ISSN: 0973-7510
E-ISSN: 2581-690X
This study focused on selection and characterization of lactic acid bacteria (LAB) strains to be used as starter and protective culture for food fermentations. Thirty LAB isolates were isolated from different fermented foods onto MRS agar medium. Two LAB isolates only CK21 and PP17 showed the highest medium acidification values pH 3.2 and 3.6 respectively and inhibited many Gram positive and Gram negative pathogenic bacteria. CK21 and PP17 isolates were selected due to their desired properties to be used as starter cultures. Based on biochemical properties as well as sequences of 16S rRNA genes, CK21 and PP17 isolates were identified as belonging to Entercoccus faecium CK21 (E.faecium) and Lactobacillus fermentum PP17 (L.fermentum) . Both strains tolerated NaCl up to 10%, bile salt up to 3% and pancreatin up to 2%. Also, these strains have the ability to grow at wide pH range (2-9) and temperature range (20-50°C). These strains inhibited many pathogenic bacteria such as Bacillus cereus (B.cereus), Streptococcus pyogens (S.pyogens), Staphylococcus aureus (S.aureus), Escherichia coli (E.coli), Listeria monocytogens (L.moncytogens), Acinetobacter baumanii (A.baumanii) and Salmonela typhimurium (S.typh). The antibacterial compound from culture supernatants of both strains was found to be proteinacious in nature, heat stable at 100ºC for 30min, and active over a wide pH ranges (2-8). All this characters coupled with the definitions of bacteriocins, so the antibacterial compound could be classified as bacteriocin. Both cell free supernatants (CFS) and neutralized CFS (NCFS) of lactic acid bacterial strains CK21 and PP17 inhibited E.coli and S.pyogenes distinctively in vitro.
Lactic acid bacteria (LAB), Starter cultures, Probiotics, Food, Bacterial pathogens
Lactic acid bacteria (LAB) include a wide group of Gram-positive fermenting bacteria which are generally non- sporulating and non-motile. They are widespread in nature, in soil, vegetables, meat, milk and the human body1. Bio preservation refers to extending shelf life and enhanced safety of foods using microorganisms and/or their metabolites 2. LAB are mainly used in this purpose as they play important role in enhancing shelf life and microbial safety, improve texture, flavor and contribute to the pleasant sensory profile of the end product 3.
Starter cultures are microbial preparations of large numbers of cells of at least one or more microorganisms to be added to a raw material to produce a fermented food by accelerating and steering its fermentation process3. LAB are used as natural or selected starters in food fermentations and apply the antimicrobial effect due to different metabolic processes (lactose metabolism, proteolytic enzymes, citrate uptake, bacteriophage resistance, bacteriocin production, polysaccharide biosynthesis, metal-ion resistance and antibiotic resistance)4.
Recently, new starter cultures of LAB bacteria with an industrially important functions are being developed and provide microbial safety and nutritional, or health advantages. Examples are LAB bacteria that produce antimicrobial substances, sugar polymers, sweeteners, aromatic compounds, vitamins, or useful enzymes, or that have probiotic properties. Nowadays the food products are considered healthy due to the characters of specific strains of LAB they contained5. There are characters of bacterial strains to be used as starter and probiotic cultures. For performing the probiotic functions, LAB have to face the low pH environment of the gastrointestinal tract. Therefore, acid resistance of LAB is important not only for their own growth, but also for fermentation and preparation of probiotic products6. Also the LAB strains must have the ability to produce antimicrobial substances such as organic acids (mainly lactic and acetic acid), hydrogen peroxide and also other compounds, such as bacteriocins and antifungal peptides7. Starter cultures consisting of LAB produce bacteriocins and other antimicrobial substances aid in extending the shelf life of food, modifying food sensory attributes and improving texture8. Also, LAB strains that can be used as probiotic and contribute in food industry as starter culture must tolerate bile salt as it is one of the most effective stress factors and it is very undesirable for survival of the probiotic strains during gastrointestinal tract9. The main aim of this study was to select LAB strains to be used as starter and protective cultures with probiotic characters in order to improve the quality of food fermentations
Collection of food samples and isolation of LAB
Thirty samples were collected from different food types such as pickles (olive, pepper, lemon, carrot and mixed samples), fermented dairy products as Kareesh cheese, white cheese and mesh, fermented fish, grape fruit and tomato salsa. All the samples are collected from different markets at Sharkia Governorate 100Km north Cairo, Egypt in the period from 1/5/2016 till 1/11/2016. Isolation of LAB was carried out using the microbial pour plate method. Different dilutions of the samples prepared in saline (0.85% NaCl) and pour plated onto MRS agar medium10 and incubated at 37°C, for 24h. Colony forming units (CFU/mL) of LAB were determined. The presumptive LAB species were purified by re-streaking on MRS agar plates and incubated to obtain single pure colony. After incubation the colonies were stored in refrigerator on MRS agar slants/stabs throughout the experiments.
Growth of LAB isolates at wide pH values and medium acidification
Growth of bacterial isolates at different pH values and their ability to acidify medium were tested. MRS broth was prepared and adjusted with either 0.1M HCL or NaOH to different pH values (3, 5, 7, and 9). The tubes were inoculated by 1% V/V fresh overnight LAB culture and incubated at 37°C for 24h. The growth was measured by reading optical density at 600nm using spectrophotometer (New Brunsweak Scientific CO.) and also final pH was detected by using pH meter11, 12.
Antagonistic effect of LAB isolates
The antimicrobial activity of the isolated LAB against pathogenic bacteia (E. coli, S. aureus, L. monocytogenes, B.cereus, A.baumanii, Salmonella typhi and S.pyogens), was performed by the well diffusion assay. The pathogenic bacteria were provided from laboratory of Applied Bacteriology, Faculty of science, Zagazig University, Egypt. The pathogenic bacteria were propagated in nutrient broth at 37°C for 24 h and streaked on to BHI (oxoid) agar plates. The plates were left for solidification then stored for 2h in a refrigerator. After that wells were made by using sterile cork porer and filled with cell-free supernatants (CFS) of LAB isolates that were prepared by centrifugation of LAB cultures at 10,000 rpm for 20 min. CFS was adjusted at pH 7 to avoid inhibition due to acidity and sterilized by filtration (0.45Mm, Milipore, Amican). After incubation at 37°C for 24h the antimicrobial activity was determined by measuring the diameter of the inhibition zone around each well (mm)13.
Identification of bacteria
Both LAB isolates CK21 and PP17 which showed the highest medium acidification and inhibited most of pathogenic bacteria were identified according to standard clinical laboratory methods recommended by Bergey’s Manual of Systematic Bacteriology14, 15.
Also the identification of both CK21 and PP17 isolates was confirmed by the fingerprinting of 16S rRNA gene(s). Total DNA was extracted from both CK21 and PP17 according to Sambrook and Russel (2001). The gene coding for 16S rRNA was amplified from each tested isolate by PCR using the forward primer 5’AGAGTTGATCMTGGCTCAG3′ and reverse primer 5′ TACGGYTACCTTGTTACGACTT 3′16. Molecular size of amplified fragments should be approximately 1485 bp. The amplification conditions were as follows: 94°C for 10 min and 35 cycles of denaturation at 95°C for 30s, annealing extension at 56°C for 1 min, 72°C for 1 min and an extension at 72 °C for 10 min. the PCR products were subjected to agarose gel electrophoresis and the bands of ampilified 16S rRNA were cleaned up using gene purification Kit (Centrisep). The amplified DNA fragments were partially sequenced at GATC Biotech (Metabion Germany) using ABI 3130, USA DNA sequencer. Sequence analysis and their comparison to deposited data in gene bank was made using the Basic Local Alignment Search Tool (BLAST) Program at http://ncbi.nlm.nih.gov/blast17.
Sodium chloride tolerance by selected LAB bacterial strains
Ability of both CK21 and PP17 strains to tolerate sodium chloride was tested. Number of test tubes contained MRS broth were adjusted to different NaCl concentrations (2, 4, 6, 7.5, 10, 12.5 and 15 %). After sterilization, each tube was inoculated by 1% V/V fresh overnight LAB bacterial strains and incubated for 24h at 37°C. After incubation, the growth of bacteria was measured by spectrophotometer at 600 nm12.
Bile salt and pancreatin tolerance by LAB strains
Sterile MRS broths were supplemented with bile (oxgal) at concentrations of (0.1, 0.5, 0.7, 1, 1.5, 2, and 2.5%) and pancreatin at concentrations of (0.5, 0.7, 1, 1.5, and 2 %) separately. After sterilization, 1% V/V of overnight culture of both CK21 and PP17 strains were inoculated into the MRS broth and incubated at 37°C for 24h. The survival rates of LAB isolates were measured by taking absorbance at 600nm using the spectrophotometer (New Brunsweak Scientific CO.)18, 19.
Antibiotic susceptibility test
The susceptibility of antibiotic discs was done by disc diffusion method as described by Bauer et al. (1966). Selected LAB strains CK21and PP17 were inoculated into MRS broth and incubated at 37 °C for 24h. Then, LAB bacterial suspension was spread onto the surface of the MRS agar plate. The antibiotic discs of fourteen antibiotics listed in Table 4 were placed onto the surface of MRS agar plates and incubated at 37°C for 24h. After incubation, inhibition zone diameters around the disc were measured using millimeter ruler20. The sensitivity, resistance and intermediacy of both LAB strains were calculated according to CLSI (2008) and NCCLS (2009).
Growth of CK21 and PP17 at wide temperature range
MRS broths were prepared and inoculated by 1%V/V of both LAB strains. The inoculated media were incubated at different temperatures (25, 30, 35, 40, 45, 50°C) for 48h. After incubation, the growth was detected by measuring the absorbance at 600 nm using spectrophotometer (New Brunsweak Scientific CO.).
Characterization of inhibitory substances produced by both LAB strains (CK21 & PP17)
Preparation of cell free supernatant
Cell-free supernatants (CFSs) were prepared by cultivating LAB strains in MRS broth for 16h at 37°C and centrifuged for 10 min at 4°C. The CFSs were used t inhibit indicator bacteria used. In another experiments, this CFS was neutralized by both 0.1N HCl and 0.1NaOH at pH 7.0 and filter sterilized (0.45 milipore, Amican) and designated NCFS 21.
Factors affecting inhibitory substances
The proteinaceous nature of the inhibitory compounds in NCFS was confirmed by testing their sensitivity to proteolytic enzymes. Aliquots of NCFS was treated by 1mg/mL from proteinase K, chemotrypsin, trypsin, pepsin, a-amylase and catalase (Sigma, USA). Antimicrobial activity was determined by using the well diffusion method as described above in Materials and Methods [22].
The pH on stability of inhibitory substance in NCFS was tested by adjusting CFS of both strain at different pH values 2,4,6,8 by using 1N HCl and 1N NaOH and incubated overnight at room temperature, pH was adjusted at pH 7 for all test and then antimicrobial activity was examined by using well diffusion method23
To test the thermal stability of inhibitory substance, aliquots of sterilized NCFS of each strain was exposed to different temperature values viz. 70, 80, 90°C, for 5 min, 100°C for 5, 10, 20, 30 min and 121°C for 15 min. Activity of inhibitory substances was detected by using well diffusion method23.
To study the effect of organic solvents on activity of the inhibitory substance, 1ml from sterilized NCFS of each LAB strain was mixed with 10% (v/v) from different organic solvents as chloroform, benzene, ethyl ether, and acetone and diethyl ether. Controls were NCFS without adding organic solvents. To allow solvents to evaporate each of samples and controls were incubated overnight at 60 °C. Then, antimicrobial activity was determined by using well diffusion method 24.
Inhibition of some pathogenic bacteria by CFS and NCFS from L. fermentum PP17 and E.faecium CK21
The effect of CFS and NCFS of CK21 and PP17 on the selected pathogenic bacteria: S.pyogens and E.coli were investigated, in vitro. BHI broths (Oxoid) were cultivated by either E.coli or S.pyogens bacteria as they were the most sensitive organisms at concentrations of about 2×105 CFU/mL. One mL from filter-sterilized CFS and NCFS of L.fermentum was added separately to 9ml from growing cells of S.pyogens and also 1ml from CFS and NCFS of E.faecium was added to E.coli. Control cells contained indicator organism without either CFS or NCFS of both LAB strains tested. After incubation at 37°C, samples were taken at time intervals; 0, 6, 12, 24, 48, 72, 96h and CFU/mL were determined at each time. The percentage of cell viability was determined in relation to control25.
The recent studies focused on selection of LAB isolates to be used as starter and protective culture in food fermentations or with probiotic capability26, 27. LAB also used in bio preservation process in order to extended shelf life and enhanced safety of foods2. In this present study, Thirty LAB isolates were isolated from different fermented food types such as pickles and fruits onto MRS agar medium. These LAB cultures were assayed for medium acidification, as production of acids is a desired property for probiotic capability and used of strain as starter cultures as most of food-borne pathogens cannot grow at acidic conditions26. Fourteen LAB isolates out of 30 tested decreased pH values to acidic levels. LAB isolates CK21, and PP17 were the most powerful in acidification of medium, they decreased pH value to almost 3.5 within 24h .This is a promising result in using of these two isolates as starter cultures for food fermentations because they could consume sugars from food matrix converting them to acid via fast fermentation process. Such produced acids are also attractive criteria for both strains to be protective cultures where pathogenic bacteria cannot grow. By consumption of food fermented by, these LAB can balance pathogenic bacteria in small intestine and improve intestinal ecosystem by their produced acids. So both CK21 and PP17 may be used as probiotics28. This is in confirm with Banwo et al.29 who reported ability of E. faecium to make shift in pH until 3.85 after 48-h incubation. The acidic medium formed due to production of lactic acid is not suitable for survival of spoilage bacteria that may found in fermenting substrate 30
The fourteen LAB isolates were tested for growth at different pH values (3, 5, 7, and 9). All of them grew well at various pH levels with higher growth value in pH range (6-7) also they were able to tolerate and grow at acidic pH values (3, 5) as shown in Table (1). It is of interest to recall herein that both CK21 and PP17 LAB isolates showed fast growth at the pH values tested. Such results enable both of these isolates to possess broader applications in food fermentations at different pH values. Similarly, Kumari et al.31 found that different LAB isolates were tolerant acidic conditions until pH 3. Also this coupled with the work of Enan et al.32, 33
Table (1):
Growth of LAB isolates at different initial pH values and their final pH values.
| pH 3 | pH 5 | pH 7 | pH 9 | LAB isolates No. & Code |
||||
|---|---|---|---|---|---|---|---|---|
| Final pH | OD600 | Final pH | OD600 | Final pH | OD600 | Final pH | OD600 | |
| 3 | 0.294 | 5 | 0.415 | 5.1 | 1.656 | 8 | 1.391 | GF7 |
| 3 | 0.528 | 4.8 | 0.822 | 5 | 1.33 | 7.4 | 0.605 | PL8 |
| 3 | 0.344 | 5.4 | 0.477 | 5 | 0.700 | 6.9 | 0.574 | PO10 |
| 3 | 0.399 | 4.8 | 0.800 | 4.5 | 1.717 | 6.4 | 1.092 | PO11 |
| 3 | 0.275 | 5 | 0.952 | 5.1 | 1.230 | 6.2 | 0.872 | PO12 |
| 3 | 0.362 | 4.7 | 0.652 | 5.3 | 1.787 | 6 | 1.11 | PM14 |
| 3.1 | 0.354 | 5 | 1.775 | 5.4 | 1.717 | 6.2 | 0,917 | PC15 |
| 2.3 | 0.433 | 4 | 0.952 | 3.4 | 1.23 | 6 | 0.752 | PP17 |
| 2.8 | 0.381 | 5.1 | 0.530 | 5 | 1.181 | 7.3 | 0.682 | CK18 |
| 3 | 0.273 | 4.2 | 0.897 | 4.2 | 1.398 | 7.2 | 0.410 | CK19 |
| 2.5 | 0.393 | 4 | 0.708 | 3.5 | 0.966 | 5.8 | 0.524 | CK21 |
| 2.9 | 0.276 | 5.4 | 0.420 | 5.2 | 0.943 | 7 | 0.760 | CM25 |
| 3 | 0.300 | 4.2 | 0.523 | 5.1 | 0.860 | 6 | 0.310 | CV28 |
| 3.1 | 0.370 | 4.8 | 0.730 | 5 | 1.272 | 5.3 | 0.672 | CV30 |
GF: Grape fruit, PL: Pickles (lemon), PO: Pickles (olive), Pickles ( mixed), PC: Pickles (carrot), PP: Pickles (pepper), CK: Cheese (Karessh) CM: Cheese (mesh), CV: Cheese (veta)
To investigate the probiotic capabilities of the 14 LAB tested, their antimicrobial activities against many Gram positive and Gram negative food –borne pathogen were studied. Results are given in Table (2) S.aureus and S.pypgens were inhibteed by all LAB tested, but S.typhimnnum was not inhibited by any of them. Most of tested LAB isolates inhibited L.monocytogens, B.cereus and Gram negative strains: E.coli and A.baumanii. This inhibition of this sensitive bacteria could be due to the inhibitory metabolites produced by LAB isolates such as lactic acid and other organic acid, hydrogen peroxide, diacetyl, carbondioxide, etanol and bacteriocin34. This greatly show the necessity of LAB in food fermentations as the antimicrobial compounds produced by these bacteria play a promising role in ensuring the safety and extending the shelf life of produced fermented food 32.
Table (2):
Antimicrobial activity of LAB isolates against some pathogenic bacteria.
| Diameter of inhibition zone (mm) | |||||||
|---|---|---|---|---|---|---|---|
| Gram negative | Gram Postive | LAB isolates | |||||
| S.typhi | A.baumanii | E.coli | S.aureus | L.monocytogens | S. pyogens | B. cereus | |
| 0 | 17 | 0 | 12 | 20 | 10 | 18 | GF7 |
| 0 | 18 | 0 | 0 | 15 | 17 | 10 | PL8 |
| 0 | 3 | 0 | 10 | 10 | 13 | 20 | PO10 |
| 0 | 15 | 20 | 15 | 17 | 12 | 18 | PO11 |
| 0 | 0 | 10 | 10 | 10 | 9 | 0 | PO12 |
| 0 | 9 | 10 | 8 | 8 | 10 | 10 | PM14 |
| 0 | 0 | 13 | 12 | 0 | 0 | 10 | PC15 |
| 0 | 0 | 15 | 10 | 10 | 0 | 0 | CK18 |
| 0 | 25 | 20 | 18 | 22 | 25 | 20 | PP17 |
| 0 | 0 | 12 | 12 | 0 | 0 | 0 | CK19 |
| 0 | 27 | 25 | 15 | 19 | 20 | 18 | CK21 |
| 0 | 10 | 0 | 12 | 0 | 0 | 10 | CM25 |
| 0 | 0 | 0 | 17 | 10 | 0 | 12 | CV28 |
| 0 | 9 | 5 | 11 | 0 | 12 | 0 | CV30 |
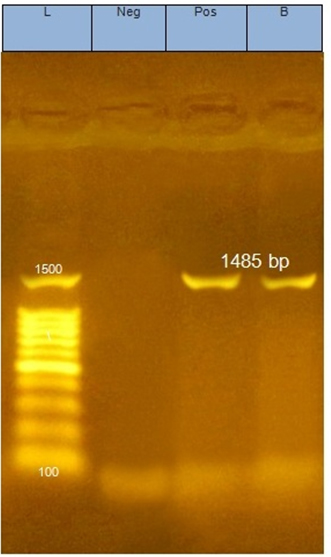
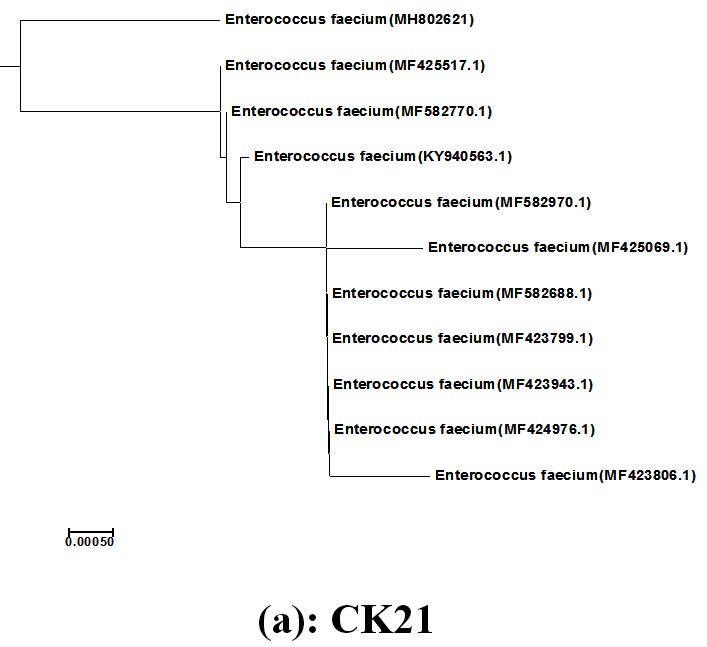
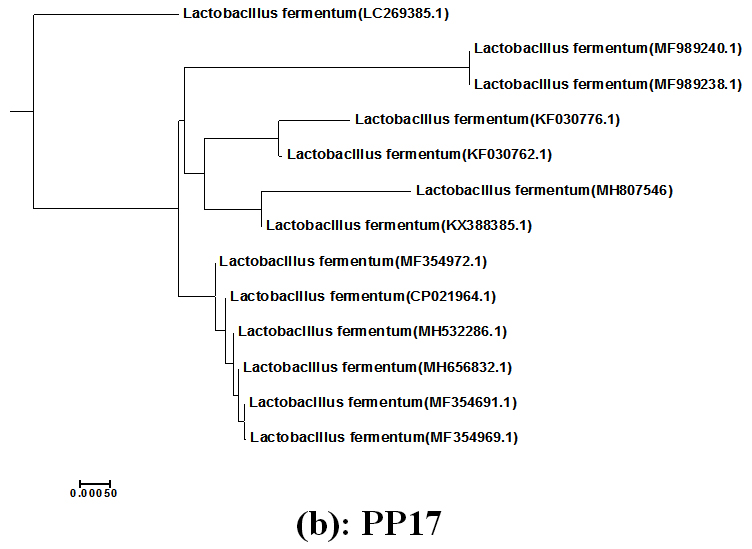
The LAB isolates; CK21 and PP17 showed the highly medium acidification, grew at wide pH range (3-9) and inhibited all the indicator bacterial pathogens tested expect S. typhimurum, they were subjected to identification using the criteria of biochemical identification that given in Bergey,s Manual of Systematic Bacteriology [14],[15]. The isolates CK21 and PP17 could be identified as belonging to E.faecium and L.fermentum respectively. Because biochemical identification showed sometimes speculative results the two isolates were subjected to molecular identification using 16S rRNA cataloging analysis26. The 16S rRNA genes of CK21 and PP17 isolates were polymerized using PCR technique, electrophoresed via agarose gel and showed molecular mass of about 1485bp Fig.(1). They were sequenced and both sequences of two strains CK21 and PP17 were submitted to GenBank (http://www.ncbi.nlm.nih.gov/GenBank/update.htm) under accession numbers MH802621 and MH807546 respectively Fig.(2) By using Basic Local Alignment Search Tool (BLAST) they showed similarities e” 98 % to E.faecium (CK21) and L.fermentum (PP17) 17 . The phylogenetic tree was displayed using treeview program Fig. (3a, b)
Fig. 1. Agarose gel electrophoresis of PCR products including the amplified 16S rRNA gene of two LAB isolates. Lane 1, DNA marker; Lane 2, negative control; Lane 3, E.faecium CK21; Lane 4 L.fermentum PP17
Fig. 2. Nucleotide sequences of 16SrRNA genes of the purified PCR products of LAB isolates (A); CK21 and (B); PP17
NaCl is an inhibitory substance that prevent the growth of certain types of bacteria. Also salt is the most widely preservative factor used in many food industry. Therefore, it was essential to test the NaCl tolerance of lactic acid bacteria [35]. In this present study L.fermentum was able to grow at MRS broth supplemented with 7% NaCl concentrations while the growth was decreased at 10% NaCl concentration or no growth detected Table (3). Meanwhile, E.faceium strain had the ability to grow in MRS broth supplemented with 12% NaCl concentrations. These results were similar to that obtained by Forhad et al.36 who reported that L.fermentum grew well until 7% NaCl concentrations but failed to grow at 10% NaCl concentration.
Table (3):
Characteristics needed for LAB strains to be used as starter and protective culture.
| Parameters | Growth of bacterial strain (O.D) | |
|---|---|---|
| Sodium chloride Conc. % | L. fermentum | E. faceium |
| 2 | 0.957 | 0.875 |
| 4 | 0.791 | 0.686 |
| 6 | 0.353 | 0.567 |
| 7.5 | 0.296 | 0.332 |
| 10 | 0.043 | 0.150 |
| 12.5 | 0 | 0.055 |
| Bile salt Conc. % | ||
| 0.1 | 0.935 | 1.06 |
| 0.5 | 0.580 | 0.981 |
| 0.7 | 0.520 | 0.761 |
| 1 | 0.362 | 0.729 |
| 1.5 | 0.206 | 0.435 |
| 2 | 0.240 | 0.350 |
| 2.5 | 0.206 | 0.123 |
| 3 | 0.133 | 0.055 |
| Pancreatin Conc. % | ||
| 0.5 | 1.130 | 1.014 |
| 0.7 | 0.947 | 0.947 |
| 1 | 0.755 | 0.654 |
| 1.5 | 0.350 | 0.223 |
| 2 | 0.124 | 0.089 |
Furthermore, CK21 and PP17 strains were tested for their ability to tolerate bile salt and pancreatin. Both strains showed high tolerance to bile salt up to 3% and pancreatin at 0.5-2% Table (3). Also this previous work37 reported that L.fermentum showed good tolerance to bile until 2.5% bile concentration. Also, Banwo et al.29 found that E.faecium tolerate bile salt up to 1% bile concentration. Differences in bile salt tolerance among LAB strains are related to their hydrolase activity .Tolerance to bile salt is one of the criteria used to select probiotic that would be able to act effectively in human gastrointestinal tract, and colonize small intestine and produce their metabolic activity37. In this study both LAB strains tolerate pancreatin up to 1.5%. Such criteria is necessary to viability of any bacterial strain to be probiotic.
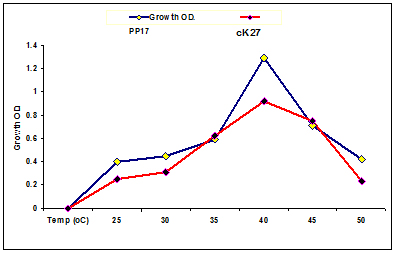
In the current study, both strains were able to grow at wide temperature growth ranged from 25°C to 50°C Fig. (4). The maximum growth of L.fermentum and E.faecium was achieved at 40°C and decreased by increasing temperature up to 50°C. Van den Berghe et al.38 reported that the optimum temperature for E.faecium was 40°C and growth observed until 50°C. The bacterial ability to grow at high temperature is a good characteristic, as it could be interpreted as showing an increased rate of growth and lactic acid production. Growth of LAB strains at wide temperature range enable the organisms to be used in food fermentations at wide temperature growth range. Moreover, the fermentation that require high temperature decreases the opportunities of contamination by other microorganisms39.
Table (4):
Antibiotic sensitivity of LAB strains E. faceium CK21 and L. fermentum PP17.
| Antibiotic | Symbol | Coc.mg/disc | Sensitivity and diameter of inhibition (mm) | |
|---|---|---|---|---|
| CK21 | PP17 | |||
| Amikacin | AK | 30 | R (0) | R (0) |
| Amoxycillin/Clavulanic acid | AMC | 20 | R (0) | S (25) |
| Ampicillin/sulbactam | SAM | 15 | R (0) | R (0) |
| Ampiclox | AX | 15 | S (20) | S (38) |
| Azithromycin | AZM | 15 | M (14) | S (19) |
| Ciprofloxacin | CIP | 15 | R (12) | M (17) |
| Ceftriaxone | CRO | 30 | R (0) | R (8) |
| Cephradine | CEP | 15 | R (12) | S (20) |
| Doxycycline | DO | 30 | S (35) | S (21) |
| Erythromycin | E | 15 | R (0) | M (15) |
| Imipenem Nitrofurantoin |
IPM F |
10 30 |
S (30) | S (40) |
| Sulphamethoxazol /trimethoprim |
SXT | 25 | S (20) | S (30) |
| Vancomycin | VA | 30 | R (0) | R (0) |
Ranges of zone of inhibition diameters exhibited by bacteria considered sensitive (𝑆), moderate (M), or resistant (𝑅) to each antibiotic
Moreover, the sensitivity of CK21 and PP17 strains to antibiotics was tested. L.fermentum (PP17) showed sensitivity and moderate sensitivity to most of antibiotic tested while it was resistant to Amikacin, Ampicillin/sulbactam, Ceftriaxone, Sulphamethoxazol/trimethoprim and Vanomycin Table (4). While, for E.faecium (CK21) the rate of antibiotic resistance is higher than that in the PP17 strain. CK21 strain was sensitive to Vanomycin, Nitrofurantoin, Imipenem, Doxycycline and Ampiclox. Our results similar with this previous work40 which found that E.faecium was sensitive to Vanomycin, Nitrofurantoin and resistant to Erythromycin and ciprofloxacin similar with our results. Melo et al.41 reported that L.fermentum TCUESC01 was resistant to Amikacin, Sulphamethoxazol/trimethoprim and Vanomycin and sensitive to most of antibiotic that PP17 strain sensitive to it. Knowledge of antibiotic sensitivity is very important by considering three important factors: the rare possibility of infection by Lactobacillus, the risk of horizontal transfer of resistance genes to other microbes, and the link between probiotic bacteria and antibiotic treatment42. The resistance profiles of our strains has the advantage that these strains can be introduced in the human diet in the same time of antibiotic treatment. Also, the antibiotic resistance is important for intestinal microbiota to recover quickly after antibiotic treatment. While this resistance is highly danger in people who suffer from bacterial infections. As LAB may act as a carrier to antibiotic resistance genes similar to those found in human pathogens. So they can transfer resistance genes to pathogenic bacteria 43.
From these results, it can be observed that both CK21 and PP17 strains have the best biotechnological properties such as inhibition of some gram positive and gram negative pathogenic bacteria, acidification of medium by production of acids, growing at wide ranges of pH and temperature and tolerance to stress conditions such as NaCl, pancreatin and bile salt. So, this strains could be used as a probiotic bacteria, starter and protective cultures for food fermentation. These results are similar with Mello et al. 41 who reported that L.fermentum can be used as a safe probiotic and food additive. Also, Banwo et al. 29 found that the E.faecium strains exhibited a good probiotic activity because of their tolerance to low pH, bile salts and their inhibitory effect against food pathogens and spoilage micro-organisms. Due to the importance of this two organisms, studies were further concentrated to characterize their inhibitory substance.
Table (5):
Characterization of the inhibitory substance produced by LAB strains CK21 and PP17.
| Diameter of inhibition zones (mm) | Sodium chloride | ||
|---|---|---|---|
| CK21 | PP17 | Enzymes | |
| 0 | 0 | Pepsin | |
| 0 | 0 | -chemotrypsin | |
| 0 | 0 | Trypsin | |
| 22 | 20 | Catalase | |
| Thermostability | |||
| 25 | 28 | For 30 min | 70 ºC |
| 25 | 28 | 30 min | 80 ºC |
| 25 | 28 | 30 min | 90 ºC |
| 25 | 28 | For 5 min | 100 ºC |
| 25 | 28 | 10 min | |
| 23 | 25 | 20 min | |
| 20 | 25 | 30 min | |
| 0 | 0 | 15 min | 120oC |
| Organic Solvent | |||
| 28 | 28 | Acetone | |
| 28 | 28 | Chloroform | |
| 25 | 28 | Ethyl lchol | |
| 28 | 25 | Diethyl ether | |
| 25 | 28 | Butanol | |
| pH | |||
| 15 | 13 | 2 | |
| 25 | 19 | 4 | |
| 28 | 25 | 6 | |
| 11 | 15 | 8 | |
| 5 | 0 | 9 | |
The inhibitory activity of NCFS from either CK21 or PP17 was inactivated after treatment of this CFS by protease enzymes (trypsin, pepsin and a-chemotrypsin), while the activity remained stable after catalase treatment Table 5. This confirming the proteinaceous nature of their antimicrobial substance as similar with Priscilia et al.44. To study the stability of the inhibitory substance (s) of both CK21 and PP17, CFS of both strains were adjusted at different pH values (2, 4, 6, 8, and 9) for 12 h and then readjusted at pH7. The activity of inhibitory substances of both strains was pH dependent with maximal activity at pH 4-6,and lower activity at slightly alkaline and lower acidic pH values . This showed that the inhibitory substance was stable at pH range 2-8. So, CK21 and PP17 strains can be used as starter and protective culture for food fermentations at acidic pH level, such acidic pH level will not effect on the stability of their bacteriocin produced. The results are similar with Ohenhen et al.45 who reported that the bacteriocin activity of some LAB was high at acidic level 2, 4, 6 and decreased at alkaline level by testing their antimicrobial activity against E.coli and S.aures
The inhibitory substances of both CK21 and PP17 strains were tested for it is thermal stability. The results showed that these substances were heat stable up to 100°C for 30 min in both strains Table (5). While, the antimicrobial activity lost in both strains after exposure to 121°C for 15 min. So, the antimicrobial compound of both strains could be classified under heat stable bacteriocin like substance. Similarly, Park et al.46 reported that enterocins produced by Enterococcus faecium JCM 5804 was heat stable up to 100°C for 30 min but the inhibitory activity was lost at 121°C for 15 min. Also, Maria and Janakiraman 47 revealed that the bacteriocin of L. acidophilusNCIM5426 was heat stable up to 100°C while part of activity was lost at 121°C for 15 min. Thermal stability of inhibitory substance of LAB is a typical characteristic for bacteriocin and bacteriocin like substance. Temperature stability is important if the bacteriocin are used as a food preservative, because many procedures of food preparation involve a heating step.
The inhibitory substances of both CK21 and PP17 strains were treated by different organic solvents but their antimicrobial activity did not affected Table 5. The fact that the bacteriocins were stable with organic solvents indicated that they were soluble in them. These results similar with the work of Ghanbari et al. 48 who found that lactocin RN78 and plantracin LC74 bacteriocins which were produced by different lactobacillus spp. kept their antimicrobial activity after treatment by organic solvents such as butanol, ethanol and methanol.
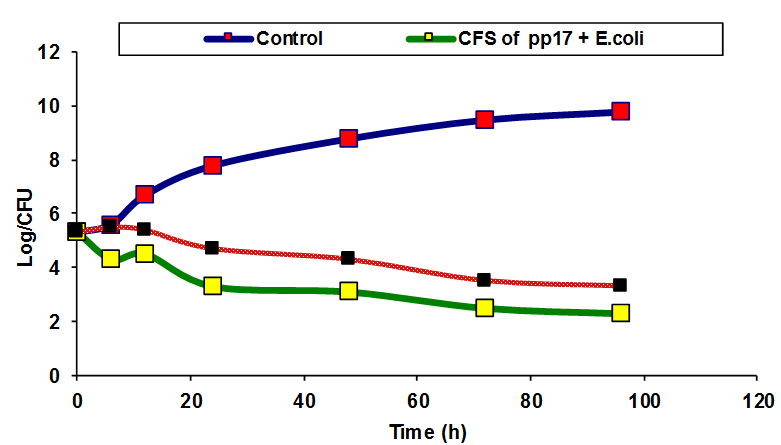
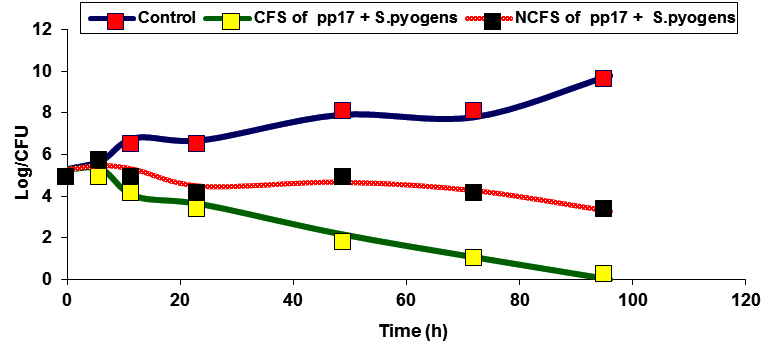
Fig. 5. Inhibitory effect of CFS and NCFS of PP17 against E.coli and S.pyogens
Inhibition of some pathogenic bacteria as E.coli and S.pyogenes by CFS and NCFS of CK21 and PP17 strains were studied, In vitro. The results showed that there was a reduction in the viable cell count of pathogenic bacteria after treatment with CFS and NCFS of CK21 and PP17 strain Fig (5a, b). The growth of pathogenic bacteria decreased distinctively, reaching almost 5-7 log cycles (growth reduction) within 72h in case in PP17 strain. While CFS of CK21 prevented growth of E.coli and S.pyogens respectively Fig (6a, b). Also NCFS of this strain inhibited both selected pathogenic bacteria vigorously and difference in colony counts among control and treated samples were almost 5-6 log cycles. In general CFS obtained from both probiotic strains (CK21 and PP17) was more active than NCFS in inhibition of both E.coli and S.pyogens. This due to CFS can contain in addition to bacteriocin, organic acids such as lactic acids, diacetyl, ethanol and acetaldehyde 31. These results are similar with that obtained by Hor and Liong 49 who found that the growth inhibition of S.aureus by CFSs of five selected LAB higher than that obtained by NCFS which make reduction in growth inhibition by 50% in the percentage of inhibition than the initial level. Bajpai et al.50 showed that there is a remarkable decline in CFU counts of S. aureus KCTC-1621 and E.coli O157:H7 when exposed to the 289 CFS of P. pentosaceus 4I1.
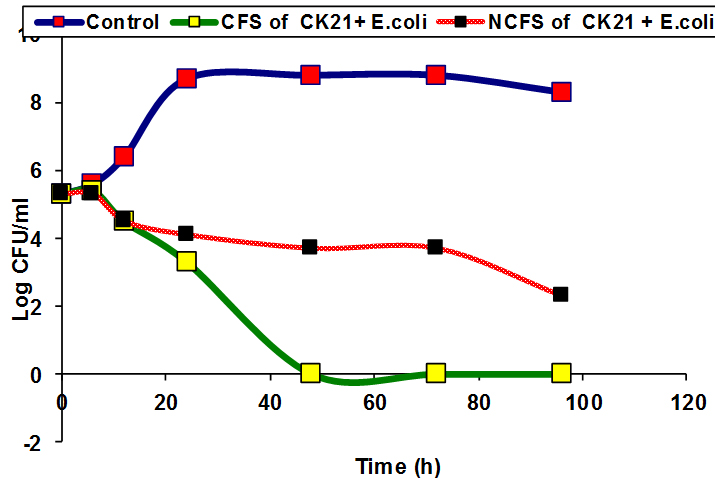
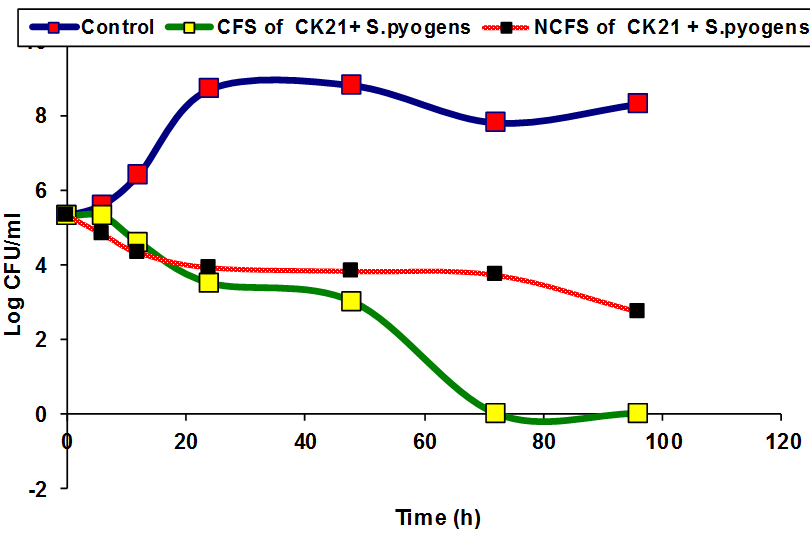
Fig. 6. Inhibition of E.coli and S.pyogens by NCFS and CFS from CK21 strain
The two lactic acid bacterial strains; CK21 and PP17 that were isolated from cheese and pickles respectively were selected due to their qualified properties as these strains are able to make medium acidification, production of some enzymes, grew well at wide pH and temperature range and make inhibition to some gram positive and gram negative foodborne pathogens in vitro. Such properties are good features for lactic acid bacterial strains to be used as starter and protective culture for food fermentations. Addition of these strains to human diet by consumption with foods fermented with them lead to improvement of many human nutritional functions due to their probiotics characters. However, further work will be necessary in the future to study the inhibition of pathogenic bacteria by these strains in situ and to study their probiotic capabilities in vivo.
ACKNOWLEDGMENTS
None.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
- Hofvendahl K & Hahn-Hagerdal B, Factors affecting the fermentative lactic acid production from renewable resources, Enzyme Microbiol Tech., 2000, 26: 87-107.
- Ross R P, Morgan S & Hill C, Preservation and fermentation: past, present and future. Int J Food Microbiol, 2002, 79: 3–16.
- Hati S, Mandal S & Prajapati JB, Novel Starters for Value Added Fermented Dairy Products, Curr Res Nutri Food Sci, 2013, 1: 83-91.
- Zotta T, Parente E & Ricciardi A, Viability staining and detection of metabolic activity of sourdough lactic acid bacteria under stress conditions, World J Microbiol and Biotech, 2009, 6: 1119–1124.
- Ali AA, Beneficial Role of Lactic Acid Bacteria in Food Preservation and Human Health, Res J Microbiolo, 2010, 5: 1213-1221.
- Wang C, Cui Y & Qu x, Mechanisms and improvement of acid resistance in lactic acid bacteria, Arch Microbiol, 2018, 2: 195-201.
- Casarotti SN, Paula AT & Reis JA, Lactic Acid Bacteria Antimicrobial Compounds: Characteristics and Applications, Food Eng Rev, 2012, 4:124
- Leroy F & Vuyst LD, Lactic acid bacteria as functional starter cultures for the food fermentation industry, Trend Food Sci Technol, 2004, 2: 67–78.
- Succi M, Tremonte P, Reale A, Sorrentino E, Grazia L, Pacifino B & Coppola R, Bile salt and acid tolerance of Lactobacillus rhamnosus strains isolates from Parmigiano Reggiano cheese, Fems Microbiol Lett, 2005, 244: 129-137.
- De Man JC, Rogosa M & Shurpe M, A medium for the cultivation of lactobacilli, Appl J Bacteriol, 1960, 23: 130-138.
- Kostinek M, Specht I, Edward VA, Schillinger U, Hertel C, Holzapfel WH & Franz CMAP, Diversity and technological properties of predominant lactic acid bacteria from fermented cassava used for the preparation of Gari, a traditional African food, Syst Appl Microbiol, 2005, 28: 527–540.
- Hoque MZF, Akter KM, Hossain MSM, Rahman MM, Billah & Islam KMD, Isolation, Identification and Analysis of Probiotic Properties of Lactobacillus Spp. From Selective Regional Yoghurts, World J. Dairy & Food Sci, 2010, 1: 39-42.
- Sumathi V. & Reetha, Screening of Lactic Acid Bacteria for Their Antimicrobial Activity against Pathogenic Bacteria, Int J Pharm Biolo Arch, 2012, 4: 802-808.
- Holt J G, Krieg N R, Sneath P H, Staley J T, Williams T & Hensyl W R, Bergey’s manual of Determinative Bacteriology, 9th ed, Williams and Willkins, Baltimore, Meryland. USA, 1994.
- Vos P D, Garrity G M, Jones D, Krieg N R, Ludwig W, Rainey F A, Schleifer K-H & Whitman W B, Bergey’s Manual of Systematic Bacteriology. 2nd ed. Vol. 3: The Firmicutes. Springer, New York, 2009.
- Lagacé L, Pitre M, Jacques M & Roy D, Identification of the Bacterial Community of Maple Sap by Using Amplified Ribosomal DNA (rDNA) Restriction Analysis and rDNA Sequencing, Appl Environ Microbiol, 2004, 70: 2052–2060.
- Altschul S F, Madden T L, Schäffer A A, Zhang J, Zhang Z, Miller W & Lipman D J, Gapped BLAST and PSI-BLAST: a new generation of protein database search programs, Nucleic Acids Res, 1997, 17: 389–402.
- Rahman S M K, Probiotic Properties Analysis of Isolated LacticAcid Bacteria from Buffalo Milk. Arch Clinical Microbiol, 2015, 7: 1-5.
- Koll P, Mandar R, Smidt I, Hutt P, Truusalu K, Mikeisaar R H, Shchepetova J, Krogh-Andersen K, Marcotte H, Hammarstrom l & Mickelsaar, Screening and evaluation of human intestinal lactobacilli for the development of novel gastrointestinal probiotics, Curr Microbiol, 2010, 61: 560-565.
- Liasi S A, Azmi T I, Hassan M D, Shuhaimi M, Rosfarizan M & Ariff A B, Antimicrobial activity and antibiotic sensitivity of three isolates of lactic acid bacteria from fermented fish product, Malaysian J Microbiol, 2009, 5: 33-37.
- Collins M D , Ropdriguez U, Ash C, Aguire M, Farrow, J A E, Martinez A, Philips B A & William A M, Phylogenetic analysis of genus Lactobacillus and related lactic acid bacteria as determined by reverse transcriptase sequences of 16S Rrna, FEMS Microbiol Lett, 1991, 77: 5-12.
- Elayaraja S, Annamalai N, Mayavu P & Balasubramanian T, Production, purification and characterization of bacteriocin from actobacillus murinus AU06 and its broad antibacterial spectrum, Asian Pac J Trop Biome, 2014, 4: 305-311.
- Mikkili I, John B D ,Chaitanya M & Kodali v p, Isolation and Characterization of Bacteriocin Producing Lactic Acid Bacteria from Curd, Int J of Chem Tech Research, 2015, 8: 388-396.
- Kumari A, Makeen K, Gory A P, Marrota F & Divya G C, Effect of bacteriocin produced from Lactobacillus lactis subsb. Lactis CCSUB 202 and mode of action Lactobacillus lactis HTCC 2028, Int j Probio, 2009; 4: 1-5.
- Todorov S D & Dicks L M T, Pediocin st18, an antilisterial bacteriocin produced by Pediocococcus pentosaceus ST18 isolated from boza, a traditional cereal beverage from Bulgaria, Process Biochem, 2005, 40: 365-370.
- Enan G, Abdel-Shafi S, Abdel-Haaliem M E F, & Negm S, Characterization of probiotic lactic acid bacteria to be used as starter and protective culture for dairy fermentations, Int J Probio Prebio, 2013, 8: 157-163.
- Reda F M, Detoxification of enterotoxigenic Bacillus cereus (JX455159) isolated from meat by a local strain of Lactobacillus plantarum (JX282192), Ann Microbiol, 2014, 64: 287–296.
- Enan G, Abo-El-Khair I A, Abdel-Shafi S, Al-Mohammadi A F, Evaluation of the use of Enterococcus faecium NM2 aprobiotic for inhibition of some urogenital pathogens, J Food Agri & Enviro, 2015, 13: 2-7.
- Banwo K, Sanni A & Tan H, Technological properites and probiotic potential of Enterococcus faecium strains isolated from cow milk, J Appl Microbiol, 2012, 114: 229-24.
- Enan G, AL-Mohammadi, AF, El-Didamony G, Abdel-Haliem M E F & Zakaria A, Antimicrobial activity of Enterococcus faecium NM2 Isolated from Urine: Purification, Characterization and Bactericidal action of Enterocin NM2, Asian J appli sci, 2014, 7: 621-634.
- Kumari A, Angmo K, Monika, Savitri & Bhalla C T, Probiotic Characterization Of Lactic Acid Bacteria Isolated From Fermented Foods And Partial Purification of its Bacteriocin, J Biotech, 2016, 5: 8-16.
- Enan G, El-Essawy A A , Uyttendaele M, & Debevere J, Antibacterial activity of Lactobacillus plantarum UG1 isolated from dry sausage: characterization, production and bactericidal action of plantaricin UG1, Int J Food Microbiol, 1996, 30: 189-215.
- Enan G , Abdellatif S H, Abdel-Shafi S, Ali A E & Ismaiel A A, Inhibitin of two Listeria strains in vitro and in situby nisin, onion and garlic juices separately and in combinations, Wulfenia J, 2018, 25: 194-231.
- Hoover D H, Microorganisms and their product in the preservation of food. In Lund, B.M.C. The microbilogical safty and quality of food aspen, Gaithers Prog, M.D, 2000, 251-276.
- Pundir K R, Kashyap N R S & Kaur k, Probiotic potential of lactic acid bacteria isolated from food samples: an in vitro study, Appl J Pharm Sci, 2013, 3: 85-93.
- Forhad M H, Rahman K S M, Rahman R S MD, Saikot K F & Biswas C K, Probiotic Properties Analysis of Isolated Lactic Acid Bacteria from Buffalo Mil, Arch Clini Microbiol, 2015, 7:1-5.
- Prabhurajeshwar C & Chandrakanth, K R, Probiotic potential of lactobacilli with antagonistic activity against pathogenic strains: An in vivo validation for the production of inhibitory substance, J biomed, 2017, 40: 270-283.
- Strompfova V & Laukova A, In vitro study on bacteriocin production of enterococci associated with chickens, Anaerobe, 2017, 13: 228–237.
- Van den Berghe E, De Winte T & De Vuyst L, Enterocin A production by Enterococcus faecium FAIR-E 406 is characterised by a temperature- and pH-dependent switch-off mechanism when growth is limited due to nutrient depletion, Int J Food Microbiol, 2006, 107: 159–170.
- Ibourahema C, Dauphin R D, Jacqueline D & Thonart P, Characterization of lactic acid bacteria isolated from poultry farms in Senegal, Afr J Biotech, 2008, 7: 2006–2012.
- Unal N, Askar S & Yildirim M, Antibiotic resistance profile of Enterococcus faecium and Enterococcus faecalis isolated from broiler cloaalc samples, Turk J Vet Anim Sci, 2017, 41: 199-203.
- Melo T A , dos Santos T F, Pereira L R, Passos H M, Rezende R P & Romano C, Functional Profile Evaluation of Lactobacillus fermentum TCUESC01: A New Potential Probiotic Strain Isolated during Cocoa Fermentation, BioMed Res Int, 2017, 2017:1-7.
- Nawaz M, Wang J & Zhou A, Characterization and transfer of antibiotic resistance in lactic acid bacteria from fermented food products, Curr Microbiol, 2011, 62: 1081–1089.
- Kim D H & Austin B, Characterization of probiotic carnobacteria isolated from rainbow trout (Oncorhynchus mykiss) intestine, Lett Appl Microbiol, 2008, 3: 141-147.
- Priscillia Y H C, Mendez-Romero A, Hernandez-Mendoz E, Acedo-Felix, Aaron F, Gonzalez-Cordova, & Belinda Vallejo-Cordoba, Antimicrobial activity and partial characterization of bacteriocin like inhibitory substances produced by lactobacillus spp. Isolated from artisanal Mexican cheese, J Dairy Sci, 2015, 98:1-9.
- Ohenhen R E, Isibor J O, Emonfonmwan G & Enablele S A, Effects of pH and storage Temperatures on Antibacterial Activity of Bacteriocin Isolated from OGI, Brit Microbiol Res J, 2015, 3: 1-9.
- Park H S, Itoh K & Fujisaw, Characteristics and identiûcation of enterocins produced by Enterococcus faecium JCM 5804T, J Appl Microbiol, 2003, 95: 294–300.
- Maria M, & Janakiraman S, Detection of heat stable bacteriocin from Lactobacillus acidophilus NCIM5426 by liquid chromatography/ mass spectrometry, Indian J Sci Tech, 2012: 5: 2325-2332.
- Ghnbari M, Jami M, Kneifel W & Domig K J, Antimicrobial activity and partial characterization of bacteriocins produced by lactobacilli isolated from sturgeon fish, Food Contr, 2013, 32: 379-385.
- Hor Y Y & Liong T M, Use of extracellular extracts of lactic acid bacteria and bifidobacteria for the inhibition of dermatological pathogen Staphylococcus aureus, Dermatologica Sinica, 2014, 32:141-147.
- Bajpai K V, Ho Han J, Rather A I , Majumder R, Jun-Nam G, ChanSeo P, Lim J, Paek K W & Ha Park y, Characterization of lactic acid bacterium Pediococcus pentosaceus 4I1 from fresh water fish Zacco koreanus and its antibacterial mode of action, PeerJ Preprints, 2016, 4: e2121v1 https://doi.org/10. 7287/ peerj. preprints.2121v1
© The Author(s) 2018. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.