ISSN: 0973-7510
E-ISSN: 2581-690X
Candida species are one of the most common causes of opportunistic infections in immunocompromised patients. An upsurge in incidence of less common species of Candida have been documented since last few years as the major cause of candidemia all over the world and they are also less susceptible to azoles, particularly fluconazole, than C. albicans. The purpose of study was speciation and antifungal susceptibility testing of Candida isolates, obtained from immunocompromised patients. The study included 150 consecutive immunocompromised patients and was initiated after the ethical approval from Sumandeep Vidyapeeth Institutional Ethical Committee (SVIEC). Informed consent was obtained from all and a Detailed Questionnaire regarding the Patient’s history and clinical findings were noted. All the specimens were subjected to a battery of microbiological examination for isolation, identification, and antifungal susceptibility testing. In our study, the incidence of candidiasis in immunocompromised patients was43.1%.C.tropicalis(n=23,37.09%) was the most common species isolated followed by C. albicans (n=20, 32.25%), C. glabrata (n=15,24.19%) and C. parapsilosis (n=4, 6.45%). The isolates showed 100% sensitivity to Amphotericin B and Nystatin, whereas 37.09% sensitivity to Ketoconazole, 20.9% sensitivity to clotrimazole, 19.35% sensitivity to itraconazole and 14.5% sensitivity to Fluconazole. The study undoubtedly indicates a substantial move in the species from C. albicans to Non-albicans Candida and with added resistance to common antifungal drugs.
Candida albicans, Non-albicans Candida, Immunocompromised Patients, Resistance
Candida species are the constituent of normal flora of human body commonly found in skin, mucous membranes, throughout the gastrointestinal tract and female genital tract, mainly in the vagina of pregnant women.1 Candidiasis is a term used for fungal infection affecting, mucosa, skin, internal organs of the body and nails. It mainly occurs as a secondary infection in an individuals with some underlying immunocompromised condition, resulting in the high mortality.1 Candida species are the 4th most common primary blood stream organism and constitute 8th most common pathogen to cause health care associated infections.2
Among candida species, Candida albicans is the most common cause of candidiasis accounting for about 60-80% of infections.3 During the last century, two significant predisposing factors have been perceived; first was the advent and indiscriminate use of antibiotics and second was the emergence of AIDS pandemic, and along with that, the changing trend of Candida species from non-pathogenic to pathogenic organism was facilitated by various virulence factors, such as adherence to host tissues, medical devices, formation of biofilms, and secretion of extracellular hydrolytic enzymes.4 This explains the fact that with the escalation of immunocompromised patients, candidiasis has become an emerging, alarming opportunistic disease. It has been noted in the recent years that there is a remarkable shift from Candida albicans to Non-albicans Candida (NAC) as a major pathogen causing infection in human, also there is change in the antifungal drug susceptibility pattern and it is mostly due to over and random use of antifungal agents.5,6 Many new antifungal drugs are available nowadays. The various manifestations of candidiasis often create dilemma for the physician, both in diagnosis and treatment, and are frequently the major cause of death in the patient. Therefore, it is very important to find out the species of Candida, and their antifungal drug susceptibility pattern in specimens for better management of the patients and also to reduce mortality. Hence, this study was conducted to identify the different species of Candida isolates and to explore its antifungal susceptibility pattern.
This Prospective cross-sectional study was undertaken for a duration of 19 months from January 2015 to July 2016 at the Tertiary Care Centre, Gujarat. Approval of Sumandeep Vidyapeeth Institutional Ethical Committee was taken prior to the initiation of work (SVIEC NO. 108). A total of 150 consecutive patients with varied immunocompromised conditions like HIV seropositivity, Diabetes mellitus, Transplant recipient, Tuberculosis, Malignancy, and haemodialysis with clinical manifestations were included in the study. Those patients, not willing to participate were excluded from the study. Different specimen like sputum, urine (Midstream and Catheterized), blood, pus, and oral swab were aseptically collected and were processed for the identification of yeast.
Methodology
Direct Microscopy
The clinical sample was subjected to Gram’s staining, for the identification of yeast cells and pus cells. Presence of hyphae and pseudohyphae were examined under the light microscope (100X magnification). The Staining was done according to the standard protocols.7
Culture
All the samples were inoculated on Sabouraud Dextrose Agar (SDA), with chloramphenicol and chlorhexidine, (Procured from HiMedia) and Blood Agar (BA) The plates were incubated at 37°C for 24- 48 hours, after which growth was identified by the following characteristics; rapidity of growth, color and morphology of the colony on the obverse and pigmentation on the reverse.
Blood culture
For blood culture, 8-10 ml(adult) or 3-5 ml(children) venous blood was collected aseptically and inoculated in BD BACTEC aerobic plus/F (Becton-Dickinson, New Jersey, USA) bottles in BD BACTEC FX40, Series FF 7004 automated blood culture system was used. The bottles were inserted either immediately or within 30 minutes inside the machine. Whenever the machine gave an alert signal, or sound, the respective bottle were removed and were sub cultured on Blood agar and Saboraud dextrose agar with chloramphenicol and chlorhexidine, (Procured from HiMedia)
Identification of the isolates
The identification of the growth as Candida was done by colony morphology (cream colored, smooth and pasty colonies) and Gram’s stain. The Speciation of Candida was done by performing various conventional tests such as Germ tube test,8 chlamydospore formation, color change on HiCromeAgar [Hichrome Candida Differential Agar (HiMedia Pvt. Ltd., Mumbai)].9 Sugar fermentation and Assimilation tests.10 HiCrome Agar is very easy and low cost method for identification of Candida species. One important advantage of HiCrome Agar is ability to detect mixed cultures.11
Antifungal Susceptibility Testing
Antifungal Susceptibility Testing was done by Disc diffusion method, which is very easy to perform and results can be interpreted by 24 hours. Therefore it can be used for diagnostic purposes on routine basis.12 MHA (Muller Hinton Agar) supplemented with glucose and methylene blue, was prepared.13,14 Following antifungal discs were used amphotericin B (100 units), fluconazole (25mcg), clotrimazole (10mcg), itraconazole (10mcg), ketoconazole (10 mcg), Nystatin (100 units) (procured from HiMedia, Mumbai). Antifungal susceptibility test was performed according to CLSI guidelines documented in M44-A.15
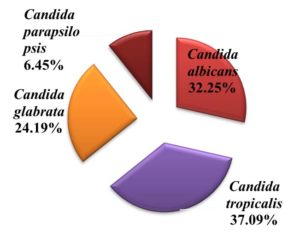
150 clinical samples were collected from 150 immunocompromised patients (3-85yrs) admitted at our institute. Out of which 62 were positive for Candida species which shows the incidence of 43.1 % in immunocompromised patients. C.tropicalis(n=23,37.09%) was the commonest isolate followed by C. albicans (n=20, 32.25%), C. glabrata (n=15,24.19%) and C. parapsilosis (n=4, 6.45%) (Figure).
Association of Candida infection with age and gender (Table 1)
Majority of the patients with Candida infection belonged to the younger category followed by the middle age group. Gender-wise, we found slight male predominance (54%) than female (46%).
Table (1):
Association of Candida infection with Age and Gender.
| Variables | With Candida infection | Without Candida infection | |
|---|---|---|---|
| Gender | Male (n=81) | 34(41.9%) | 47(58.02 %) |
| Female(n=69) | 28(40.57 %) | 41(59.42 %) | |
|
Age-Group |
0-10(n=15) | 5(33.3 %) | 10(66.6 %) |
| 11-20(n=10) | 2(20 %) | 8(80%) | |
| 21-30(n=20) | 6(30%) | 14(70%) | |
| 31-40(n=30) | 10(33.3%) | 20(66.6%) | |
| 41-50(n=40) | 15(37.5%) | 25(62.5%) | |
| 51-60(n=12) | 10(83.3%) | 2(16.6%) | |
| 61-70(n=17) | 9(53 %) | 8(47 %) | |
| 71-80(n=4 ) | 3(75 %) | 1(25 %) | |
| 81-90(n=2) | 2(100 %) | 0(0 %) |
Ward wise emergence of Candida infection (Table 2)
Most of the Candida species were isolated from ICU (42/80, 52.5 %) followed by Medicine ward (13/32, 40.6 %). TB ward (2/8, 25%), Surgery ward (3/12, 25%) and Paediatric ward (2/15, 13.3%).
Table (2):
Ward wise distribution of Candida infection.
| Ward | Total Sample | With Candida n=62 |
Without Candida N=88 |
|||
|---|---|---|---|---|---|---|
| ICU | 80 | 42 | 38 | |||
| Medicine | 32 | 13 | 19 | |||
| Paediatric ward | 15 | 02 | 13 | |||
| Surgery ward(n=6) | 12 | 03 | 09 | |||
| Neuro ward | 01 | 00 | 01 | |||
| TB ward | 08 | 02 | 06 | |||
| Ortho ward | 02 | 00 | 02 | |||
| Total | 150 | 62 | 88 | |||
Sample wise distribution of Candida species (Table 3)
Most of the Candida species were isolated from urine sample (40/87, 45.9%). C tropicalis (15/87) was the most common isolates followed by C. albicans (14/87), C. glabrata (7/87) and C. parapsilosis (4/87). From sputum sample, C. glabrata (6/45) and C. tropicalis (6/45) were predominant followed by C.albicans (4/45). C. glabrata (2/12) and C. tropicalis (2/12) were isolated from blood sample. From oral swab C. albicans (2/2) was isolated.
Table (3):
Association of Candida species with various clinical specimens.
| Culture positivity | Total positive | ||||
|---|---|---|---|---|---|
| C.albicans | C.glabrata | C.tropicalis | C.parapsilosis | ||
| Urine (n=87) | 14 | 7 | 15 | 4 | 40(45.9%) |
| Sputum (n=45) | 4 | 6 | 6 | – | 16(35.5%) |
| Blood (n=12) | – | 2 | 2 | – | 4(33.3%) |
| Pus (n=4) | – | – | – | – | 0 |
| Oral Swab (n=2) | 2 | – | – | – | 2(100%) |
| Total | 20(32.2%) | 15(24.1%) | 23(37.05%) | 4(6.45%) | 28( 48%) |
Antifungal Susceptibility Pattern of Candida Species (Table 4)
All the isolated species of Candida were 100% susceptible to amphotericin B, and nystatin. All the isolated species of C. albicans (100%), C. parapsilosis (75 %) and C. glabrata (20%) were susceptible to fluconazole whereas C. tropicalis isolates showed 91 % resistance to fluconazole. All the Candida isolates showed varying degree of resistance to ketoconazole, such as C. albicans (70), C. tropicalis (86.9), C. glabrata (33.3) and C. parapsilosis. The sensitivity of Clotrimazole for C. albicans was 10%, for C. tropicalis was 9.52%, for C. glabrata was 66.6% and for C. parapsilosis was 50%. C. tropicalis and C. albicans showed 100% resistance to Itraconazole, while C. glabrata showed 53.3% and C. parapsilosis showed 50% resistance to Itraconazole.
Table (4):
Antifungal Susceptibility Pattern of Candida Species.
| Candida species | Antifungal | Sensitivity | (%) | Resistance | (%) |
|---|---|---|---|---|---|
| C. tropicalis(n=23) | Fluconazole | 2 | 8.69% | 21 | 91.30% |
| Ketoconazole | 3 | 13.04% | 20 | 86.95% | |
| Itraconazole | – | 23 | 100% | ||
| Clotrimazole | 2 | 9.52% | 21 | 91.30% | |
| Amphotericin-B | 23 | 100% | – | ||
| Nystatin | 23 | 100% | – | ||
| C.albicans(n=20) | Fluconazole | 20 | 100% | – | |
| Ketoconazole | 6 | 30% | 14 | 70% | |
| Itraconazole | – | 20 | 100% | ||
| Clotrimazole | 2 | 10% | 18 | 90% | |
| Amphotericin-B | 20 | 100% | – | ||
| Nystatin | 20 | 100% | – | ||
| C.glabrata(n=15) | Fluconazole | 3 | 20% | 12 | 80% |
| Ketoconazole | 10 | 66.66% | 5 | 33.33% | |
| Itraconazole | 7 | 46.66% | 8 | 53.33% | |
| Clotrimazole | 10 | 66.66% | 5 | 33.33% | |
| Amphotericin-B | 15 | 100% | – | ||
| Nystatin | 15 | 100% | – | ||
| C.parapsilosis(n=4) | Fluconazole | 3 | 75% | 1 | 25% |
| Ketoconazole | 3 | 75% | 1 | 25% | |
| Itraconazole | 2 | 50% | 2 | 50% | |
| Clotrimazole | 2 | 50% | 2 | 50% | |
| Amphotericin-B | 4 | 100% | – | ||
| Nystatin | 4 | 100% | – |
In recent years, Candida spp. have emerged as a main pathogen of a variety of human infections which clearly proves that there is an extreme change in model of infectious disease. Those microorganisms previously non-pathogenic are now the commonest cause of morbidity and mortality. Among candida species, C. auris have emerged as a pandrug resistant organism which lead to prolong hospital stay and healthcare cost.16 In the present study we found 62 isolates of Candida species from 150 clinical specimens. About 80% of the Candida species were isolated from urine and sputum sample, which indicates the higher incidence and distribution of Candida species causing urinary tract and respiratory tract infections. Among Non-albicans Candida, Candida tropicalis was the most common species isolated followed by C. albicans, C. glabrata and C. parapsilosis respectively. Our results were in accordance with the studies done by D. Kumar et al.17 Lata R Patel et al.18 and Ayushi Jain et al.19 which revealed C. tropicalis as the most prevalent species. among the Candida isolates. There were many studies which showed that the incidence of NAC (Non-albicans Candida) ranges from 54-75%. Golia et al.20 A study done by Vijaya et al.21 showed C. albicans (45.9%) have a lower incidence than Non- albicans Candida (54.1%). A study done by Manchanda et al.22 also presented Non- albicans Candida (72.4%) with a higher incidence than C.albicans (27.5%). Recently a study done by Urvashi Chongtham et al.23 showed that Non-albicans Candida (NAC) species have replaced Candida albicans with decreased susceptibility to commonly used antifungal agents. In the latest decade, increasing incidence of candidemia caused by Non-albicans Candida (NAC) species have been reported, which has led to the rise of NAC investigations. Their ability to cause recurrent infections, higher resistance to antifungal drugs, and potential to cause outbreaks infections has led to further research. In the present study, the prevalence of Non-albicans Candida was significantly high, this might be due to random use of antifungal drugs, which lead to elimination of more sensitive C. albicans and selection of azole resistant Non-albicans Candida. The antifungal susceptibility pattern was different for different Candida spp Such as, all Candida isolates tested were susceptible to Amphotericin B and Nystatin. All the Candida isolates were tested for Azole group showed reduce susceptibility. Whereas all C. albicans were found sensitive to fluconazole. D. Kumar et al.17 reported the susceptibility profile of Candida isolates as 100% sensitive to Amphotericin B and Voriconazole and 89.3% were sensitive to fluconazole. Sonu Panwar et al.24 reported the susceptibility profile of Candida isolates as 94.1% were sensitive to AMB, 58.3% to Fluconazole, 72.5 % to Ketoconazole, and 85 % to Nystatin which is very much similar to our study. From above listed study it is clear that these NAC species are resistance to most of the commonly prescribed antifungal drugs. Therefore, The emergence of multidrug and pandrug-resistant C. albicans and NAC noted in several parts of the world necessitates continuous antifungal susceptibility testing and monitoring.25
In the present study, the incidence of candidiasis was 43.1 % with C. tropicalis as the most common species isolated. Identification of Candida species helps to start early suitable antifungal dugs and also it helps to reduces the morbidity and mortality. The antifungal drugs fluconazole and ketoconazole were most commonly used drugs but least effective against NAC species, higher resistance against them were shown by C. tropicalis and C. glabrata. This Changing trends in the antifungal susceptibility pattern recommends for antifungal susceptibility testing of Candida isolates on routine basis in clinical microbiology laboratories.
ACKNOWLEDGMENTS
The authors would like to thank Sumandeep University for giving permission to conduct this research study successfully.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
None.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
This study was approved by the Sumandeep Vidyapeeth Institutional Ethical Committee with reference number SVIEC NO-108.
INFORMED CONSENT
Written informed consent was obtained from the participants before enrolling in the study.
- Chander JA. Textbook of Medical Mycology, 4th edition, Jaypee Brothers Medical Publishers 2018;401-433.
- Ahmed S, Shahid M, Fatima N, Khan F, Tayyaba U. Candidemia – Changing trends from Candida albicans to Non-albicans Candida from a tertiary care center in western UP, India. CHRISMED J Health Res. 2020;7(3):167-172.
Crossref - Manjunath V, Vidya GS, Sharma A, Prakash MR, Murugesh. Speciation of Candida by Hicrome agar and sugar assimilation test in both HIV infected and non infected patients. Int J Biol Med Res. 2012;3(2):1778-1782.
- Sardi JCO, Scorzoni L, Bernardi T, Fusco-Almeida AM, Mendes Giannini MJS. Candida species: current epidemiology, pathogenicity, biofilm formation, natural antifungal products and new therapeutic options. J Med Microbiol. 2013;62(1):10-24.
Crossref - Gullo A. Invasive fungal infections: the challenge continues. Drugs. 2009;69(Suppl 1):65-73.
Crossref - Yang YL, Cheng HH, Ho YA, Hsiao CF, Lo HJ. Fluconazole resistance rate of Candida species from different regions and hospital types in Taiwan. J Microbiol Immunol Infect. 2003;36(3):187-191.
- Tripathi N, Sapra A. Gram Staining. Treasure Island (FL): Stat Pearls Publishing. 2022. https://www.ncbi.nlm.nih.gov/books/NBK562156/?report=reader.
- Forbes BA, Sahm DF, Weissfield AS. Laboratory methods in basic Mycology. Bailey and Scott’s diagnostic Microbiology 12th edition, USA: Mosby Elsevier. 2007:629-716.
- Mehta R, Wyawahare AS. Evaluation of Hicrome Candida differential agar for species identification of Candida isolates from various clinical samples. Int J Cont Med Res. 2016;3(4):1219-1222.
- Land GA, Vinton EC, Adcock GB, Hopkins JM. Improved Auxanographic Method for Yeast Assimilations: a Comparison with Other Approaches. J Clin Microbiol. 1975;2(3):206-217.
Crossref - Kanna BV, Kumar GA, Swapna M, Joshy M. Easow Isolation and identification of candida species from various clinical samples in a tertiary care hospital. Int J Res Med Sci. 2017;5(8):3520-3522.
Crossref - Jayachandran AL, Katragadda R, Ravinder T, et al. Antifungal Susceptibility Pattern among Candida species: An Evaluation of Disc Diffusion and Broth Micro-dilution Method. J Microbiol Infect Dis. 2018;8(3):97-101.
Crossref - Clinical Laboratory Standards Institute. Method for antifungal disk diffusion susceptibility testing of yeasts; Approved Guideline. CLSI document 2009; 29(17):M 44-A2 .https://clsi.org/media/1634/m44a2 sample.pdf
- Lee SC, Fung CP, Lee N, et al. Fluconazoledisk diffusion test with methylene blue- and glucose-enrichedMueller-Hinton agar for determining susceptibility of Candida species. J Clin Microbiol. 2001;39(4):1615-1617.
Crossref - Pfaller MA, Boyken L, Messer SA, Hollis RJ, Diekema DJ. Stability of Mueller-Hinton Agar Supplemented with Glucose and Methylene Blue for Disk Diffusion Testing of Fluconazole and Voriconazole. J Clin Microbiol. 2004;42(3):1288-1289.
Crossref - Musyoki VM, Mutai W, Ngugi N, Otieno F, Masika MM. Speciation and antifungal susceptibility of Candida isolates from diabetic foot ulcer patients in a tertiary hospital in Kenya. PAMJ. 2022;41(34).
Crossref - Kumar D, Banerjee T, Chakravarty J, Singh SK, Dwivedi A, Tilak A. Identification, antifungal resistance profile, in vitrobiofilm formation andultrastructural characteristics of Candida species isolated from diabetic foot patientsin Northern India. Indian J Med Microbiol. 2016;34(3):308-314.
Crossref - Patel LR, Pethani JD, Bhatia P, Rathod SD, Shah PD. Prevalence of Candida Infection and its Antifungal Susceptibility Pattern in Tertiary Care Hospital, Ahmedabad. Natl J Med Res. 2012;2(4):439-441.
Crossref - Jain A, Farooq U, Singh S, Sharma V, Sharma SR, Ahamad I.Frequency of candida infection among intensive care unit patients and their susceptibility profile. IP International Journal of Medical Microbiology and Tropical Diseases. 2021;7(3):133-136.
Crossref - Golia S, Reddy KM, Karjigi KS, Hittinahalli V. Speciation of Candida using chromogenic and cornmeal agar with determination of fluconazole sensitivity. Al Ameen J Med Sci. 2013;6(2):163-166.
- Vijaya D, Harsha TR, Nagaratnamma T. Candida speciation using chrom agar. J Clin Diag Res. 2011;5(4):75-57.
- Manchanda V, Agarwal N, Verma. Yeast identification in routine clinical Microbiology laboratory and its clinical relevance. Indian J Med Microbiol. 2011;29(2):172.
Crossref - Chongtham U, Athokpam DC, Singh RM. Isolation, Identification and Antifungal Susceptibility Testing of Candida Species: A Cross-sectional Study from Manipur, India. J Clin Diagn Res. 2022;16(4):DC09-DC14.
Crossref - Panwar S, Faujdar SS. Prevalence, Distribution, Risk factors and Antifungal Susceptibility Profiles of Candida species in a Tertiary Care Hospital. Int J Curr Microbiol App Sci. 2016;5(4):329-337.
Crossref - Gade N, Noval A. Antifungal susceptibility and speciation of clinical isolates of Candida at Tertiary Care Hospital, Central India. Int J Infect Dis. 2020;101(S1):394.
Crossref
© The Author(s) 2023. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.