ISSN: 0973-7510
E-ISSN: 2581-690X
Halophilic bacteria which adapted to live under extreme conditions are worthy sources of enzymes in industrial microbiology and bioremediation. This study aimed to isolate halophilic bacteria which are able to use crude oil as the only carbon source. Fifteen soil samples were collected from Alborz oil field near city Qom, Iran. Liquid mediums containing soil samples, 5,10 and 15% of artificial sea water (ASW) and crude oil as the sole carbon source incubated at 37°C for one week. Two gram negative moderately halophilic bacteria designated BM1 and BM2 were isolated. Supplementary molecular analysis using 16S rDNA sequencing revealed that they belong to Halomonas and Ochrobactrum genera, respectively. Biochemical tests showed they are both positive for extracellular lipase activity. Considering to cleanup the majority of oil contamination at Alborz oil field, further efforts should be focused on research to understand the degradation mechanism and identification of the enzymes involved in this process.
Biodegradation; crude oil; Halomonas; Iran; Ochrobactrum.
Hypersaline environments are distributed in a variety of terrestrial and aquatic ecosystems1. Halophiles, the microorganisms dwelling at high salt concentrations are members of Archaea, Bacteria, and Eukarya domains. They have evolved properties to adopt high osmotic pressure, pH and salinity2. In recent years, a verity of industrial, biotechnological and commercial applications has been described for halophiles3. A common propose is finding new strains to design functional consortium to cleanup greater amounts of hydrocarbon contamination4. The efficiency of bioremediation of oil spill ranged from 1 to 100 percent depending on the type of halophilic microorganisms5.
Many studies have been planned to cleanup the polluted sites after the hydrocarbon contamination6. A variety number of expensive technologies like soil washing and filling, land farming and soil vapor extraction are used for contaminated water and soil remediation although most of them lead to incomplete decomposition of hydrocarbons7. Detoxification or removing of complex contaminant using microorganisms are the main approaches to oil spill bioremediation8; the growth and proliferation of oil degrader bacteria stimulated by adding growth-promoting substrates to the polluted sites (biostimulation) and inserting them to the contaminated locations (bioaugmentation)7. With the aim of microbial bioremediation, screening and introducing microorganisms which are able to degrade hydrocarbons efficiently has found a priority in recent industrial microbiology studies9. Many hypersaline environments belong to oil rich sites and the inhibitory effects of salt on microbial growth is the serious obstacle in bioremediation procedures in these area. As a result, salt resistant microorganisms are the incomparable source to cleanup petroleum contaminated soil10.
The major salt sequence in the central plateau of Iran (between the Alborz mountains in the north and the Zagros mountains in the south) is known as Kuh-e-namak (mountain of salt in Persian) near the city of Qom which formed of thick beds of KCl and NaCl. Alborz oil field situated 15 kilometers to the east of city Qom, Iran, had been discovered in the early 1960s11. The well number 5 located on the crest of the Alborz Anticline blew out through salt after drill had penetrated five centimeters into hard rock at a depth of 2600 meters. Oil and gas shot about 50 meters in the air and sprinkled the countryside for many kilometers. In addition to closing beside road and railway, a lake of oil with considerable branches flowing into the surrounding salty lands and plains caused an emergency state in the city Qom12. Despite the partial control after 25 days, flowing oil last for 80 days and it was estimated 80,000 barrels per day13. With the passage of nearly six decades, only a trace of contamination remains. Considering the slow rate of natural degradation of oil spill, the halophilic microorganisms inhabiting the soil would play a key role in decontaminating the area. In accordance with this view, screening and characterization of halophilic microorganisms which were able to breakdown oil contamination at location around well number five had been carried out in this study.
Saline soil samples were collected from fifteen different zones of Kuh Sefid area near to Alborz oil field located 15 kilometers east of city Qom, Iran, (longitude 50º 88′ E and latitude
34º 72′ N) on 24 May 2013. All samples were put in sterile plastic bags and transported to the laboratory for further analysis. Samples were taken from the surface and various depths (5-30 cm) with maximum distance of 10 km.
The medium containing 5, 10 and 15% of artificial sea water with the following composition (g/L): 175 g NaCl, 20 g MgCl2.6H2O, 5 g K2SO4, 0.1 g CaCl2.2H2O, adjusted pH to 7–7.5, were amended with 1% filter sterilized crude oil (v/v). 1 g of each soil sample was added into each medium and incubated for a week by continuous shaking with 120 rpm at 37°C. The range of NaCl concentration for growth was tested in solid medium with NaCl concentration between 5% to 15%. To determine the optimal temperature and pH for growth, broth cultures were incubated at 4-37°C at intervals of 5°C and at pH 6–8 at intervals of 0.5 pH units.
Cell morphology and spore formation were examined by using light microscopy (model BH 2; Olympus) and phase-contrast microscopy (Zeiss), respectively. Gram staining was carried out according to the method described by Doetsch et al. (1981)14. Catalase production was determined by adding 3% (v/v) H2O2 and observing its hydrolysis and the consequent gas formation15. The oxidase activity was detected according to Kovacs (1956)16. Tests for use of glucose and sucrose as sole carbon sources at a concentration of 1 g were performed in 5 ml liquid media in which yeast extract was reduced to 0.1 g, amended with 0.5 g NH4Cl. Extracellular amylase activity was assayed using 1% starch agar medium (Merck) following the protocols of Gonzalez et al. (1978)17. To detect the hydrolysis of starch, I2–0.6% KI solution or Lugol’s iodine was used. After incubation at 37°C for 7 days, presence of dark blue, purple, or black color represented amylolytic activity. The urease activities were checked as recommended by Smibert & Krieg (1994)18. Lipase activity was detected using sterilized basal nutrient agar medium containing 2.5% olive oil (w/v) at 60°C. Then, 0.001% rhodamine B was added to homogenized mixture at 50°C and poured. Observation of shinny colonies under UV irradiation after incubation of bacteria at 37°C for 7 days indicated lipolytic activity17.
The 16S rRNA gene of the isolates were amplified using the universal primers 8F (5´-AGAGTTTGATCCTGGCTCAG-3´) and 1492R (5´-GGTTACCTTGTTACGACTT-3´). PCR mixtures of 50 µL contained 17 µL dH2O, 1 µL primers, 1 µL dNTP, 0.75 µL MgCl2 (50mM), 25 µL PCR amplification buffer (1X), 2 U Taq DNA polymerase, 5 µL template DNA. The amplification was done by initial denaturation 95°C for 5 min and {95°C 1 min, 56°C 1 min, 72°C 1.5 min (30 cycles)} and 72°C for 10 min (final cycle). PCR products of 16S rRNA gene were sequenced in both directions using an automated sequencer by bioron (Korea). Multiple sequence alignment was performed using BLAST and Ribosomal Database Project (RDP) with the published sequences of closely related bacteria in the GenBank database of the National Center for Biotechnology Information. Phylogenetic tree was constructed by neighbor-joining and UPGMA method with MEGA719-20. Bootstraping analysis21 was used to evaluate the tree topology by performing 1000 resampling. The 16S rRNA gene sequences of the strains have been deposited in the GenBank database.
Using crude oil as sole carbon source, only two halophilic oil-degrading bacterial strains designated BM1 and BM2 were isolated from 15 samples. Both were gram negative rods which produced similar cream colonies. Table 1 lists the biochemical features of the two isolates. BM1 colonies were observed in all mediums containing 5 to 15 percent of ASW in less than an overnight. While BM2 colonies were appeared after one week in mediums with 5 and 10 percent of ASW concentration.
Phenotypic characteristics of two isolates from Alborz oil field, Qom, Iran
BM1 |
BM2 |
|
|---|---|---|
Color |
Cream |
Cream |
Gram staining |
– |
– |
Shape |
Rod |
Short rod |
Spore forming |
– |
– |
Optimum pH |
7.5 |
7.5 |
Optimum temperature |
37°C |
37°C |
Optimum salinity |
10% (ASW) |
10% (ASW) |
Glucose |
+ |
+ |
Sucrose |
+ |
+ |
Catalase |
– |
+ |
Oxidase |
– |
+ |
Urease |
– |
– |
Amylase |
– |
+ |
Lipase |
+ |
+ |
Phylogenetic analysis of 16S rDNA sequences of the isolates revealed two completely different taxonomic groups. With bootstrap values (22-100%), BM1 belongs to class Gammaproteobacteria, genus Halomonas and BM2 was a member of class Alphaproteobacteria, genus Ochrobactrum. Figure 1 shows the phylogenetic tree of the isolates. Numerous studies in this regard have been conducted in recent decades. Similar study in Gulf of Mexico Beach Sands identified members of the Gammaproteobacteria (Alcanivorax, Marinobacter) and Alphaproteobacteria (Rhodobacteraceae) as fundamental players in petroleum degradation22.
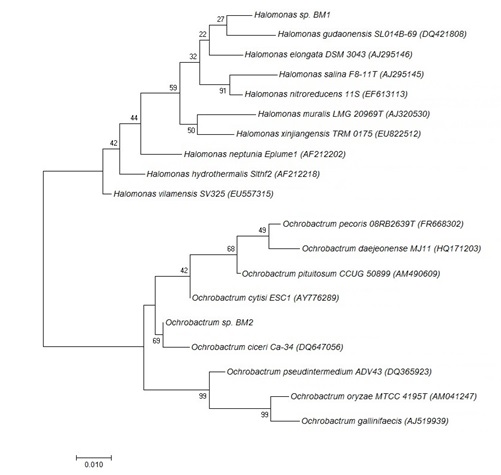
Fig. 1. Phylogenetic tree showing the position of the halophilic isolates, based on the partial 16S rDNA sequence comparison, obtained by the neighbor-joining method. The accession numbers for the reference strains are included in brackets. Bootstrap values are indicated on the branches.
Although a great number of halophilic and halotolerant microorganisms have been found to be hydrocarbon degrader, most of them affiliated to the class Gammaproteobacteria23. To exemplify, characterization of polycyclic aromatic hydrocarbon (PAH)-degrading bacteria from sediments of the Bizerte lagoon, Tunisia, led to screening 100 bacteria which three-quarters of them were affiliated to the Gammaproteobacteria24. A Halomonas strain obtained from saline wastewater samples collected in a petroleum rig was able to use crude oil, diesel and lubricant oil as the only carbon source25. Halomonas sp. ANT-3b isolated from seawater in Antarctica used diesel fuel as a sole carbon and energy sources and was able to break down n-hexadecane to nanodroplets 26. Increasingly, Halomonas was among 25 isolated strains from Persian Gulf and the Caspian Sea based on a high growth rate on crude oil and on hydrocarbon degradation ability27. Aromatic compounds were degraded as carbon and energy sources by Halomonas campisalis in high pH and salt systems28. Besides, Halomonas shengliensis sp. SL014B-85T isolated from saline soil Shengli oilfield, China, showed to be a crude oil degrader29. Equally, a mixture of Halomonas sp. and Alcanivorax sp. strains isolated from the spilled oil in Japan Sea displayed a high rate degradation of heavy oil30.
In spite of the fact that the pioneer oil-degrader bacteria are mostly Gammaproteobacteria, Betaproteobacteria and Actinobacteria, in present study we found a new member of Alphaproteobacteria which was able to use crude oil as the sole source of carbon9. In harmony with our results, an Ochrobactrum intermedium was obtained from soils contaminated by transformer oil31. Also, a 79% crude oil degradation rate in saline solution was observed by microbial consortium including Ochrobactrum sp. N132. Further to this, in a consortium composed of Ochrobactrum sp., Rhodococcus strain NTU-1 and Bacillus fusiformis L-1 which was isolated from an oil-contaminated field, the degradation of the alkane was stimulated by Ochrobactrum sp. 33. Ochrobactrum anthropi strain AD2 was identified as an efficient oil degrader and isolated from the waste water treatment plant of an oil refinery34. In contrast, an Ochromonas sp. isolated from a diesel oil contaminated soil which was not an oil degrader showed the inhibitory effect on the degradation of diesel oil by P. aeruginosa strain WatG35.
Our isolates were positive for extracellular lipase production which is one of the key enzymes in oil degradation process36. As some studies reported, Halomonas bacteria isolated from hypersaline environments showed hydrolase producing ability particularly amylase and lipase37-39. However, in this study Halomonas sp. BM1 was negative for amylase activity. The Ochrobactrum isolates from saline sediment of mangrove areas of São Paulo State, Brazil, showed extracellular lipase activity but was negative for amylase production40. On the contrary, in a screening study, Ochrobactrum anthropi BMO-111 had not expressed any extracellular activities such as amylase and lipase production41.
Two distant bacteria belonging to genus halomonas (Gammaproteobacteria) and genus ochrobactrum (Alphaproteobacteria) were isolated. Both were able to use crude oil as the sole source of carbon. Halomonas sp. BM1 showed speed growth rate on mediums containing 5, 10 and 15% artificial sea water. Ochrobactrum sp. BM2 appeared on mediums with 5 and 10% ASW concentration after one week. They were able to produce extracellular lipase which is one of the key enzymes in petroleum degradation. Further study on hydrocarbons degradation processes and lipase structure make them potentially useful for application in bioremediation of oil contaminated area.
- S.A. Cantrell, L. Casillas-Martínez and M. Molina. Characterization of fungi from hypersaline environments of solar salterns using morphological and molecular techniques. Mycological Research, 2006;110(8): 962-70.
- A. Hedi, B. Essghaier, J-L. Cayol, M-L. Fardeau and N. Sadfi. Prokaryotic biodiversity of halophilic microorganisms isolated from Sehline Sebkha Salt Lake (Tunisia). African Journal of Microbiology Research, 2014; 8(4): 355-67.
- L.R. Brown. Microbial enhanced oil recovery (MEOR). Current Opinion in Microbiology, 2010; 13(3): 316-20.
- D. Zhaoa, Ch. Liua, L. Liub, Y. Zhanga, Q. Liua and W-M. Wuc. Selection of functional consortium for crude oil-contaminated soil remediation. International Biodeterioration & Biodegradation, 2011; 65(8): 1244–48.
- A. Moneke and C. Nwangwu. Studies on the bioutilization of some petroleum hydrocarbons by single and mixed cultures of some bacterial species. African Journal of Microbiology Research, 2011; 5(2): 1457-66.
- G.S. Sayler and S. Ripp. Field applications of genetically engineered microorganisms for bioremediation processes. Current Opinion in Biotechnology, 2000; 11(3): 286-9.
- N. Das and P. Chandran. Microbial degradation of petroleum hydrocarbon contaminants – an overview. Biotechnology Research International, 2011; 13 pages.
- G.M. Gadd. Metals, minerals and microbes: geomicrobiology and bioremediation. Microbiology, 2010; 156(3): 609–43.
- YC. Gao, SH. Guo, JN. Wang, D. Li, H. Wang H, DH. Zeng. Effects of different remediation treatments on crude oil contaminated saline soil. Chemosphere, 2104; 117: 486-93.
- A. Oren, P. Gurevich, M. Azachi, Y. Henis. Microbial degradation of pollutants at high salt concentrations. Biodegradation, 1992; 3(2): 387–98.
- XU. Guoqiang, Z. Shaonan, LI. Zhongdong and S. Lailiang. Carbonate Sequence Stratigraphy of a Back-Arc Basin: A Case Study of the Qom Formation in the Kashan Area, Central Iran. Acta Geologica Sinica, 2007; 81(3): 488-500.
- RL. Rosenberg. Qum-1956: A Misadventure in Iranian Oil. Business History Review, 1975; 49(1): 81-104.
- I. Abaie, H. J. Ansari, A. Badakhshan and A. Jaafari. History and development of the Alborz and Sarajeh fields of Central Iran. 6th World Petroleum Congress, 1963, pp. 697-713.
- R. N. Doetsch. Determinative methods of light microscopy. In Manual of Methods for General Bacteriology, pp. 21–33. Edited by P. Gerhardt, R. G. E. Murray, R. N. Costilow, E. W. Nester, W. A. Wood, N. R. Krieg & G. B. Philips. Washington, DC: American Society for Microbiology, 1981.
- L. J. Bradshaw. Laboratory Microbiology, 4th edn. Fort Worth, TX: Saunders College Publishing, 1992.
- N. Kovacs. Identiûcation of Pseudomonas pyocyanea by the oxidase reaction, Nature, 1956: 178(4535): 703–4.
- C. Gonzalez, C. Gutierrez, and C. Ramirez. Halobacterium vallismortis sp. nov., an amylolytic and carbohydrate-metabolising extremely halophilic bacterium. Canadian Journal of Microbiology, 1978; 24(6): 710–5.
- R. M. Smibert and N. R. Krieg. Phenotypic characterization in Methods for General and Molecular Bacteriology, pp. 607–654. Edited by P. Gerhardt, R. G. E. Murray, W. A. Wood & N. R. Krieg. Washington, DC: American Society for Microbiology, 1994.
- K. Tamura, G. Stecher, D. Peterson, A. Filipski and S. Kumar. MEGA6: molecular evolutionary genetics analysis version 60, Molecular Biology and Evolution, 2013; 30(12): 2725-9.
- N. Saitou and M. Nei. The neighbor joining method: a new method for reconstructing phylogenetic trees, Molecular Biology and Evolution, 1987; 4(4): 406-25.
- J. Felsenstein. Confidence limits on phylogenies: an approach using the bootstrap. Evolution, 1985; 39(4): 783–91.
- J. E. Kostka, O. Prakash, W. A. Overholt, S. J. Green, G. Freyer, A. Canion, et al. Hydrocarbon-Degrading Bacteria and the Bacterial Community Response in Gulf of Mexico Beach Sands Impacted by the Deepwater Horizon Oil Spill. Applied and Environmental Microbiology, 2011; 77(22): 7962-74.
- L. F. Martins and R. S. Peixoto. Biodegradation of petroleum hydrocarbons in hypersaline environments. Brazilian Journal of Microbiology, 2012; 43(3): 865-72.
- O. Ben Said, M.S. Goñi-Urriza, M. El Bour, M. Dellali, P. Aissa and R. Duran. Characterization of aerobic polycyclic aromatic hydrocarbon-degrading bacteria from Bizerte lagoon sediments, Tunisia. Journal of Applied Microbiology, 2008; 104(4): 987-97.
- S. Mnif, M. Chamkha and S. Sayadi. Isolation and characterization of Halomonas sp. strain C2SS100, a hydrocarbon-degrading bacterium under hypersaline conditions. Journal of Applied Microbiology, 2009; 107(3): 785-94.
- M. Pepi, A. Cesàro, G. Liut and F. Baldi. An antarctic psychrotrophic bacterium Halomonas sp. ANT-3b, growing on n-hexadecane, produces a new emulsifying glycolipid. FEMS Microbiology Ecology, 2005; 53(1): 157-66.
- M. Hassanshahian, G. Emtiazi and S. Cappello. Isolation and characterization of crude-oil-degrading bacteria from the Persian Gulf and the Caspian Sea. Marine Pollution Bulletin, 2012; 64(1): 7-12.
- CS. Oie, CE. Albaugh and BM. Peyton. Benzoate and salicylate degradation by Halomonas campisalis, an alkaliphilic and moderately halophilic microorganism. Water Research, 2007; 41(6): 1235-42.
- YN. Wang, H. Cai, CQ. Chi, AH. Lu, XG. Lin, ZF. Jiang, et al. Halomonas shengliensis sp. nov., a moderately halophilic, denitrifying, crude-oil-utilizing bacterium. International Journal of Systematic and Evolution Microbiology, 2007; 57(6): 1222-6.
- S. Nakamuraa, Y. Sakamotoa, M. Ishiyamaa, D. Tanakaa, K. Kuniib, K. Kubob, et al. Characterization of two oil-degrading bacterial groups in the Nakhodka oil spill. International Biodeterioration and Biodegradation, 2007; 60(3): 202-7.
- J. A. Z.E. Liz, J. J. Roblero, J. Z. D. de la Serna, A. V. P. de León and C. H. Rodríguez. Degradation of polychlorinated biphenyl (PCB) by a consortium obtained from a contaminated soil composed of Brevibacterium, Pandoraea and Ochrobactrum. World Journal of Microbiology and Biotechnology, 2009; 25(1): 165-70.
- MT. Bao, LN. Wang, PY. Sun, LX. Cao, J. Zou and YM. Li. Biodegradation of crude oil using an efficient microbial consortium in a simulated marine environment. Marine Pollution Bulletin, 2012; 64(6): 1177-85.
- LA. Sayavedra-Soto, WN. Chang, TK. Lin, CL. Ho and HS. Liu. Alkane Utilization by Rhodococcus Strain NTU-1 Alone and in Its Natural Association with Bacillus fusiformis L-1 and Ochrobactrum sp. Biotechnology Progress, 2006; 22(5): 1368-73.
- C. Calvo, G. A. Silva-Castro, I. Uad, C. García Fandiño, J. Laguna and J. González-López. Efficiency of the EPS emulsifier produced by Ochrobactrum anthropi in different hydrocarbon bioremediation assays. Journal of Industrial Microbiology & Biotechnology, 2008; 35(11): 1493-501.
- A. Ueno, Y. Ito, I. Yumoto and H. Okuyama. Isolation and characterization of bacteria from soil contaminated with diesel oil and the possible use of these in autochthonous bioaugmentation. World Journal of Microbiology and Biotechnology, 2007; 23(12): 1739-45.
- R. Margesin, A. Zimmerbauer and F. Schinner. Soil lipase activity – a useful indicator of oil biodegradation. Biotechnology Techniques, 1999; 13(12): 859–63.
- A. Ventosa, J.J. Nieto and A. Oren. “Biology of moderately halophilic aerobic bacteria”. Microbiology and Molecular Biology Reviews, 1998; 62(2): 504-44.
- H. Baati, R. Amdouni, N. Gharsallah, A. Sghir and E. Ammar. Isolation and characterization of moderately halophilic bacteria from Tunisian solar saltern, Current Microbiology, 2010; 60(3): 157-161.
- C. Sánchez-Porro, S. Martín, E. Mellado and A. Ventosa. Diversity of moderately halophilic bacteria producing extracellular hydrolytic enzymes, Journal of Applied Microbiology, 2003; 94(2): 295-300.
- RA. Castro, MC. Quecine, PT. Lacava, BD. Batista, DM. Luvizotto, J. Marcon, et al. Isolation and enzyme bioprospection of endophytic bacteria associated with plants of Brazilian mangrove ecosystem. SpringerPlus. 2014; 3(1): 382.
- K. Sowndhararajan, S. Marimuthu and S. Manian. Biocontrol potential of phylloplane bacterium Ochrobactrum anthropi BMO-111 against blister blight disease of tea. Journal of Applied Microbiology, 2013; 114(1): 209-18.
© The Author(s) 2017. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


