ISSN: 0973-7510
E-ISSN: 2581-690X
The present study was included isolating alkaloid from the fruit of the nutmeg (Myristica fragrans) where observed the presence one of alkaloid compound in the fruit of the nutmeg , which is the nature of the aromatic and has carbonyl and phenol groups in addition to the presence of nitrogen . The isolated alkaloid was showed antibacterial activity against Gram-positive bacteria (Staphylococcus aureus) and Gram- negative bacteria (Escherichia coli) while the alkaloid compound was showed antifungal effect against Candida albicans and Cryptococcous neoformans. The isolated alkaloid compound was not showed any cytotoxicity of human blood solution in concentration of (10-200 ppm). From this result we concluded that the alkaloid isolated from Myristica fragrans can be used in treatment of various disorders because of the effectiveness of the compound against bacteria and fungi.
Myristica fragrans, antibacterial activity, antifungal activity, alkaloid.
Myristica fragrans, is a tropical evergreen tree (family Myristicaceae), nutmeg was used as a sachet, and the Romans used it as incense native of the E. Moluccas. It is found only as a specimen tree in Botanical gardens. The seed of the plant is known as “nutmeg” and the spice made of its seed and the arillus of the seed is called “mace”. Both nutmeg and mace contain many volatile oils. The contents of these oils have a variety of individual pharmacological effects, some of which disagree others (Jellin et al.; 2005). The fruit contains ethereal oil-cells often with phenolic and myristicin; the seed and the aril are used for flavouring food (Pal et al., 2011).
Nutmeg is diffuse as a spice and also possesses various therapeutic properties. It has a characteristic pleasant fragrance and a slightly warm taste. It is used to flavour many kinds of baked foods, confections, confectionery, meats, sausages, vegetables and drinks. Medicinally, Nutmeg is used as an anti-diarrheal agent for patients with medullar carcinoma of the thyroid. The effectiveness of the treatment may be due to the inhibition of prostaglandin synthesis in the mucosa and sub mucosa of the colon. It is sometimes used as a stomachic, tonic, carminative as well as for intestinal colic and catarrh, to stimulate appetite, to control flatulence, it also promotes and regulates the flow of menstruation and abortion (Olaleye et al., 2006).
Plant Materials & Chemical
The Nutmeg Myristica fragrans plant used in this study was obtained from the local markets. All the chemicals were buy from Sigma & Alderich Co.
The reagents were prepared in deionized distilled water to eliminate the pollution of metal ions.
Preparation of extracts
Alkaloid extract
Twelve (12 g) of plant powder defending oil was mixed with 150 ml of ethanol (10% acetic acid) and left on continuous mixing under room temperature for 4 h. The mixture was filtered by using filter paper (Whatmann No, 31) and Buchner funnel; then the filtrate was concentrated to a quarter of its size by a rotary evaporate at 70°C. The sulfuric acid (2%) was added to the concentrated filtrate and the concentrated filtrate was treated with concentrated ammonium hydroxide until pH9,then the solution was filtered and separated by addition 20ml of chloroform. The precipitate was removed by filtration, through filter paper, then filtrates was concentrated under vacuum then dried at room temperature (Yubin et al., 2014), the weight of yellow amorphous compound was 1.935g.
Preliminary qualitative test
Preliminary tests were carried out on the alkaloid compound, as showed in the table (1).
Table (1):
Preliminary qualitative test for isolated alkaloids.
| Test | Test Result | Notes | Conclusion | ||
|---|---|---|---|---|---|
| Dragendroff | + | Formation of the orange precipitate | Presence of alkaloids | ||
| Molish | – | No violet ring | No carbohydrate | ||
| Benedict | – | No red precipitate | No glycosides | ||
| FeCl3(1%) | – | No blusich green colour | No phenols | ||
| HgCl2(5%) | – | No white precipitate | No saponins | ||
| Ninhydrin (1%) | – | No violet colour | No amino acids | ||
| Libermann-burchard | – | No violet colour | No triterpenes and sterols | ||
Thin layer chromatography
TLC was transferred out on the alkaloid extract by using (Ethanol: H2O: HAC) (5:1:1).
Solubility test
Uses several polar and nonpolar solvent to solubility test to alkaloid compound.
Active groups test
After ensuring the one alkaloid compound, then uses active groups test to do it .
FTIR and UV-Visible spectroscopy
Fourier transform Infrared using PyE-UNICAM-30300S, and UV-Visible analysis using JASCOUV, were done to alkaloid compound.
Biological activity
Two bacterial isolates, namely Escherichia coli and Staphylococcus aureus were used to establishment the antibacterial activity, in addition to two isolates of yeasts, Candida albicans and Cryptococcous neoformans that were used to discernment of antifungal activity. All clinical bacterial with yeast isolates were obtained from microbiology laboratories of Biology Department, Science College, Basrah University.
Antibacterial and Antifungal tests
The minimum inhibitory concentration, (MICs) for extract of alkaloid was evaluated by broth microdilution method (CLSI, 1996 and CLSI, 2002). The extract was dissolved in dimethyl sulfoxide (DMSO) with concentrations ranging from 100-0.05 µg/ml, while the suspension for each isolate was prepared and incorporated to a turbidity of 0.5 McFarland scale (1×106 cfu/ml ) for yeasts and (1×108 cfu/ml) for bacteria, and diluted with broth microdilution technique to reach the final concentration 1-5×103 cfu/ml for yeasts and 1-5 x104 cfu/ml for bacteria.
The test was performed by using microtiter plates and each concentration was tested in duplicate with positive and negative controls (the microorganism and pure media respectively). The plates were incubated at 37°C for 24 h for bacteria and at 30°C for 48 h for yeasts.
The MIC was measured by the lowest concentration of extract that yield an 80% reduction in observable growth was compared with the control, as well as, when MIC value was 100 µg/ml or less, the antimicrobial activity was considered significant.
Cytotoxicity test
Biocompatibility test was carried out for the prepared alkaloid extract against fresh human blood according to Nair et. al. (1989) method that including in briefly:- Different concentration of extract were prepared (200, 100, 50 and 10 ppm), then 100 µl of each tube of human blood solution. The tubes were left at room temperature and the formation of turbidity of blood solution was tested after 15, 30 and 60 min. as an indication for the cytotoxicity of extract.
In the existing study, alkaloids extract was separated and purified with extraction percentage equal to (16.39%). Table (1) appears the qualitative chemical analysis of alkaloids segregate from Myristica fragrans. The results showed the presence of alkaloid only but carbohydrate, glycosides, phenols, saponins, and amino acids were not found. It is known that alkaloids are a lot in many medicinal plants. Table (2) show the thin layer chromatography (TLC) result of an alkaloid extracted from Myristica fragrans. There one spot was separated have rate of flow(Rf) value equal to 0.59 was tested and developed by Dragendroff reagent as a qualitative and specific developer for alkaloid, also by using iodine vapour the alkaloid separated showed brown colour and by using 40% H2SO4 which showed the alkaloid was attached with sugar (Harborne, 1984).
Table (2):
TLC of alkaloid extracted from myristica fragrans.
| Eluent system | Reagent | Spot.NO | Flow rate (Rf) | conclusion |
|---|---|---|---|---|
| (EtOH:H2O:HAC) (5:1:1) | I2-Vapour | 1 | 0.592 | Presence of nitrogen organic compounds |
| Dragendroff | 1 | 0.592 | Presence of alkaloid | |
| 40% H2SO4 | 1 | 0.592 | Presence sugar with alkaloid |
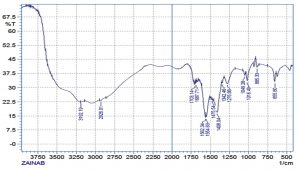
For making sure of purification alkaloid compound isolated from Myristica fragrans and determination of chemical groups for it, where observed the isolated compound contains aromatic structure, carbonyl group, nitrogen and phenol groups as shown in table (3), Fig.(1) and table (5) shows the full scan of IR spectrum of alkaloid extract we deduce from the spectrum that the alkaloid extract is molecular that has an aromatic composition and contains a carbonyl group. The isolated alkaloid compound also showed solubility in ethanol, methanol and n-hexane as indicated in the table (4).
Table (3):
Statements the effective groups of alkaloid compound extracted from myristica fragrans.
Double bond |
Alcohol |
Carboxylic acid |
Aldehyde &ketone |
Amines |
Phenols |
|---|---|---|---|---|---|
+ |
– |
– |
+ |
+ |
+ |
Brown solution |
– |
– |
Yellow precipitation |
yellow solution |
Purple solution |
Fig. 2. Cytotoxicity assay indicated that Myristica fragrans alkaloid extract have no cytotoxicity effect against different concentrations of human blood solution during 15, 30 and 60 min. at room temperature (1= 200 ppm, 2=100 ppm, 3=50, 4=10 ppm and 5= control tube)
Table (4):
Solubility of alkaloid compound extracted from Myristica fragrans.
water |
Ethanol |
methanol |
n-hexane |
chloroforme |
Petroleumether |
|
|---|---|---|---|---|---|---|
Alkaloid compound |
– |
+ |
+ |
+ |
– |
– |
The UV-Spectrum shows maximum absorption at 298 nm due to (p-p*) transition, which is the characteristic of the unsaturated double bond.
Table (5):
Full scan of the IR spectrum of alkaloid extract.
Band frequency cm-1 |
Bond |
Function group |
|---|---|---|
3500 |
O-H |
Aliphatic & aromatic |
3192 |
C-H |
Aromatic(C-H) |
3500 |
NH2 |
Amine group |
1554,1562 |
C=O |
Carbonyl group |
1276 |
C-O |
Phenol(C-O) |
The antibacterial and antifungal activities of Myristica fragrans alkaloid extract against two bacterial and two yeast isolates were investigated in this study. The result has reported the potency of alkaloid extract as antibacterial and antifungal activities with concentration ranging from 100-50 µg/ml (Table 6).
Table (6):
Antibacterial and antifungal activity of Myristica fragrans alkaloid extract against some bacterial and yeast isolates.
Isolates |
MIC (µg/ml) |
|---|---|
Bacteria |
|
Staphylococcus aureus |
100 |
Escherichia coli |
50 |
Yeasts |
|
Candida albicans |
50 |
Cryptococcous neoformans |
100 |
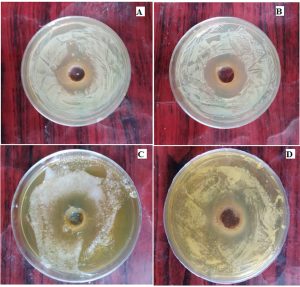
The current study clearly indicates that Myristica fragrans alkaloid extract exhibited excellent antimicrobial activity against both gram positive and gram negative bacteria in addition to significant antifungal activity against yeast isolates (Fig. 3).
Fig. 3. Antibacterial and antifungal activity of isolated alkaloid compound
A-Staphylococcous aureus
B-E.coli
C-Candida albicans
D-Cryptococcous neoformans
The present work was revealed that no any cytotoxic effect of Myristica fragrans alkaloid extract against human blood solution in all its concentrations that used in cytotoxicity assay conditions (Fig. 2).
ACKNOWLEDGMENTS
The authors are grateful to Departments of Chemistry and Biology, College of Science, University of Basra for providing materials and instrument used in the completion of the present study.
CONFLICT OF INTEREST
The authors declares that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors have made substantial contribution to the work and approved it for publication.
FUNDING
None.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
This study does not include tests on humans or animals except cytotoxicity test which performed by using 5 ml of human blood was taken from author Dr. Inaam Alrubayae as volunteer.
- Jellin, JM, Gregory, PJ, Batz, F, Hitchens, K et al. Pharmacist’s Letter/ Prescriber’s Letter Natural Medicines Comprehensive Database. 7th ed. Stockton, CA: Therapeutic Research Faculty, 2005. p 918 -919.
- Pal M, Srivastava M, Soni DK, Kumar A, Tewari SK. Composition and anti-microbial activity of essential oil of Myristica fragrans from Andaman Nicobar Island. Int. J. Pharm. Life Sci., 2011; 2(10): 1115-1117.
- Anon. The Wealth of India Raw Materials, Volume 6. CSIR, New Delhi, 1962, pp. 473-480.
- Jan R. Assa, Simon B. Widjanarko, Joni Kusnadi, Siegfried Berhimpon. Antioxidant Potential of Flesh, Seed anMace of Nutmeg (Myristica fragrans Houtt) International Journal of ChemTech Research CODEN (USA): IJCRGG ISSN : 0974-4290, 2014; 6(4): pp 2460-2468.
- Barceloux, D.G ., Medical. Toxicology of Natural Substances: foods, fungi, medicinal herbs, plants, and venomous animals. John Wiley and Sons Inc. U.S.A., 2008; pp: 67-70.
Crossref - DeMilto, L. and Frey, R.J. In: J. L. Longe (ed.), the Gale Encyclopedia of Alternative Medicine. Farmington Hills, Mich: Thomson/Gale. 2005.
- Pal M, Srivastava M, Soni DK, Kumar A, Tewari SK. Composition and anti-microbial activity of essential oil of Myristica fragrans from Andaman Nicobar Island. Int. J. Pharm. Life Sci., 2011; 2(10): 1115-1117
- Olaleye, T. Afolabi C. Akinmoladun and Akindahunsi A.A. Antioxidant properties of Myristica fragrans (Houtt) and its effect on selected organs of albino rats African Journal of Biotechnology, 2006; 5(13): pp. 1274-1278.
- Jellin, JM, Gregory, PJ, Batz, F, Hitchens, K et al. Phamacist’s Letter/ Prescriber’s Letter Natural Medicines Comprehensive Database. 7th ed. Stockton, CA: Therapeutic Research Faculty, 2005.
- Parimala N. and Amerjothy S. Histological and Histochemical Investigations of Myristica fragrans Houtt. Myristicaceae, 2013; 1(5): Page 106.
- Yubin J., Yu Miao Y, WangBing and Zhang Yao (The extraction, separation and purification of alkaloids in the natural medicine) Journal of Chemical and Pharmaceutical Research, 2014; 6(1): 338-345.
© The Author(s) 2019. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.