ISSN: 0973-7510
E-ISSN: 2581-690X
Diabetic ulceration is a multi-factorial problem which is responsible for considerable morbidity threatening the health care system. By knowing the clinical profile and bio-burden on diabetic ulcer, it is highly beneficial for health treatment. The purpose of the present study was to evaluate the diversity of major bacterial etiology in diabetic ulcer patients. The different samples like pus, swab, and infected tissues were collected from diabetic ulcer patients aseptically and samples were transported through cold chain to the laboratory. The samples were cultured in nutrient agar, mannitol salt agar, macConkey agar and blood agar. Suspected colonies were biochemically confirmed for the isolation of Staphylococcus aureus, Pseudomonas aeruginosa, E.coli and Klebsiella spp. In total 150 diabetic ulcer patients suffering from diabetic foot ulcers, gangrene, burn cases and accidental ulcer cases were analyzed. However, most of the patients developed mono-microbial infection; S.aureus was the most prevalent microbe in diabetic ulcer cases, which were positive for nucA gene.
S. aureus, Diabetic foot ulcer, Etiology
Diabetes is a major health issue that has reached alarming levels with nearly half a billion people living with diabetes worldwide. At present, 463 million adults are living with diabetes and by 2045, this will rise to 700 million1. The proportion of people with type 2 diabetes is increasing in most countries. Along with the rising prevalence of diabetes, an increase in its complications is also expected. Patients with diabetes are more susceptible to infections due to increased glucose levels and suppressed immune response as well as the neuropathy and decreased blood flow to the extremities that lead to slow-healing wounds2. Diabetic foot ulcerations and infections are one of the major medical, social, economic problem and the leading cause of morbidity and mortality, especially in the developing countries like India3. It is estimated that around 15% of diabetic patients develop foot ulcers in their life time. Diabetes accounts for more than 50% of amputation of which 85% of lower amputation in diabetes patients are preceded by foot ulcers4. The global prevalence of diabetic foot ulcers varies from 3% in Oceania to 13% in North America, with a global average of 6.4%.The annual incidence of diabetic foot ulcer (DFUs) or necrosis in diabetic patients is known to be about 2% to 5% and the lifetime risk ranges from 15% to 20%5. Diabetic foot ulcers are most commonly caused by irritated or wounded feet, nerve damage, poor circulation and hyperglycaemia. Unusual swelling, redness, irritation and stenches on one or both feet are common initial symptoms of foot ulcers6. Prevalence of diabetic foot ulcer in the clinical population of India is found to be 3.6%7. There are scanty reports available regarding the incidence of different bacterial pathogens associated with DFU in Odisha. So the present study has been envisaged to document the spectrum of bacterial pathogens associated with diabetic ulcer in this region.
Study subjects
One hundred and fifty patients with diabetes attending general surgery ward for diabetic foot ulcer management at different tertiary care Hospital, Bhubaneswar were included during the study period from November 2019 to March 2020. Patients willing to participate in the study were enrolled. Samples were collected from patients with different grades of wounds/ulcers and gangrene after obtaining written informed consent among the age group of ≥18 years.
Data collection
Socio-demographic and anthropological data [age, marital status, literacy status, occupation, life style (sedentary/ active), familial history (parents/siblings), reasons for stress, duration and severity of disease, along with type of treatment (oral anti-diabetic/ insulin), etc. were collected from patients with diabetes using standardized questionnaires.
Sample collection
Samples were collected from patients with diabetes having ulcers, surgical sites with infection and other wounds by needle aspirate method. In case of closed wounds, the skin or mucosal surface were disinfected with 2% chlorhexidine or 70% alcohol followed by iodine solution (1-2% tincture iodine or 10% solution of povidone-iodine). Prior to specimen collection, removal of iodine with alcohol was done. In case of open wounds, debridement, was thoroughly rinsed with sterile saline prior to collection8. Tissue samples were obtained from depth of ulcers and transferred aseptically into labelled sampling vials with sterile saline and processed in the Microbiology laboratory Regional Medical Research Centre, Bhubaneswar.
Microbial analysis
Samples were streaked on nutrient agar, mannitol salt agar, macConkey Agar (MCA) and incubated aerobically for 18-24 h at 37°C. Plates with no growth or comparatively low growth will be re-incubated for another 18-48 h for isolation of bacteria that require extended incubation. Blood Agar plates were incubated in aerobic conditions. Cultural characteristics, morphological appearances of colonies on selective media, gram staining and standard biochemical tests were performed to characterize the bacteria.
DNA extraction
DNA was prepared from bacteria as described previously9. In brief, a single colony forming unit(CFU) was suspended in 20 µl of lysis buffer containing 0.25% (vol/vol) sodium dodecyl sulfate and 0.05 N NaOH. After heating for 15 min at 95°C, 180µl of high-performance liquid chromatography-grade H2O (GCC Biotech) was added and the lysis suspension was stored at 20°C for further analysis.
PCR analysis for S.aureus
PCR reaction contained 2μl of DNA which was added to 23 μl of master mix containing 0.1μl of Taq polymerase , 0.5 μl of dNTPs , 0.625 μl of primers were used for amplification of the nucA gene (nucA1 5’GCGATTGATGG TGATACGGTT3’ and nucA2 5’AGCCAAGCCTTGACGAACTAAAGC3’), 1.5μl of MgCl2 , 2.5μl of 10X PCR buffer and 17.15μl of water (GCC Biotech). PCR was performed using the program including initial denaturation at 95°C for 10 mins, followed by 30 cycles of annealing with an temperature of 55°C was used for nucA gene amplification. Final extension was carried out for 5 mins at 72°C. The PCR products were visualized using agarose gel (2%) electrophoresis. Gel images were captured using the Gel documentation system.
A total of 150 patients with diabetic ulcers were enrolled. Most of them developed diabetic foot ulcers; whereas few patients developed gangrene, some also developed ulcers after an accident or burns. Out of the total patients studied 106 (73%) people developed diabetic foot ulcers; whereas 24 (16%) patients developed gangrene due to loss of blood supplies to the extremities, 9 (6%) people suffered from ulcers accidentally and only 7 (4.6%) people developed ulcers after burns. Further 85 (71%) males and 21 (67%) females developed DFUs whereas; 7 (22%) females and 17 (14%) males developed gangrene. About 5% each of males suffered from DFUs either due to accident and/or burns (Table-1and Fig. 1). As per the age group wise distribution with signs and symptoms of patients, 119 (79.4%) were males and 31 (20.6%) were females. 112 (74.6%) patients in the age group ≥45-55 years and 34 (22%) in the age group ≥55-65 years developed DFUs. About 105 (70%) of patients had infection from single bacterial species; whereas 45 (30%) patients had polymicrobial infection. In this study, different age groups, duration of diabetes, types of infection, duration of infection and medication were the significant risk factors in DFUs (Table 2). The microbiological profile of samples from patients with diabetic foot ulcers showed a total number of 195 isolates were detected from 150 ulcer specimens. S.aureus was isolated from 114 (76%) of the samples followed by 42 (28%) Pseudomonas aeruginosa. E.coli was found 34 (22%) of the samples. The infection status of samples from patients with diabetic foot ulcers.78 (52%) of the samples were infected with S.aureus, 19 (12%) with P.aeruginosa, 6 (4%) with E.coli whereas; 2 (1%) had Klebsiella spp. only (Table-3). It was found that the major etiological agent was S.aureus followed by P.aeruginosa and E.coli. S.aureus was one of the most important microorganism that manifests a range of clinical problems resulting from high-resistance to anti-microbial agents. PCR was performed for the confirmation of S.aureus. All the samples were positive for nucA gene which confirmed the presence of S.aureus in diabetic ulcer patients. (Fig. 2)
Table (1):
Percentage of clinical cases in patients with diabetic ulcers.
| Clinical cases [n=150, (%)] | Male [n = 119, (%)] | Female [n = 31 (%)] | |
|---|---|---|---|
| DFU | 106 (73.33) | 85 (71.42) | 21 (67.74) |
| Gangrene | 24 (16) | 17 (14.28) | 7(22.58) |
| Accidental | 9 (6) | 7 (5.88) | 2(6.45) |
| Burns | 7(4.66) | 6 (5.04) | 1(3.22) |
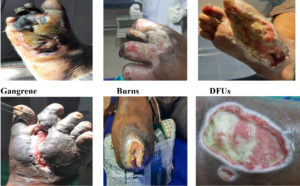
Fig. 1. Different types of ulcers in patients with diabetes (Gangrene, DFUs, Burn cases, Accidental cases and Amputation cases).
Table (2):
Prevalence of age group and Sepsis patient related to diabetic foot ulcer.
Bacterial isolates |
No. of Diabetic Ulcer Patients [n =150 (%)] |
|---|---|
S.aureus |
78 (52) |
S.aureus + P.aeruginosa |
15 (10) |
S.aureus + E.coli |
21 (14) |
P.aeruginosa |
19 (12.66) |
P.aeruginosa + E.coli |
6 (4) |
P.aeruginosa + Klebsiellasp |
2 (1.33) |
E.coli |
6 (4) |
E.coli + Klebsiellasp |
1 (0.66) |
Klebsiellasp |
2 (1.33) |
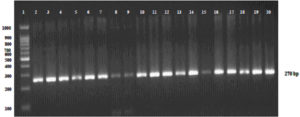
Fig. 2. Agarose gel electrophoresis showing PCR amplification of nucA gene of S. aureus isolates. [Lane 1:100bp ladder, lane2-19 S.aureus DNA, lane 20: ATCC strain of S. aureus (25923)].
Table (3):
Bacterial etiology related to diabetic ulcer patients.
| Parameters | No. of patients with Diabetic Ulcers n =150, (%) |
|
|---|---|---|
| Age Group (in years) |
>30-45 | 2(1.33%) |
| >45-60 | 138(92%) | |
| >60 | 10 (6.66%) | |
| Duration of Diabetes (in years) | <10 | 57(38%) |
| >10 | 93 (62%) | |
| Types of Infection | Mono-microbial | 105 (70) |
| Poly-microbial | 45 (30) | |
| Duration of Infection (in months) |
1-9 | 71(55.33%) |
| 10-19 | 72(40%) | |
| 20-29 | 4(2.66%) | |
| ≥30 | 3 (2%) | |
| Medication | Insulin | 74 (49.33%) |
| Oral | 76 (50.66%) | |
It was observed that S.aureus was the major etiological agent among one hundred fifty diabetic ulcers samples, S.aureus were concomitantly isolated from diabetic foot ulcer patients. Our finding is consistent with previous studies reported by Mottola et al10. S.aureus is one of the most important micro-organisms that cause clinical problems resulting high-resistance to different antimicrobial agents. Though it is rarely found in the normal flora of humans, it is frequently isolated from patients with burns, cystic fibrosis and neutropenia11. Diabetic foot ulcer is one of the most common devastating complications of diabetes mellitus and the leading cause of agonizing amputation throughout the world12. These infections may be colonized by pathogenic and anti-microbial resistant bacteria, harbouring several virulence factors that could impair its successful treatment13. Certain socio-cultural practices in India like barefoot walking, poor hygiene habits, inadequate facilities for diabetic care, low level of education and poor socio-economic conditions often lead to foot lesions and hospitalizations14. The present study showed 80% male ascendancy followed by 20% female. However, the present study is also comparable with a multicentric study from Saudi Arabia15,16. Direct antibiotic treatment cannot be recommended as per clinical signs or symptoms of infection as there is a very fine line between colonization of micro-organisms and problematic bio-burden and the direct antibiotic treatment efficacy remains unclear on the basis of only these two major determinants. Whereas, treatment options based upon targeting microbial population to promote healing and determining infection related complications might be a novel one17. S.aureus may cause severe tissue damage in diabetic patients and should never be ignored as insignificant in diabetic foot ulcers. Moreover, it should never be considered a contaminant or normal flora, and it should clearly be considered a pathogen, because it may result in sepsis and amputation18. The numerous virulence factors and toxins secreted by S.aureus during infection that evade host immune defences are few of the challenges in managing S.aureus infections is an inherent resistance mechanism, referred to as intrinsic resistance. Its multiplicity of resistance mechanisms may render this microbe less amenable to control by antibiotic cycling. S.aureus is noted for its metabolic versatility and its exceptional ability to colonize a wide variety of environments and also for its intrinsic resistance to a wide variety of antimicrobial agent19.
There are many studies that suggest that apart from clinical factors, the socio-demographic variables play an important role in diabetic foot ulceration. It was reported by DeBerardis et al (2005) that the prevalence of diabetic foot complications was higher in older patients, those with limited formal education and a low socio-demographic status20. Our results couldn’t substantiate the claim mentioned in the above study. One ninety five micro-organisms were isolated from one fifty clinical samples of diabetic foot ulcers, which showed multiple bacterial infections that represent an average of 1.3 organisms per ulcer which is slightly lower than other studies21,22, and showed an average of 1.52 organisms per ulcer. Diabetic foot infections are usually polymicrobial in nature and this has been well documented in the literature. S.aureus was the most common isolate observed in diabetic foot ulcers that was in accordance with findings of previous studies23,24. The emergence of S.aureus in the DFUs caused severe wound infection and worsened of the wound25. However, the bacterial diversity and prevalence of specific bacteria vary greatly from studies to studies. Identification of diabetes with DFUs and its associated factors are the key to reduce further complications and to have baseline information to initiate appropriate interventions.
The present study reports that S.aureus was the major etiological agent with socio-demographic and clinical profile of patients with diabetic ulcer. There was a predominance of mono-microbial growth with gram-positive organisms. Healthcare should be made more accessible to facilitate early diagnosis of DFU and its complications to minimize the rate of amputations. This type of study should be continued for a longer period both in coastal and tribal areas of Odisha.
ACKNOWLEDGMENTS
Thanks to ICMR, NEW DELHI, [NO.2019 – 4588/NCD-III], for their support.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
The project was funded by ICMR-NEW DELHI [NO.2019 – 4588/NCD-III]
ETHICS STATEMENT
The study was approved by the institutional human ethics committee of the ICMR- Regional medical research centre, Bhubaneswar and was carried out in accordance with the approved guidelines.
AVAILABILITY OF DATA
All datasets generated or analyzed during this study are included in the manuscript.
- International Diabetes Federation. IDF DIABETES ATLAS. Ninth edition 2019.
- Enoch S, Harding K, Wound bed preparation: the science behind the removal of barriers to healing. Wounds. 2003;15(7):213-229.
- Gadepalli R, Dhawan B, Sreenivas V, Kapil A, Ammini AC, Chaudhry R. A Clinico microbiological study of diabetic foot ulcers in an Indian tertiary care hospital. Diabetes Care. 2006;29(8):1727-1732.
Crossref - Shankhdhar K, Shankhdhar LK, Shankhdhar U, Shankhdhar S. Diabetic foot problems in India: an overview and potential simple approaches in a developing country. Curr Diab Rep. 2008;8(6):452-457.
Crossref - Chun DI, Kim S, Kim J, et al. Epidemiology and burden of diabetic foot ulcer and peripheral arterial disease in Korea. J Clin Med. 2019;8(5):748.
Crossref - Gardner SE, Frantz RA, Doebbeling BN. The validity of the clinical signs and symptoms used to identify localized chronic wound infection. Wound Repair and Regen. 2001;9(3):178-186.
Crossref - Singh N, Armstrong DG, Lipsky BA. Preventing foot ulcer in patients with diabetes. JAMA. 2005;293(2):217-228.
Crossref - Standard operating procedures Bacteriology. 1st Edition. Antimicrobial Resistance Surveillance and Research Network, ICMR. 2015;40-43.
- Sambrook J, Fritsch EF, Maniatis T. Molecular cloning: a laboratory manual. Cold spring harbor laboratory. 1989.
- Mottola C, Matias CS, Mendes JJ, et al. Susceptibility patterns of Staphylococcus aureus bio-films in diabetic foot infections. BMC Microbiology. 2016;16(1):119.
Crossref - Zubair M, Malik A, Ahmad J. Incidence, risk factors for amputation among patients with diabetic foot ulcer in a North Indian tertiary care hospital. The Foot. 2012;22(1):24-30.
Crossref - Roberts AD, Simon GL. Diabetic foot infections: the role of microbiology and antibiotic treatment. Seminars in Vasc Surg. 2012;25(2,):75-81.
Crossref - Dunyach-Remy C, Ngba Essebe C, Sotto A, Lavigne J-P. Staphylococcus aureus toxins and diabetic foot ulcers: role in pathogenesis and interest in diagnosis. Toxins. 2016;8(7):209.
Crossref - Shahi SK, Kumar A, Kumar S, Singh SK, Gupta SK, Singh TB. Prevalence of diabetic foot ulcer and associated risk factors in diabetic patients from North India. J Diabetic Foot Complication. 2012;4(3):83-91.
- Viswanathan V, Thomas N, Tandon N, Asirvatham A, Rajasekar S. Profile of diabetic foot complications and its associated complications-a multi-centric study from India. JAPI. 2005;53:933-936.
- Al-Rubeaan K, Al Derwish M, Ouizi S, et al. Diabetic foot complications and their risk factors from a large retrospective cohort study. PloS One. 2015;10(5):e124446
Crossref - Lipsky BA, Aragon-Sanchez J, Diggle M, et al. IWGDF guidance on the diagnosis and management of foot infections in persons with diabetes. Diabetes Metab Res Rev. 2016;32(Suppl. 1):45-74.
Crossref - Jyothylekshmy V, Menon AS, Abraham S. Epidemiology of diabetic foot complications in a podiatry clinic of a tertiary hospital in South India. Indian Journal of Health Sciences and Biomedical Research (KLEU). 2015;8(1):48-51.
Crossref - Sangeeta J, Ray P, Manchanda V, et al. Methicillin resistant Staphylococcus aureus (MRSA) in India: prevalence & susceptibility pattern. Indian J Med Res. 2013;137(2):363-369.
- De Berardis G, Pellegrini F, Franciosi M, et al. Are Type 2 diabetic patients offered adequate foot care? The role of physician and patient characteristics. J Diabetes Complications. 2005;19(6):319-327.
Crossref - Raja NS. Microbiology of diabetic foot infections in a teaching hospital in Malaysia: a retrospective study of 194 cases. J Microbiol Immunol Infect. 2007;40(1):39-44.
- Bansal E, Garg A, Bhatia S, Attri AK, Chander J. Spectrum of microbial flora in diabetic foot ulcers. Indian J Pathol Microbiol. 2008;51(2):204-208.
Crossref - Abdulrazak A, Bitar ZI, Al-Shamali AA, Mobasher LA. Bacteriological study of diabetic foot infections. J Diabetes Complications. 2005;19(3):138-141.
Crossref - Sharma VK, Khadka PB, Joshi A, Sharma R. Common pathogens isolated in diabetic foot infection in Bir Hospital. Kathmandu Univ Med J (KUMJ). 2006;4(3):295-301.
- Shettigar K, Murali TS. Virulence factors and clonal diversity of Staphylococcus aureus in colonization and wound infection with emphasis on diabetic foot infection. European Eur J Clin Microbiol Infect Dis. 2020;39:2235-2246.
Crossref
© The Author(s) 2021. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.