ISSN: 0973-7510
E-ISSN: 2581-690X
The extensive use of synthetic textile dyes raises serious environmental and health concerns due to their toxicity and recalcitrancy. This research investigates the possibility of Luffa sponge (LS), a widely available biodegradable agricultural product, as an immobilization matrix for dye removal. The study focuses on isolation and characterization of dye-degrading bacteria from surface water source which is immobilized on Luffa surface to facilitate dye removal via adsorption and degradation processes. Escherichia fergusonii KD01 and Staphylococcus epidermidis KSD02 are two such isolated and tested dye degrading immobilized strains on LS. The performance of the bacteria individually in suspension culture and immobilized on LS (Bc-LS) was tested under different parameters, shaking or static, contact time (0-8 hrs), pH (4, 6, 8, and 11) and temperatures (24, 37, 42 and 45 °C). The results displayed synergistic effect of physical adsorption and microbial breakdown by Bc-LS surpassing individual bacterial performance and untreated LS. These findings were supported by FTIR (Fourier Transform Infrared) which validated the interactions between dye molecules and functional groups on the Bc-LS, while SEM (Scanning Electron Microscopy) photos showed successful bacterial immobilization on LS after treatment. Furthermore, cytotoxicity and phytotoxicity tests revealed a significant reduction in dye toxicity into less hazardous by-products. Reusability studies demonstrated LS was functional for dye adsorption after five cycles also. These findings display Bc-LS as an effective, long-lasting, and environmentally acceptable material for dye remediation, making it a suitable choice for dye wastewater treatment applications.
Luffa Sponge, Dye, Bacteria, Immobilization, Adsorption, Biodegradation, Toxicity
The discharge of synthetic dyes, notably from the textile industry, raises major environmental and health problems. These colors are highly visible, poisonous, and chemically stable, making them resistant to standard wastewater treatment methods. Their persistence in aquatic habitats affects ecosystems by lowering light penetration, impeding photosynthesis and depleting dissolved oxygen, which can result in hypoxic conditions and severe toxicity of aquatic lives.1 Methylene blue (MB), is one such cationic, inert, triphenylmethane dye bearing the chemical name 3,7-bis(dimethylamino)-phenothiazin-5-ium chloride, frequently used in paper, fabric, food and cosmetics industry.2 The partial or untreated MB in wastewaters is highly soluble and becomes positively charged due to ionization of the nitrogen atoms in the dimethylamino groups forming a quaternary ammonium ion. The dye imparts acute toxicity to phytoplanktons affecting their growth and chlorophyll content and disturbs the aquatic trophic system by diminishing zooplanktons like Daphnia magna, the ecological balancer.3,4 Further, the untreated dye is carcinogenic to humans, affecting digestive tract, neuro-systems, skin, heart and genitourinary organs.5 Another cationic dye, Malachite Green (MG), often used to dye fabrics such as silk, wool, and leather, consist of complex aromatic compounds that resist biodegradation and can bioaccumulate in species, resulting in toxicity across the food chain from lower to higher species. Both MG and its reduced form, leuco-malachite green, can accumulate in tissues, and are potential teratogenic and carcinogenic compounds. A prolonged period of exposure can result in genetic changes, neurological, respiratory, and reproductive problems.6-9 Bacterial decolorization of synthetic dyes has attained a lucrative status due to its rapid growth, ease in genome manipulation, eco-friendliness and versatility in decolorizing various dyes.10-13 This approach presents a sustainable alternative to traditional physical and chemical procedures like coagulation, advanced oxidation and membrane filtration which bear constraints of exorbitant operational costs, generation of harmful by-products and formation of excessive sludge.14
Consequently, application of dye degrading bacterial isolates is more impeccable for treatment of waste effluents dyes in green and economical approach.15-18 Several studies have highlighted dye degrading bacteria like Lysinibacillus sphaericus, Bacillus albus, Enterobacter aerogenes, and Stenotrophomonas maltophilia in decolourization of wide spectrum of dyes efficiently.19-23 To enhance stability and efficiency of these biocatalysts, microbial immobilization along with adsorption techniques, has emerged as a preferred strategy for speedy and sustainable dye removal.24,25 Whole cell immobilization is more practical than bacterial enzymes for large scale applications as enzyme immobilization has technical and economic challenges like enzyme isolation and purification.26,27
The sponge gourd, Luffa cylindrica, of the Cucurbitaceae family is a natural abundant crop found in tropical and subtropical countries and offers an ideal substrate for bioremediation. The fibrous three-dimensional strong matrix with extensive porous vascular system provides excellent scaffold for immobilization of dye degrading bacteria as well as waste water treatment.28-31 The lignocellulosic components of Luffa portray 60% cellulose, 30% hemi-cellulose and 10% lignin.32 The bacteria successfully immobilizes on Luffa due to the interaction between bacterial cell membrane components and the surface functional groups like carboxyl (-COOH) and hydroxyl (-OH) group of the matrix. Bacterial cells cling to the surface and interiors of the support matrix via hydrophobic interactions primarily with its lignin component and further attainable through electrostatic forces, often mediated by ionic strength and pH. Biofilm formation is also reported on different lignocellulosic carriers as a means of immobilization which can withstand changes in the surrounding environment with less damage. Once adsorbed, bacteria can grow and form stable biofilms in the three-dimensional structure of LS, turning into a highly active and reusable biocomposite for wastewater bioremediation.32-35
This study addresses key United Nations Sustainable Development Goals (SDGs) by emphasizing on clean water technologies and valorization of agricultural waste (LS) to sustainable treat textile dye effluents. While several investigations focused on dye degradation studies by bacterial isolates alone or in consortium, whole cell immobilized on Luffa biocomposite remains unexplored. To address the issue, two dye degrading bacteria isolated from surface water of Kamarhati region, Agarapara, Kolkata was immobilized on LS to design a novel, low-cost remediation system that synergistically combines biosorption and biodegradation for enhanced dye decolourization. After 16S rRNA, the bacteria were identified as Escherichia fergusonii KD01 (S4 strain) and Staphylococcus epidermidis KSD02 (S5 strain) which were immobilized on the surface of LS to assess their synergistic ability to remove MG and MB dye from wastewater quickly. E. fergusonii, a Gram-negative rod shaped bacterium, has displayed remarkable dye removal capability surpassing 98% MB removal utilizing a diverse spectrum of dye-degrading enzymes, including azoreductases, laccases, and peroxidases. In addition to enzymatic biodegradation, the bacteria via surface biosorption can also aid in dye decolourization with negligible intracellular bioaccumulation. S. epidermidis, a Gram-positive cocci strain, however, displayed modest dye removal (about 50%-70%) predominantly by biosorption, facilitated by the strong binding affinity of its thick peptidoglycan layer and negatively charged teichoic acids for cationic dye molecules.36-38
Novelty statement
This study reports for the first time the whole cell immobilization of E. fergusonii KD01 and Staphylococcus epidermidis KSD02 on LS for textile dye wastewater treatment. The novel approach involves enhancing high adsorption capacity of LS conjugated with dye degrading bacteria to encourage rapid decolorization of complex textile dyes by simultaneous biosorption and biodegradative approach.
Substrate, dyes and chemicals
Luffa Sponge (LS), Distilled water, Malachite Green (MG) and Methylene Blue (MB) dyes, Nutrient Broth (NB), nutrient agar (NA). The stock dye solution (1%) was prepared using distilled water and subsequently diluted. LS were obtained from local market, MG and MB dyes from SRL, India and all the chemicals were procured from Hi-Media, India.
Isolation and screening of Dye degrading bacteria
To isolate dye degrading bacteria, water sample from Loknath Mandir Lake of Kamarhati region, Kolkata, West Bengal was chosen which approximately bears location of 22.654° N latitude and 88.388° E longitude. The collected water samples were stored at 4 °C and then aseptically serial diluted for dye degradation studies. The highest diluent was plated on Nutrient agar plates supplemented with Malachite Green (MG) and Methylene Blue (MB) dyes at 37 °C overnight. Colonies from these plates were screened for dye degradation in Nutrient broth containing MB and MG dyes in a shaker incubator. From several tested isolates, two bacteria indicating superior dye removal capacity was selected for further studies. These isolated colonies were further streaked on NA to obtain pure cultures and named as S4 and S5.
Identification and Biochemical characterization of Dye degrading bacteria
The pure cultures of isolated strains (S4 and S5) were analyzed for 16S rRNA gene by Barcode Biosciences, India. DNA was isolated from respective cultures and 16S rRNA gene fragment was amplified using 16S rRNA-F and 16S rRNA-R primers which revealed a single discrete PCR amplicon of 1500 bp on agarose gel. The band was extracted, purified and using forward and reverse primers were sequenced using BDT v3.1 Cycle sequencing kit on ABI 3730xl Genetic Analyzer. Applying aligner software, consensus sequence of 16S rRNA gene was generated from forward and reverse sequence data. The obtained gene sequence of 16S rRNA underwent BLAST with the ‘nr’ database of NCBI GenBank database. Based on the maximum identity score, first ten sequences were selected and aligned using Clustal W software.39 MEGA 10 was used to calculate the Distance matrix and phylogenetic tree was constructed based on the data.40
Standard procedures of Biochemical characterization like Gram staining, IMViC, catalase and TSI were performed. Genbank submission of 16S rRNA sequencing confirmed these bacterial isolates as E. fergunosii (S4) KD01 (NCBI GenBank Accession no: PQ001941) and S. epidermidis (S5) KSD02 (NCBI GenBank Accession No.: PQ478040). The working bacterial culture was maintained in nutrient agar plates and sub-cultured in sterilized NB at 37 °C in incubator shaker. For long term storage, glycerol stock was made and the bacteria revived in NB at 37 °C when required.
Physio-chemical parameters of Dye degrading bacteria
Shaking and Static conditions
The bacterial strains were kept in shaking (120 rpm) and static conditions at 37 °C for 24 hrs to visualize the dye decolourization performance.
Temperature and pH optimization
Temperature and pH parameters were tested to determine the optimum culture conditions desirable for effective decolourization of the MB and MG dyes. For the temperature optima, the bacterial strains (S4 and S5) were grown at 4 different temperatures (25, 35, 40 and 45 °C) in shaking conditions containing sterile NB. 2% inoculum from overnight grown bacterial cells supplemented with MB and MG dyes at respective temperatures were grown for 24 hours and thereafter the spectroscopic evaluation done at specific wavelengths. Similarly, for optimum pH evaluation, pH range (4, 6, 8 and 9) was taken. The different pH was adjusted using HCl and NaOH and individual bacteria with respective dyes were subjected to overnight incubation at 37 °C in incubator shaker. The following day, dye degradation was measured spectrophotometrically at respective absorption wavelengths of each dye in UV-Vis Double Beam Spectrophotometer (Shimadzu UV1800, Japan).
Preparation of the LS
Luffa Sponges (LS) were collected and cut into uniform pieces (2 × 1.5 cm and 1-1.5 g in weight), rinsed with distilled water and disinfected in 70% ethanol for 20-30 minutes in water bath. This cleared off adhered dirt particles and loosened the porous structure, facilitating better attachment for bacterial immobilization. The LS pieces were dried in oven, autoclaved and two sets were prepared: one set for immobilizing with bacterial strains (S4) and (S5) and another set as un-immobilized LS, to assess physical adsorption alone.
Preparation of Bacteria immobilized LS (Bc-LS)
The following process was adapted from Behera et al.41 with slight modifications. Bacterial cells (S4) and (S5) were revived and freshly inoculated in sterile flasks containing NB and allowed to reach log phase (OD of ~0.3). The ensuing cells served as seed inoculum for immobilization on LS. By batch adsorption approach, LS pieces were added to separate sterile beakers containing NB, inoculated with bacterial seed inoculum. These were incubated at static conditions for 48 hours at 37 °C to allow bacterial growth and immobilization on LS creating S4-LS and S5-LS by natural adsorption process.
Dye degradation studies by Batch Adsorption
After 48 hours incubation, the LS pieces were gently rinsed with sterile distilled water to remove non-adherent cells. Each LS piece was immersed individually into 20 mL of aqueous MG and MB solution of known initial concentration. The samples were incubated under shaking conditions at 37 °C to allow sufficient contact time for the adsorption process to reach equilibrium. The incubation period was maintained from 5-12 hours, based on preliminary trials that established the approximate time required for maximum dye uptake. After equilibrium was reached, the remaining solution was analyzed to determine the residual dye concentration by measuring the optical density (OD) at 617 nm for MG and 668 nm for MB. The dye degradation was calculated by the following formula:
Degradation (%) = (Initial absorbance-Final absorbance) / (Initial absorbance) × 100
Characterization studies
Spectroscopic studies
UV-Vis spectrophotometer (Shimadzu UV1800, Japan) was used to determine the residual dyes post biosorption on LS surface at specific wavelengths. After the dye decolourization was over, samples were taken out and centrifuged to precipitate out the bacterial biomass. The supernatant was used to measure the respective O.D of the remaining solution. The degradation value was calculated based on the outputs from the UV-Vis and comparing them with initial respective dye concentrations.
The surface chemistry between dyes and LS, before and after biosorption, was analyzed using a Fourier Transform Infrared (FTIR) spectrometer in the range of 4000-400 cm-1 and resolution of 4 cm-1. Following standard procedures, the samples were prepared by homogenizing the powdered biosorbent (LS and LS-dyes) with KBr. The resulting IR spectra identified the functional groups present, revealing vibrational frequency shifts that indicate which groups were involved in the adsorption process.
Field Emission Scanning Electron Microscopy (FESEM)
To visualize bacterial immobilization, LS matrix was prepared for Field Emission Scanning Electron Microscopy (Zeiss Gemini, Germany). LS samples immobilized with the bacteria (S4 and S5) were fixed in 3.5% glutaraldehyde for 6 hour, washed with a phosphate buffer, and dehydrated in a graded ethanol series before being vacuum-dried and gold-coated. These prepared samples, along with unimmobilized LS, were imaged at magnifications between 0.5 and 10 KX.
LS reusability study
Post-dye adsorption, the LS pieces were regenerated for further dye removal studies. For dye desorption, LS pieces were soaked in acidic (H2SO4) and organic solvent (ethanol) for 1 hour under shaking condition. Then they were washed thoroughly with distilled water and air-dried at room temperature. The desorbed LS were re-used in fresh MB and MG dye solutions for further dye adsorption under same conditions. Number of adsorption-desorption cycles was repeated 5 times and dye removal efficiency was measured after each cycle.
Toxicity study
The Phytotoxicity assay, a part of ecotoxicological assessment was performed with the raw dyes (MG and MB) and degraded form of the dye on Cicer seeds (Bengal gram). Seed germination rate, plumule, and radicle lengths were measured post-treatment with dyes and its degraded form along with water as control. For the study, healthy seeds were chosen, surface sterilized with HgCl2 and again washed with distilled water. The seeds after treatment with raw dye and its degraded products were placed between wet tissue papers and allowed to germinate at room temperature. Control set was kept using tap water of the same volume at the same time. Thereafter all the parameters were recorded till 12 days’ time. The seed germination (%) formulae for phytotoxicity assay calculated is:
Germination %= Number of Sowed seeds-Number of Germinated seeds / Number of Sowed seeds × 100
Cytotoxicity assay
Lactate Dehydrogenase Activity (LDH release assay) was performed to assess the degree of cytotoxicity of the raw vs. degraded form of the dyes at defined concentrations. Cells release LDH after death into the medium; the released LDH reduces NAD to NADH, which is specifically detected colorimetrically at 450 nm through microplate reader. Peripheral blood mononuclear cells isolated from healthy donors by Ficoll density gradient centrifugation, were treated with intact and degraded form of the MB and MG dye at defined concentrations in a 96 well plate (1 x 106 cells/ well). Each concentration was plated in triplicate. Positive control (PBMCs treated with 10% Triton X-100) and negative control (untreated PBMCs) were plated alongside the test samples in triplicates. The plate was incubated for 24 hours at 37 °C with 5% CO2 in cell culture incubator. Thereafter the cytotoxicity was assessed by measuring the release of LDH is cell free supernatants and the cytotoxicity (%) was calculated by the formula-
Cytotoxicity % = (Experimental LDH Release- Spontaneous LDH Release) / (Maximum LDH Release – Spontaneous LDH Release) × 100
Statistical analysis
All the data existing are mean values of triplicate experiment results represented as ± standard error. Microsoft Office Professional 2013 was used to calculate significance level and standard error.
Bacterial strain isolation and characterization
Burgeoning evidence across literature pinpoint the entry of unbound textile dyes into surface waters apart from other sources. The adverse impact is visible in the aquatic biotic communities and the tiny microbial world. With speculation of presence of dye degrading bacteria from such cradles, the isolation source of bacteria was determined from a local surface water body (Loknath Mandir Lake) of Kamarhati region, Kolkata. After initial screening from 21 colonies, 5 colonies were chosen having dye degrading properties for both MB and MG dyes. Those selected colonies were further grown in dye supplemented NA plates from where two bacteria with superior dye degradation performance were chosen (Figures 1a,b). The bacteria displayed Gram- negative and Gram-positive features after gram staining and named subsequently as S4 and S5 (Figure 1c).
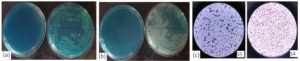 Figure 1. Screening of dye degrading bacteria on dye supplemented plates. a) Growth of S4 and S5 on MB, b) Growth of S4 and S5 on MG, c) Gram-positive (S5) and Gram-negative (S4)
Figure 1. Screening of dye degrading bacteria on dye supplemented plates. a) Growth of S4 and S5 on MB, b) Growth of S4 and S5 on MG, c) Gram-positive (S5) and Gram-negative (S4)
The S4 colonies displayed white smooth, round cells, opaque features. They are Gram-negative, rod-shaped, facultative anaerobic, and non-spore-forming bacteria. S5 demonstrated flat white smooth dense colonies on NA plates. S5 are Gram-positive cocci, clustered, facultative anaerobic, and non-spore-forming bacteria. Both bacteria are citrate and catalase positive indicating citrate is used as carbon and ammonium as nitrogen source; also catalase enzyme is produced by these bacteria to protect cell from toxic metabolites. S4 is both Indole and methyl red negative. Table demonstrates the other biochemical properties of S4 and S5.
Table:
Biochemical characterization of S4 and S5
Biochemical tests |
E. fergusonii (S4) |
S. epidermidis (S5) |
|---|---|---|
Indole |
-ve |
+ve |
Methyl Red |
-ve |
+ve |
Voges proskaur |
-ve |
+ve |
Citrate |
+ve |
+ve |
Catalase |
+ve |
+ve |
TSI |
Lactose not fermented, but small amount of glucose is fermented at butt, H2S generated at slant |
No sugars fermented, No gas, No H2S |
Optimization parameters for dye decolourization by S4 and S5
Shaking and static conditions
The dye decolourization activity of the bacterial strains were strongly dependent on agitation. Under static conditions both bacterial strains S4 and S5 displayed negligible decolourization of MB and MG after 24 hours of incubation at 37 °C. In shaking conditions at 120 rpm, both strains achieved significant decolourization for both the dyes within same time frame and temperature conditions (data not shown). Agitation creates aerobic conditions which activates oxygen dependent enzymes like laccases, azoreductases and peroxidases. These enzymes require oxygen as terminal electron acceptor and reducing powers like NADH and FADH2 to carry out robust dye degradation with high yield of bacterial biomass. Static conditions display slow oxygen diffusion from the air-liquid interface and with incubation time the dissolved oxygen is rapidly depleted creating an anoxic environment throughout the container. Bacterial growth though visible but oxygen deprivation blocks the functioning of the aerobic-dependent dye degrading enzymes. Similar reports were visible in Reactive Blue 160 degradation studies by Bacillus subtilis.29,42
Temperature and pH optimization
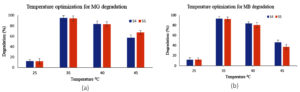
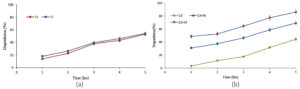
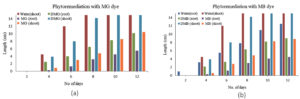
Temperature critically influences dye degradation, as it affects the microbial enzymes. To optimize the temperature influence on dye degradation studies, range of temperature (24, 37, 42 and 45 °C) was setup for working of S4 and S5 bacteria. For MG dye, S4 showed peak degradation at 37 °C (97%), while S5 demonstrated 94% at the same temperature. These results confirm 37 °C as the optimal temperature for dye degradation for both S4 and S5 (Figure 2a, b).
The dye degradation activity decreased when the temperature was raised above 37 °C or below. The optimal performance at 37 °C aligns with physiological adaptation of E. fergusonii and S. epidermidis which colonize warm blooded bodies. Reduced dyed degradation capability at lower temperatures (<37 °C) results from enzyme deactivation and reduced cellular metabolism. At higher temperature dye degrading enzymes are deactivated with loss in cell viability.43
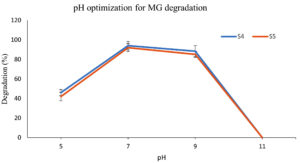
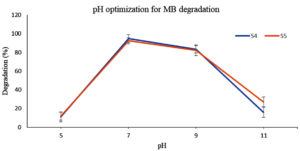
The effect of pH on both the dye removal by E. fergusonii (S4) and S5 was studied following a pH range from (5, 7, 9, and 11 for MB) and (4, 6 and 8 for MG). Beyond pH 8.0, MG transforms into leuco-malachite stage (colourless). The bacterial survivability and enzyme activity strongly depends on pH and primarily rely on the isoelectric point of the major bacterial proteins. The results in this study depicts pH 8.0-9.0 best for S4 activity which is in agreement with the data obtained by Raj et al.,20 where the optimum pH was found to be 8.7 with 98.4% dye decolorization. E. fergunosii bears 64% similarity with E. coli whereby the isoelectric point of majority of the proteins reported in E. coli lies at pH 5.5 and pH 9.5.43 E. fergunosii (S4) also displayed better dye degrading activity between pH 6.0 and pH 9.0, with pH 7.0 maximum decolourization of 94% was achieved for both dyes (Figure 3a, b).
The reduction in MB dye degrading efficiency at alkaline pH (>9.0) could be attributed to inhibition of bacterial growth or reduced passage of dye molecules across the S4 bacteria cell membrane. At pH range between 8.0-9.0, the bacterial membrane possibly had negative charge which facilitated the transport of positive dye molecules. Similarly, at low pH (4.0) the MB decolorization was insignificant due to the possibility of accumulation of surface positive charge on the bacterial membrane, which might electrostatically resist the cationic MB dye molecules. At higher pH, the issue could be minimized as the bacterial cell surface facilitates the transmembrane transport of the positively charged dye molecules.44,45 Similar results were obtained by Pillai et al.,46 where pH 7.0 demonstrated highest azo dye degradation by the fungus Streptomyces.
Dye removal capacity of LS
The dye concentration for both MB and MG was varied from 0.0125% to 1% and tested against single and double LS without any bacterial immobilization for their dye removal capacity. Single LS biosorbed both the dyes completely at their lowest concentration within 2 hours whereas double LS showed dye removal efficacy within 1 hour (data not shown). After several trials, 0.05% dye concentration was chosen in this study for complete dye decolourization within time frame of 8 hrs. At this concentration, 98.02% and 98.56% of both the dyes was adsorbed by double LS.
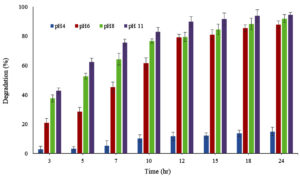
Dye removal capacity of LS at different pH
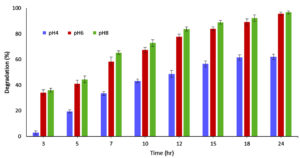
To study the effect of pH on adsorption capacity of LS, different pH range was chosen, considering the pH preference of S4 and S5 bacteria and the ionization capacity of MG dye. pH concentrations range (4.0, 6.0, 8.0 and 11.0) for MB and pH (4.0, 6.0, and 8.0) for MG were considered. MB adsorption by LS was 94.55%, 92.0%, 87.87% and 14.94% for pH 11, 8.0, 6.0 and 4. Similarly, for MG dye adsorption on LS, 96%.75%, 95.7% and 62.12% was displayed at pH 8.0, 6.0 and 4 (Figure 4a, b).
The results imply that dye sorption capacity of LS improved in basic pH region (pH 8-11) far better than at acidic pH 4.0. At basic pH, the dye adsorption is improved on the LS due to the deprotonation of hydroxyl groups (-OH) resulting in negatively charged -O– groups that attract the cationic MG dye through electrostatic interactions. At acidic pH, hydroxyl groups remain protonated and neutral, limiting the number of active binding sites for MG and resulting in decreased adsorption. The color loss is attributed to the chemical change in presence of mild reducing agents and disruption of the conjugated system.45,46
Bacterial immobilization on LS (Bc-LS)
The dye degrading bacteria S4 and S5 was immobilized on Luffa to enhance the process of dye removal both by physical adsorption of LS and enzymatic decolourization by these bacterial species. The interaction between bacterial cell membrane components and the surface functional groups like carboxyl (-COOH) and hydroxyl (-OH) of the matrix helped in the immobilization process. The successful immobilization through static incubation in NB media was demonstrated in the NA plate where the LS displayed bacterial growth after 37 °C incubation for 24 hours (Figures 5a,b).
FESEM studies
Further FESEM analysis confirmed the successful immobilization of S4 and S5 onto LS (Figures 6a, b, c).
Figure 6. Scanning electron micrographs. a) Unimmobilized LS, b) S4 bacteria immobilized on LS, c) S5 bacteria immobilized on LS
Prior to dye adsorption, the unimmobilized Luffa displays network of long fiber which are uneven and non-uniform (Figure 6a). The uniform distribution of S4 and S5 bacteria across the LS was visible and cell morphology revealed rod shaped S4 bacteria (Figure 6b) and cocci shape of S5 (Figure 6c). Attachment and bacterial spread over the complete uneven surface area of LS are crucial factors for effective biosorption of MG and MB dyes. The firm attachment of bacterial cells to the fibrous network of the LS can be credited to the existence of different functional groups like carboxyl, hydroxyl and amino in the matrix surface. Concurrent studies by Zainab et al.47 also demonstrated LS as an excellent surface carrier for adhering methanogens and their biofilm.
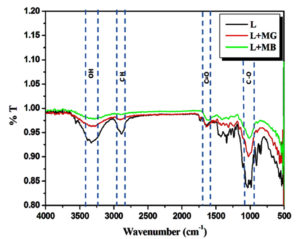
FTIR studies
FTIR spectroscopy suggested the presence of functional groups involved in LS-dye interactions. Following the adsorption of MG and MB dyes on LS, the dried and powdered LS were subjected to FTIR analysis (Figure 7). The visible spectra peak at 3,330 cm-1 is associated with the stretching of hydroxyl (OH) groups that exist in the cellulose structure of the material. The peaks found within the range of 1650-1735 cm-1 and 1100-1373 cm-1 was attributed to the C=O stretching vibration of the carbonyl group and the C-O stretching vibration of the acetyl groups, respectively. The bond observed at 2,800 cm-1 is associated with the aliphatic C–H group found in the alkyl group. After adsorption, changes in the peak intensity of LS denote the role of respective groups (O-H, C-H and C-O bonds) in the adsorption process of MG and MB dye onto LS. Similar microscopic features of the cellulosic polysaccharide were observed for absorption of MB and MG dyes while immobilizing Gluconacetobacter kombuchae on Luffa aegyptica support for bacterial cellulose production.48,49
Dye degradation studies
After the successful immobilization process, the dye degradation experiments were set up based on 37 °C temperature where the S4 and S5 showed highest dye degradation properties, otherwise temperature had no influence separately on LS adsorption. The pH was also considered between 7.5-8.5, as the best dye degradation was exhibited by S4 and S5 and good adsorption by LS in these ranges. The MG and MB dye concentration chosen was 0.05% in this study. Initially the bacteria were grown to lag phase prior to immobilization as the larger inoculum size with active seed culture minimizes the length of adaptation (lag period) phase and facilitates the biomass concentration within a short time leading. Figure 8 (a,b) displays the experimental set up of dye degradation and adsorption by S4 and S5 bacteria after 5 hours.
Figure 8. Dye adsorption and degradation studies. a) MG dye, LS and immobilized LS (Bc-LS-S4, Bc-LS-S5); b) MB dye, LS and immobilized LS (Bc-LS-S4, Bc-LS-S5) after 5 hours
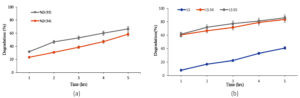
E. fergusonii (S4) individually exhibited high dye-degrading capacity when used alone displaying 54.34% and 60.04% for MB and MG dye. Whereas S. epidermidis (S5) exhibited superior dye degradation capacity with 58.16% and 62.45% for MB and MG dye.
The reason could be attributed to powerful presence of dye degrading enzymes like azoreductase, laccase, and peroxidase enzymes, facilitating breakdown of dye molecules into less toxic forms. S. epidermidis, was more active enzymatically, contributed significantly to adsorption through its thick peptidoglycan layer which enhanced surface contact and dye retention. When used in conjunction with LS, there was an enhanced degradation of 83.6% and 85.84% for MG dye by S4 and S5 within 5 hours compared to 54.34% and 58.16% for MB dye. Thus within 5 hours, dye degradation heightened by 19.06% and 14.6% by S4 for MB dye. MG dye degradation was 25.54% quicker when S4 and 19.84% when S5 was immobilized on LS than used alone (Figures 9a, b and 10a, b). Thus immobilization enhanced dye degradation in synergistic way than the use of bacteria alone.
Figure 9. Demonstrates comparison between MB degradation capabilities of (a) individual bacteria S4 and S5 with that of (b) LS alone and bacteria immobilized LS (LS-S4 and LS-S5)
Figure 10. Demonstrates comparison between MG degradation capabilities of (a) individual bacteria S4 and S5 with (b) LS alone and bacteria immobilized LS (LS-S4 and LS-S5)
Toxicity assays
Phytotoxicity
Effect of raw dye and microbial treated dye on germination and growth of Cicer seedlings were observed. Seed germination showed a variance of 60% with DMG (degraded malachite green) and 40% growth with DMB (degraded methylene blue) compared to 100% germination in water whereas no seed germination was observed with the raw dyes (Figure 11). In the pot treated with the DMG solution, healthy plant growth was observed, with an average root length of 10 cm and shoot length of 22 cm after 12 days growth period. In contrast, seeds irrigated with the raw MG solution showed stunted growth, with an average root length of 5.5 cm and shoot length of 10 cm (Figure 12a, b).
Figure 12. Comparison of root and shoot length of germinated Cicer seeds when treated with (a) MG dye and degraded MG, (b) MB dye and degraded MB
In case of DMB also similar pattern in phytotoxicity was observed whereby 9 and 20 cm were the lengths of root and shoot compared to diminished growth of raw MB treated dyes. Thus dye treatment with S4 and S5 bacteria reduced the inherent toxicity and mitigated the negative impacts on ecosystem and agricultural crops. Similar results were reported by Sudarshan et al.,50 where the germination index of Solanum lycopersicum seeds increased to 71.42% when the MG dye was treated with green synthesis compound. Kishor et al.,51 reported 100% seed germination when the toxic MB dye was degraded by Bacillus albus MW407057. Alaya et al.,52 reported 83% seed germination with degraded MG produced by a compost bacterium, Stenotrophomonas maltophilia compared to 30% germination with MG treated seeds. Thus the bacteria helped to reduce the toxicity of the dyes post-treatment compared to the raw dye which is normally present in textile effluents.
Cytotoxicity
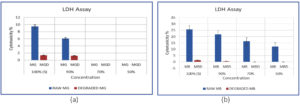
LDH, a soluble cytoplasmic enzyme found in most cells including blood, is released extracellularly upon damage to plasma membrane. The released LDH is detected by tetrazolium salt in presence of NADH which is formed when LDH oxidizes lactate to pyruvate. A colored product (formazan) is formed which is detected by standard spectroscopic techniques. In the LDH assay graph, the results imply that raw MG and MB are cytotoxic to some extents to the white WBCs, however it is pretty minimal (25% for MB and 9.5% for MG). In contrast, degraded MG (DMG) and degraded MB (DMB) has far lower cytotoxicity (~1%), even at full strength (Figures 13a, b and 14a, b).
Figure 13. Cytotoxicity test on WBCs. a) Different concentration of MG and (b) its degraded concentration, c) Different concentration of MB and (d) its degraded concentrations, (e) 96 well LDH assay plate with MB, MG, DMB and DMG in consecutive rows. Each dye concentration were examined in triplicate and the average plotted in LDH assay graph
Figure 14. Graphical representation of LDH assay. (a) MG and DMG, (b) MB and degraded MB. The calculated P values between different control and treatment groups were ≤0.05
The cytoxicity effect lies on various factors like the model system, dye concentration and the time of exposure. However the cytotoxicity effect of MG has been demonstrated earlier by works of de Almada et al.,53 on 4 different human cell lines. They cytotoxic effect of MG was effective in dose dependent manner whereby high MG caused tissue necrosis and low MG led to cellular apoptosis. Similar confirmation on MG toxicity has been reported on aquatic fishes, human cell lines and marine Vibrio fisheri.54
MB also imparts cytotoxicity to several human cell lines in dose dependent manner. As proven by earlier researchers working on the cytotoxic mechanism of MB; the dye is carcinogenic with serious implications on human brain cells, murine tumor cell line, and mouse fibroblast cells at high doses.55,56 Thus frequent exposure to MB and MG can be deleterious and the released dye should be degraded sustainable to protect lives and ecosystem.
Reusability studies
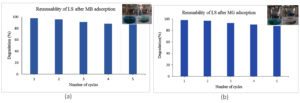
To evaluate the economic viability and practical implementation of immobilized cell systems on Luffa (Bc-LS), the carrier displayed potential for multiple use for the dye decolourization studies. LS matrix was exposed to different acid-organic solvent treatment which helped in the release of the dye from the matrix. Prior to that the immobilized cells were killed after autoclaving the Bc-LS for a stipulated time. Post acid and organic solvent treatment showed difference in color intensity between the first and 5th wash indicating a decrease in dye adsorption capacity of LS.
The matrix after the first wash cycle imparts strong dye colour, indicating strong MB and MG loading, whereas the Luffa after the 5th cycle appears much lighter, showing active site saturation or decreased binding efficiency over time (Figure 15a,b). The desorption process, reinforces the concept of renewal and reuse of the biodegradable matrix. Even after five cycles, Luffa has 86%-88% dye degradation efficiency, indicating that it can be reused more than five times for effective dye adsorption. Alvarado-Gomez et al.,57 reports 12 times reuse of LS for elimination of oils from water.
Presence of stable dye families in the effluents poses an increasing risk to the environment, and efforts to find practical and environmentally friendly ways to remove them are urgent. Luffa has attracted interest because of its porous nature, being renewable and its strong adsorption properties. Surface modification of Luffa by immobilizing dye degrading microbes brings together physical binding and biological processes. Moreover, immobilization provides enhanced stability, reusability and resistance for these microbes. Ongoing efforts aim to improvise surface properties of Luffa further to make it suitable for domestic and industrial uses. With this synergistic method, not only dye uptake efficiently increased, but provide a low-cost, environmentally friendly solution to dye remediation, making it ideal for decentralized wastewater treatment or small-scale companies. However, more research is required to improve operational settings, ensure long-term microbial viability, and evaluate system performance under real-world industrial effluent circumstances.
ACKNOWLEDGMENTS
The authors acknowledge the contribution of Mr. Kolimo Prashanth Reddy and Dr. Pallab Dutta, NIPER, Kolkata for FE-SEM works and UGC-DAE, Kolkata for FTIR data. The authors sincerely acknowledge Dr. Saptak Banerjee, Senior Scientific Officer, Chittaranjan National Cancer Institute for providing valuable guidance and resources to conduct studies on human blood samples. The authors also thank JIS University for providing facilities and support to carry out the research work.
CONFLICT OF INTEREST
The authors declare no conflict of interest.
AUTHORS’ CONTRIBUTION
DP conceptualized and supervised the study. KD, BD and SB performed the research work. DP and PM wrote the manuscript. SD reviewed and revised the manuscript. All authors read and approved the final manuscript for publication.
FUNDING
This study was partially funded by the Scottish Church College, Faculty Research Grant.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
Not applicable.
- Dutta S, Adhikary S, Bhattacharya S, et al. Contamination of textile dyes in aquatic environment: Adverse impacts on aquatic ecosystem and human health, and its management using bioremediation. J Environ Manag. 2024;353:120103.
Crossref - Contreras M, Grande-Tovar CD, Vallejo W, Chaves-Lopez, C. Bio-removal of methylene blue from aqueous solution by Galactomyces geotrichum KL20A. Water. 2019;11(2):282.
Crossref - Moorthy AK, Rathi BG, Shukla SP, Kumar K, Bharti VS. Acute toxicity of textile dye Methylene blue on growth and metabolism of selected freshwater microalgae. Environ Toxicol Pharmacol. 2020;82:103552.
Crossref - Xiang X, Zhou J, Lin S, et al. Dual drive acute lethal toxicity of methylene blue to Daphnia magna by polystyrene microplastics and light. Sci Total Environ. 2022;840:156681.
Crossref - Oladoye PO, Ajiboye TO, Omotola EO, Oyewola OJ. Methylene blue dye:Toxicity and potential elimination technology from wastewater. Results in Engineering. 2022;16:100678.
Crossref - Islam T, Repon MdR, Islam T, Sarwar Z, Rahman MM. Impact of textile dyes on health and ecosystem: a review of structure, causes, and potential solutions. Environ Sci Poll Res. 2022;30(4):9207-9242.
Crossref - Hemashenpagam N, Selvajeyanthi S. Textile Dyes and Their Effect on Human Beings. In: Ahmad A, Jawaid M, Mohamad Ibrahim MN, Yaqoob AA, Alshammari MB. (eds) Nanohybrid Materials for Treatment of Textiles Dyes. Smart Nanomaterials Technology. Springer, Singapore 2023:41-60
Crossref - Sarkar S, Bayen S, Samanta S, Pal D. Spent Mushroom Substrate- Prospects and Challenges of Agrowaste management into sustainable solutions: A Review. Int J Ag Env Biotech. 2024;17(04):731-741.
Crossref - Gharavi-Nakhjavani MS, Niazi A, Hosseini H, et al. Malachite green and leucomalachite green in fish: a global systematic review and meta-analysis. Environ Sci Poll Res. 2023;30(17):48911-48927.
Crossref - Hamad MTMH, Soliman MSS. Application of Immobilized Aspergillus niger in Alginate for Decolourization of Congo Red Dye by Using Kinetics Studies. J Polym Environ. 2020;28:3164-3180.
Crossref - Cheng CM, Patel AK, Singhania RR, et al. Heterologous expression of bacterial CotA-laccase, characterization and its application for biodegradation of malachite green. Bioresour Technol. 2021;340:125708.
Crossref - Mishra A, Takkar S, Joshi NC, et al. An integrative approach to study bacterial enzymatic degradation of toxic dyes. Front Microbiol. 2022;12:802544.
Crossref - Najim AA, Radeef AY, Al Doori I, Jabbar ZH. Immobilization: the promising technique to protect and increase the efficiency of microorganisms to remove contaminants. J Chem Technol Biotechnol. 2024;99(8):1707-1733.
Crossref - Maji P, Pal D. Nanotechnology in Advanced Oxidation Processes for Water Remediation. In: Garg MC, Rajput, VD, Minkina T, Himanshu SK. (eds) Nano-solutions for Sustainable Water and Wastewater Management. Nanotechnology in the Life Sciences. Springer, Cham. 2025.
Crossref - Khan A, Nayarisseri A, Singh SK. Characterization and optimization of azo dyes degrading microbes isolated from textile effluent. Sci Rep. 2025;15(1):11241.
Crossref - Mburu A, Njuguna D, Nzila C, et al. Comparative treatment of textile dye wastewater with chemical coagulants and bacterial exopolysaccharides. The Journal of The Textile Institute. 2025;116(9):2100-2110.
Crossref - Ariffin F, Latif MAS. Biological treatment of azo dye in textile wastewater:A comprehensive review. Nutrients and Colored Compounds in Wastewater. 2025;495-514.
Crossref - Pandey A, Kumar S, Bithel N, Kumar S, Mir MA. Sustainable biodegradation of malachite green dye by novel non-pathogenic Pseudomonas aeruginosa ED24. World J Microbiol Biotechnol. 2025;41(2):44.
Crossref - Srinivasan S, Bankole PO, Sadasivam SK. Biodecolorization and degradation of textile azo dyes using Lysinibacillus sphaericus MTCC 9523. Front Environ Sci. 2022;10:990855.
Crossref - Raj DS, Nagarajan SV, Raman T, Venkatachalam P, Parthasarathy M. Remediation of textile effluents for water reuse:decolorization and desalination using Escherichia fergusonii followed by detoxification with activated charcoal. J Environ Manag. 2021;277:111406.
Crossref - Pandey A, Kumar S, Bithel N, Kumar S, Mir A. Sustainable biodegradation of malachite green dye by novel non-pathogenic Pseudomonas aeruginosa ED24. World J Microbiol Biotechnol. 2025;41(2):44.
Crossref - Wanyonyi WC, Onyan JM, Shiundu PW, Mulaa FJ. Biodegradation and detoxification of melachite green dye using novelenzymes from Bacillus cereus strain KM201428: Kinetic and Metabolite Analysis. Energy Procedia 2017;119:38-51.
Crossref - Al-Ansari MM, Li Z, Masood A, Rajaselvam J. Decolourization of azo dye using a batch bioreactor by an indigenous bacterium Enterobacter aerogenes ES014 from the waste water dye effluent and toxicity analysis. Environ Res. 2022;205:112189.
Crossref - Asranudin, Purnomo AS, Holilah, et al. Adsorption and biodegradation of the azo dye methyl orange using Ralstonia pickettii immobilized in polyvinyl alcohol (PVA)–alginate–hectorite beads (BHec-RP). RSC Adv. 2024;14(26):18277-18290.
Crossref - Hasanien YA, Zaki AG, Abdel-Razek AS. Employment of collective physical pretreatment and immobilization of Actinomucor biomass for prospective crystal violet remediation efficiency. Biomass Conv Bioref. 2023;15(2):3235-3249.
Crossref - Zdarta J, Jesionowski T, Pinelo M, et al. Free and Immobilized Biocatalysts for Removing Micropollutants from Water and Wastewater: Recent Progress and Challenges. Bioresour Technol. 2022;344:126201.
Crossref - Tite TE, Ngema PT, Makhathini TP. Exploration of free and immobilized biocatalysts for the treatment of paper industry effluents. Separation & Purification Reviews. 2025;54(3):258-275.
Crossref - Oun AA, Kamal KH, Farroh K, Ali EF, Hassan MA. Development of fast and high-efficiency sponge-gourd fibers (Luffa cylindrica)/hydroxyapatite composites for removal of lead and methylene blue. Arab J Chem. 2021;14(8):103281.
Crossref - Rahman SSA, Vaishnavi T, Vidyasri GS. et al. Production of bacterial cellulose using Gluconacetobacter kombuchae immobilized on Luffa aegyptiaca support. Sci Rep. 2021;11:2912
Crossref - Xu R, Wei J, Cheng D, et al. Abundant porous biochar derived from luffa vine for removal of methylene blue:Selective adsorption and mechanistic studies. Industrial Crops and Products. 2024;219:119114.
Crossref - Chen L, Lu H, Jiang X, et al. Microorganisms immobilized hydroxyethyl cellulose/luffa composite sponge for selective adsorption and biodegradation of oils in wastewater. Int J Biol Macromol. 2024;277(Part 1):133477.
Crossref - Lago A, Rocha V, Barros O, Silva B, Tavares T. Bacterial biofilm attachment to sustainable carriers as a clean-up strategy for wastewater treatment:A review. Journal of Water Process Engineering. 2024;63:105368.
Crossref - Dzionek A, Wojcieszynska D, Hupert-Kocurek K, Adamczyk-Habrajska M, Guzik U. Immobilization of Planococcus sp. S5 Strain on the Loofah Sponge and Its Application in Naproxen Removal. Catalysts. 2018;8(5):176.
Crossref - Mishra A, Singh D, Singh RS, Mishra V, Kumar M, Giri BS. Performance study of the bioreactor for the biodegradation of methyl orange dye by luffa immobilized Stenotrophomonas maltophilia and kinetic studies: A sustainable approach. Groundw Sustain Dev. 2024;27:101378.
Crossref - Patil NP, Bholay AD, Kapadnis BP, Gaikwad VB. Biodegradation of Model Azo Dye Methyl Red and other Textile Dyes by Isolate Bacillus circulans NPP1. J Pure Appl Microbiol. 2016;10(4):2793-2800.
Crossref - Ayed L, Chaieb K, Cheref A, and Bakhrouf A. Biodegradation and decolorization of triphenylmethane dyes by Staphylococcus epidermidis. Desalination. 2010;260(1-3):137-146.
Crossref - Stingley RL, Zou W, Heinze TM, Chen H, Cerniglia CE. Metabolism of azo dyes by human skin microbiota. J Med Microbiol. 2010;59(Pt 1):108-114.
Crossref - Agha HM, Jawad AH, Wilson LD, Al-Essa K, ALOthman ZA. A chitosan-Staphylococcus epidermidis bacterial biocomposite adsorbent for removal of reactive orange 16 dye: Box–Behnken design optimization. Int J Biol Macromol. 2025;309(Pt 1):142752.
Crossref - Thompson JD, Higgins DG, Gibson TJ. CLUSTALW:improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrixchoice. Nucleic Acids Res. 1994;22(22):4673-4680.
Crossref - Kumar S. MEGA3: Integrated software for Molecular Evolutionary Genetics Analysis and sequence alignment. Brief Bioinform. 2004;5(2):150-163.
Crossref - Behera S, Mohanty RC, Ray RC. Ethanol production from mahula (Madhuca latifolia L.) flowers using free and immobilized (in Luffa cylindrical L. sponge discs) cells of Zymomonas mobilis MTCC 92. Ann Microbiol 2011;61(1);469-474.
Crossref - Barathi S, Aruljothi KN, Karthik C, Padikasan IA. Optimization for enhanced ecofriendly decolorization and detoxification of Reactive Blue160 textile dye by Bacillus subtilis. Biotechnol Rep (Amst). 2020;28:e00522.
Crossref - Jeon W-B. Retrospective analyses of the bottleneck in purification of eukaryotic proteins from Escherichia coli as affected by molecular weight, cysteine content and isoelectric point. BMB Rep. 2010;43(5):319-324.
Crossref - Thornton LA, Burchell RK, Burton SE, et al. The effect of urine concentration and pH on the growth of Escherichia coli in canine urine in vitro. J Vet Int Med. 2018;32(2):752-756.
Crossref - Arab C, El Kurdi R, Patra D. Effect of pH on the removal of anionic and cationic dyes using zinc curcumin oxide nanoparticles as adsorbent. Mater Chem Phys. 2022;277:125504.
Crossref - Pillai HPJS. Optimization of Process Conditions for Effective Degradation of Azo Blue Dye by Streptomyces DJP15. J Pure Appl Microbiol. 2017;11(4):1757-1765.
Crossref - Zainab A, Meraj S, Liaquat R. Study on Natural Organic Materials as Biofilm Carriers for the Optimization of Anaerobic Digestion. Waste Biomass Valor. 2020;11:2521-2531.
Crossref - Ngwenya V, Ndebele NR, Ncube LK, Nkomo NZ, Gadlula S, Ndlovu LN. Enhanced technique for removal of methylene blue dye from water using Luffa microcrystalline cellulose. S Afr J Chem. 2024;78:113-121.
Crossref - Mashkoor F, Nasar A. Preparation, characterization and adsorption studies of the chemically modified Luffa aegyptica peel as a potential adsorbent for the removal of malachite green from aqueous solution. J Mol Liq. 2019;274:315-327.
Crossref - Sudarsan S, Murugesan G, Varadavenkatesan T, Vinayagam R, Selvaraj R. Phytotoxicity mitigation and malachite green removal from wastewater using superparamagnetic activated carbon. ACS omega. 2025;1(30):33788-33805.
Crossref - Kishor R, Saratale GD, Saratale RG, et al. Efficient degradation and detoxification of methylene blue dye by a newly isolated ligninolytic enzyme producing bacterium Bacillus albus MW407057. Colloids Surf B: Biointerfaces.2021;206:111947.
Crossref - Alaya V, Kodi RK, Ninganna E, Gowda B, Shivanna MB. Decolorization of Malachite green dye by Stenotrophomonas maltophiliaa compost bacterium. Bull Natl Res Cent. 2021;45:81
Crossref - de Almada Vilhena AO, Lima KMM, de Azevedo LFC, et al. The synthetic dye malachite green found in food induces cytotoxicity and genotoxicity in four different mammalian cell lines from distinct tissues. Toxicol Res. 2023 12(4):693-701.
Crossref - Radko L, Minta M, Trebas S. Cellular toxicity of malachite green and leucomalachite green evaluated on two rat cell lines by MTT, NRU, LDH, and protein assays. Bull Vet Inst in Pulawy. 2011;55(2):347-353.
- Lee YS, Wurster RD. Methylene blue induces cytotoxicity in human brain tumor cells. Cancer Letters. 1995;88(2):141-145.
Crossref - Sanchala D, Bhatt LK, Pethe P, Shelat R, Kulkarni YA. Anticancer activity of methylene blue via inhibition of heat shock protein 70. Biomed Pharmacother. 2018;107:1037-1045.
Crossref - Alvarado-Gomez E, Tapia JI, Encinas A. A sustainable hydrophobic luffa sponge for efficient removal of oils from water. Sustain Mater Technol. 2021;28:e00273.
Crossref
© The Author(s) 2026. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.