ISSN: 0973-7510
E-ISSN: 2581-690X
The Indian gooseberry is a monoecious tree and bears a unisexual flower where a high degree of cross-pollination is required for an ideal fruit set as well as better productivity. The fruit set, retention and yield in gooseberry depends on the nutritional and hormonal balance of the tree. The current work is based on the objective to estimate the fruit set, retention, sex ratio and yield in gooseberry after application of a recommended dose of fertilization (RDF) in association with farm yard manure (FYM) and biofertilizers (Azotobacter, Azospirillum and phosphate solubilizing bacteria). The maximum sex ratio was estimated in control (250.22 and 251.09) due to sufficient nitrogen fertilization. However, the highest fruit set, retention percent and yield per tree were reported with the treatment containing three-fourth of RDF in combination with Azotobacter, Azospirillum and PSB (phosphate solubilizing bacteria). Further, it was observed that about 25 to 50% replacement in RDF could be possible through biofertilizer application as a constant source of nutrients to trees.
Biofertilizers, flowering, fruiting, fruit yield, Indian gooseberry, microbial consortium, sex ratio
The Indian gooseberry, known as aonla, is native to tropical South – East Asia and belongs to family Euphorbiaceae. It is characterized by a phase of dormancy soon after flowering and fruit set in spring which continue throughout summer till the onset of monsoon. This dormancy phase has minimum water requirement so there is no need of irrigation during summer season and this makes aonla the most ideal crop for arid region.1 There are two critical phases for fertlization of aonla trees: immediate after harvesting for better flowering and fruiting, and during monsoon for better fruit development. Like the other crops, aonla trees also respond very well to chemical fertilizers when applied as per recommended dose of fertlizers. Howerver, indiscriminate application of chemical fertilizers had adversely affected the soil fertility, water quality, yield and quality of the produce in long run.2 Further, the methodologies for application of fertilizers had also damaged the health of soil which can be improved through fertigation3,4 or soiless cultivation5-7 or aeroponics8 or protected cultivation9 for better growth and quality production of fruits.10
There is need of an urgent and active approach for eco-friendly management of crops including traditional knowledge like panchgavya or effective microbial technology (EMT).11 The EMT approach includes a number of associative and free-living microorganisms like Azospirillum, Azotobacter, Cyanobacteria, etc. which are present in soil and can fix atmospheric nitrogen. In addition to nitrogen fixation, these organisms are also known to synthesize plant growth promoting substance such as vitamin B-complex, Indole Acetic Acid, Gibberellins and cytokinin.12 Inoculation of seeds with efficient strains of Azotobacter and Azospirillum contribute only about 15-20 kg fertilizer equivalent N/ha in different crops. Integrated use of phophorus solublizing bacteria (PSB) with low graded rock phosphate contributes about 30-35kg P2O5 /ha in neutral to slightly alkaline soils. Further, application of plant growth promoting microbes brings improvement in microbial population beneficial for soil health as these can enable the plants for managing soil alkalinity and acidity stress.13 These microbes have great potential in enhancing the plant growth and crop productivity, on which very little work has been done.14
Integrated use of chemical fertilizers with biofertilizers markedly increased fertilizer use efficiency and minimize their losses and leakage. The present studies emphasized to evaluate the response of gooseberry fruit trees for flowering, fruiting and yield attributes after application of integrated nutrient management practices in cv. NA-7.
Study Materials
The present study was conducted during 2019 and 2020 in ten-years-old orchard of gooseberry, located at the Department of Horticulture, ITM University Gwalior, MP (India). The soil can be characterised as sandy loam with average proporation of fine sand (64.77%), silt (22.76%) and clay (14.95%) with average pH of 7.71. Thirty-three trees of uniform vigour from ten-years-old Aonla (cv ‘NA-7’) orchard plantedat 8.0 x 8.0 m distance were used for the experiment.
Nutrients Sources for Treatments
The treatments include recommended dose of fertilizer (RDF) as 1.0 kg of nitrogen, 0.5 kg of phosphorus and 1.0 kg of potassium per tree. Farm yard manure (FYM) @ 100 kg/tree along with biofertilizers (Azotobacter, Azospirillum and PSB @ 100 g/tree each) was applied during the second week of January. The recommended dose of fertilizers was applied in three different proporation i.e., 100%, 75% and 50% in form of urea, single super phosphate, and murrete of potash, respectively. After harvesting of previous crops, two-third of the total nitrogen and complete phosphorus and potassium were applied while remaining dose of nitrogen was applied during onset of monsson. The fertilizer was well mixed with the soil in the trenches and then levelled. The statistical analysis for randomized block design was carried out with eleven treatments and three replications. The treatments were, T0 – Full dose of NPK (1000:500:1000 g/tree) control; T1 – three-fourth dose of NPK/tree + 100 kg FYM; T2 – three-fourth dose of NPK/tree + 100 kg FYM + Azotobacter; T3 – three-fourth dose of NPK/tree + 100 kg FYM + Azospirillum; T4 – three-fourth dose of NPK/tree + 100 kg FYM + PSB; T5 – three-fourth dose of NPK/tree + 100 kg FYM +Azotobacter + Azospirillum+ PSB; T6 – half dose of NPK/tree +100 kg FYM; T7 – half dose of NPK/tree +100 kg FYM + Azotobacter; T8 – half dose of NPK/tree +100 kg FYM + Azospirillum; T9 – half dose of NPK/tree +100 kg FYM + PSB; T10 – half dose of NPK/tree +100 kg FYM + Azotobacter + Azospirillum + PSB.
Observations and Objective
The observation on flowering and fruiting was measured in terms of the flower initiation (recorded during the month of March), sex ratio (male flowers: female flowers), fruit set (%), retention (%) and fruit yield (kg/tree). The observation recorded on various parameters were analysed statistically as per the randomized block design for one way analysis of variance. Further, correlation and regression studies were also carried out to understand the relation between the dependent variables as yield and the independent variables under study.
Flowering Attributes
The observations on time of flower initiation clearly reflects that the early flower initiation was revealed in treatment T5 (15 March and 12 March) followed by T10 (17 March and 14 March) during the years of experiment i.e., 2019 and 2020, respectively. While, the late flowering was observed in control (20 March and 24 March) during both the years. A profound combined effect of NPK, FYM and biofertilizers was reported on the flowering on Aonla cv. NA-7 (Table 1). The higher sex ratio was observed in control (250.22 and 251.09) followed by T1 (three-fourth dose of NPK/tree + 100 kg FYM) and T6 (half dose of NPK/tree + 100 kg FYM) during both the years i.e., 2019 and 2020. T10 (half dose of NPK/tree + 100 kg FYM + Azotobacter + Azospirillum + PSB) showed comparatively lower sex ratio (245.62 and 244.08) followed by T9, T5 and T8 during both the years. All the other treatments except T0, T1 and T6 did not exhibit any significant difference in first year with treatment T10.
Table (1):
Sex ratio and fruit set in aonla trees as function of INM practices consisiting of biofertilizers.
| Treatments | Sex ratio | Fruit set (%) | ||
|---|---|---|---|---|
| 2019 | 2020 | 2019 | 2020 | |
| T0 | 250.22 | 251.09 | 23.23 | 24.24 |
| T1 | 249.88 | 250.22 | 26.88 | 30.21 |
| T2 | 247.12 | 247.65 | 33.66 | 43.56 |
| T3 | 247.4 | 246.39 | 30.92 | 39.79 |
| T4 | 246.44 | 246.66 | 30.16 | 38.38 |
| T5 | 246.09 | 245.62 | 44.55 | 50.16 |
| T6 | 248.06 | 248.14 | 27.27 | 29.29 |
| T7 | 247.00 | 247.2 | 28.00 | 35.43 |
| T8 | 246.20 | 246.26 | 27.72 | 32.67 |
| T9 | 245.82 | 245.42 | 29.21 | 35.64 |
| T10 | 246.62 | 244.08 | 35.30 | 44.46 |
| Mean | 247.26 | 247.16 | 30.60 | 36.61 |
| S.E. (diff) | 0.8855 | 0.954 | 1.37 | 1.009 |
| CD at 5% level | 1.8471 | 1.99 | 2.858 | 2.106 |
The present observations on the time of flower initiation and sex ratio have shown that application of inorganic fertilizers and biofertilizers had significant effect. Time of flower initiation reduced after combined application of inorganic fertilizers and biofertilizers. It was noticed that early flowering was in T5 (three-fourth dose of NPK/tree + 100 kg FYM + Azotobacter + Azospirillum + PSB) followed by T10 (half dose of NPK/tree + 100 kg FYM + Azotobacter + Azospirillum + PSB). Although, sex ratio is varietal character but it has been reported to vary from year to year and as a function of climatic changes during flower bud development. Moreover, the variations of sex ratio also depend upon the mineral nutrients, day length, temperature, etc. The higher sex ratio in case of 100% RDF is associated with sufficient availability of nitrogrn to plants and a balanced C:N ration in soil. The lower sex ratio in the treatments with 50% RDF is associated with poor N-availability which might have caused reproductive stress in aonla trees due to high value of C:N ratio resulting microbial immobilization.15 The treatments where reduction in RDF application was not supplemented with application of Azotobacter and/or Azospirillum had reflected lower sex ratio which might be due to poor availability of nitrogen in absence of free atmospheric nitrogen fixation in the rhizosphere of aonla trees as in case of T9 (half dose of NPK/tree + 100 kg FYM + PSB). Further, the high female flower production is resource intensive biological phenomenon resulting shift of sex ration in favour of male fowers.16
Fruiting Attributes
The analysis of data on fruit set percentage (Table 1) clearly indicates that all combinations significantly influenced the fruit set (%) as compared with control. The treatment T5 was found to be reasonably superior to the rest of the treatments during both the years. The highest value of fruit set was observed in T5 (44.55 and 50.16 %) followed by T10, T2, T3 and T4 during the experimental years i.e., 2019 and 2020, respectively. Similarly, the treatment T5 also exhibited highest fruit retention (35.55 and 43.17 %) followed by T10, T4, T3 and T2 whereas, the minimum was observed in control (10.14 and 8.99%) during the year of 2019 and 2020, respectively (Table 2). However, all the other treatments were estimated with significantly better fruit retention over the control during both the years.
Table (2):
Gooseberry fruit retention and yield as function of INM practices consisiting of biofertilizers.
| Treatments | Fruit retention (%) | Fruit yield (kg/tree) | ||
|---|---|---|---|---|
| 2019 | 2020 | 2019 | 2020 | |
| T0 | 10.14 | 8.99 | 132.6 | 131.84 |
| T1 | 14.81 | 16.66 | 132.76 | 134.64 |
| T2 | 26.64 | 33.32 | 146.39 | 147.56 |
| T3 | 26.66 | 33.42 | 140.47 | 140.87 |
| T4 | 28.57 | 34.08 | 148.75 | 151.49 |
| T5 | 35.55 | 43.17 | 159.6 | 161.68 |
| T6 | 18.49 | 27 | 133.69 | 134.86 |
| T7 | 21.42 | 28.56 | 136.96 | 139.12 |
| T8 | 22.71 | 26.31 | 140.47 | 141.39 |
| T9 | 24.99 | 30.3 | 137.22 | 137.3 |
| T10 | 29.41 | 41.36 | 155.32 | 156.92 |
| Mean | 23.58 | 29.38 | 142.20 | 143.42 |
| S.E.(diff) | 0.6379 | 2.0448 | 4.255 | 4.958 |
| CD at 5% level | 1.3295 | 4.2654 | 8.877 | 10.343 |
A significant improvement in fruit set and retention was reported after the application of treatment combination of inorganic fertilizers and biofertilizers sources of nutrients. Interestingly, no relation was reported in sex ratio and fruit set or fruit retentions parameters in aonla. The sex ratio was significantly influenced by nitrogen application and uptake while the fruit set or retention was largely influenced due to balanced fertilization approach in T5, T10, T2, T3 and T4. This could also be attributed to hormonal balance, particularly auxin, where biofertilizers were applied resulting reduced fruit drops and greater retention17.
Thus, balanced dose of inorganic fertilizersin combination with biofertilizers is necessary for achieving higher fruit set, retention and lowest fruit drop. The increase in fruit set might be associated to auxin synthesisand translocation due to presence of Azotobacter. The increased fruit set might also be due to improvement in the emergence of a greater proportion of female flowers and reduced fruit drop resulting in higher number of fruits per shoot. Further, the balanced supply of nutrients through combined application of inorganic fertilizers and biofertilizers has resulted in timely induction of flowering andimproved retention of fruits which in turn account for improved synthesis and translocation of photosynthates at critical requirements1.
Fruit Yield (kg/tree)
The estimation of fruit yield (Table 2) clearly shows an improvement in yield in T5 over other treatments except T10 and exhibiting maximum fruit yield (159.60 and 161.68 kg/tree) during the year 2019 and 2020, respectively; however, the lowest fruit yield was estimated with control (132.60 and 131.43 kg/ tree). Thus, it can be concluded that half dose of NPK + 100 kg FYM + Azotobacter + Azospirillum + PSB was the most economical and effective in terms of yield as it reduced the NPK requirements as compared to third fourth dose of NPK + 100kg FYM + Azotobacter + Azospirillum + PSB.
The better yield in the treatments with combination of NPK, FYM and biofertilizers might bean outcome of better growth and development of the plants under the treatments as compared to control. Application of biofertlizers might also be responsible for conditioning of soil and improvement of soil health, free atmospheric nitrogen synthesis, increased nutrient uptake and efficient utilization of nutrients for photosynthetic activities resulting improvement in yield.18-20 Singh et al.21 has also reported increased nutrient status in leaves of aonla trees due to INM practices during intercropping which confims a correlation between nutrient mobilization, nutrient uptake and nutrient utilization during INM approach in aonla orchard.22 The current studies on flowering and fruiting attributes of aonla revealed that better yield in aonla was realized when the levels of leaf N, P and K as well as micro-nutrients improved in soil and leaves.23,24 The positive correlation between leaf nutrients and fruit yield was also noticed by Rattan et al. in kinnow25.
Correlation and Regression Study for Yield
The correlation and regression analysis for the flowering, fruiting and yield attributes reflected high residual value which confirms existance some more factors beyond the study which can influence the yield of aonla fruits after application of biofertilizers (Table 3). Also, a strong significant effect of sex ratio, fruit set and retention were reported for aonla fruit yield. The strong and positive correlation was noticed for fruit set with fruit retention (0.92) and fruit yield (0.92). However, a negative correlation of sex ration was noticed with all other traits in aonla trees provided with biofertilizer application.26 Similar findings have also been reported by Singh et al.27 in mango as influenced by different chemicals.
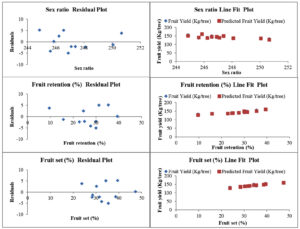
Fig. 1. Residual plots and line fit plots for yield of aonla fruits (Y) as a function of sex ratio, fruit retention (%) and fruit set (%) after application of biofertilizers treatments
The residual plot of all attributes for fruit yield (Fig. 1) confirms similarity between fruit set and retention percentage with negative value for T5, T6 and T7 and zero value for T10. This confirms accuracy if prediction for T10 where 50% of RDF was supplemented with biofertilizers.28 Further, the line fit plot of all traits for fruit yield confirmed the coincidence of expected value with actual value of fruit yield so these attributes can be effectively used to predict the aonla fruit yield.29
Table (3):
Correlation and regression study for yield of gooseberry fruits after biofertilizers application.
Correlation Statistics |
Sex ratio |
Fruit Set (%) |
Fruit retention (%) |
Fruit Yield (kg/tree) |
|---|---|---|---|---|
Sex ratio |
1 |
|||
Fruit Set (%) |
-0.69 |
1 |
||
Fruit retention (%) |
-0.88 |
0.92 |
1 |
|
Fruit Yield (kg/tree) |
-0.69 |
0.92 |
0.88 |
1 |
| Regression Statistics | |
|---|---|
| Multiple R | 0.9997 |
| R Square | 0.9994 |
| Adjusted R Square | 0.8742 |
| Standard Error | 4.0946 |
| Observations | 11 |
| ANOVA | ||||||
|---|---|---|---|---|---|---|
| df | SS | MS | F | Significance F | ||
| Regression | 3 | 225114.46 | 75038.15 | 4475.72 | 7.51693 x 10-12 | |
| Residual | 8 | 134.12 | 16.76 | |||
| Total | 11 | 225248.59 | ||||
Application of three-fourth recommended dose of NPK/tree + 100 kg FYM along with biofertilizers (Azotobacter, Azospirillum and PSB) was recorded highest fruit set, retention and yield in gooseberry, while half dose of NPK/tree + 100 kg FYM and biofertilizers was next best treatment. Further, the sex ratio, fruit set and retention have strong and significant influence over fruit yield of gooseberry trees subjected to biofertlizers application as supplementary dose with 50-75% of RDF.
ACKNOWLEDGMENTS
Authors are thankful to ITM University, Gwalior for constant support for resources as and when required.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
None.
ETHICS STATEMENT
Not applicable.
AVAILABILITY OF DATA
All datasets generated or analyzed during this study are included in the manuscript.
- Sharma A, Prasad D, Chaurasiya R, et al. Influence of foliar application of Zinc, Calcium and GA3 on fruit drop, yield and quality attributes of aonla cv. NA-7. Int J Curr Microbial Appl Sci. 2019;(8):135-146.
- Lin W, Lin M, Zhou H, Wu H, Li Z, Lin W. The effects of chemical and organic fertilizer usage on rhizosphere soil in tea orchards. PLoS ONE. 2019;14(5):e0217018.
Crossref - Singh D, Singh SK. Efficient Usage of Water and Fertilizers for Papaya Production-A review. Annals of Biology. 2019;35(2):258-267.
- Bahadur L, Anmol, Singh SK. Growth potential of banana (Musa) plants after fertigation treatments under polynet house condition. Annals of Biology. 2021;37(1):82-85.
- Spehia RS, Singh SK, Devi M, et al. Estimation of drip irrigation water requirement in different soilless growing media for protected cultivation of tomato (Solanum lycopersicum L.). Annals of Biology. 2019a;35(1):78-82.
- Spehia RS, Singh SK, Devi M, et al. Standardization of growing media and its effects on nutrient uptake in tomato (Solanum lycopersicum L.) under protected conditions. Annals of Agri-bio Research. 2019b;24(1):71-75.
- Spehia RS, Singh SK, Devi M, et al. Effect of soilless media on nutrient uptake and yield of tomato (Solanum lycopersicum). Indian J Agric Sci. 2020;90(4):732-735.
- Singh P, Singh SK, Anmol, Sharma KK. Evaluation of performance of popular cultivars of potato under aeroponic conditions. Annals of Biology. 2020;36(3):412-417.
- Bahadur L, Anmol, Singh D, Singh SK. A review on successful protected cultivation of banana (Musa). Plant Archives. 2020;20(Suppl 2):1570-1573.
- Singh D, Singh SK, Damathia LB. Impact of fertigation on Papaya crop under protected conditions. Ecology Environment and Conservation. 2019a;25(1):295-299.
- Rohitha MS, Sharma R, Singh SK. Integration of panchagavya, neemcake, and vermicompost improves the quality of chilli production. Journal of Applied Horticulture. 2021;23(2):212-218.
Crossref - Sumbul A, Ansari RA, Rizvi R, Mahmood I. Azotobacter: A potential bio-fertilizer for soil and plant health management. Saudi J Biol Sci. 2020;27(12):3634-3640.
Crossref - Msimbira LA, Smith DL. The roles of plant growth promoting microbes in enhancing plant tolerance to acidity and alkalinity stresses. Front Sustain Food Syst. 2020;4:106.
Crossref - Trivedi P, Schenk PM, Wallenstein MD, Singh BK. Tiny microbes, big yields: enhancing food crop production with biological solutions. Microbial Biotechnol. 2017;10(5):999-1003.
Crossref - Brust GE. Management strategies for organic vegetable fertility. In Safety and practice for organic food. Academic Press. 2019:193-212.
Crossref - Delph LF, Herlihy CR. Sexual, fecundity, and viability selection on flower size and number in a sexually dimorphic plant. International Journal of Organic Evolution. 2012;66(4):1154-1166.
Crossref - Singh R, Bakshi M, Singh SK. Effective management of fruit drop and quality improvement in Kinnow mandarin using auxin, fungicide and micronutrients. Annals of Agri-bio Research. 2019b;24(1):88-90.
- Dynarski KA, Houlton BZ. Nutrient limitation of terrestrial free-living nitrogen fixation. New Phytologist. 2018;217(3):1050-1061.
Crossref - Jugnake MO, Patil MB, Shinde SE. Effect of biofertilizer and chemical fertilizer on growth and yield of sweet orange (Citrus sinensis L. Osbeck). International Journal of Chemical Studies. 2017;5(6):1061-1064.
- Kumar M, Kathayat K, Singh SK, Singh L, Singh T. Influence of bio-fertilizers application on growth, yield and quality attributes of cucumber (Cucumis sativus L.): A review. Plant Archives. 2018;18(2):2329-2334.
- Singh SK, Sharma M, Singh PK. Yield, fruit quality and leaf nutrient status of aonla as influenced by intercropping under integrated nutrient management. Journal of Crop and Weeds. 2018;14(1):09-13.
- Anmol, Singh SK. Yield improvement in cucumber through integrated nutrient management practices in central plain zone (Pb-3) of Punjab, India. International Journal of Research and Analytical Reviews. 2018;5(4):766-772.
- Singh SK, Sharma M, Singh PK. Combined approach of intercropping and INM to improve availability of soil and leaf nutrients in fruit trees. Journal of Chemical and Pharmaceutical Sciences. 2016;9(2):823-829.
- Subash M, Rafat H. Effect of plant growth promoters and biofertilizers on yield and yield components of sesame (Sesamum indicum L). Life Science Archives (LSA). 2016;2(4):622-627.
- Rattan CS, Singh SK, Badhan BS. Influence of tree age on vegetative growth, leaf nutrient content and yield of Kinnow trees. Plant Archives. 2020;20(2):5257-5262.
- Allesh SP, Varma S, Reshmi KS, et al. Effect of flower sex ratio on fruit set in pumpkin (Cucurbita maxima). Scientia Horticulturae. 2019;246:1005-1008.
Crossref - Singh MK, Singh VB, Singh SS, Singh AK. Floral biology and fruit set of mango (Mangifera indica L.) as influenced by different chemicals. Int J Curr Microbial Appl Sci. 2019;8(1):1106-1117.
Crossref - Law M, Jackson D. Residual plots for linear regression models with censored outcome data: a refined method for visualizing residual uncertainty. Communications in Statistics-Simulation and Computation. 2017;46(4):3159-3171.
Crossref - Montgomery DC, Peck EA, Vining GG. Introduction to linear regression analysis. John Wiley & Sons. 2021.
© The Author(s) 2021. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.