Visible Spectrophotometer from Thermo Fisher Scientific Inc., Madison, USA) and CFUs/ml. The bacterial cells were transferred into 50 ml polypropylene conical VWR® high-performance centrifuge tubes with plug caps (VWR International, LLC Radnor, PA, USA) and harvested by centrifugation at 5000 rpm. The bacterial pellets were washed three times with 10 ml of 0.9% NaCl normal saline and centrifuged at 5000 rpm for 7 min at 25°C. After the third wash, the microbial pellets were re-suspended in 10 ml of 0.9% NaCl normal saline and the CFUs/ml and OD600 were measured to determine the optimal growth of viable cells before adding the TiO2 NPs.
Antibacterial Activity of TiO2 Nanoparticles
Five doses of the synthesized TiO2 NPs (100, 200, 400, 600 and 800 μg) were used and sterilized by UV light for 45 min. Bacterial growth was monitored using drop-plating method for counting the CFUs/ml (Miles et al., 1938). Each concentration of every NPs sample was dissolved in 5 ml of bacterial suspension and mixed gently by vortex. Serial dilutions (1:10) were accomplished for the five concentrations of TiO2 NPs by adding 100 μL of the bacterial cells to 900 μL 0.9% NaCl normal saline. Three 10 μL aliquots of the proper dilution were plated onto LB agar plate and incubated overnight at 37°C. Samples were incubated at 37°C shaker incubator with 150 rpm at different time intervals (60, 120 and 150 min). The schematic diagram for the whole experimental protocol of antibacterial activity of the synthesized TiO2 NPs was illustrated in (Fig. 2).
Monitoring the Growth Curve of the Bacterial Strains Exposed to TiO2 Nanoparticles
TiO2 NPs were added at different doses (100, 200, 400, 600 and 800 µg/ml) to measure their effect on the growth of bacterial cells. This was handled by processing the effect of presenting the TiO2 NPs on the viable bacterial cells at different time intervals. Drop-plate method was implemented for the recovered samples by spotting 10 μL aliquots in triplicates on LB agar and incubated at 37°C overnight. The dose-response curve experiment was completed after (150 min) when the bacterial cells reached to the decline phase. The antibacterial activity of TiO2 NPs against tested bacterial strains was evaluated in dose-response manner (using different concentrations) by counting the CFUs/ml versus time to detect the minimum inhibitory concentration (MIC). A comparison was done from the plotting dose-response curves of CFUs/ml versus time (min) to investigate the ideal TiO2 NPs concentration that exhibited antibacterial effect at a certain exposure time.
The Mutagenicity and Toxicity Assessment of TiO2 Nanoparticles
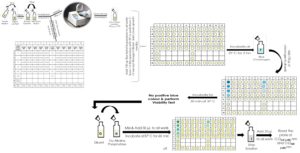
The genotoxicity of TiO2 NPs was conducted using an analytical Genotoxicity SOS – Chromo TestTM Kit purchased from EBPI (Environmental Bio-Detection Products Inc., Mississauga, Ontario, Canada). It is an enzymatic colorimetric assay to detect DNA damaging agents after incubating the tested TiO2 NPs samples with a genetically engineered bacterium E. coli PQ37 (Jabbour et al., 2016). The test was performed to detect the genotoxic samples using β-galactosidase (β-gal) and alkaline phosphatase (AP) as a signal of SOS response activation. The amount of β-gal induction is revealing the level of SOS induction and bacterial genotoxicity whereas the AP activity was used to detect the range of bacterial cytotoxicity (Kocak, 2015). Rat liver S-9 fraction was simulated the liver function metabolism for measuring the mutagenic potential of any chemical substances such as TiO2 NPs. The lyophilized bacteria were resuscitated by transferring 10 ml of growth media to the dried bacteria and roughly mixed for 30 seconds. Then, 100 μL from bacterial suspension was transferred to a new bacterial growth medium and incubated overnight for (8 – 12) hours in a rotary shaker at 150 rpm at 37°C. The overnight bacterial inoculum was diluted with fresh growth medium using the equation.1 to a final OD600 of 0.05. Various concentrations (100, 200, 400, 600 and 800 μg) of each sample of TiO2 NPs were dissolved in 1 ml of 50% dimethyl-sulfoxide (DMSO). The SOS – Chromo Test was performed with and without metabolic activation S-9.
The Required Volume of Culture = 0.5 / OD of overnight culture
Equation 1. The Required Volume for Bacterial Dilution.
The first and seventh columns of the 96 – wells microplate contained the six, two-fold dilutions of the positive control, 4-Nitro-Quinoline-N-Oxide (4-NQO) and S-9 positive control, 2-Amino – Anthracene (2-AA), in 10% DMSO. The last raw in the plate was used as negative controls while serial dilution was performed for positive controls. Other columns contained 10 μL aliquots of 10% DMSO and TiO2 NPs in a dose-response manner for each sample without performing serial dilutions. The experimental design of SOS – Chromo Test was illustrated in Fig. 3.
Statistical Analysis
The experiments were achieved in triplicate for each strain. All statistical analysis was carried out using Minitab® Statistics software for Windows, version 17.3.1 (Minitab, Inc. USA). The prediction of antibacterial activity of different concentrations of TiO2 NPs to show a reduction in the cell growth (CFUs/ml) were accomplished by analyzing the data points at several period intervals of all experiments for each strain. The non-parametric test Kruskal-Wallis and the T-test analysis were conducted to evaluate statistically significant differences (p < 0.05). A 95% confidence level was used for all statistical analysis and the curves were plotted using Excel 2010 for Windows.
Nanoparticles Structure Investigation
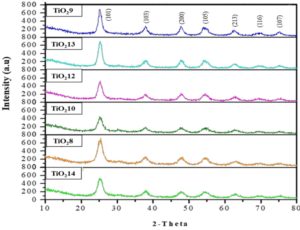
The X-ray diffractometer was used to analyze the crystalline nature, the size and the shape of atoms structure in the sample (Garino et al., 2014). The spectra of TiO2 NPs in Fig. 4 showed multiple crystalline shapes that located at several positions in the sample. The size of TiO2 NPS was calculated by using Scherrer formula (Equation 2), which constant (K) for spherical shape equals (0.9).
D= Kλ / (β cosθ)
Equation 2. The Required Volume for Bacterial Dilution.
The broad peaks illustrated relatively small size particles whereas their intensities varied among the several TiO2 NPs. Phases (101), (103), (200), (105), (213), (116) and (107) were parallel to the diffraction peaks displayed at 2θ of 25.3, 37.8, 47.8, 54.1, 62.6, 70.4 and 75.5, respectively. The calculated samples size was found to be exceeded the excitation Boher diameter by about 1.5 nm. Obviously, the reported nano-crystallite sizes revealed that sample TiO2 10 has the smallest size (3.4 nm) compared to other TiO2 NPs mentioned in Table 1.
Table (1):
TiO2 NPs Categories, Size, Solvents Percentage and Titanium Precursor Concentration.
| TiO2 NPs Categories | Samples | Size (nm) | Water: Ethanol | Concentration of Ti[OCH(CH3)2]4 (mmol) |
|---|---|---|---|---|
| Large size More than (> 5 nm) | TiO2 8 | 7.6 | (25: 75) % | 3.38 |
| TiO2 9 | 6 | (50: 50) % | 3.38 | |
| Medium Size Between (4 – 5 nm) | TiO2 12 | 4.6 | (75: 25) % | 3.38 |
| TiO2 13 | 4.9 | (100: 0) % | 3.38 | |
| Small Size Less (< 5 nm) | TiO2 10 | 3.4 | (0: 100) % | 3.38 |
| TiO2 14 | 3.7 | (100: 0) % | 1.96 |
Nanoparticles Morphological Features
Surface morphology, topography and chemical composition of the synthesized TiO2 (8-14) had been recorded using field emission scanning electron microscope (FESEM- JSM-7600F). The examined nanoparticles powder deposited on carbon tripe and demonstrated high 3- dimensional resolution images of materials. However, the images exposed condensed aggregated spherical like clusters of TiO2 NPs with approximate average sizes between 3-15 nm. Sample TiO2 10 had the smallest particle size as shown in Fig. 5 while other sample’s sizes varied between medium to large size. Thus, this variation in the particle sizes might be correlated to different ratio of solvents and deionized water used in the synthesis of the TiO2 NPs.
Fig. 5. FESEM micrograph of the TiO2 samples. All the images were taken at magnification of 120k with SEI detector and the scale shown is 100 nm.
Antibacterial activity of TiO2 nanoparticles with particle size more than 5 nm
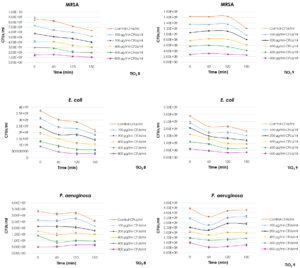
The results of antibacterial activity for TiO2 8 and TiO2 9 NPs against MRSA, P. aeruginosa and E. coli are illustrated in Fig. 6. There was a significant reduction in the number of CFUs for MRSA exposed to both TiO2 8 and TiO2 9 NPs at the lower (100 µg/ml) and the highest concentrations (800 µg/ml) after 150 min of exposure time (p = 0.018 and 0.016, respectively). Similarly, TiO2 8 NPs confirmed a significant antibacterial activity against E. coli at the maximum concentration (800 µg/ml) only after 60 min of exposure time (p = 0.034) while TiO2 9 NPs showed a significant antibacterial activity with the lowest (100 µg/ml) and the highest (800 µg/ml) after 150 min (p = 0.028). There was a significant reduction in the number of CFUs/ml of P. aeruginosa exposed to 800 µg/ml TiO2 8 NPs after 150 min (p = 0.043). Similarly, 100 µg/ml TiO2 9 NPs caused a significant reduction in the number of CFUs/ml of P. aeruginosa after 60 min of exposure time.
Fig.6. Antibacterial Activity of TiO2 Nanoparticles with particle size more than 5 nm against MRSA, P. aeruginosa and E. coli.
Antibacterial activity of TiO2 nanoparticles with particle size between 4 – 5 nm
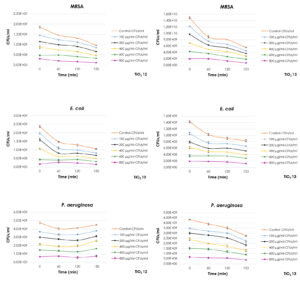
The antibacterial activity results for TiO2 12 and TiO2 13 NPs with particles size between 4-5 nm against MRSA, E. coli and P. aeruginosa were presented in Fig. 7. The antibacterial activity of TiO2 NPs was monitored in a dose-response curve using different concentrations of the NPs (100, 200, 400, 600 and 800 µg/ml). TiO2 12 NPs had superior antibacterial activity against all of three bacterial strains (Fig. 7). The maximum concentration (800 µg/ml) was more effective against MRSA and E. coli after 120 min of exposure time. The effect of NPs significantly increased after 150 min of exposure time (p = 0.009 and 0.019, respectively). There was a significant reduction in the number of CFUs/ml of P. aeruginosa after exposure to 100 µg/ml TiO2 12 NPs (p = 0.028). Similarly, TiO2 13 NPs have illustrated superior antibacterial activity against MRSA P. aeruginosa and E. coli at the lowest (100 µg/ml) and the highest (800 µg/ml) concentrations after only 60 min exposure time (p = 0.001 and 0.034, respectively).
Fig. 7. Antibacterial Activity of TiO2 Nanoparticles with particle size between 4-5 nm against MRSA, P. aeruginosa and E. coli.
Antibacterial activity of TiO2 nanoparticles with particle size less than 5 nm
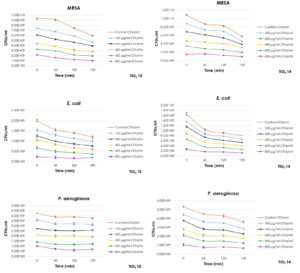
The antibacterial activities of TiO2 10 and TiO2 14 NPs with particles size less than 5 nm were illustrated in Fig. 8. There was a significant decrease in the number of CFUs/ml of MRSA exposed to different concentrations (100 µg/ml – 800 µg/ml) of TiO2 10 NPs only after 60 min exposure time (p = 0.002 and 0.006, respectively). Similar observation was reported for TiO2 10 NPs tested against E. coli. Nevertheless, TiO2 10 NPs demonstrated limited antibacterial activity against P. aeruginosa (Fig. 8).
Fig. 8. Antibacterial Activity of TiO2 Nanoparticles with particle size less than 5 nm against MRSA, P. aeruginosa and E. coli.
TiO2 14 NPs displayed a remarkable antibacterial activity against MRSA and E. coli only after 60 min of exposure time with all of the tested concentrations: 100, 200, 400, 600 and 800 µg/ml (Fig. 8). Nevertheless, the effect was limited with P. aeruginosa as only 800 µg/ml of TiO2 14 NPs showed antibacterial activity after 150 min of exposure time (p = 0.020) (Fig. 8).
Table (2):
The SOSIF Classification.
SOSIF |
Interpretation |
Comments |
|---|---|---|
SOSIF ˂ 1.5 |
Non-Genotoxic |
Safe to be used |
SOSIF = 1.5 – 2.0 |
Inconclusive |
Required further investigations |
SOSIF ˃ 2.0 |
Genotoxic |
Required more testing |
Table (3):
The Results of SOSIF Genotoxicity / Mutagenicity (orange colour is Non-Genotoxic/Mutagenic, yellow colour represents Genotoxicity/Mutagenicity, and white colour indicates inconclusive results).
| Genotoxicity without S-9 | ||||||||
|---|---|---|---|---|---|---|---|---|
| TiO28 | TiO29 | TiO210 | TiO211 | TiO212 | TiO213 | TiO214 | PC | |
| Conc 100 | 1.8994 | 1.79998 | 1.58027 | 0.97077 | 1.86769 | 1.85486 | 1.78688 | 16.9241 |
| Conc 200 | 1.36073 | 1.60371 | 1.30767 | 0.90812 | 1.59203 | 1.41954 | 1.31452 | 21.2512 |
| Conc 400 | 1.35139 | 1.72513 | 1.42012 | 1.41259 | 1.85806 | 2.09103 | 1.55882 | 14.3697 |
| Conc 600 | 1.84057 | 1.99856 | 1.58817 | 2.2077 | 1.76223 | 2.08029 | 2.01408 | 12.4203 |
| Conc 800 | 1.79617 | 1.40874 | 1.5017 | 1.28735 | 1.39187 | 1.30633 | 2.06113 | 8.39894 |
| 4.69399 | ||||||||
| Genotoxicity with S-9 | ||||||||
| TiO28 | TiO29 | TiO210 | TiO211 | TiO212 | TiO213 | TiO214 | (S-9) PC | |
| Conc 100 | 1.10219 | 1.15893 | 1.11566 | 1.04904 | 1.1013 | 1.75615 | 1.27575 | 3.38622 |
| Conc 200 | 1.34008 | 1.3729 | 1.22645 | 1.21673 | 1.37219 | 2.92085 | 2.39201 | 2.34017 |
| Conc 400 | 1.15793 | 1.3046 | 1.11214 | 1.35654 | 1.08676 | 2.46638 | 1.40647 | 1.91376 |
| Conc 600 | 0.82746 | 1.18808 | 1.0468 | 1.21316 | 1.34458 | 1.57243 | 1.30031 | 1.39771 |
| Conc 800 | 1.21262 | 1.10052 | 1.12215 | 1.11284 | 1.15748 | 1.96345 | 1.58716 | 1.22269 |
| 1.12751 | ||||||||
Mutagenicity and Toxicity Assessment of TiO2 NPs
The mutagenicity and genotoxicity results were assessed by calculating the SOS – Induction Factor (SOSIF) (Equation.3). The calculated optical density of all TiO2 NPs in the absence and the presence of S-9 activation enzyme were classified according to SOSIF classification (Table 2). Most concentrations (100, 200, 400, 600 and 800 µg/ml) of the synthesized TiO2 NPs were non-genotoxic and non-mutagenic (Fig. 9). However, concentrations reported inconclusive, require more investigations to figure out their toxicity (Table 3).
SOSIF= (OD630i ) ÷ (OD405i) / (OD630NC) ÷ (OD405NC) / (OD630NC) ÷ (OD405NC)
Equation…(3)
The emergence of antimicrobial resistance pathogenic strains considered as one of the major concerns World-Wide for human health and dramatically raised economic costs. MRSA, P. aeruginosa and E. coli are highly resistance to broad – spectrum of antibiotics which considered as the most causative agents of nosocomial infections. These strains become an endemic in hospitals and long – term care facilities because they show a dramatic increase in resistance to antimicrobial agents, especially vancomycin (John et al., 2015). Multi-drug-resistance (MDR) bacteria are tremendously hard to eradicate and guide researchers towards discovering novel strategies for treatment of wound infection. Therefore, introducing new antimicrobial agents can control the rate of morbidity and mortality that result from infectious diseases such as wound infections. Metallic NPs have been studied as highly promising alternative approach to treat wound infection (Huh and Kwon, 2011, Pachaiappan et al., 2020). TiO2 NPs are inexpensive, biologically and chemically stable, and corrosion-resistive (Xiao et al., 2015). Nowadays, the field of materials science consider TiO2 as an eco-friendly material and promising semiconductor with antimicrobial activity (Gopinath et al., 2016, Periyat et al., 2016)
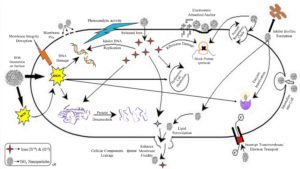
In this study, the antibacterial activity of different concentrations and sizes of the synthesized anatase TiO2 nanoparticles (NPs) was investigated against MDR strains. Our findings showed that all samples of TiO2 NPs possessed antibacterial activity against the tested strains. Nevertheless, TiO2 12 (4.6 nm) and TiO2 13 (4.9 nm) with medium size had the best antibacterial activity against all the three strains at the minimum concentration (100 µg/ml). These findings were in agreement with previous study of the antibacterial activity of metallic oxide NPs (Alkaim, 2017, Dadi et al., 2019, Kaushik et al., 2019). Antibacterial activity of TiO2 NPs is very complicated and several factors such as NPs physicochemical properties might affect their activity (Hajipour et al., 2012). The exact mechanisms of bacterial cell inhibition or death due to NPs effect were completely unclear and not fully understood. Many studies conducted to investigate the exact mechanisms of bacterial cell inhibition or death and suggested several possible scenarios. The possible inhibitory mechanism can be through electrostatic interaction and oxidative stress. Fig. 10 summarizes all possible antibacterial mechanisms of TiO2 NPs against bacterial strains.
The physiochemical properties of TiO2 NPs play a role in their antibacterial activity against bacterial community as reported in several studies (Zhao et al., 2010, Jesline et al., 2015, Hoseinzadeh et al., 2017, Kumar et al., 2017a). Many factors influenced the bacterial cell death mechanism of NPs included size, shape, concentration, electrical charge, surface structure, solvents and the exposure time (Sirelkhatim et al., 2015). Moreover, different ratio of the solvents and the concentrations of titanium used to synthesize TiO2 NPs may influence their antibacterial activity as reported in previous studies (Hu et al., 2012). Several studies showed the effect of using different solvents, precursor concentrations and conditions on the size, shape, crystal distribution, surface properties and antibacterial activity of NPs (Kumar et al., 2017b). The mixture percentage of deionized water (H2O) to Ethanol (CH3CH2OH or C2H6O) and concentrations of Titanium (IV) Isopropoxide (Ti [OCH (CH3)2]4) in each NPs sample was illustrated in Table.1.
The nanoparticles with large size (> 5 nm) such as TiO2 8 (7.6 nm) and TiO2 9 (6 nm) were prepared using half or less percentage of water to ethanol. These NPs illustrated more antibacterial activity against MRSA and E. coli with limited activity against P. aeruginosa. Samples with small size (< 5 nm) such as TiO2 10 (3.4 nm) was synthesized using ethanol only as a solvent had greater antibacterial activity against MRSA and E. coli and least effect against P. aeruginosa. Small size, fine shape and narrow distribution of particles are correlated with low precursor concentration such as sample TiO2 14 (3.7 nm) that prepared using only water as solvent and half concentration of titanium precursor (1.96 mmol). This sample had superior antibacterial activity against all of the three bacterial strains (Hu et al., 2012). On the other hand, the medium sized NPs between (4 – 5 nm) were prepared with high percentage of water to ethanol such as TiO2 12 (4.6 nm) and TiO2 13 (4.9 nm) have shown a significant antibacterial activity against MRSA, E. coli and P. aeruginosa at all concentrations. Antibacterial activity limitation towards P. aeruginosa could be correlated to the nature of resistance mechanism which is multi-factorial (Chatterjee et al., 2016). This strain possessed an intrinsic resistance and able to develop a resistance readily and rapidly resulting in decreased membrane permeability 12-100-fold than other bacteria (Taylor et al., 2014, Ramirez-Estrada et al., 2016).
Many studies reported that nearly most of the TiO2 NPs are non-genotoxic/mutagenic (Chen et al., 2014). Most concentrations of our synthesized TiO2 NPs showed non-genotoxic and non-mutagenic effect at the maximum concentration (800 µg/ml). Though, some concentrations of our particles displayed genotoxic effect and this could be due to the solvent used for dissolving the NPs which was 50% dimethyl-sulfoxide (DMSO), and 2% of it considered toxic for the cells (Alhadrami and Paton, 2013).
Nowadays, there is a great competition in finding novel technologies against MDR bacteria. Nanoparticles are widely used as antibacterial agents against several MDR pathogens. Thus, titanium oxide NPs can be a proper alternative antibacterial agent. This study sheds light on the antibacterial activity of TiO2 NPs on MDR microorganisms that cause wound infections. The TiO2 NPs exhibited high efficacy as a strong antibacterial agent towards the tested strains. Their antibacterial activity against MDR pathogens was as follows: MRSA (Gram-positive) > E. coli (Gram-negative) > P. aeruginosa (Gram-negative). Thus, the most effective samples that demonstrated superior antibacterial activity is ranked as TiO2 12 > TiO2 13 > TiO2 14 > TiO2 10 > TiO2 9 ≥ TiO2 8. The synthesized TiO2 NPs were non genotoxic/mutagenic. Thus, these NPs can be great alternative to antibiotics for the treatment of wound infection. This demonstrates potential applications of these NPs in medical and biomedical fields.
ACKNOWLEDGMENTS
The authors acknowledge with thanks DSR for technical and financial support.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
Both authors have been equally contributed towards the experimental work and writing the manuscript.
FUNDING
This project was funded by Deanship of Scientific Research (DSR), King Abdulaziz University, Jeddah, Saudi Arabia, under grant G-1439/142/248.
ETHICS STATEMENT
This article does not contain any studies with human participants or animals performed by any of the authors.
AVAILABILITY OF DATA
All datasets generated or analyzed during this study are included in the manuscript.
- Alhadrami HA, Paton GI. The potential applications of SOS-lux biosensors for rapid screening of mutagenic chemicals. FEMS Microbiol Lett. 2013;344(1):69-76.
Crossref - Hussain AJA, Abd FG, Alkaim AF, Al-Azzawi A. Eco Friendly Synthesis, Characterization and Antibacterial Activity of ZnO Nan particles using Bacillus Subtilis against Multi-Drug Resistant Bacteria. Journal of Global Pharma Technology. 2017;9(7):207-213.
- Atef NM, Shanab SM, Negm SI, Abbas YA. Evaluation of antimicrobial activity of some plant extracts against antibiotic susceptible and resistant bacterial strains causing wound infection. Bulletin of the National Research Centre. 2019;43:144.
Crossref - Bassetti M, Pecori D, Sibani M, Corcione S, De Rosa FG. Epidemiology and Treatment of MDR Enterobacteriaceae. Curr Treat Options Infect Dis. 2015;7:291-316.
Crossref - Baym M, Stone LK, Kishony R. Multidrug evolutionary strategies to reverse antibiotic resistance. Science. 2016;351(6268):aad3292.
Crossref - Bui VKH, Park D, Lee Y-CJP. Chitosan combined with ZnO, TiO2 and Ag nanoparticles for antimicrobial wound healing applications: a mini review of the research trends. Polymers (Basel). 2017;9(1):21.
Crossref - Chatterjee M, Anju C, Biswas L, Kumar VA, Mohan CG, Biswas R. Antibiotic resistance in Pseudomonas aeruginosa and alternative therapeutic options. Int J Med Microbiol. 2016;306(1):48-58.
Crossref - Chen T, Yan J, Li Y. Genotoxicity of titanium dioxide nanoparticles. J Food Drug Anal. 2014;22(1):95-104.
Crossref - Chowdhury AHMSK, Husain MA, Akter N, et al. Prevalence of Extended Spectrum b-Lactamases (ESBL) Producers Among Gram-Negative Bacilli in Wound Infection. Chattagram Maa-O-Shishu Hospital Medical College Journal. 2016;15(1):26-30.
Crossref - Chudobova D, Cihalova K, Guran R, et al. Influence of microbiome species in hard-to-heal wounds on disease severity and treatment duration. Braz J Infect Dis. 2015;19(6):604-613.
Crossref - Dadi R, Azouani R, Traore M, Mielcarek C, Kanaev A. Antibacterial activity of ZnO and CuO nanoparticles against gram positive and gram negative strains. Materials Science and Engineering: C. 2019;104:109968.
Crossref - Dhar Y, Han Y. Current developments in biofilm treatments: Wound and implant infections. Engineered Regeneration. 2020;1:64-75.
Crossref - Friaes A, Resina C, Manuel V, Lito L, Ramirez M, Melo-Cristino J. Epidemiological survey of the first case of vancomycin-resistant Staphylococcus aureus infection in Europe. Epidemiol infect. 2015;143(4):745-748.
Crossref - Gainza G, Villullas S, Pedraz JL, Hernandez RM, Igartua M. Advances in drug delivery systems (DDSs) to release growth factors for wound healing and skin regeneration. Nanomedicine: Nanotechnology, Biology and Medicine. 2015;11(6):1551-1573.
Crossref - Garino C, Borfecchia E, Gobetto R, Van Bokhoven JA, Lamberti C. Determination of the electronic and structural configuration of coordination compounds by synchrotron-radiation techniques. Coordination Chemistry Reviews. 2014;277-278:130-186.
Crossref - Gopinath K, Kumaraguru S, Bhakyaraj K, Thirumal S, Arumugam A. Eco-friendly synthesis of TiO2, Au and Pt doped TiO2 nanoparticles for dye sensitized solar cell applications and evaluation of toxicity. Superlattices and Microstructures. 2016;92:100-110.
Crossref - Gupta AK, Batra P, Mathur P, et al. Microbial epidemiology and antimicrobial susceptibility profile of wound infections in out-patients at a level 1 trauma centre. Journal of Patient Safety & Infection Control. 2015;3(3):126-129.
Crossref - Hajipour MJ, Fromm KM, Ashkarran AA, et al. Antibacterial properties of nanoparticles. Trends Biotechnol. 2012;30(10):499-511.
Crossref - Hoseinzadeh E, Makhdoumi P, Taha P, et al. A Review on Nano-Antimicrobials: Metal Nanoparticles, Methods and Mechanisms. Curr Drug Metab. 2017;18(2):120-128.
Crossref - Hu M, Bai C, Song M, Lv X, Zhang S, Qiu G. Preparation of spherical monodispersed titanium dioxide by microwave assistance. International Journal of Remote Sensing Applications. 2012;2:31-33.
- Huh AJ, Kwon YJ. “Nanoantibiotics”: A new paradigm for treating infectious diseases using nanomaterials in the antibiotics resistant era. J Control Release. 2011;156(2):128-145.
Crossref - Insan NG, Hodiwala AVB, Vashisth R, Yadav A, Danu M. Antibiotic Sensitivity Pattern of Aerobic Bacterial Isolates in Wound Infections in Navi Mumbai, India. Br Microbiol Res J. 2015;10:1-6.
Crossref - Jabbour J-F, Farah J, Abdel-Massih RM. Hospital wastewater genotoxicity: A comparison study between an urban and rural university hospital with and without metabolic activation. Journal of Environmental Engineering and Ecological Science. 2016;5:2.
Crossref - Jesline A, John NP, Narayanan P, Vani C, Murugan S. Antimicrobial activity of zinc and titanium dioxide nanoparticles against biofilm-producing methicillin-resistant Staphylococcus aureus. Applied Nanoscience. 2015;5:157-162.
Crossref - John G, Kumar KP, Gopal SS, Kumari S, Reddy BK. Enterococcus faecalis, a nightmare to endodontist: A systematic review. Afr J Microbiol Res. 2015;9:898-908.
Crossref - Kaushik M, Niranjan R, Thangam R, et al. Investigations on the antimicrobial activity and wound healing potential of ZnO nanoparticles. Applied Surface Science. 2019;479:1169-1177.
Crossref - Khansa I, Schoenbrunner AR, Kraft CT, Janis JEJP, Open RSG. Silver in wound care-friend or foe?: a comprehensive review. Plast Reconstr Surg Glob Open. 2019;7(8):e2390.
Crossref - Kline KA, Bowdish DME. Infection in an aging population. Curr Opin Microbiol. 2016;29:63-67.
Crossref - Kocak E. Investigation of potential genotoxic activity using the SOS Chromotest for real paracetamol wastewater and the wastewater treated by the Fenton process. J Environ Health Sci Eng. 2015;13:66.
Crossref - Kumar V, Singh K, Kumar A, Kumar M, Singh K, Vij A, Thakur A. Effect of solvent on crystallographic, morphological and optical properties of SnO2 nanoparticles. Materials Research Bulletin. 2017a;85:202-208.
Crossref - Mihai MM, Dima MB, Dima B, Holban AM. Nanomaterials for wound healing and infection control. Materials (Basel). 2019;12(13):2176.
Crossref - Miles AA, Misra S, Irwin J. The estimation of the bactericidal power of the blood. Epidemiology and Infection. 1938;38(6):732-749.
Crossref - Mirzaei A, Neri G. Microwave-assisted synthesis of metal oxide nanostructures for gas sensing application: a review. Sensors and Actuators B: Chemical. 2016;237:749-775.
Crossref - Morton LM, Phillips TJ. Wound healing and treating wounds: Differential diagnosis and evaluation of chronic wounds. J Am Acad Dermatol. 2016;74(4):589-605.
Crossref - Naskar A, Kim K-SJP. Recent advances in nanomaterial-based wound-healing therapeutics. Pharmaceutics. 2020;12(6):499.
Crossref - Pachaiappan R, Rajendran S, Show PL, Manavalan K, Naushad M. Metal/metal oxide nanocomposites for bactericidal effect: A review. Chemosphere. 2020;128607.
Crossref - Periyat P, Naufal B, Ullattil SG. A Review on High Temperature Stable Anatase TiO2 Photocatalysts. Materials Science Forum. Trans Tech Publ. 2016;855:78-93.
Crossref - Priyanka KP, Sukirtha TH, Balakrishna KM, Varghese T. Microbicidal activity of TiO2 nanoparticles synthesised by sol-gel method. IET Nanobiotechnol. 2016;10(2):81-86.
Crossref - Ramirez-Estrada S, Borgatta B, Rello J. Pseudomonas aeruginosa ventilator-associated pneumonia management. Infect Drug Resis. 2016;9:7-18.
Crossref - Rai RV, Bai AJ. Nanoparticles and their potential application as antimicrobials. Science Against Microbial Pathogens: Communicating Current Research and Technological Advances, Mendez-Vilas, A.(Ed.). University of Mysore, India. 2011:197-209.
- Serra R, Grande R, Butrico L, et al. Chronic wound infections: the role of Pseudomonas aeruginosa and Staphylococcus aureus. Expert Rev Anti Infect Ther. 2015;13(5):605-613.
Crossref - Singh K, Panghal M, Kadyan S, Chaudhary U, Yadav J. Antibacterial Activity of Synthesized Silver Nanoparticles from Tinospora cordifolia against Multi Drug Resistant Strains of Pseudomonas aeruginosa Isolated from Burn Patients. J Nanomed Nanotechnol. 2014;5(2):1000192.
Crossref - Sirelkhatim A, Mahmud S, Seeni A, et al. Review on zinc oxide nanoparticles: antibacterial activity and toxicity mechanism. Nanomicro Lett. 2015;7(3):219-242.
Crossref - Song Z, Sun H, Yang Y, et al. Enhanced efficacy and anti-biofilm activity of novel nanoemulsions against skin burn wound multi-drug resistant MRSA infections. Nanomedicine: Nanotechnology, Biology and Medicine. 2016;12(6):1543-1555.
Crossref - Sportelli MC, Picca RA, Cioffi N. Recent advances in the synthesis and characterization of nano-antimicrobials. TrAC Trends in Analytical Chemistry. 2016;84(Part A):131-138.
Crossref - Taylor PK, Yeung AT, Hancock RE. Antibiotic resistance in Pseudomonas aeruginosa biofilms: towards the development of novel anti-biofilm therapies. Journal of Biotechnology. 2014;191:121-130.
Crossref - Xiao G, Zhang X, Zhang W, Zhang S, Su H, Tan T. Visible-light-mediated synergistic photocatalytic antimicrobial effects and mechanism of Ag-nanoparticles@ chitosan-TiO2 organic-inorganic composites for water disinfection. Applied Catalysis B: Environmental. 2015;170-171:255-262.
Crossref - Yadav AR, Mohite, Shrinivas K. A Brief Review: Microwave Chemistry and its Applications. Research Journal of Pharmaceutical Dosage Forms and Technology. 2020;12:191-197.
Crossref - Yah CS, Simate GS. Nanoparticles as potential new generation broad spectrum antimicrobial agents. DARU Journal of Pharmaceutical Sciences. 2015;23:43.
Crossref - Zhao Z-G, Liu Z-F, Miyauchi M. Nature-inspired construction, characterization, and photocatalytic properties of single-crystalline tungsten oxide octahedra. Chemical Communications. 2010;46:3321-3323.
Crossref
© The Author(s) 2021. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.