ISSN: 0973-7510
E-ISSN: 2581-690X
Hepatitis C virus (HCV) infection in hemodialysis patients is mainly associated with insufficient precautionary measures. Compared with the general population, the prevalence of HCV infection is high among hemodialysis patients. HCV infection in these patients is typically associated with increased morbidity and mortality; however, the early detection of HCV may retard disease progression and reduce morbidity and mortality. In this study, we sought to estimate the seroprevalence of HCV infection among patients undergoing hemodialysis in a tertiary care hospital in Chennai, Tamil Nadu, South India. This was a prospective, observational, cross-sectional study that included 82 hemodialysis patients. Blood samples were collected from each patient, and the serum was separated for detection of the presence of HCV antibodies, HCV core antigen, and HCV RNA using an enzyme-linked immunosorbent assay, enzyme immunoassay, and polymerase chain reaction, respectively. Relevant socio-demographic, clinical, and laboratory information was also obtained. Among samples collected from the 82 patients, seven were found to be positive for HCV antibodies, whereas nine and eleven samples were found to be positive for HCV core antigen and HCV RNA, respectively. Long durations of renal failure and hemodialysis were identified as being positively associated with HCV infection in these hemodialysis patients, and a statistically significant reduction in hemoglobin levels and an increase in creatinine levels were detected among the HCV-positive patients. The overall seroprevalence of HCV infection in hemodialysis patients was found to be 13.4%. Given that high rate false-negative reactions were detected using ELISA and EIA, screening of HCV RNA during the early stages of HCV infection can contribute to preventing its nosocomial spread in these patients. Moreover, it is imperative that stringent universal precautionary measures are implemented in all health care settings and hemodialysis units to minimize the risk of HCV transmission.
Seroprevalence, Hepatitis C, Haemodialysis, ELISA
Hepatitis C virus (HCV) is a single-stranded, enveloped, positive-sense RNA virus that causes hepatitis and can lead to serious liver complications, including cirrhosis and hepatocellular carcinoma, particularly in patients undergoing hemodialysis (HD), in whom immunity is typically low.1
Whereas in the general population, the prevalence of HCV infection ranges from 0.5% to 1.5%, in patients undergoing HD, the prevalence can exceed 50% in both developed and developing countries.2-7 A range of risk factors have been identified for the high prevalence of HCV infection in HD patients, the most important of which are an increase in the number of blood transfusions, an increase in the duration of HD, and improper sterilization or internal contamination of dialysis equipment.8 Given the associated high rates of morbidity and mortality, HCV infection in HD patients is recognized as a significant health problem.9
The detection of HCV infection in HD patients poses considerable challenge, as the poor immune response of these patients means that they have a longer window of infection.10 The earliest infection-related marker to appear in the blood is HCV RNA, which occurs at approximately 1 to 3 weeks after onset, followed by the appearance of HCV core antigen at 2 to 6 weeks. However, it is only at 8 to 12 weeks after exposure to HCV that HCV antibodies are generally detected in the blood.11 With respect to the diagnosis of HCV infection; many laboratories preferentially perform HCV antibody detection. However, the main disadvantage of this procedure is that when used independently it fails to detect all HCV-infected patients who are in the early stages of infection, and thus may lead to high false-negative rates.12
The gold standard method for the early diagnosis of HCV infection is PCR-based HCV RNA detection; however, this approach tends to be limited by its higher costs and low availability.13,14 An alternative method for detecting HCV infection during its early stages is the detection of HCV core antigen in the blood.15,16
In India, although numerous studies have been conducted to determine the seroprevalence of HCV and to assess the efficacy of different serological tests among patients undergoing HD, either the data remain scarce or the tests conducted are based exclusively on the detection of HCV antibodies.
AIM
To estimate the seroprevalence of HCV infection among hemodialysis patients attending a tertiary care hospital in Chennai, Tamil Nadu, South India
Objective
To assess the efficacy of different serological tests in diagnosing HCV infection and also to identify the risk factors for HCV infection in patients undergoing hemodialysis.
The study was initiated after having initially gained approval from the Institutional Human Ethics Committee (IEC No: 40102015).
Study design
This was a prospective, observational, cross-sectional study.
Study Duration
6 months (July 2016 to December 2016).
Number of samples
Blood samples were collected from 82 patients undergoing HD in a tertiary care hospital in Chennai, South India.
Inclusion criteria
(a) Both genders were included, and the age range of participants was set to 18–60 years
(b) Those willing to give written, informed consent to participate in the study
Exclusion criteria
(a) Patients who have K/C/O HCV infection and were receiving treatment
(b) Pregnant women
(c) Patients co-infected with HIV
Details regarding the study and study procedures were clearly explained to the patients in their native language, and written informed consent were obtained. In no part of this manuscript is the personal identity of the patients revealed.
Relevant socio-demographic, clinical, and laboratory information was obtained from all patients, and having collected the necessary information, detailed physical examinations were performed.
Sample collection and processing
Samples of blood (5 mL) were collected from patients under aseptic conditions and transferred to collection tubes containing EDTA. Serum was subsequently separated and stored at -80°C until further use. All the samples were subjected to the following diagnostic tests:
- HCV RNA detection using polymerase chain reaction (PCR)
- HCV core antigen detection using an enzyme immunoassay (EIA)
- HCV antibody detection using a 3rd generation enzyme-linked immunosorbent assay (ELISA)
All tests were performed in accordance with the manufacturer’s instructions.
Statistical analysis
Statistical analysis was performed using SPSS 20.0 software. For all continuous variables, the results are presented as mean values ± standard deviation, and for the categorical variables, data are expressed as percentages. Continuous and categorical variables were compared using Student’s t-tests and chi-square tests, respectively. A probability value (P value) of less than 0.05 was considered statistically significant.
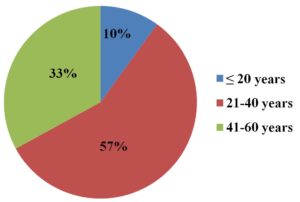
A total of 82 patients were included in this study, among whom 65 (79%) were males and 17 (21%) were females. A majority of the patients [47 (57%)] were in the 21–40-year age group and the mean age of patients was 36 ± 10.32 years. Details of a percentage analysis of participant age groups are presented in Figure 1.
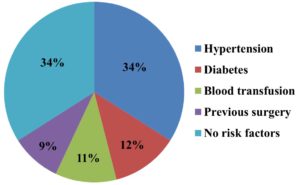
Most common risk factors for HCV infection in HD patients were found to be hypertension (34%), followed by diabetes (12%). The percentage analysis for the risk factors of the study participants is given below in Figure 2.
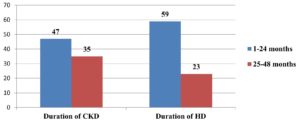
Analysis of the duration of renal failure revealed that in 57% of the patients, the disease had been present for more than 24 months. Similarly, analysis of the duration and frequency of HD revealed that a majority (72%) of the patients had been receiving HD for more than 24 months, and 90% were undergoing seven to nine HD sessions per month. Furthermore, approximately 67% of the patients had a history of changing dialysis center. Co-infection with HBV was found in 4% of the patients. Data obtained from a frequency analysis of the duration of CKD and HD are presented in Figure 3.
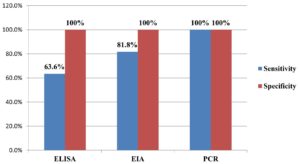
The sensitivity and specificity of the various diagnostic tests in detecting the HCV infection is given below in Figure 4.
Of the 82 patients, 11 (13.4%) were found to be HCV-positive, among whom 10 (91%) were male and one (9%) was female. A majority (82%) of the HCV-positive patients were in the 21–40-year age group, among whom the mean age was 38 ± 13.36 years. The summary of the seroprevalence of HCV by various diagnostic methods is presented below in Table 1.
Table (1):
Seroprevalence of HCV by various diagnostic methods.
No |
Tests |
Frequency (n=82) |
Percentage (%) |
|---|---|---|---|
1 |
ELISA – Positive for HCV antibody |
7 |
8.5 |
2 |
EIA – Positive for both HCV core antigen |
9 |
11 |
3 |
PCR – Positive for HCV RNA
(Gold standard) |
11 |
13.4 |
Analysis of the risk factors of HCV infection revealed that HCV positive-patients were more likely to have a history of diabetes, blood transfusion, and surgery, although the associations were not significant. The mean duration of renal failure and HD were found to be significantly longer among the HCV-positive patients, and even though these patients frequently changed dialysis center, this does not appear to have any significant association with HCV infection. A comparison of the different characteristics of HCV-positive and -negative patients are shown below in Table 2.
Table (2):
Comparison of various characteristics of HCV positive and HCV negative patients.
| No | Characteristics | HCV Positive (N=11) | HCV Negative (N=71) | P value |
|---|---|---|---|---|
| 1 | Age | |||
| ≤ 20 years | 1 (9%) | 7 (9%) | 0.17 | |
| 21-40 years | 9 (82%) | 38 (54%) | ||
| 41-60 years | 1 (9%) | 26 (37%) | ||
| 2 | Gender | |||
| Male | 10 (91%) | 55 (77%) | 0.44 | |
| Female | 1 (9%) | 16 (23%) | ||
| 3 | Risk factors | |||
| Hypertension | 3 (27.3%) | 25 (35%) | 0.09 | |
| Diabetes | 3 (27.3%) | 7 (10%) | ||
| Blood transfusion | 3 (27.3%) | 6 (8.5%) | ||
| Surgery in the past | 1 (9.05%) | 6 (8.5%) | ||
| No risk factors | 1 (9.05%) | 27 (38%) | ||
| 4 | Duration of renal failure (in months), Mean ± SD | 29 ± 7.16 | 19 ± 13.06 | 0.01 |
| 5 | Duration of HD (in months), Mean ± SD | 28 ± 7.62 | 18 ± 13.39 | 0.02 |
| 6 | Change in dialysis centre | |||
| Yes | 9 (82%) | 46 (65%) | 0.26 | |
| No | 2 (18%) | 25 (35%) | ||
Analysis of different biochemical parameters revealed a statistically significant reduction in hemoglobin levels and an increase in creatinine levels among HCV-positive patients. Although the levels of AST and ALT were normal in HCV-positive patients, they were found to have a positive association with HCV infection. In both HCV-positive and -negative patients, the levels of urea was found to be particularly high, although the differences were not statistically significant. Other parameters, such as total bilirubin and total protein, were found to be within the normal range in both groups. A comparison of the assessed biochemical parameters in HCV-positive and -negative patients are shown in Table 3.
Table (3):
Comparison of various biochemical parameters among HCV positive and HCV negative patients.
No |
Parameters |
HCV Positive (N=11) Mean ± SD |
HCV Negative (N=71) Mean ± SD |
P value |
Normal range |
|---|---|---|---|---|---|
1 |
Haemoglobin (g/dl) |
10.1 ± 1.18 |
9.1 ± 1.48 |
0.03* |
12-16 |
2 |
Urea (mg/dl) |
113 ± 36.38 |
113 ± 49.44 |
0.97 |
14-23 |
3 |
Creatinine (mg/dl) |
7.2 ± 4.72 |
4.6 ± 3.60 |
0.04* |
0.7-1.3 |
4 |
Total Bilirubin (mg/dl) |
0.4 ± 0.09 |
0.5 ± 0.25 |
0.12 |
0.1-1.2 |
5 |
AST (U/L) |
20 ± 8.84 |
26 ± 9.16 |
0.05* |
8-48 |
6 |
ALT (U/L) |
14 ± 3.26 |
24 ± 11.45 |
0.007* |
7-55 |
7 |
Total Protein (g/dl) |
6.2 ± 0.60 |
6.3 ± 0.46 |
0.68 |
6-8.3 |
Hepatitis C virus (HCV) infection occurs mainly as a consequence of failures to comply with universal precautionary measures.17 Among hemodialysis (HD) patients, HCV infection is associated with an increased risk of progression to end-stage renal disease and cardiovascular events.18 Poor compliance with HCV-antiviral therapy, the lack of an effective vaccine, and adverse complications have contributed to HCV infection becoming a major global health problem. Hence, identifying HCV infection in HD patients at the earliest stage possible and the timely initiation of treatment may contribute to reducing progression and also the incidence of morbidity and mortality.
Seroprevalence of HCV
In the HD patient cohort examined in this study, we detected an overall HCV prevalence of 13.4%, which is comparable to the proportions reported in studies conducted in other regions of India. Table 4 presents a list of the HCV seroprevalence studies conducted on HD patients in different regions of India.
Table (4):
Seroprevalence studies of HCV infection in HD patients done in India.
Author |
Place |
Year |
Prevalence (%) |
Method |
|---|---|---|---|---|
Chadha MS et al.19 |
Maharashtra |
1993 |
12.1 |
ELISA/PCR |
Jaiswal SP et al.20 |
Madhya Pradesh |
1996 |
41.9 |
ELISA |
Gosavi MS et al.21 |
Maharashtra |
1997 |
27.8 |
ELISA |
Chandra M et al.22 |
Andhra Pradesh |
2004 |
46 |
ELISA |
Reddy AK et al.23 |
Andhra Pradesh |
2005 |
13.2 |
EIA |
Medhi S et al.24 |
Delhi |
2008 |
17.2 |
EIA |
Jasuja S et al.25 |
Delhi |
2009 |
27.7 |
ELISA/PCR |
Surendra kumar P et al.26 |
Tamilnadu |
2011 |
12.4 |
Enhanced chemiluminescence |
Prakash S et al.27 |
Uttar Pradesh |
2012 |
6.9 |
ELISA/PCR |
Jamil M et al.28 |
Meghalaya |
2016 |
1.38 |
ELISA |
Subramanian G et al.29 |
Gujarat |
2016 |
2.7 |
ELISA |
Anitha madhavan et al.30 |
Kerala |
2018 |
8 |
ELISA/PCR |
Dhar A et al.31 |
Jammu |
2016 |
31.4 |
ELISA/PCR |
Present study |
Tamilnadu |
2016 |
13.4 |
ELISA/EIA/PCR |
Studies on the prevalence of HCV infection among HD patients in other countries have reported prevalences ranging from 4.8% to 39% (Table 5), which is accordingly broadly consistent with our findings in the present study.
Table (5):
Seroprevalence studies of HCV infection in HD patients done in other countries.
Author |
Place |
Year |
Prevalence (%) |
Method |
|---|---|---|---|---|
Muller GY et al.32 |
South America |
1992 |
39 |
ELISA |
Soni PN et al.33 |
South Africa |
1993 |
4.8 |
ELISA |
Fabrizi et al.34 |
Italy |
1994 |
20 |
ELISA |
Hinrichsen S et al.35 |
Europe |
1996 |
7 |
ELISA |
Seck SM et al.36 |
West Africa |
2011 |
5.6 |
ELISA/PCR |
Present study |
Tamilnadu, South India |
2016 |
13.4 |
ELISA/PCR |
A majority of the HCV prevalence studies conducted in both India and other countries have been based on the detection of HCV antibodies using ELISA. However, when used alone, antibody detection may be ineffective in detecting early HCV infection. As an alternative, PCR can detect HCV during the early stages of infection, although the high cost of analysis tends to limit its use. Consequently, we believe that detection of the HCV core antigen using EIA would enable a sufficiently reliable early diagnosis of HCV infection, such that measures can be taken to reduce the spread of infection and prevent nosocomial dissemination.
To date, there have been few studies on the seroprevalence of HCV based on the detection of HCV core antigen using EIA. Among those studies that have been conducted, Reddy et al.23 and Medhi et al.24 used HCV core antigen EIA for the detection of HCV infection in patients undergoing HD and reported prevalences of 13.2% and 17.2%, respectively, which are comparable to seroprevalence of 13.4% detected in the present study.
The data presented in Table 4 indicate that during the period from 1993 to 2010, the seroprevalence of HCV in HD patients ranged from 12.1% to 46%,19-25 whereas studies conducted after 2010 reveal a general decline in prevalence to 12.4%.26-30 One exception in this regard, however, is a study conducted in Jammu in 2016, which reported a considerably higher prevalence of 31.4%.31 We speculate that this wide divergence in prevalence could be attributed to a number of contributory factors, including different geographical locations, a different study duration, or differences in the sensitivity and specificity of the methods used for detecting HCV infection. In this latter regard, prior to 2010, such studies tended to rely heavily on 1st generation ELISAs, which were characterized by comparatively low sensitivity and specificity. These assays have, however, been superseded by 2nd and 3rd generation ELISAs, the sensitivities and specificities of which are considerably enhanced.
In the present study, we used a combination of three diagnostic approaches, on the basis of which, we detected prevalences ranging from 8.5% to 13.4%. HCV antibody detection using a 3rd generation ELISA revealed an 8.5% prevalence of HCV infection among HD patients, with 63.6% sensitivity and 100% specificity. Comparatively, the prevalence of HCV infection detected using an HCV core antigen EIA was 11%, with 81.8% sensitivity and 100% specificity. The sensitivity of a test is defined as its ability to yield a positive result for a subject who has a particular disease. Given that the sensitivities of both ELISA and EIA are comparatively low, there is a high likelihood of obtaining false negative results when using these techniques. In general, PCR assays are considered to be the most sensitive tests for detecting HCV infection.
In this study, we indeed obtained high false negative rates of 36.4% and 18.2% using ELISA and EIA, respectively, and hence if HCV is suspected, it is highly recommended that HCV RNA is obtained from all patients undergoing HD.
Using the gold standard PCR test, we detected an overall seroprevalence of HCV infection of 13.4% among HD patients in a tertiary care hospital in South India. The most important risk factors associated with the acquisition of HCV infection among HD patients were identified as a prolonged duration of renal failure and a long duration of HD. We detected a significant reduction and increase in the levels of hemoglobin and creatinine, respectively, among HCV-positive patients. Analysis based on ELISA was found to be moderately sensitive and highly specific in detecting HCV infection, whereas EIA showed both high sensitivity and specificity. Hence, given that seroconversion to HCV antibodies does not occur in all HD patients, on account of their poor immune response, the detection of antibodies based exclusively on ELISA would be insufficiently sensitive for establishing a diagnosis of HCV infection; consequently, in cases in which PCR tests are either unavailable or unaffordable. EIA would be a reasonable alternative as a diagnostic test for detecting HCV infection. In addition, providing relevant education and creating awareness among health care providers regarding the stringent screening of blood and blood products, adopting strict universal precautionary measures in HD units, proper management of HD machines, and appropriate waste management may contribute to minimizing the risk of the nosocomial spread of HCV infection.
ACKNOWLEDGMENTS
The authors would like to thank the participants for giving consent to obtain the samples used for analysis in this study.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
None.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
The study was approved by the Institutional Ethics Committee, Madras Medical College, Chennai, India, with IEC No: 40102015.
INFORMED CONSENT
Written informed consent was obtained from the participants before enrolling in the study.
- Ahmad J, Eng FJ, Branch AD. HCV and HCC: clinical update and a review of HCC-associated viral mutations in the core gene. Semin Liver Dis. 2011;31(4):347-355.
Crossref - Bhattacharya PK, Roy A. Management of hepatitis C in the Indian context: an update. J Liver. 2015;4(187):2167-0889.
Crossref - Ladino M, Pedraza F, Roth D. Hepatitis C Virus Infection in Chronic Kidney Disease. J Am Soc Nephrol. 2016;27(8):2238-2246.
Crossref - Ozer Etik D, Ocal S, Boyacioglu AS. Hepatitis C infection in hemodialysis patients: A review. World J Hepatol. 2015;7(6):885-895.
Crossref - Saab S, Brezina M, Gitnick G, Martin P, Jr. Yee HF. Hepatitis C screening strategies in haemodialysis patients. Am J Kidney Dis. 2001;38(1):91-97.
Crossref - Ghorbani NR, Djalalinia S, Modirian M, et al. Prevalence of hepatitis C infection in Iranian hemodialysis patients: An updated systematic review and meta-analysis. J Res Med Sci. 2017;22:123.
Crossref - Jakupi X, Mlakar J, Lunar MM, et al. A very high prevalence of hepatitis C virus infection among patients undergoing hemodialysis in Kosovo: a nationwide study. BMC Nephrol. 2018;19(1):304.
Crossref - Natov SN, Lau JY, Bouthot BA, et al. Serologic and virologic profiles of hepatitis C infection in renal transplant candidates. New England Organ Bank Hepatitis C Study Group. Am J Kidney Dis. 1998;31(6):920-927.
Crossref - Fissell RB, Bragg-Gresham JL, Woods JD, et al. Patterns of hepatitis C prevalence and seroconversion in haemodialysis units from three continents: the DOPPS. Kidney Int. 2004;65(6):2335-2342.
Crossref - Lok AS, Chien D, Choo QL, et al. Antibody response to core, envelope and nonstructural hepatitis C virus antigens: comparison of immunocompetent and immunosuppressed patients. Hepatology. 1993;18(3):497-502.
Crossref - Xiang Y, Lai XF, Chen P, Yang Y. The correlation of HCV RNA and HCV core antigen in different genotypes of HCV. J Clin Lab Anal. 2019;33(1):e22632.
Crossref - Glynn SA, Wright DJ, Kleinman SH, et al. Dynamics of viremia in early hepatitis C virus infection. Transfusion. 2005;45(6):994-1002.
Crossref - Gretch DR. Diagnostic tests for hepatitis C. Hepatology. 1997;26(3 Suppl 1):43S-47S.
Crossref - Duong MC, Nguyen VTT, Otsu S, McLaws ML. Prevalence of hepatitis B and C virus infections in hemodialysis patients in Vietnam: A systematic review and meta-analysis. JGH Open. 2019;4(1):29-38.
Crossref - Jadoul M, Bieber BA, Martin P, et al. Prevalence, incidence, and risk factors for hepatitis C virus infection in hemodialysis patients. Kidney Int. 2019;95(4):939-947.
Crossref - Ashkani-Esfahani S, Alavian SM, Salehi-Marzijarani M. Prevalence of hepatitis C virus infection among hemodialysis patients in the Middle-East: A systematic review and meta-analysis. World J Gastroenterol. 2017;23(1):151-166.
Crossref - Martin P, Fabrizi F. Hepatitis C virus and kidney disease. J Hepatol. 2008;49(4):613-624.
Crossref - Park H, Adeyemi A, Henry L, Stepanova M, Younossi Z. A meta-analytic assessment of the risk of chronic kidney disease in patients with chronic hepatitis C virus infection. J Viral Hepat. 2015;22(11):897-905.
Crossref - Chadha MS, Arankalle VA, Jha J, Banerjee K. Prevalence of hepatitis B and C virus infections among haemodialysis patients in Pune (western India). Vox Sang. 1993;64(2):127-128.
Crossref - Jaiswal SP, Chitnis DS, Naik G et al. Prevalence of antiHCV antibodies in central India. Indian J Med Res. 1996;104:177-181.
- Gosavi MS, Shah SK, Shah SR, Pal RB, Saldanha JA, Banker DD. Prevalence of hepatitis C virus (HCV) infection in Mumbai. Indian J Med Sci. 1997;51(10):378-385.
- Chandra M, Khaja MN, Hussain MM, et al. Prevalence of hepatitis B and hepatitis C viral infections in Indian patients with chronic renal failure. Intervirology. 2004;47(6):374-376.
Crossref - Reddy AK, Murthy KV, Lakshmi V. Prevalence of HCV infection in patients on haemodialysis: survey by antibody and core antigen detection. Indian J Med Microbiol. 2005;23(2):106-10.
Crossref - Medhi S, Potukuchi SK, Polipalli SK et al. Diagnostic utility of hepatitis C virus core antigen in hemodialysis patients. Clin Biochem. 2008;41(7- 8):447-52.
Crossref - Jasuja S, Gupta AK, Choudhry R, et al. Prevalence and associations of hepatitis C viremia in hemodialysis patients at a tertiary care hospital. Indian J Nephrol. 2009;19(2):62-67.
Crossref - Kumar PS, Venu G, Rao AM, Balakrishnan N, SaraVanan T, Sofiarani A. Prevalence and risk factors of hepatitis C among maintenance hemo- dialysis patients at a tertiary care hospital in Coimbatore. India J Clin Diagnostic Res. 2011;5:725-728.
- Prakash S, Jain A, Sankhwar SN, et al. Prevalence of hepatitis B & C viruses among patients on hemodialysis in Lucknow, Uttar Pradesh. Clin Epidemiol Glob Health. 2014;2:19-23.
Crossref - Jamil M, Bhattacharya PK, Yunus M, Lyngdoh CJ, Roy A, Talukdar KK. Prevalence of hepatitis B and hepatitis C in haemodialysis population in a tertiary care centre in north eastern India. Int J Biomed Amp Adv Res. 2016;7:267-269.
Crossref - Subramanian G, Vegad MM, Vadsmiya MG, Murawala SM, Patel FV. Seroprevalence of hepatitis C in renal failure patients on maintenance hemodialysis – Study in a tertiary care hospital – Western India. Int J Med Sci Public Health. 2016;5(4):678-680.
Crossref - Madhavan A, Sachu A, Balakrishnan AK, Vasudevan A, Balakrishnan S, Vasudevapanicker J. Prevalence of hepatitis C among haemodialysis patients in a tertiary care hospital in south India. Iran J Microbiol. 2020;12(6):644-649.
Crossref - Dhar A, Chandail VS, Sambyal V, Jamwal V. Prevalence of hepatitis C in patients with chronic kidney disease at a tertiary care hospital in north India: a retrospective analysis. Int J Res Med Sci. 2019;7:2198-203.
Crossref - Muller GY, Zabaleta ME, Arminio A, et al. Risk factors for dialysis associated hepatitis C in Venezuela. Kidney Int. 1992;41:1055-1058.
Crossref - Soni PN, Tait DR, Kenoyer DG, et al. Hepatitis C virus antibodies among risk groups in a South African area endemic for hepatitis B virus. J Med Virol. 1993;40(1):65-68.
Crossref - Fabirizi F, Lunghi G, Raffaele L, et al. Serologic survey for control of hepatitis C in hemodialysis patients: Third-generation assays and analysis of costs. Nephrol Dial Transplant. 1997;12(2):298-303.
Crossref - Hinrichsen H, Leimenstoll G, Stegen G, et al. Prevalence and risk factors of hepatitis C virus infection in haemodialysis patients: a multicentre study in 2796 patients. Gut. 2002;51(3):429-433.
Crossref - Seck SM, Dahaba M, Gueye S, Ka EF. Trends in hepatitis C infection among hemodialysis patients in Senegal: results of a decade of prevention. Saudi J Kidney Dis Transpl. 2014;25(6):1341-1345.
Crossref
© The Author(s) 2023. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.