ISSN: 0973-7510
E-ISSN: 2581-690X
The study included investigation virulence factors that product from urease positive bacteria species isolated from urinary tract infection. a total of 60 clinical specimens, taken from urine sample had been cultured and identified pure isolates result show like Klebsiella.pneumonia (n = 5, 8.3%), Staphylococcus.sp (n = 3, 5%) and Pseudomonas aeruginosa (n = 1, 1.6%). Most isolated of urease positive bacteria were negative gram stains excepted Staphylococcus sp. was positive gram stain. All isolates gave positive result of catalase test, while coagulase, oxidase, indole, Simmon citrate and kligler test was performed for all isolated gave variable result. Also all urease positive bacteria were identified by using VITEK-2 system. Klebsiella.pneumonia, Staphylococcus.aureus and Pseudomonas.aeruginosa non fermentation for lactose sugar test while Staphylococcus.Saprophyticus had ability lactose fermentation. Antibiotic susceptibility for all urease positive bacteria results showed that all highly sensitive towards (Amikacin, Gentamycin, Imipenem, Norfloxacin, Ciprofloxacin and Vancomycin) while showed low sensitivity towards (Ceftriaxone, Cefotaxime, Nalidixic acid and Nitrofurantoin). In addition all bacteria produce urease showed had Clear difference in another virulence factors like (DNase, Lipase, Protease).
Virulence Factors, Urease positive bacteria, UTI, Clinical specimens
Urinary tract infection (UTI) caused by different Gram-negative bacteria is the most common infections. About 150 million patients are suffering UTI each year worldwide. Antibiotics resistance in uropathogens bacteria are a major global attention which can lead to failure of treatment, genetically modified of bacteremia, increase for requirement therapy, and lengthening of hospital accommodation UTI is the second infectious disease after the respiratory disorder. Catheterization process is the main risk factor of urinary tract infection1-3. The high risk of UTI at women’s pregnancy. They are common, reminder diagnosis and treatment as even occasional bacteria can be related with reverse pregnancy result. Complications of embryo include early labour, stillbirth and depressed birth weight. The pyelonephritis is a maternal complication with has a high return rate4. The urease positive bacteria are five species with urease activity (Proteus.mirabilis, Pseudomonas.areginousa, Klebsiella pneumoniae, Enterobacter.cloacae and Staphylococcus aureus) were subsequently isolated from different patient clinical samples5. Coagulase-negative staphylococci (CNS) are a many of Gram-positive bacteria. They are consisting of 16 Subspecies are known to cause human infection. Makeup about 9% of nosocomial infections. The species are most recurrent causes diseases in humans are Staphylococcus epidermidis, Staphylococcus. haemolyticus and Staphylococcus. Saprophyticus. Whereas Staphylococcus aureus is the best known and has been frequently implicated in the etiology of a series of infections and intoxications in humans6-8. Pseudomonas. aeruginousa has many types of virulence factors that have important roles in the pathogenicity of this bacterium. The major virulence factors are toxin A, alkaline protease (aprA), elastase, and have many exoenzymes. Two phospholipases C enzymes may also have roles in the hydrolysis of phospholipids in lung tissue9,10. The potency virulence factors of bacterial pathogenesis associated with the disease pathway, including swarming, growth rates, fimbria expression, flagella, and the production of hemolysins, ureases, proteases, lipase in addition Dnase have been described in many studies11 Lipopolysaccharide (LPS) on their exterior is the essential composition of the outer membrane and exclusive of the central virulence factors of the microorganism It composed of a polysaccharide part, containing an O-specific chain (O-antigen, O-PS), and a core region, as well as a lipophilic region, termed lipid A, which anchors the LPS to the bacterial outer membrane12-14. Staphylococcus.epidermidis and other CONS are infected bacteria, it contains the extracellular products like protease, DNase, lipase, hemolysis and external enzymes (exoenzymes) probably responsible for human cell tissues degradation and spreading of a disease caused by these germ organism15-17.
Samples
Sixty samples were obtained from AL Ramadi teaching hospital for Maternity and Children from the period June to August /2018. Collected from suspected patients suffering from urinary tract infection in a different age.
Collection and Culture Samples
All specimens were collected in sterilized recommended containers. The urine specimens were run by centrifuge machine at 1500 rpm for 5 minutes. The discarded of the supernatant then cultured the precipitate by streaking on MacConkey agar, blood agar then incubated at 37°C for 24-48 hour18.
Laboratory diagnosis
It was included microscopic examination of the growing bacterial cells were carried out by transferring a pure single colony to glass slide and procedure gram staining19. The selected colonies undergo biochemical tests to diagnose the species level and as follows:
Catalase test
Application of this test to differentiate between staphylococci and streptococci bacteria. Transfer single colony by age (18-24) hour to slide and add few drop of 3% hydrogen peroxide. Immediate bubble formation indicating a positive test20.
Oxidase test
Pick up some colonies from the growth media by the sterile wooden sticks loop into the moistened filter paper by 1% oxidase reagent. The change in the color of the zone to purple color after (30) second indicating for a positive result.
Coagulase test (slide method)
Was rapid diagnosis used as the following: add two drops of sterile saline on each area of the divided slide. Then transfer the bacterial colonies to make a suspension. And was treated with undiluted plasma. Observed the result by clumping within 10-20 second of the bacterial suspension was positive result21.
Mannitol Fermentation test
Mannitol salt agar (MSA) was used for investigation staphylococci pathogenic types mainly Staphylococcus aureus. Furthermore, inoculated with a clear colony of growing bacteria and incubated at 37C° for 24 hours. the ph phenol read indicator (MSA) through changed to a yellow color mean positive result22.
Indole test
The peptone water inoculated by single colony form isolated bacteria, incubated for 24-48 hours at 37ºC added after that 0.5ml of Kovacs reagent. And recorded the result23. 2.3.6 Citrate utilization test: (Simmon´s citrate agar) bacterial inoculated in agar and incubated for 24-48hours at 37ºC the sodium carbonate generated were change the indicator color (bromothymol blue) from green to blue color23.
Kligler iron agar (KIA)
All isolated species was inoculated and incubated at 37Cº for overnight, change in color phenol-red indicator from red to yellow color means a positive result24.
Vitek 2 system for identification of bacteria
The bacterial isolates from urine were identified by Vitek 2 compact System. The selected cards were used depended upon result of the gram stain and conditions growth of the microorganism to be tested25.
Sugar Fermentation
Broth Fermentation Tests were used to differentiate between microorganism ability to carbohydrate fermentation by a metabolic process. Each kind of following sugar: glucose, fructose, sucrose, maltose, lactose, xylose, mannose and sorbitol were placed in tubes containing 5 ml Purple Broth Base medium, the results appear after 24-48 hour of incubation at 37°C24,26.
Antibiotic Susceptibility test
To determined antimicrobial agent we used Muller – Hinton (MH) agar by Kirby-Bauer Disk diffusion technique. Transferred the bacterial growth and spread by sterile glass spreader L – shape. The antibiotics disc was placed. The plates were incubated for 24 hours at 37 °C. And the determined result by measuring the millimeter zone inhibition27.
Statement of some virulence Factors
Determination of DNase test
Prepared DNase agar according to the instruction of Manufacture Company. Inoculated the plates from growth clear colonies of bacterial isolated and incubated for 18-24 hour at 37°C, the plate flooded with 1N HCl reagent and leave the plates for few minute reactions by absorbing the Hydrochloric acid. The positive reaction indicates through a clear zone surrounding growth colonies on the DNase agar28.
Determination of Urease test
Inoculating the isolated bacteria onto the urea agar slant tubes prepared according to the instruction of Manufacture Company and incubated at 37ºC for 18-24 hours, observe any change of color in the inoculated urea agar to a pink color is a positive result of urease bacterial activity23.
Determination of Lipase production
Inoculating the isolated bacteria into lipase activity agar Tributryin agar and the growth colonies had the opacity after 24-48hour incubation time at 37ºC defined the positive result test29.
Determination of Protease production
Detected of protease production test on skim milk (SM) agar. The agar was prepared according to the instruction of Manufacture Company. The isolated bacteria were inoculated and the clear zone surrounding colonies through hydrolysis of casein main component in milk is a positive reaction30.
A total of sixty clinical patients samples were investigated as urinary tract infection tabulated samples from urine culture. Also, the samples distributed as the gender group female and male. 28% from all cultures are the positive percentage as shown in Table 131. The risk factors estimation which associated with kinds of culture samples and appeared the urine sample cultures is more represented because of many acquired urinary tract infection32.
Table (1):
The percentage of risk factors associated with a patient infected.
| Variables | Groups | Number Patient |
Cultures +ve | Cultures -ve | ||
|---|---|---|---|---|---|---|
| N | % | N | % | |||
| Samples | Urine | 60 | 17 | 28.33 | 43 | 71.66 |
| Age | ≤ 2 year | 39 | 11 | 28.20 | 28 | 71.79 |
| >2 year | 21 | 6 | 28.57 | 15 | 71.42 | |
| Gender | Male | 20 | 5 | 25 | 15 | 75 |
| Female | 40 | 12 | 30 | 28 | 70 | |
| Total | 60 | 17 | 28% | 43 | 72% | |
Statistical analysis revealed the age and gender was considered as a risk factor. The age is more significant risk factors. As well as another studies they were reported such as linked to the surgical process, a hospital long time stay, and used the urinary devices33,34.
We studied important virulence factors in urease positive bacteria species which causes different urinary inflammation for the human being. In this study, the specimens were collected from different patients represented to AL Ramadi Teaching Hospital for Maternity and Children. The microscopical examination for all isolates bacteria were done by Gram stain. The results showed the most predominant isolated bacteria pathogen was Escherichia coli (n = 8, 13.3%) the second major causative microorganism Klebsiella.pneumonia (n = 5, 8.3%) followed by Staphylococcus.sp (n = 3, 5%) and Pseudomonas aeruginosa (n = 1, 1.6%)Table 2.19
Table (2):
Number and percentage of bacteria isolated from UTI patients.
| Bacterial Isolates | Number and percent of bacteria isolates (from 60 patients) | ||
|---|---|---|---|
| Number | Gram Stain | % | |
| E.coli | 8 | – | 13.3 |
| Klebsiella pneumonia | 5 | – | 8.3 |
| Staphylococcus.aureus | 2 | + | 5 |
| Staphylococcus.Saprophyticus | 1 | + | |
| Pseudomonas.aeruginosa | 1 | – | 1.6 |
| Negative culture result | 43 | ||
| Total | 60 | ||
The results constant with the previous studies of feedback in which E. coli Pseudomonas aeruginosa Klebsiella pneumonia and others were the major causative pathogen isolated from urinary tract disorder patients31.
Table (3):
Biochemical tests of bacterial isolated.
| bacterial isolated |
Biochemical test | |||||
|---|---|---|---|---|---|---|
| Catalase | Coagulase | Oxidase | Indole | Citrate | Kligler Iron | |
| Klebsiella. pneumonia |
+ | – | – | + | + | + |
| Staphylococcus. aureus |
+ | + | – | – | – | + |
| Staphylococcus. Saprophyticus |
+ | – | – | – | – | + |
| Pseudomonas. aeruginosa |
+ | – | + | – | + | – |
Table 3 showed all isolates of bacteria species were given a different result for biochemical identification. Klebsiella.Pneumonia gave positive result for catalase, indole, Simmon citrate and kligler iron test while gave negative result for oxidase and coagulase test.
Staphylococcus aureus gave positive result for catalase test; coagulase test is indicator test for Staphylococcus. aureus from another Staphylococcus sp. and had capability to ferment mannitol salt agar to produce large yellow zone colonies like a golden stain (Fig. 1)35. In addition to a positive result for kligler iron test and a negative result for Simmon citrate and indol test. Staphylococcus.Saprophyticus species gave positive result for catalase test and kligler iron.
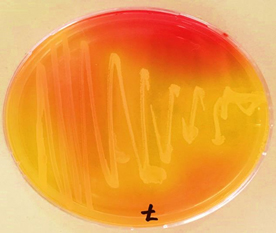
Fig. 1. Staph.aurues isolated (golden stain) on Mannitol salt agar
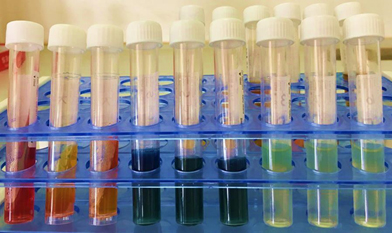
Fig. 2. Biochemical test for Urease +ve isolated bacteria
Pseudomonas aeruginosa gave positive result for catalase and oxidase test that is differentiates test for Pseudomonas sp. these results agreement with the study of [10] who found that all Pseudomonas aeruginosa isolated positive reaction for Simmon citrate while negative reaction for kligler iron test, indole test and glucose fermentation test (Fig 2). Additionally for identification of isolated bacteria we used Vitek 2 compact system and the percentage of bacterial tested was ranged from (85-97%),
Table (4):
Sugar fermentation for the bacterial strain.
| Bacterial Strain |
Sugar Fermentation | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Glucose | Lactose | Sucrose | Maltose | Fructose | Xylose | Mannose | sorbitol | ||
| Klebsiella pneumonia |
+ | – | + | + | + | + | + | + | |
| Staphylococcus. aureus |
+ | – | + | + | + | + | + | + | |
| Staphylococcus. saprophyticus |
– | + | + | – | + | + | + | + | |
| Pseudomonas. aeruginosa |
– | – | + | + | + | + | – | + | |
+: positive result; – : negative result.
Table 4 showed all isolated were fermented some types of sugar through change the indicator color as a positive result for sucrose, fructose, xylose, and sorbitol. Klebsiella pneumonia gave positive fermentation for glucose, maltose ,mannose and helpless to lactose fermentation test this results in agreement with [36].
While Staphylococcus aureus had various result for sugar type’s fermentation glucose, maltose and mannose. All isolates were unable to ferment lactose exception Staphylococcus.saprophyticus and had ability less for glucose and maltose fermentation. (Fig: 2) Also Pseudomonas aeruginosa gave positive result for maltose fermentation and negative result to ferment glucose, lactose and mannose.
Table (5):
Antibiotic sensitivity test.
| Urease PS bacteria Isolated | Antimicrobial agent | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| AK | IPM | VA | CRO | CTX | NOR | NA | CIP | NI | GM | |
| Klebsiella pneumonia |
++ | – | – | R | R | +++ | + | +++ | R | + |
| Staphylococcus. aureus |
++ | ++ | ++ | – | R | – | – | – | – | + |
| Staphylococcus. Saprophyticus |
++ | +++ | ++ | R | R | – | – | – | – | ++ |
| Pseudomonas. aeruginosa |
++ | +++ | – | +++ | ++ | ++ | R | R | R | + |
AK \Amikacin, IPM \ Impienem VA \Vancomycin , CRO \Ceftriaxone, CTX\Cefotaxime, NOR \Norfloxacin, NA \Nalidxic acid, CIP\Ciprofloxacin , NI\Nitrofurantion, GM \Gentamycin
Seventeen selected isolates from urine samples were tested for resistance toward ten antibiotics and only urease positive bacteria were tested Table 5. In all urinary tract infections studies, Klebsiella pneumonia is present37. and the sensitivity test result show with Amikacin, Norfloxacin, Gentamycin and Ciprofloxacin, sensitive which is agreement with the previous data of studies38 while resistance for Cephalosporines group (Ceftriaxone and Cefotaxime).

Fig. 3. Sugar fermentation test for Urease +ve bacteria isolated
Staphylococcus aureus and Staphylococcus. Saprophyticus isolated show clear sensitive result aminoglycoside antibiotic group with (Amikacin, Gentamycin) and good sensitive for Imipenem, Vancomycin39 Beta-lactam antibiotics Ceftriaxone, Cefotaxime are actually often multi-resistant with staphylococcal infections40.
In this project, Pseudomonas aeruginosa showed the variable sensitive result for Amikacin and Gentamycin. Imipenem agent gave quite broad sensitive reaction against Pseudomonas aeruginosa. this results in consistent with41 The major reason for resistance Aminoglycoside antibiotic group through Aminoglycoside-modifying enzymes (AMEs) secretion42 (Fig. 4).
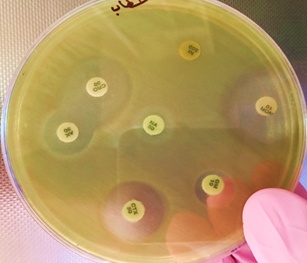
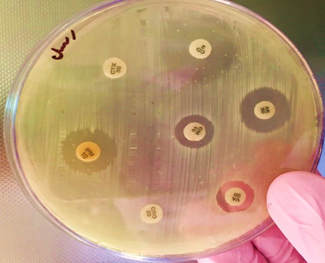
Fig. 4. Antibiotic Resistance Test for Staphylococcus sp. and Pseudomonas sp
Table (6):
Virulence factors which produce by Urease PS bacterial isolated from UTI samples.
| Bacterial Isolates | Virulence Factor | |||
|---|---|---|---|---|
| DNase | Urease | Lipase | Proteases | |
| Klebsiella pneumonia | – | + | – | – |
| Staphylococcus.aureus | + | + | + | – |
| Staphylococcus Saprophyticus | – | + | – | – |
| Pseudomonas.aeruginosa | – | + | + | + |

Fig. 5. Urease positive test
Results in table 6 showed all isolated bacteria were positive for Urease test because of the most predominant clinical samples from urine (Fig. 5). Staphylococcus aureus. produces many types of a virulent factor. Showed of all species isolated production DNAse (Fig. 6) and Lipase (Fig. 7)17. Lipase activity has main properties for nutrition of the bacteria. The correlation between lipase extracellular activity and pathogenicity of staphylococci is a very equal role in patients with the Staphylococcus aureus infections also interfering with phagocytosis process43. while the isolated gave a negative result with Protease.

Fig. 6. DNase positive result for Staph.aureus isolated
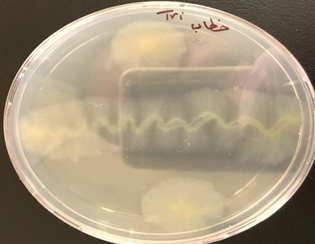
Fig. 7. Lipase test for Staph.aureus isolated
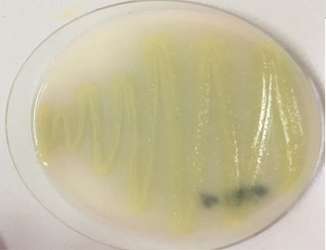
Fig. 8. Protease test for Pseud aeruginosa
Pseudomonas aeruginosa isolated release a large number of factors showed cultured positive result for media used Lipase and exactly for protease enzyme factor (Fig. 8)44. while gave negative result for DNAse, from the result appear various production of protease enzyme depending on the source isolation. The enzyme has importance role for the resistance of phagocytosis by neutrophils inhibition and destroys surface-bound IgG and C3.
The study appears different bacterial isolation from urinary tract infection in different parameter. The female patients are more prone to infection from male. Antibiotic sensitivity for bacterial isolated appeared bigger variation result compare with bacteria in same species. The variation in produce virulence factors depended on the species types but all isolated were positive for urease test because the urine contains urea composed.
ACKNOWLEDGMENTS
None.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
- Khoshnood, S., et al., Drug-resistant gram-negative uropathogens: a review. Biomedicine & Pharmacotherapy, 2017. 94: p. 982-994.
- Badamchi, A., et al., Molecular detection of six virulence genes in Pseudomonas aeruginosa isolates detected in children with urinary tract infection. Microbial pathogenesis, 2017. 107: p. 44-47.
- Gad, G.F.M., et al., Detection of icaA, icaD genes and biofilm production by Staphylococcus aureus and Staphylococcus epidermidis isolated from urinary tract catheterized patients. The Journal of Infection in Developing Countries, 2009; 3(05): p. 342-351.
- Curtiss, N., I. Meththananda, and J. Duckett, Urinary tract infection in obstetrics and gynaecology. Obstetrics, Gynaecology & Reproductive Medicine, 2017. 27(9): p. 261-265.
- Osaki, T., et al., Urease-positive bacteria in the stomach induce a false-positive reaction in a urea breath test for diagnosis of Helicobacter pylori infection. Journal of medical microbiology, 2008. 57(7): p. 814-819.
- Goudarzi, M., et al., Characterization of coagulase-negative staphylococci isolated from hospitalized patients in Tehran, Iran. Journal of Paramedical Sciences (JPS) Spring, 2014. 5(2): p. 2008-4978.
- Piette, A. and G. Verschraegen, Role of coagulase-negative staphylococci in human disease. Veterinary microbiology, 2009. 134(1-2): p. 45-54.
- Layer, F., et al., Comparative study using various methods for identification of Staphylococcus species in clinical specimens. Journal of clinical microbiology, 2006. 44(8): p. 2824-2830.
- Krall, R., et al., Pseudomonas aeruginosa ExoT is a Rho GTPase-activating protein. Infection and immunity, 2000. 68(10): p. 6066-6068.
- Fazeli, N. and H. Momtaz, Virulence gene profiles of multidrug-resistant Pseudomonas aeruginosa isolated from Iranian hospital infections. Iranian Red Crescent Medical Journal, 2014. 16(10).
- Jacobsen, S.M., et al., The high-affinity phosphate transporter Pst is a virulence factor for Proteus mirabilis during complicated urinary tract infection. FEMS Immunology & Medical Microbiology, 2008. 52(2): p. 180-193.
- Torzewska, A., P. St¹czek, and A. RóŸalski, Crystallization of urine mineral components may depend on the chemical nature of Proteus endotoxin polysaccharides. Journal of medical microbiology, 2003. 52(6): p. 471-477.
- Kaca, W., et al., Human complement activation by smooth and rough Proteus mirabilis lipopolysaccharides. Archivum immunologiae et therapiae experimentalis, 2009. 57(5): p. 383-391.
- Ró¿alski, A., Lipopolysaccharide (LPS, endotoxin) of Proteus bacteria-chemical structure, serological specificity and the role in pathogenicity. Acta Universitatis Lodziensis. Folia Biologica et Oecologica, 2008(Acta Universitatis Lodziensis. Folia Biologica et Oecologica 04/2008).
- Vuong, C., et al., Polysaccharide intercellular adhesin (PIA) protects Staphylococcus epidermidis against major components of the human innate immune system. Cellular microbiology, 2004. 6(3): p. 269-275.
- Begun, J., et al., Staphylococcal biofilm exopolysaccharide protects against Caenorhabditis elegans immune defenses. PLoS pathogens, 2007. 3(4): p. e57.
- Cunha, M.d.L.R.d., L.M.S.d.S. Rugolo, and C.A.d.M. Lopes, Study of virulence factors in coagulase-negative staphylococci isolated from newborns. Memórias do Instituto Oswaldo Cruz, 2006. 101(6): p. 661-668.
- Colle, J., et al., Practical Medical Microbiology (Makie & McCartne). International Student Ed. Of, 1996.
- Tille, P., Bailey & Scott’s Diagnostic Microbiology-E-Book. 2015: Elsevier Health Sciences.
- Josephine, A., Morella, A. M., Helen-Eckel,, laboratory manual and work book in microbiology application to patient care. . 8 ed., 2006.
- Vandepitte, J., Basic Laboratory Procedure in clinical Bacteriology/. J. Vandepitte (et al) World Health Organisation., 2003.
- Brooks, G.F.C., K. C., :Morse, S. A. and Mietzner, T.A. , Staphylococcus. (Jawetz). Melnick and delberg’s, Medical Microbiology. . 2010.
- MacFaddin, J.F., Biochemical tests for identification of medical bacteria. 1st ed. Williams and Wilkins.Baltimore, USA., 2000.
- Pierce, M.J.L.B.E., A Photographic Atlas for the Microbiology laboratory 2010. 4th EDITION: p. 71-74.
- Pincus, D.H., Microbial identification using the bioMerieux VITEK® 2 System. Encyclopedia of Rapid Microbiological Methods. Bethesda, MD: Parenteral Drug Association, 2006.
- Kools, W.E.a.s., T.L., Update on clinical significance of coagulase negative staphylococci. Clin. Microbiol., 1994. Rev., 7: p. 117-140.
- Cockerill, F.R., Clinical, and L.S. Institute, Performance standards for antimicrobial susceptibility testing: twenty-second informational supplement;[… provides updated tables for… M02-A11 and M07-A9]. 2012: National Committee for Clinical Laboratory Standards.
- Brown, A., Benson’s Microbiological Applications: Laboratory Manual in General Microbiology, Short Version. 2007: McGraw-Hill HigherEducation.
- Chamberlain , N.R.a.B., S.A., Characterization and expression of fatty acids modifying enzymes produce by Staphylococcus epidermidis. . J.Med.Microb, 1997. 46: p. 693-697.
- Benson, H.J., Microbiology applictions laboratory manual in general microbiology Vol. 7 ed. 1998, McGraw-Hill
- Dimitrov, T.S.U., E.E. Emara, M. Awni, F. Passadilla,R., Etiology and Antibiotic Susceptibility Patterns of Community-Acquired Urinary Tract Infections in a Kuwait Hospital. Med Princ Pract, 2004(13): p. 334 –339.
- Al-Kazaz, A.-K. and W.W. Al-Bassam, The isolation and characterization of Proteus mirabilis from different clinical samples. Jornal of Biotechnology Research Center, 2013. 7(2): p. 24-30.
- Jroundi, I., et al., Prevalence of hospital-acquired infection in a Moroccan university hospital. American journal of infection control, 2007. 35(6): p. 412-416.
- Guerado, E., et al., Role of mental disorders in nosocomial infections after hip fracture treatment. Interdisciplinary perspectives on infectious diseases, 2010. 2010.
- Thorberg, B.-M., et al., Bovine subclinical mastitis caused by different types of coagulase-negative staphylococci. Journal of dairy science, 2009. 92(10): p. 4962-4970.
- Holt, J.G., et al., Group 19. Regular, nonsporing Gram-positive rods. Bergey’s manual of determinative bacteriology, 9th ed. Williams & Wilkins Co., Baltimore, Md, 1994: p. 566.
- Barnett, B.J. and D.S. Stephens, Urinary tract infection: an overview. The American journal of the medical sciences, 1997. 314(4): p. 245-249.
- Meharwal, S., et al., Complicated nosocomial UTI caused by nonfermenters. Indian Journal of Urology, 2002. 18(2): p. 123.
- Dheyab, A.S., Antibiotic Resistance and Molecular DNA Fingerprinting of Staphylococcus aureus from Clinical Samples. 2013.
- Oliveira, D.C. and H. De Lencastre, Methicillin-resistance in Staphylococcus aureus is not affected by the overexpression in trans of the mecA gene repressor: a surprising observation. PLoS One, 2011. 6(8): p. e23287.
- Cox, G., et al., Structural and molecular basis for resistance to aminoglycoside antibiotics by the adenylyltransferase ANT (2 3 )-Ia. MBio, 2015. 6(1): p. e02180-14.
- AL-Kaisse, A.A., A.N. AL-Thwani, and R.Q. AL-Segar, Incidence and Antibiotics Sensitivity of Multidrug-Resistance of Pseudomonas aeruginosa Isolated from Burn’s Patients and Environmental Samples fromThree Hospitals in Baghdad. Jornal of Biotechnology Research Center, 2015. 9(2): p. 67-73.
- Dinges, M.M., P.M. Orwin, and P.M. Schlievert, Exotoxins of Staphylococcus aureus. Clinical microbiology reviews, 2000. 13(1): p. 16-34.
- Martins, V.V., et al., Pathogenic potential and genetic diversity of environmental and clinical isolates of P seudomonas aeruginosa. Apmis, 2014. 122(2): p. 92-100.
© The Author(s) 2018. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


