ISSN: 0973-7510
E-ISSN: 2581-690X
Biosurfactant obtained from microorganisms isolated from different locations presents an alternative source to chemical surfactant, due to its structural and functional similarity, as well as its biodegradability properties. The goal of this work was to isolate biosurfactant-producing bacterial strains in seven sites with oil-contaminated soils, in district Lucknow, Uttar, India. Drop collapse assay, oil displacement method, emulsification activity and haemolytic assay were used to screen the total of 13 distinct colonies obtained from this study. Isolates GAR-1, TP-2, PET-1 & PET-3 were screened and evaluated to be more potent for biosurfactant production. The molecular identification of GAR-1, TP-2, PET-1 & PET-3 isolates were identified by 16S rRNA sequencing and characterized by FT-IR analysis. A total of 13 bacteria were isolated in the oil-contaminated soils, these four strains with high biosurfactant-production, identified as Bacillus paralicheniformis HRA-IU, B. velezensis NWR-20, Bacillus sp. HUR-IU and P. aeruginosa HUR-IA, respectively. The present study confirms that isolated microorganisms have biosurfactant property and screens the most potent biosurfactant (rhamnolipid) producing strain which is Bacillus sp. HUR-IU.
Biosurfactant, Drop Collapse Assay, Emulsification, Oil-contaminated Soils, Isolated Microorganisms
Disasters like oil spills can have detrimental effects on the environment, the economy, and society. They are the discharge of refined petroleum products or crude oil from offshore platforms, rigs, wells, and tankers. While they can happen on land, marine environments are where these leaks happen most frequently. They can be costly owing to the loss of oil and the ecosystems.1 Tanker crashes in 2022 caused the environment to lose around 15,000 tons of oil. Roughly 14,000 tons of this is present in the three major events (ITOPF, 2022).2 Low molecular weight hydrocarbons are volatilized and polar components are dissolved in water as oil spreads in the environment.3 Despite their low solubility, oil hydrocarbons are mostly found in the water or adhere to soil particles. It is essential to develop cost-effective and high-efficiency remediation methods.4
Often, synthetic surfactants are employed to minimize oil spills by spreading the oil and expediting its mineralization. Research indicates that surfactants can increase the surface area of hydrophobic pollutants in soil or water, leading to increased aqueous solubility and microbial breakdown.5 Chemical surfactants are toxic and do not break down easily, so researchers are looking for better alternatives. Despite their origin in microorganisms, biosurfactants exhibit numerous characteristics that are common with chemical surfactant. The advantages of their biodegradability, biocompatibility, low toxicity, and excellent substrate specificity over chemical surfactants are evident. Biosurfactants such as surfactin and rhamnolipids from Bacillus and Pseudomonas species have been proven to be effective in eliminating contaminants from contaminated areas.6 Various industries, including pharmaceuticals and petroleum products, as well as agricultural techniques like agriculture and food processing use biosurfactants for their wide range of applications. In addition, they can be used to increase well oil extraction, reduce the viscosity of heavy oils, clean oil storage tanks, increase pipeline flow and even stabilize fuel water oil emulsions.7
Although there has been more research on producing biosurfactants, industrial scale production has not yet been accomplished because of high production costs and limited yield. This makes it necessary to isolate new, highly productive microorganisms that produce biosurfactants.8 Hence, the objectives of present study are to: (i) isolate microorganisms which produce biosurfactant from oil contaminated sites; (ii) characterize the surface tension properties of their metabolites; (iii) Molecular basis Identification of biosurfactant producing microorganisms (iv) Characterization of produced microbial biosurfactant.
Experiment sampling
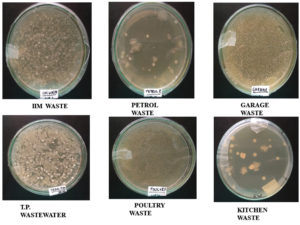
The collection of soil samples were done from various oil-contaminated waste sites (Table 1) in Lucknow, Uttar Pradesh, India by digging the soil up to 2-3 inches. They were kept in air-tight sterile polythene zip-lock packets and acclimatized in nutrient medium for future purpose.
Table (1):
Geographic coordinates of oil-contaminated sites
No. |
Oil-Contaminated Site |
Geographic Coordinates |
|---|---|---|
1 |
IIM dump site |
26.8981° N, 80.8715° E |
2 |
Poultry waste site |
27.01° N, 81.039° E |
3 |
Petrochemical waste site |
26.5439° N, 80.5644° E |
4 |
Kitchen waste site |
26.55°N, 80.5746°E |
5 |
Automobile garage waste site |
26.5438° N, 80.5644° E |
6 |
Tedhipuliya wastewater site |
26.9170° N, 80.9543° E |
7 |
Two-wheeler garage waste site |
26.8702° N, 80.9042° E |
Isolation and selection of biosurfactant producing bacterial strains
Oil-contaminated soil was taken as a bacterial inoculum. 10 grams of oil contaminated soil and 90 mL of deionized water was mixed in a shaker at 120 rpm for 30 minutes and then kept still for 30 minutes.9
Screening of Biosurfactant producing bacterial strains
The following four qualitative methods were used to screen the bacterial strains which produce biosurfactant:
Oil spreading assay
This technique was performed according to the method described by Youssef et al.,10 and Plaza et al.11 Briefly, distilled water (10 ml) was added to the petri dishes followed by addition of crude oil (20 ml) to the water surface. Then 2 ml of supernatant was dropped with the help of pipette on to the crude oil. Then the diameter of clear zone formed on the oil surface was compared to distilled water (2 ml) as negative control.
Drop collapse method
The method of collapsing the drop was developed by Jain et al. The method is based on the destabilization of liquid droplets by surfactants. Therefore, drops of a cell suspension or of culture supernatant are placed on an oil droplet on glass surface. The polar water molecules are repelled from the hydrophobic surface and the drops remain stable if the liquid does not contain surfactants. If the surfactants are present in the fluid, these drops will be dispersed or even collapsed due to a decrease of force or interfacial tension within the water droplet and its hydrophobic surface. The stability of drops is determined by the concentration of surfactants and their correlation to surface and interfacial tension.12
Haemolytic Activity
Supernatant extracted from isolated strains culture was screened on Petri plates containing drops of human blood on water. Haemolytic activity was checked with the occurrence of a formation of define clear zone. This protocol is self-trial procedure so it has no reference.
Emulsification test
Emulsification activity was measured according to the method of Cooper et al. To 2 ml of culture supernatant or biosurfactant crude extract, 2 ml of each crude oil i.e., kitchen waste oil and automobile waste oil respectively, were added and vortexed at high speed for 2 min. The mixture was allowed to stand for 24 h prior to measurement.13
The emulsification activity is defined as the height of the emulsion layer divided by the total height and expressed as percentage.14
E24% = Height of emulsified layer /Height of aqueous layer × 100
Where, E24 is emulsification index
The emulsification stability can be defined as the ability of emulsion to resist changes to its structure over a defined time period.
ES % = Remained volume of emulsion layer/ Initial volume of emulsion layer × 100
Where, ES is emulsification stability
The molecular identification of biosurfactant producing isolates
Extraction of bacterial genomic DNA
Selected bacterial isolates were maintained in Bushnell Hass mineral salt broth supplemented with the glucose (1%) for 24 hrs at 37°C. For DNA extraction, precipitation and purification, HiPer Bacterial Genomic DNA Extraction Kit (Himedia Laboratory Pvt. Ltd. Mumbai) was used. Electrophoreses of isolated DNA was done in 0.8 % Agarose gel and visualized on a Gel Documentation System in Genie, Bangalore.
Identification of isolates by 16S rRNA sequencing method
The selected isolates were sent to BioKart Lab, Karnataka, India for 16S rRNA sequencing. The sequence thus obtain was subjected to similarity search using nucleotide BLAST 2.2.31 (blastn) software available at NCBI,15 and deposited in GenBank NCBI. The identification was made by evaluating total score, query coverage, maximum score, E-value and percentage.16
Phylogenetic position of isolate with respect to their closely related sequence
The closely related sequence obtain from blast were downloaded and multiple sequence alignment were conducted using Clustal Omega tool available at EMBL-EBI. Phylogenetic tree were developed to determine taxonomic position of the bacterial isolates using Neighbor Joining algorithm.17
Structural characterization
Biosurfactants produced from Bacillus sp. HUR-IU was characterized by using Alpha II (210966) – Sample Compartment *36F175832D -OPUS Version 8.5(SP1) Build: 8, 7, 10 20200710 with wavenumber range 400-4000 cm-1. To determine the functional groups and the chemical bonds, samples were prepared for infrared analysis by mixing approximately 1 mg of crude biosurfactant with 100 mg of KBr and pressing the mixture into the form of a pellet at 134MPa for 2–3 min to obtain transparent pellets. The IR spectrum of the pellet was collected from 400 to 4000 wavenumbers (cm-1) by using a FTIR spectrometer with sample dispersed in the pellets of KBr.
Isolation and selection of biosurfactant producing strains
Spreading & streaking
Spreading-One milliliter of each supernatant was spread on the surface of sterile nutrient agar medium plates and incubated at 37°C for 24 h. Further microbial colonies appeared in nutrient agar medium were used for the screening as shown in Figure 1.18
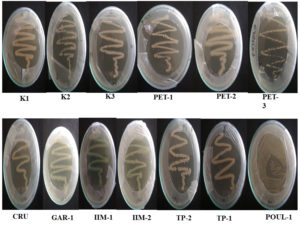
Streaking- Single bacterial colony was maintained in nutrient agar medium; by picking up by sterilized inoculating needle, incubated at 37°C for 24 h (Figure 2).19,20
Screening of Biosurfactant producing bacterial strains
The biosurfactant producing bacterial strains were screened by following qualitative methods:
Oil-spreading method
Out of 13 strains, 4 strains shows maximum clear zone in oil spreading method. This result reveals that these strains show maximum surfactant properties and can go for biosurfactant production (Figure 3, 4).
Drop collapse assay
All the strains show the positive result in drop collapse assay as shown in Figure 5. The drop collapse assay is indicative of surface wetting activity. This means that the strains exhibits the property of biosurfactant and can be explored further to utilize their potential in bioremediation, therapeutics and agriculture.
Haemolytic activity
All the strains show the positive result in haemolytic activity method as shown in Figure 6. This indicates that these strains are good producers of biosurfactant and can be used in pharmaceutical industry for inhibition of fibrin clot.
Emulsification activity of isolated strains
Figure 7 shows emulsification activity of isolated microbial strains. Emulsification index aids in quantitative screening of biosurfactant producers. This test predicts a higher hydrophilic-lipophilic balance value in the formation of the emulsion. The emulsification index measures how efficiently a biosurfactant strengthens contact between oil and water. A high emulsification index value means there is also high contact between oil and water.
Biosurfactant production by isolated strains
Tentative identification of 13 isolated bacterial strains was done on the basis of biosurfactant activity. Preliminary identification of isolated bacteria species were found to be showing emulsification activity after 24 h (E24 %), oil spreading assay, oil drop collapse assay, haemolytic assay and stability.
Out of 13 isolated strains, 4 strains show positive result for biosurfactant production on the basis of EI, oil spreading assay & emulsification stability as shown in Table 2. Further, these 4 strains i.e., PET-1, PET-3, GAR-1 & TP-2 were identified by 16S RNA.
Table (2):
Different isolated bacterial strains and their qualitative analysis
| Bacterial isolates | Oil Displacement Assay | Drop Collapse Assay | Haemolytic assay | Emulsification Activity (E24%) | Emulsification Stability (in %) | Stability | |||
|---|---|---|---|---|---|---|---|---|---|
| Kitchen oil | Mobile oil | Petrol | Terpene | ||||||
| Petrol | Terpene | ||||||||
| PET -1 | ++++ | +++ | ++ | ++ | 66 | 93.33 | 92.3 | 88.23 | Highly stable |
| PET -3 | ++ | +++ | + | + | 52 | 71.88 | 25 | 16.66 | Stable |
| GAR-1 | ++ | + | + | + | 72 | 80 | 16.66 | 33.33 | Stable |
| TP-2 | + | ++ | + | + | 50 | 66 | 32 | 57.14 | Stable |
Keys: (-)= no result, (+)=weak result, (++)=average result, (+++)=good result, (++++)=best result
Molecular basis Identification of biosurfactant producing bacteria
Total four DNA isolation were performed and validated by Agarose gel electrophoresis.
- The 16S rRNA sequencing of GAR-1 was conducted using 16S rRNA forward primer (27F: 5’-GCAACAATGC-3’). By analyzing the resulting sequence, the GAR-1 bacterium was identified as Bacillus paralicheniformis strain HRA-IU 16S ribosomal RNA gene, partial sequence. The past work also determined that Bacillus paralicheniformis had ability to produce biosurfactant. Phylogenetic tree of the strain of Bacillus paralicheniformis with closest similarity using neighbor joining method was analyzed by Clustal omega is shown as Figure 8. The 16S rRNA sequence was published at NCBI with accession number OK668297.
- The 16S rRNA sequencing of PET-1 was conducted using 16S rRNA forward primer (27F: 5’-GAGGTAACGG-3’). By analyzing the resulting sequence, the PET-1 bacterium was identified as Bacillus sp. (in: Bacteria) strain HUR-IU 16S ribosomal RNA gene, partial sequence. The past work also determined that Bacillus sp. had ability to produce biosurfactant. Phylogenetic tree of the strain of Bacillus sp. with closest similarity using neighbor joining method was analyzed by Clustal omega is shown as Figure 9. The 16S rRNA sequence was published at NCBI with accession number OL347993.
- The 16S rRNA sequencing of TP-2 was conducted using 16S rRNA primer (5’-TGGGATAACT-3’). By analyzing the resulting sequence, the TP-2 bacterium was identified as Bacillus velezensis strain NWR-20 16S ribosomal RNA gene, partial sequence. The past work also determined that Bacillus velezensis had ability to produce biosurfactant. Phylogenetic tree of the strain of Bacillus velezensis with closest similarity using neighbor joining method was analyzed by Clustal omega is shown as Figure 10. The 16S rRNA sequence was published at NCBI with accession number OL348203.
- The 16S rRNA sequencing of PET-3 was conducted using 16S rRNA forward primer (63F: 5’-GAATCTGCCT-3’). By analyzing the resulting sequence, the PET-3 bacterium was identified as Pseudomonas aeruginosa strain HUR-IA 16S ribosomal RNA gene, partial sequence. The past work also determined that Pseudomonas aeruginosa had ability to produce biosurfactant. Phylogenetic tree of the strain of Pseudomonas aeruginosa with closest similarity using neighbor joining method was analyzed by Clustal omega is shown as Figure 11. The 16S rRNA sequence with accession number OL348062 was published at NCBI.
Fourier Transform Infrared Spectroscopy (FTIR)
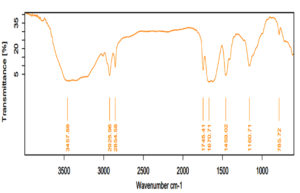
The presence of biosurfactant was further confirmed through FTIR spectra of the crude biosurfactant. As shown in Figure 12, a absorption band was seen at 3457 cm-1 due to the C–H bonds, O–H groups of carboxyl OH and hydrogen vibration of amide N–H functions, at 2854-2925 cm-1 matches to the C–H (CH3) and (CH2) stretch (aliphatic chain), at 1670-1745 cm-1 is similar to C=O stretching bond (presence of lactone ring), a single band near 1443 cm-1 is because of the vibration deformation δ(CH), the presence of an aliphatic chain-CH at 1160 cm-1 and at 785 cm-1 corresponds to the –COC–group vibrations in the cyclic structures of carbohydrates and confirmed the presence of bonds formed between Carbon atom and hydroxyl groups in chemical structures of rhamnose rings. This result confirms that the biosurfactant produced by Bacillus sp. HUR-IU corresponds to rhamnolipid.
The present study was conducted to isolate and screen biosurfactant-producing bacteria from various oil contaminated soil sites such as poultry waste site, garage waste site, IIM dumping site, kitchen waste site and petrol pump site in Lucknow city, India. 13 strains isolated as a pure culture from these contaminated soil sites and out of 13 strains, 4 strains (PET-1, PET-3, GAR-1 & TP-2) show positive result for biosurfactant production on the basis of EI, oil spreading assay & emulsification stability.
In the present study, three qualitative methods (drop collapse, haemolytic activity and oil dispersion) and two quantitative methods (emulsification Index and emulsification stability) were applied. E24 index with petrol for PET-1, PET-3, GAR-1 & TP-2 were 66%, 52%, 72% and 50%, respectively, and with terpene oil E24 were 93.33%, 71.88%, 80 and 60%, respectively. Qualitative analysis (drop collapse, haemolytic activity and oil dispersion) for PET-1, PET-3, GAR-1 & TP-2 isolates were also shown in Table 2. Further molecular identification for PET-1, PET-3, GAR-1 & TP-2 isolates were done by 16S RNA method and its result reveal that PET-1 was Bacillus sp. HUR-IU, PET-3 was Pseudomonas aeruginosa strain HUR-IA, GAR-1 was Bacillus paralicheniformis strain HRA-IU and TP-2 was Bacillus velezensis strain NWR-20. Among all the isolates best emulsification activity was shown by Bacillus sp. HUR-IU and biosurfactant produced by Bacillus sp. HUR-IU was characterized by FTIR analysis on the basis of functional group present in a produced biosurfactant was rhamnolipid.
In this study, thirteen biosurfactant-producing bacterial strains were isolated from different oil contaminated soil sites in Lucknow, India. The four strains were identified in which one strain was found to produce maximum biosurfactant. The biosurfactant produced by isolate Bacillus sp. HUR-IU had a strong ability to solubilize oil, which has great potential for commercial exploitation in the area of enhanced bioremediation. On the basis of functional groups present in biosurfactant produced from Bacillus sp. HUR-IU was characterized as rhamnolipid by FTIR analysis and can be used to improve bioremediation of oil contaminated soil. To assess the feasibility potential of these applications, further investigation is ongoing.
ACKNOWLEDGMENTS
The authors would like to acknowledge the support and encouragement given by the Integral University, Lucknow, for writing this research article with reference number IU/R&D/2024-MCN0002384.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
AS conceptualized the study and performed supervision. HR applied methodology, performed investigation, data interpretation, formal analysis and wrote the manuscript. VS reviewed and edited the manuscript. All authors read and approved the final manuscript for publication.
FUNDING
None.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
Not applicable.
- Ritchie, Hannah, Veronika Samborska, and Max Roser. “Oil Spills.” Our World in Data. 2022.
- ITOPF. Oil Tanker Spill Statistics 2023. https://www.itopf.org/knowledge-resources/data-statistics/statistics/. Accessed date: 2023
- Bhadra S, Dixita C, Verma AK. Biosurfactants: Secondary metabolites involved in the process of bioremediation and biofilm removal. Appl Biochem Biotechnol. 2023;195(9):5541-5567.
Crossref - Uyar E, Avci T. Screening and molecular identification of biosurfactant/bioemulsifier producing bacteria from crude oil contaminated soils samples. Biologia. 2023:1-15.
Crossref - Amber JP, Bharti B, Benton MG. Nano-enhanced bioremediation for oil spills: a review. ACS ES&T Engineering. 2021;1(6):928-946.
Crossref - Pardhi DS, Panchal RR, Raval VH, et al. Microbial surfactants: A journey from fundamentals to recent advances. Front Microbiol. 2022;13:982603.
Crossref - Ahmadi-Ashtiani H-R, Baldisserotto A, Cesa E, et al. Microbial biosurfactants as key multifunctional ingredients for sustainable cosmetics. Cosmetics. 2020;7(2):46.
Crossref - Mohanty SS, Koul Y, Varjani S, et al. A critical review on various feedstocks as sustainable substrates for biosurfactants production: a way towards cleaner production. Microb Cell Fact. 2021;20(1):120.
Crossref - Rani M, Weadge JT, Jabaji S. Isolation and characterization of biosurfactant-producing bacteria from oil well batteries with antimicrobial activities against food-borne and plant pathogens. Front Microbiol. 2020;11:64.
Crossref - Youssef NH, Duncan KE, Nagle DP, Savage KN, Knapp RM, McInerney MJ. Comparison of methods to detect biosurfactant production by diverse microorganisms. J Microbiol Methods. 2004;56(3):339-347.
Crossref - Plaza GA, Zjawiony I, Banat IM. Use of different methods for detection of thermophilic biosurfactant-producing bacteria from hydrocarbon-contaminated and bioremediated soils. Journal of Petroleum Science and Engineering. 2006;50(1):71-77.
Crossref - Jain DK, Collins-Thompson DL, Lee H, Trevors JT. A drop-collapsing test for screening surfactant-producing microorganisms. J Microbiol Methods. 1991;13(4):271-279.
Crossref - Zouari O, Lecouturier D, Rochex A, et al. Bio-emulsifying and biodegradation activities of syringafactin producing Pseudomonas spp. strains isolated from oil contaminated soils. Biodegradation. 2019;30(4):259-272.
Crossref - Chooklin CS, Sakulsawasdipan K, Sawain A, Saimmai A, Rattanapan N, Kitcha W. The Enhancing Oil Pollution Remediation Sites with Effective Emulsifying Property of Biosurfactant-Producing Bacillus oceanisediminis PM 08. ASEAN Journal of Scientific and Technological Reports. 2023;26(3):52-62.
Crossref - Kishan G, Sharma SK, Holkar SK, et al. Diverse spectra of virus infection identified through high throughput sequencing in nursery plants of two Indian grapevine cultivars. Physiol Mol Plant Pathol. 2023;128:102135.
Crossref - Erasmus DJ. DNA barcoding: A different perspective to introducing undergraduate students to DNA sequence analysis. Biochem Mol Biol Educ. 2021;49(3):416-421.
Crossref - Kandeel M, Mohamed MEM, El Lateef HMA, Venugopala KN, El Beltagi HS. Omicron variant genome evolution and phylogenetics. J Med Virol. 2022;94(4):1627-1632.
Crossref - Shagufta SM, Dharani PV. Screening and Isolation of Bacteria Producing Biosurfactants from Waste. J Pure Appl Microbiol. 2022;16(1):567-576.
Crossref - Shamim A, Tripathi G, Ansari JA, et al. Effect of pH on aluminum uptake and differential aluminum tolerance in cyanobacterial strains: A bioresource for agricultural and environmental sustainability. Bioresource Technology Reports. 2022;18:100999.
Crossref - Nawar, Haider H., and Hurria H. Al-Juboory. Isolation and Diagnosis of the Bacterium Pectobacterium Caratovorium, the Causative Agent of Soft Rot Disease on Potatoes, Macroscopically and Biochemically. IOP Conf Ser : Earth Environ Sci. 2023;1262(3):032062.
Crossref
© The Author(s) 2024. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.