ISSN: 0973-7510
E-ISSN: 2581-690X
Coronavirus disease (COVID) is highly contagious, and negligence of it causes high morbidity and mortality globally. The highly infectious viral disease caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) was abbreviated as COVID-19 (Coronavirus disease 2019) by World Health Organization first time on February 11, 2020, and later on, WHO declared COVID-19 as a global pandemic on 11/3/2020. Epidemiological studies demonstrated that the SARS CoV-2 infects the overall population, irrespective of age, gender, or ethnic variation, but it was observed in clinical studies that older and compromised immunity population is much more prone to COVID-19. SARS-CoV-2 majorly spread through aeration route in droplet form on sneezing and coughing, or by contact when touching eyes, nose or mouth with the infected hands or any other organs, resulting from mild to severe range of SARS-CoV-2 infection. This literature-based review was done by searching the relevant SCI and SCOPUS papers on the pandemic, SARS-CoV-2 and COVID-19, herbal formulation, and Ayurveda from the databases, Academia, Google Scholar, PubMed, and ResearchGate. The present review attempts to recognize the therapeutic strategies to combat COVID-19 because of the current human risk. Indian system of medicine, including herbals, has immense potential in treating and managing various viral infections and provides evidence to utilize Ayurvedic medication to improve immunity. Cumulative research findings suggest that Ayurvedic formulations and herbal immunomodulators (Tino sporacordifolia, Withania somnifera, Crocus sativus, Zafran, Allium sativum, Zingiber officinale, Albizia lebbek, Terminalia chebula, Piper longum, Mangifera indica, Ocimum sanctum, Centella asiatica ) are promising in the treatment of outrageous viral infections without exerting adverse effects. Considering the ancient wisdom of knowledge, the herbal formulations would compel healthcare policymakers to endorse Ayurveda formulations to control the COVID-19 pandemic significantly.
COVID-19, Ayurveda, Immunomodulators, Herbals, Viral infections, anti-viral
Mounting research findings suggest that COVID-19 is primarily a respiratory syndrome chronically affecting whole-body organ; caused by a novel coronavirus know as severe acute respiratory syndrome coronavirus 2 [SARS-CoV-2], and have affected the quality of life and is a burden on the socio-economic system of the nation. SARS-CoV-2 is a highly fatal ailment, and to date, there is no specific vaccine or drug in the treatment market. Supportive treatment is the only choice for disease management and prevention. Various studies are being performed worldwide, and few selected drugs are in clinical trials but still far from the final goal.
As per the current scenario, only those having strong immunity can survive, fight, and protect themselves from COVID-19. Several herbal medicines as Immunomodulators have been utilized by humankind since time immemorial or ancient period. Although complete scientific validations of these herbs Immunomodulators have not been evaluated, still several trials are in progress. Fundamentally, the Immune system can differentiate between host proteins/cells and foreign particles or antigens. Further, the body’s immune system fights against various infectious diseases/agents after foreign bodies/agents’ invasion. Some other infectious agents are also involved in the immunization, including external stimuli. The immune system can be categorized into two broad classes based on function, i.e., one is the innate immune system known as the non-specific immune system, and another one is an adaptive immune system that is commonly known as acquired/specific immune system 1,2.
Various studies demonstrated that different types of barriers, including microbial, chemical, and physical are innate immunity. Additionally, some other imperative mediators of the defense system instantly distribute defense bio-chemicals like inflammatory markers/ immune-proteins and macrophages, monocytes, complement, and neutrophils, collectively recognized as pattern recognition receptors (PRRs). These pattern receptors for host sensors can be identified as pathogen-associated molecular patterns [PAMPs]. Furthermore, when pathogens entered the host, they trigger the expression of PAMPs, further release a mixture of immune molecules such as interferons, chemokines, and cytokines, nitric oxide [NO], dendritic cells, and also involved in the establishment of the phagocytosis process. These immune-components are also concerned with the activation of memory B and naive T cells3,4.
Globally, the human community faces unprecedented health problems in the form of a novel Corona virus-19(CoVs-19) disease documented as COVID-19. CoV-19 originated from Coronavirus, known as severe acute respiratory syndrome corona virus-2 [SARS-CoV- 2] and is associated with respiratory damages. At this instant, the world health organization (WHO) has declared the COVID-19 pandemic and has realized it as a significant health issue in the world. The first time Covs-19 infected person was identified at Wuhan city, Hubei Province, China, with an outbreak of respiratory sickness or damage. In the beginning, it was reported to the WHO on December 31/2019, which was declared COVID-19 on January 30, 2020, by the WHO. In the current scenario, COVID-19 spread worldwide within very short time duration and was declared a pandemic by the world health organization 1–4.
Recent findings suggested that COVID-19 infection affects the population with weak immune system attacks with the worst outcomes that affect the respiratory system and result in complete organ failure and become lethal in certain cases5,6.
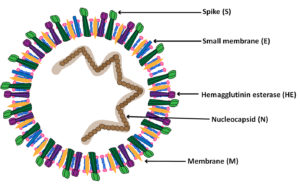
In the current scenario, COVID-19 has affected approximately 215 countries and territories around the world, still have reported, with more than 63,228,544 confirmed cases and more than 1,467,830 deaths until December 2, 2020. As per the WHO report, COVID-19 is one reason for causing health issues and ascending wealth problems1–5,7. Phylogenetic evolutionary studies have shown resemblance of COVID-19 with other already available coronavirus strains because COVID-19 have shared identical nucleotide with SARS-CoV, bats SARS-CoV and MERS- CoV. Previous studies have reported the structure of COVID-19 as multiple layered spherical structures with crown-like spikes all over the surface, providing well-known crown morphology 1–4,7,8 [figure-1a & b].
Fig. 1a. the structure of Corona virus virion and its genome RNA contain (trans-membrane proteins) the N protein (outward appearance a helical cased within membrane of the viral), hemagglutinin-esterase (HE); spike(S); Small membrane envelope(E); Membrane (M)
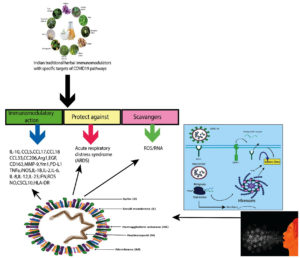
Fig. 1b. Systematic effect of traditional immunomodulatory on the suppression of different inflammatory marker and protective effect against the scavengers
The spike protein trimers form the peplomers embedded inside the envelope that provides a down-like shape to the virion. Merely, few members of CoVs, including the hemagglutinin-esterase -HE protein involved in the formation of smaller spikes on the membrane[M] with small membrane [E] protein (Minor component of the viral envelope). These are also transmembrane proteins that are associated with CoVs assembly(Fig. 1a &b). According to reports, CoVs have various proteins that perform different tasks, such as the S glycoprotein, which acts as the receptor-binding protein that causes infection and targets neutralizing antibodies. Similarly, the M glycoprotein involves virus gathering at intracellular membranes in the region of the endoplasmic reticulum Golgi complex [ERGIC]9–14.
Fig. 4b. The potential mechanism of bioactive (safranal and crocin from C.Sativus) molecules against infectious agents through immune response.
To date, only a few vaccines and anti-viral drugs such as Hydroxychloroquine, Favinapir, Ritonavir, Oseltamivir, Lopinavir, Ganciclovir, and Remdesivirare the highly and exclusively effective therapeutic approaches to treat viral disease; whereas, there are no existing viable vaccines to eliminate or suppress COVID-19 infections 15–17. Based on its previous clinical application, assorted anti-virals in different countries and regions [include Lopinavir/Ritonavir [400/100mg.12hourly], Chloroquine [500 mg 12 hourly] and Hydroxychloroquine [200 mg 12 hourly]and Azithromycin 500mg in 24 hours antibiotic for secondary infection, used to curtail the COVID 1918,19, whereas these drugs have minimal effect; moreover, as per WHO guideline some precaution make to safe from COVID-19 such as social distancing, quarantine, and isolation of suspected or infected person, are the best tools. Also, maintenance of proper hygiene, frequent hand wash with soap or alcohol [> 70 or 80 %] based sanitizers, and a mask 7,18–21. Recently, WHO has declared that eight COVID-19 antivirus vaccines enter preclinical and human clinical trials, but these vaccines might require months/years to launch into the market. Simultaneously, to negate the homicide situation until the vaccine approval and launch therapeutic herbal medications must be explored18–21.
In Indian ancient Ayurveda methodology, modern scientific study, and our practical knowledge are in the parallel clinical setting and, like other several studies, we have assessed the plan for evaluation of the immunomodulatory activity of herbal medicines. In the current time, Ayurveda is more popular and applicable because of its elaborative description of the root cause and management strategies of several epidemics (Janapadodhwamsa) in the Ayurvedic therapeutic system. Still, maximum research study and development have a target to identify such biochemical, biological moieties or solitary compounds could lead to precise target to withstand and overcome diseases associated with COVID-19, worldwide 19,21.
Research and development should be focused on herbal or natural source/molecules with high selectivity and potency. Low toxicity for targeted molecular or cellular targets and potency of the drug to overcome the COVID-19 situation must also be considered part of the study. After much consent, several studies have been designed to develop herbal medicine as alternative medicine, gaining global interest. In this consideration, researchers and scientists have recommended immune-enhancing herbs like Phyllanthus Emblica, Azadirachta indica, Tino sporacordifolia, Withania somnifera, Crocus sativus, Zafran, and Swertia chirata that would be helpful for the improvement of body defense mechanism to fight COVID-19 infection / to inhibit its replication inside the host cells 18–21. Several herbal formulations have been reported in the Ayurveda history or ancient cargo-space for the hindrance and management of COVID-19.
In traditional medicinal science, various parts of different plants or herbs were used as an herbal drug against several clinical ailments. Some such bioactive molecules – vinblastine, vincristine and its semi-synthetic offshoots isolated from the Madagascar periwinkle (Catharan thusroseus), capsaicin from chili peppers (Capsicum species), paclitaxel from Pacific yew (Taxusbrevifolia), and galantamine from the Caucasian snowdrop (Galanthus caucasicus) are examples of herbal medicines molecules 1–3,7,19,22,23.
We believe this review will provide details about plants based medicine, which adds up to boost the immune system, act as Immunomodulators, and their essential features that might express potent anti-viral activity.
For extensive pandemic information, SARS-CoV-2, Coronavirus, its structure and pathology, its prevalence, occurrence, traditional medicine, herbal formulation Tino sporacordifolia, Withania somnifera, Crocus sativus, Zafran, Allium sativum, Zingiber officinale, Albizia lebbek, Terminalia chebula, Piper longum, Mangifera indica, Ocimum sanctum, Centella asiatica, and its mechanism, anti-COVID drug, all authors equally, searched for the most recent information about this pandemic using several search engines such as Science Direct & Scopus, PubMed, Springer Link, Research Gate, Wiley Online Library, and Google Scholar databases and Elsevier. Between May and December 2020, the current review search was performed, and cited literature was screened from various SCI and Scopus index papers. Further treatment and management of COVID-19 by using herbal formulation was the current study that met this review’s purpose.
Herbals medinces and COVID-19
A literature search was performed using a search engine like Science Direct & Scopus, PubMed, SpringerLink, Wiley Online Library, and Google Scholar databases to identify immunomodulatory effects of Tino sporacordifolia, Withania somnifera, Crocus sativus Zafran, Allium sativum, Zingiber officinale Albizia lebbek, Terminalia chebula, Piper longum, Mangifera indica, Ocimum sanctum, Centella asiatica and their active constituents with experimental evidence of involvement in the immune system and also using keywords like Coronavirus, pandemic, its pathogenesis, clinical features and structure of COVID 19, Ayurveda, mechanism of various anti-viral plants, and medicinal plants, herbal Immunomodulators and its mechanism. The present review search was conducted between August and October 2020, and obtained cited literature from different SCI and Scopus index journals were screened. The present study that met this review’s aim was further screened and examined the herbal Immunomodulators against COVID-19 11.
The author should mention the term (keywords), database, and search engine used for the manuscript’s literature survey.
Natural immunomodulatory products or their extracts (containing bioactive compounds) with immunomodulatory potential may significantly impact infection management with COVID-19 and may establish a novel immunomodulatory agent. World Health Organization has currently permitted a few herbal-based immunomodulator medicines undergoing preclinical trial and clinical trials 1–3, 18,19,21,24. Concurrently, In India, the Ministry of AYUSH has also recommended using herbal medicine as preventive healthcare services and enhancing immunity with particular reference to respiratory health or COVID-19.
Rasayana Chikitsa
Ayurveda, or ancient science of life, dealt with immunity as a holistic approach and a potentially promising immunotherapy approach. Ayurveda, deal defense of health as a healthy character and also suppress/cure the diseases. Specifically, Ayurveda emphasizes promoting and improving health by boosting the strengthened power of host defenses and acting as a resistive force against day-to-day physiological extremes and opportunistic maladies 22.
In Ayurveda, human beings’ daily literature wellness, known as “Vyadhiksamatwa,” means fighting against a specific infectious disease. The terms protective medicine and curative aspects are management terms as the self-explanatory-”Vyadhibalavirodhitvam” and “Vyadyutpadapratibandhakatvam” – resistance against the loss of the integrity, proportion, and interrelationship amongst the individual’s Dosas andDhatus22. Several useful dravya or liquid, preparation or formulations, the mechanism involved in enhancing immunity (Balaor Vyadhiksamatwa) [classical Ayurveda]22.
In Ayurveda, epidemics or pandemics were already described as “Janapadodhvansa” 25or “Marak” 22,26.RasayanaChikitsais is known to proRasa, Rakta, Mamsa, Medha, Asthi, Majja, and Sukra [essential seven vital tissues] and Ayana [the path or channel]. In Rasayanatherapy, there was some point described for a healthy life or boost up of immunity [the Ojas][Vyadhiksamatwa] such as SatmyaAhara, Nidrarejuvenate the physiology of the body and produce resistance against infection at both physically and mentally- bring about proper uptake, growth, and improvement of essential Saptadhatus] along with SwasthavrittaParipalana [Dinacharya and Ritucharya] and that collectively called Rasayana therapy. Rasayana[Rasa and Ayana][Rasa means Saptadhatue (proper sleep), Yogic Asanas, and Brahmacharya22,25,26. According to Acharya Charaka, a person undergoing Rasayana therapy achieves long life, memory, mental power, freedom from infection25. Some studies have also suggested Rasayana therapy will provide better treatment by boosting immunity power in this pandemic time or safe from COVID-19 infection 22,25,2728. The mode of action of Rasayanatherapy can be performed through different actions like nutrition, immunomodulatory agent, an antioxidant, anti-aging, and neuroprotective. Some medicinal plants play an essential role in enhancing the Immune described below.
Guduchi (Giloy,Gilu,Tinosporacordifolia)
In Unani, Guduchi is famous as Gilu (figure-2), while in Ayurveda’s medicine system has shown remarkable medicinal properties like antioxidant, phagocytic activities, and anti-sedative antipyretic action as well as immunomodulatory properties 27. RasayanaChikitsa uses fevers (different form) and acts as a blood purifier, immune enhancer, including analgesic effect. Also, provide anti-inflammatory benefits and chronic cough or respiratory relief. In Unani medicine system use in the treatment of tuberculosis, leprosy, syphilis, and gonorrhea. It contains (as shown in table-1) bioactive Rasayana constituents tinosporin, tinosporide, and cordifolide. It has antioxidant, phagocytic activities, antipyretic action as well as immunomodulatory properties 25,27.
Table (1):
Showing most important and sub groups of natural products present in different parts of Tinospora cordifolia and their biological activities.
Plant Part |
Chemical molecules (Active Component Compound) |
Biological activity |
References |
|---|---|---|---|
Stem, Root |
Alkaloids (Berberine, Magnoflorine, Tetrahydropalmatine, Choline, Tembetarine, , Tinosporin, Palmetine, Isocolumbin, Aporphine alkaloids, Jatrorrhizine,) |
Neurological, immunomodulatory, psychiatric conditions Anti-viral infections, Anticancer, anti-diabetes, inflammation, |
(32-36) |
Whole Plant |
Diterpenoid Lactones (Columbin, Furanolactone, Clerodane derivatives [[5R,10R]-4R-8Rdihydroxy-2S-3R:15,16- diepoxy-cleroda-13 [16], 14-dieno-17,12S:18,1Sdilactone], Tinosporon, Tinosporides, Jateorine) |
Induce apoptosis in leukemia by activating caspase-3and bax, inhibits bcl-2Anti-inflammatory, antimicrobial, antihypertensive, anti-viral. |
(37-41) |
Stem |
Glycosides (Palmatosides, Tinocordiside, 18-norclerodane glucoside, Furanoid diterpene glucoside, Tinocordifolioside, Cordioside, Cordifolioside Syringin, Syringinapiosylglycoside, Pregnane glycoside, Cordifolioside A, B, C, D and E) Sesquiterpenoid (TinocordifolinAliphatic compoundOctacosanol, Heptacosanol Nonacosan-15-one dichloromethane) |
Parkinsons, Dementia, Immunomodulator |
(42-51) |
Shoot |
Steroids (β–sitosterol, δ-sitosterol, 20 β-hydroxyecdysone, Ecdysterone, Makisterone A, Giloinsterol ) |
Immunomodulator and For neurodegenerative disorder IgA neuropathy, |
(52-53) |
Root,
Whole Plant |
Others (3,[a,4-di hydroxy-3- methoxy-benzyl]-4-[4- compounds hydroxy-3- methoxy-benzyl]- tetrahydrofuran, Jatrorrhizine, Tinosporidine, Cordifol, Cordifelone, Giloinin, Giloin, N-transferuloyltyramine as diacetate, Tinosporic acid) |
Anti-viral infections, Anticancer, Anti-inflammatory Protease inhibitors for HIV and drug resistant HIV |
(54-57)
|
Few studies support the evidence for activation of macrophages like NFêB translocation and cytokine synthesis followed by activation of the immune system in HIV positive patients and Infectious bursal disease in young chicks were also get relief by its effect 29–31. The studies have supported that it is involved to activate macrophages like NFêB translocation and inflammatory synthesis, even though, ultimately, activates the immune system and Human immunodeficiency virus (HIV) positive patients and Infectious bursal disease in young chicks were also get relief by its effect29–31.
Ashwagandha– Withania somnifera (W.S.)
Ashwagandha (figure-3) is well-known as Withania somnifera (W.S.), which means ’Smell of Horse,’ which may be because of two main reasons 1) the root itself smells like a horse, 2) the root is supposed to imbibe with the strength and virility of a horse. It brings into being Kamashakti as like horse in human and its plant bears smell like a horse. Except for this use, various traditional reports have supported that W.S is very useful in treating several diseases like inflammation, cancer, stress, fatigue, diabetes, and cardiovascular complications. W.S includes an analgesic, astringent, antispasmodic, and immune-stimulant compound, with particular emphasis on a stress-related suppression in immunity. Some studies have shown that its adaptogenic usage is emphasized for stress-insomnia, incapacity, and nervous fatigue58.
In the Indian medicinal or therapeutic system, Withania somnifera is a beneficial medicinal plant used to treat various ailments but is mainly concerned with neuro-related diseases such as stress or insomnia, depression, anxiety, neurodegenerative 58. W.S is also supportive medicine for age-related disease; for better improvement of the immune-system, historically, it involves detoxification when consumed its root extract with food 58–60.
Kumkum (Crocus sativusZafran) Saffron
In India, Kumkum Saffron (Crocus sativus) (figure-4) is a valuable or exclusive, expensive species61. Iranian medical books have major descriptions of Saffron (the dried and dark red stigma) of Crocus sativus Zafran. Crocus sativus Zafran is a famous medicinal plant globally and cultured in many countries like Central Asia, Europe, India, Turkey, and China. It contains bioactive molecules safranol, crocin, and picrocrocin include 63 percent sugar, 12 percent proteins, 5 percent fat, 5 percent minerals, 10 percent water, few fibers, and including anthocyanins, glycosides, alkaloids, and some flavonoids, including quercetin 61-62. In vitro & In vivo studies have shown its potent anti-aging 62, anti-inflammatory63, antioxidant64, antimicrobial 17,65 anti-asthmatic, anti-viral, and free radical scavenging effect. Additionally, it acts as a cardio-protective, neuro-protective nephron-protective, and hepatoprotective herbal remedy 62-65. It acts as a potent immune booster on humoral and cellular immunity65. Several studies suggested that crocin and crocetin (major bioactive components of Saffron) are affecting the natural killer cell (NK-cell) activity or could increase NK-cell activity in the elderly; through this mechanism, both are involved in modulating the immune response 65. On the other hand, different in vivo and in vitro supportive findings explained its immunoregulatory (figure-4b) properties and constituents. Therefore, compounds with immunoregulatory properties may help prevent and treat such communicable diseases and affect COVID-19 treatment.
Rasona[Allium sativum]Garlic
Rasona or Garlic is known as Lehsun (figure-5), (tunicate bulb belongs to Lillaceae family, is hot and dry in temperament as per the Greco-Arab system of medicine 66. From ancient times due to blistering properties, lehsun helps recover from respiratory damage, cough problem, arthritis, and constipation. It protects from epidemic diseases 69because of its active chemical ingredients -alliin, allicin, ajoene, vinyldithin, S-allylcycsteine, and diallyl sulfides 67.
Multiple studies have documented its biological effects: antioxidant, anti-inflammatory, immunomodulatory, antibiotic, bacteriostatic, antifungal, anti-viral, antihelminthic, antithrombic, hypotensive, hypoglycemic, and hypercholesterolemic, as well as anti-influenza B, herpes simplex, and coxsackievirus anti-viral activity69-70. Some studies reported that Garlic molecules could modulate cytokine expression in lipopolysaccharide-activated human blood and inhibit NF-êB from which show its immune-modulatory effect is evident 71.
It is also involved in activating macrophages, promoting immunoglobulins71, and reducing polymorphonuclear cells’ migration through endothelial cell layers 72. Some studies have observed that it involved inhibiting the fabrication of NO and prostaglandin-E2, suppresses inducible form of NO synthase and COX-2 expression, decreases the production of inflammatory cytokines like TNF-a, interleukin 6, and
interferon ã 70-72. Some other finding has supported that involved to improve lung function in smokers,73 and reduce tracheal exudates in horses 73.
On the other hand, in the case of COVID-19, proinflammatory cytokines ( Tumor necrosis factor-alpha (TNF-á), interleukin-1 (IL-1), interleukin-2 (IL-2), interleukin-6 (IL-6), interferon-gamma (IFN-ã) and leptin) are among those exhibiting an increasing tendency. In contrast, Garlic contains countless compounds that have the potential to influence immunity, and alternately, are involved in the modulation of cytokine secretion, which may provide a mechanism of action for many of its therapeutic effects and other infectious disease treatment73. Therefore, Garlic has beneficial effects immunological and hormonal, causes significant increases in CD4 + and CD8 + cells, and stimulates NK. Additionally, cells decreased leptin, leptin receptor, peroxisome proliferator-activated receptor-gamma (PPAR-ã), and IL-6 concentrations73. Through this beneficial effect of Garlic play an essential role in the treatment of patients with COVID-19 infection.
Shunthi[Adraka, Zingiberofficinale]Ginger
According to the Greco-Arab medicine system, ginger or adrak(figure-6) is a rhizome, [brown color and bittertaste] has the hot and dry temperament and is commonly used an appetizer and general tonic. It has been used to treat anorexia, flatulence, high blood pressure, arthritis, and the common cold from the ancient period. It contains many bioactive molecules that act as immunomodulators like zingiberine, gingerol, gingerdione, shogaol, paradol, hexahydrocurcumin, and gingerenone A 74.
Some studies have supported that its extract is involved in reducing oxidative stress and inhibit superoxide production; it also affects anti-inflammatory and analgesic activities 58. Recently a report has observed that its effect against swine flu [H1N1] and human respiratory syncytial virus in human respiratory tract cell lines 75-77 and including bronchodilator effect ameliorates allergic
asthma74-79.
Numerous studies have suggested that gingerols showed anti-viral properties against the human respiratory syncytial virus on HEp2 and A549 cell line and participate in promoting bronchodilation (relieving fluid in the lungs, preventing lung fibrosis, naso or trachea/pharyngeal clogging, alleviating cough, and also relieving chest pain, and make easy to breathing). Thus, its active molecules play a promising role against the novel COVID-19 for blocking receptor binding and viral phagocytotic internalization of the SARS-CoV-2 (the binding of the S- (spike) protein to the angiotensin-converting enzyme 2 (ACE-2) receptors) and significantly relieving to COVID-19 patients79.
Shirisa (Albizialebbek)
Generally known as Shirishor Siris (figure-7) contain Albigensian, albiziagenin, lebbecacidin, myricitrin as major chemical constituents, and bioactive molecules, its effect as an anti-inflammatory, antihistamine, antioxidant, and anticancer activities27,80-83.
Both in vivo and in vitro studies have shown the immunomodulatory effect (on the humoral and cell-mediated immune responses) of the bark of Albizzia lebbeck (Sirisha). Numerous studies have shown that hyperinflammatory response (cytokine Strom) sign of COVID-19; therefore, reducing inflammatory markers could be considered a potential therapeutic target against COVID-19. Thus, Albizzia lebbeck is involved to decreases the level of pro-cytokines – leptin, leptin receptor, peroxisome proliferator-activated receptor-gamma (PPAR-ã), and IL-6. On the other hand, the extract of sirisha increased the serum antibody titer, several plasma cells in the spleen with a concomitant decrease in DTH response, MPO enzyme activity in the spleen, and serum level of TNF-alpha, IFN-g, and IL-6 in rodent studies81-83.
Haritaki [Terminalia chebula]
It is known as Haritakior Harad (King of medicine’ in Tibet) (figure-8) and is mentioned in Ayurvedic literature, Rasayana, Tridoshahara indicated for Swasaand Kasa. Various studies isolated some essential bioactive ingredients such aschebulinic acid, anthraquinone, arachidic acid, etc., along with other phytoconstituents such as polyphenols, terpenes, anthocyanins, flavonoids, alkaloids, and glycosides. They play a significant role as antibacterial, antispasmodic, antioxidant, and immunomodulatory activities 25,84-88. The previous finding supported that its fruits hold various active chemical compounds responsible for different health benefits like geriatric diseases and improve memory and brain function. It is useful to treat various clinical diseases like cancer, cardiovascular diseases, paralysis, leprosy, ulcers, gout, arthritis, epilepsy, cough, fever, diarrhea, gastroenteritis, skin disorders, urinary tract infection, and wound infections84-86. Therefore, its extract contains various active compounds involved in the treatment of diabetes, bacterial and fungal infections, immunodeficiency diseases, hyperlipidemia, and possess other pharmacological beneficial effects87-88, so that its supplement might be responsible for fights against COVID-19
Pippali[Piper longum]
Pippali (figure-9) is locally famous as Long pepper, and with a long history, it is mentioned in Atharvaveda, Pippaliis mentioned as Rasayana. Acharya Charaka and Susruta. Numerous finding has supported that it acts as anti-inflammatory, antispasmodic and also showed immunomodulatory properties. Its chemical profile contains various significant active molecules like Piperine, Piperine, pellitorine, caryophyllene, etc., as major chemical constituents 27,89-91. The extract of P. longum was significantly involved in improving the immune system through activating macrophages in vitro study. In another in vitro study, its extract caused an increase in the total WBC count, Bone marrow cellularity, along with á-esterase positive cells. Some other findings supported its immunomodulatory property and were proven by using the macrophage migration index (MMI), haem-agglutination titre (H.A.), and phagocytic index (P.I.) in mice90-91. Numerous studies have proven that SARS-CoV-2 mainly damages the immune system and induces oxidative stress, cause damage to airway epithelial cells.
Additionally, an increased cytokine level causes damages to tissue damage of the lungs, kidney, and heart and leads to severe complications like respiratory failure, etc91. Other findings supported that the extract of P. longum as a supplement acts as anti-viral activity against different viruses, reduced stress, anti-inflammatory effect, and reduced the level of cytokines89. Therefore, the previous finding supported that it might take responsibility to inhibit the replication of SARSCov-2 RNA(COVID-19) in the host cells to benefit COVID-19 prevention.
Amra[Mangiferaindica]
Mangifera Indica or Amraor Mango (figure-10) contain mangiferin, quercetin, gallic acid as chemical constituents and show anti-viral and anti-influenzal activities 27,92-97. However, numerous studies have explained its inhibitory activity against other than COVID-19 19, infectious disease, causing viruses viz., Human Immunodeficiency Virus (HIV), Herpes Simplex Virus (HSV) (C) Hepatitis B Virus (HBV), Epstein-Barr Virus (EBV), Foot and Mouth Disease Virus (FMDV), Newcastle disease virus (NDV) etc92. Another finding also supported that its bioactive molecules have been shown to inhibit cyclophosphamide bone marrow suppressive effect and phagocytic activity in rodents and thus possess as herbal immunomodulatory. It could be responsible for inhibiting SARS-Corona Virus replication97.
Tulsi [Ocimum sanctum]
Locally famous name as Tulsi or Holy basil (figure-11) contains bornyl acetate, cadinene, camphene, eugenol, and limonene as major chemical constituents. Previous scientific studies have shown that Tulsi is antimicrobial, hypoglycemic, hypolipidemic, hepatoprotective, immunomodulatory, antioxidant, anticarcinogenic, analgesic, wound healing, anti-inflammatory, antipyretic, antidiarrheal, anti-asthmatic, anti-ulcerogenic and adaptogenic,98-100.
The most active components are eugenol and eugenol epoxide and may prevent HIV-1 replication with two different mechanisms (increase in IL-4, IgE, PLA2, and T.P. levels, all pathological lung indices as well as a significant decrease in IFN-ã/IL-4 ratio) was seen in the asthmatic compared to the control rats. The most active components are eugenol and eugenol epoxide and may prevent HIV-1 replication with two different mechanisms (increase in IL-4, IgE, PLA2, and T.P. levels, all pathological lung indices as well as a significant decrease in IFN-ã/IL-4 ratio) was seen in the asthmatic compared to the control rats. Its extract causes decreased IL-4, IgE, PLA2, and T.P. levels, whereas increased IFN-ã/IL-4 ratio in vivo study. Higher concentrations of the extract were significantly more than those of dexamethasone. Its extract causes pathological changes, immunological and inflammatory markers, and acts as a potent anti-viral activity against hepatitis B virus, adenoviruses, African swine fever virus, and some RNA viruses in vitro study 100. Therefore, its active phytochemicals are predicted to have the potential to be repurposed as anti- COVID-19 herbal immunomodulatory or ayurvedic therapeutics.
Mandukaparni [Centellaasiatica]
It is commonly known as Mandukaparnior Gotukola (figure-12); contains Asiatic acid, centric acid, centellic acid, carotene, betulinic acid, thankunic acid, and isothankunic acid, etc. as major chemical constituents. Furthermore, there are some other triterpenes like brahmic acid, centellin, centellicin, asiaticin, bayogenin, terminolic acid, 3²,6²,23-trihydroxyolean-12-en-28-oic acid, 3²,6²,23-trihydroxyurs-12-en-28-oic acid, 3-O-[±-L-arabinofuranosyl] 2±,3²,6²,23-± tetrahydroxyurs-12-en-28-oic acid, centellasapogenol A, centellasaponins A-D, ursolic acid, pomolic acid, 3-epimaslinic acid, 23-O-acetylmadecassoside, and 23-O-acetylasiaticoside B101
Various studies suggested that Centella asiatica displayed antioxidative, anti-inflammatory, and effective drug for neurodegenerative disease induced by glutamate- or b-amyloid 102. Its inhibitory activities against fibroproliferative disorders (Keloids) through blocking the TGF-â/Smad pathway and major active molecules suppress HIV-1 LTR transcription and viral replication (vital function to inhibit herpes simplex virus)103. on the previous anti-viral effect, it might be responsible for inhibiting the replication of COVID-19. It has established Rasayana[anabolic], analgesic, and immunomodulatory properties. It has been mentioned as MedhyaRasayanain Ayurvedic classics 101.
Additionally, the potential use as immunomodulatory agents, modes of action, and plant origin important a plant-based compound has also been comprehensively target point for curtail of COVID-19. Several previous finding has supported that some compounds, other than phytochemicals including essential oils, steroids, terpenoids, phenolics, pigments, ûavonoids, and alkaloids, etc. have exhibited immunomodulatory effect. Plant-derived compounds showing promising potential as immunomodulatory agents against COVID-19 are listed in table-2.
Table (2):
Herbal immune-modulator with their source and mechanism with proven evidence.
S.No. |
Plants name |
Bioactive molecules [Immunomodulators] |
Immunomodulatory Mechanism |
Reference |
|---|---|---|---|---|
1. |
Coptischinensis Franch |
Alkaloids Berberine |
Down-regulate the development of T-helpercellscytokines[Th1(TNF-a,IL-2),andTh2 (IL-4)] |
104 |
2. |
Chelidonium majus L. |
Chelerythrine |
Inhibit the release of PGE2 by regulation of cyclooxygenase-2 activity |
105 |
3. |
Piper longum Linn |
Piperine |
Reduce the levels of IL-1β, IL-6, and TNF-alpha proinflammatory cytokines. COX-2, NOS-2, and NF-3B expressions are governed by Down. Inhibit eicosanoide generation by inhibiting phospholipase A2 and TXA2 synthase activity. |
106-107 |
4. |
Picea crassifolia |
Chrysin |
Inhibited production of cytokine pro – inflammatory (TNF, IL-1β, and IL-1β) IL-6’s). By intracellular calcium modulation, the release of histamine from mast cells is decreased. |
108-109 |
5. |
Lithospermum erythrorhyzon Sieb. etZucc. |
Shikonin |
Inhibit NF-κB activity,inhibit Th1 cytokines expression and induce Th2 cytokines |
110
|
6. |
Nigella sativa L. |
Thymoquinone |
The LPS-induced proliferation of fibroblasts prevented and H2O2-induced Generation of 4-hydroxynonenal. IL-1β, TNF-a, MMP-13, COX-2, and PGE2 are inhibited when blocking MAPK p38, ERK1/2, and NF-kBp65 phosphorylation. |
111 |
7. |
Andrographis paniculata |
Terpenoid 14-deoxyandrographolide |
Enhanced lymphocyte proliferation. Enhanced activation of IL-2 in lymphocytes. |
112 |
8. |
Tripterygium wilfordii |
Demethylzelasteral |
It prevents vascular endothelial cell proliferation. |
111 |
9. |
Luffa cylindrica |
Echinocystic acid |
Enhanced phagocytic index of macrophages in humoral and cell-mediated immune responses |
112 |
10. |
Tripterygium wilfordii |
Demethylzelastera |
Inhibits vascular endothelial cell proliferation |
113 |
11. |
Luffa cylindrica, Phytolacca americana |
Oleanolic acid |
Reduce the level of IL-1 alpha, IL-6, and TNF-alpha and their effect on the complement pathway through C3 convertase inhibition. Inhibits the activity of adenosine deaminase |
114 |
12. |
Panax ginseng |
Ginsang |
Enhances the production of cytokines and species of reactive oxygen by Including macrophages. The phagocytic behavior of macrophages is induced. |
115 |
13. |
Tripterygium wilfordii |
Triptolide |
Inhibit recruitment of lymphocytes and hereditary proinflammatory cytokines (IL-2, iNOS, TNF-alpha, COX-2, and IFN-γ) expressions. It also prevents transcription factors such as NF-kB, NFAT, and STAT3 from activating. |
116 |
14. |
Tripterygium wilfordii |
Celastrol |
Inhibit proinflammatory cytokine expression, adhesive molecules, Regulation of the proteasome, and topoisomerase II. |
117 |
Despite the few vaccines against COVID-19 available in the market; still, a psychological stigma against COVID-19 remains the same. To date, there are no satisfactory remedial measures against COVID-19 treatment, early diagnostic tools, and efficient treatment modalities in the modern health system warrants extensive research. Further, the pharmaceutical industries face serious challenges towards the drug discovery/vaccine development against highly versatile COVID-19. There is an urgent need to develop new drugs and Immunomodulators in combination with traditional knowledge validated through modern science against the treatment and management of COVID-19. These herbal immune-modulators drugs show a promising approach to promote the immune system and healthy living. However, necessary to use herbal medicinal products to alleviate COVID-19’s burden and promote healthy living. These traditional medicines are regarded as new and promising sources of potential anti-viral drugs. These encouraging preclinical and clinical trials suggested that the traditional medicines are promising drug candidates in the treatment and management of COVID-19. In a nutshell, to obtain better therapeutic benefits, less expensive, and minor adverse reactions, search for alternative medicine from the natural source, i.e., herbal remedies, used traditionally, now documented with the safety profile. It is high time to validate herbal medicines as primary or adjuvant therapy to prevent and manage COVID-19 infection.
Further, extensive research is required to validate herbal Immunomodulators and their molecular mechanisms in in-silico, preclinical, and well-designed clinical trials. It would be interesting to design future therapeutic approaches for immunomodulatory pathways with a synergistic combination of natural herbal drugs.
ACKNOWLEDGMENTS
We are grateful to all author’s affiliations, including health organizations and Corona Warriors, who provide great help to peoples.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
This review was carried out in collaboration with all the authors, and they have fully contributed to the literature review planed, preparation, and editing of the manuscript.
FUNDING
None
ETHICS STATEMENT
Not applicable.
AVAILABILITY OF DATA
All datasets generated or analyzed during this study are included in the manuscript and/or the Supplementary Files.
- Baxter D. Active and passive immunity, vaccine types, excipients and licensing. Occup Med. 2007;57(8):552-556.
Crossref - Vesely MD, Kershaw MH, Schreiber RD, Smyth MJ. Naturalinnate and adaptive immunity to cancer. Annu Rev Immunol. 2011;29:235-271.
Crossref - Moradali MF, Mostafavi H, Ghods S, Hedjaroude GA. Immunomodulating and anticancer agents in the realm ofmacromycetes fungi [macrofungi]. Int Immunopharmacol.2007;7(6):701-724.
Crossref - Oberlies NH, Kroll DJ. Camptothecin and taxol: historic achievements in natural products research. J Nat Prod. 2004;67(2):129-135.
Crossref - Lai C, Shih TP, Ko WC, Tang HJ, Hsueh PR. Severe acute respiratory syndrome coronavirus 2 [SARS-CoV-2] and coronavirus disease-2019 [COVID-19]: The epidemic and the challenges. Int J Antimicrob Agents. 2020;55:105924.
- Shereen MA, Khan S, Kazmi A, Bashir N, Siddique R. COVID-19 infection: origin, transmission, and characteristics of human corona viruses. Journal of Advanced Research. 2020;24:91-98.
Crossref - Ren L, Y-M, Wu Z-Q, et al. Identication of a novel coronavirus causing severe pneumonia in human: a descriptive study. Chin Med J. 2020;133(9):1015-1024.
Crossref - Zhou P, Yang X-L, Wang X-G, et al. A pneumonia out break associated with a new coronavirus of probable bat origin. Nature. 2020;579:270-273.
Crossref - Gallagher TM, Buchmeier MJ. Coronavirus spike proteins in viral entry and pathogenesis. Virology. 2001;279(2):371-374.
Crossref - Tooze J, Tooze SA. Infection of AtT20 murine pituitary tumour cells by mouse hepatitis virus strain A59: virus budding is restricted to the Golgi region. Eur J Cell Biol. 1985;37:203-212
- Klumperman J, Locker JK, Meijer A, Horzinek MC, Geuze HJ, Rottier PJ. Coronavirus M proteins accumulate in the Golgi complex beyond the site of virion budding. J Virol. 1994;68(10):6523-6534.
Crossref - Krijnse-Locker J, Ericsson M, Rottier PJ, Griffiths G. Characterization of the budding compartment of mouse hepatitis virus: evidence that transport from the RER to the Golgi complex requires only one vesicular transport step. J Cell Biol. 1994;124(1):55-70.
Crossref - Opstelten DJ, Raamsman MJ, Wolfs K, Horzinek MC, Rottier PJ. Envelope glycoprotein interactions in coronavirus assembly. J Cell Biol. 1995;131(2):339-349.
Crossref - Bassetti M, Vena A, Giacobbe DR. The Novel Chinese Coronavirus [2019-nCoV] Infections: challenges for fighting the storm. Eur J Clin Invest. 2020;50(3)e13209.
Crossref - Finlay BB, Hancock REW. Can innate immunity be enhanced to treat microbial infections? Nat Rev Microbiol. 2004;2(6):497-504.
Crossref - Li G, De Clercq E. Therapeutic options for the 2019 novel coronavirus [2019-nCoV]. Nat Rev Drug Discov. 2020;19:149-150.
Crossref - Gautret P, Lagier J-C, Parola P, et al. Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial. Int J Antimicrob Agents. 2020;56(1):105949.
Crossref - Cynthia Z, Qiongqiong L, Yingzhu, et al. Research and Development on therapeutic agents and vaccines for COVID-19 and related human coronavirus diseases. ACS Cent Sci. 2020;6(3):315-331.
Crossref - Chakraborty C, Sharma AR, Bhattacharya M, Sharma G, Lee SS. The 2019 novel coronavirus disease [COVID-19] pandemic: A zoonotic prospective. Asian Pac J Trop Med. 2020;13(6):242-246.
Crossref - Ranjan A, Chauhan A, Gurnani M, Jindal T. Potential Phytochemicals as Efficient Protease Inhibitors of 2019-nCoV. Preprints. 2020;2020:040240.
Crossref - Pandey DN. Seven shields of Ayurveda between health and diseases. Ann Ayurvedic Med. 2019;8(1-2):6-10.
Crossref - Guo YR, Cao QD, Hong ZS, et al. The origin, transmission and clinical therapies on coronavorus disease 2019 [COVID-19] outbreak-an update on the status. Mil Med Res. 2020;7(1):11.
Crossref - Cascella M, Rajnik M, Cuomo A, et al. features, evaluation and treatment coronavirus [COVID-19] [updated 2020 Mar 20]. In: Statpearls [internet]. Treasure Island [FL]: Statpearls Publishing. 2020 Jan.
- Singh K, Verma B. ‘The Concept of Vyadhikshamatva in ayurveda’. Ayurpharm- International Journal of Ayurveda and Allied Sciences. 2012;1(5):99-108.
- World Health Organization. “Q&A on coronaviruses” [https://www.who.int/news-room/q-a-detail/q-acoronaviruses]. April 23, 2020. Retrieved April 23, 2020.
- Anonymous. “Coronavirus Disease 2019 [COVID-19] – Transmission” [https://www.cdc.gov/coronavirus/201 9-ncov/prepare/transmission.html]. Centers for Disease Control and Prevention. Updated on April 13, 2020. Retrieved April 23 2020.
- Sharma RK, Dash B. Caraka Samhita, Chowkhamba Sanskrit Series Office, Varanasi, Vol-II. 2013:140.
- Singhal GD, Susruta Samhita, Chaukhamba Sanskrit Pratishthan, Delhi,Vol-I, 2007:60.
- Sharma RK, Dash B. Caraka Samhita, Chowkhamba Sanskrit Series Office, Varanasi, Vol-III , 2013:8.
- Sastry JLN, Dravyaguna Vigyana, Chaukhamba Orientalia, Varanasi, Vol-II, 2010:33-49.
- Sharma RK, Dash B., Caraka Samhita, Chowkhamba Sanskrit Series Office, Varanasi, Vol-III. 2013:46.
- Nagarkatti DS, Rege NN, Desai NK, Dahanukar SA. Modulation of Kupffer cell activity by Tinospora cordifolia in liver damage. J Postgrad Med. 1994;40:65-67.
- Nair PK, Rodriguez S, Ramachandran R, et al. Immune stimulating properties of a novel polysaccharide from the medicinal plant Tinospora cordifolia. Int Immunopharmacol. 2004;4(13):1645-1659.
Crossref - Sachan S, Dhama K, Latheef SK, et al. Immunomodulatory potential of Tinospora cordifolia and CpG ODN [TLR21 Agonist] against the very virulent infectious Bursal disease virus in SPF Chicks. Vaccines [Basel]. 2019;7(3):106.
Crossref - Upadhaya AK, Kumar K, Kumar A, Mishra HS. Tinospora cordifolia [Willd.] Hook. F. and Thoms. [Guduchi]-alidation of the Ayurvedic pharmacology through experimental and clinical studies. Int J Ayurveda Res. 2010;1(2):112-121.
Crossref - Rout GR. Identification of Tinospora cordifolia [Willd.] Miers ex Hook F & Thoms using RAPD markers. Z Naturforsch C. 2006;61:118-122.
Crossref - Patel SS, Shah RS, Goyal RK. Antihyperglycemic, anti-hyperlipidemic and antioxidant effects of Dihar, a poly herbal ayurvedic formulation in streptozotocin induced diabetic rats. Indian J Exp Biology. 2009;47:564-570.
- Gupta R, Sharma V. Ameliorative effects of Tinospora cordifolia root extract on histopathological and biochemical changes induced by aflatoxin-b [1] in mice kidney. Toxicol Int. 2011;18(2):94-98.
Crossref - Jagetia GC, Rao SK. Evaluation of the antineoplastic activity of guduchi [Tinospora cordifolia] in ehrlich ascites carcinoma bearing mice. Biol Pharm Bull. 2006;29(3):460-466.
Crossref - Patel MB, Mishra S. Hypoglycemic activity of alkaloidal fraction of Tinospora cordifolia. Phytomedicine. 2011;18(12):1045-1052.
Crossref - Sriramaneni RN, Omar AZ, Ibrahim SM, Amirin S, Mohd ZA. Vasorelaxant effect of diterpenoid lactones from and rographis paniculata chloroform exract on rat aortic rings. Pharmacognosy Res. 2010;2(4):242-246.
Crossref - Yang S, Evens AM, Prachands S, et al. Mitochondrial-mediated apoptosis in lymphoma cells by the Diterpenoid lactone and rographolide, the active component of and rographis paniculata. Clin Cancer Res. 2010;16(19):4755-4768.
Crossref - Zhao F, He EQ, Wang L, Liu K. Anti-tumor activities of and rographolide, a diterpene from And rographis paniculata, by inducing apoptosis and inhibiting VEGF level. J Asian Nat Prod Res. 2008;10(5-6):467-473.
Crossref - Kohno H, Maeda M, Tanino M, et al. A bitter diterpenoid furano lactone columbine from calumbae Radix inhibits azoxy methane-induced rat colon carcinogenesis. Cancer Let. 2002;183(2):131-139.
Crossref - Dhanasekaran M, Baskar AA, Ignacimuthu S, Agastian P, Duraipandiyan V. Chemopreventive potential of Epoxy clerodane diterpene from Tinospora cordifolia against diethyl nitrosamineinduced hepyocellular carcinoma. Invest New Drugs. 2009;27(4):347-355.
Crossref - Ly PT, Singh S, Shaw CA. Novel environmental toxins: Steryl glycosides as a potential etiological factor for age- related neurodegenerative diseases. J Nrurosci Res. 2007;85(2):231-237.
Crossref - Karpova EA, Voznyi YV, Dudukina TV, Tsvetkva IV. 4-Trifluoromethylumbelliferyl glycosides as new substrates form revealing diseases connected with hereditary deficiency of lysosome glycosidases. Biochem Int. 1991;24:1135-1144.
- Kapil A, Sharma S. Immunopotentiating compounds from Tinospora cordifolia. J Ethopharmacol. 1997;58(2):89-95.
Crossref - Chen S, Wu K, Knox R. Structure-function studies of DT-diaphorase [NQO1] and NRH: Quinone oxidoreductase [NQO2]. Free Radic Biol Med. 2001;29(3-4):276-284.
Crossref - Badwin AS. Control of oncogenesis and cancer therapy resistance by the transcription factor NF-kappa B. J of Clin Invest. 2001;107(3):241-246.
Crossref - Yang JH, Kondratyuk TP, Marler LE, et al. Isolation and evaluation of kaempferol glycosides from the fern neocheiropteris palmatopedata. Phytochemistry. 2010;71(5-6):641-647.
Crossref - Kim SK, Kim HJ, Choi SE, Park KH, Choi HK, Lee MW. Antioxidative and inhibitory activities on nitric oxide [NO] and prostaglandin E2 [COX-2] production of flavonoids from seeds of prunus tomentosa Thunberg. Arch Pharm Res. 2008;31(4):424-428.
Crossref - Lv J, Xu D, Perkovic V, et al. Corticosteroid therapy in IgA nephropathy. J Am Soc Nephrol. 2012;23(6):1108-1116.
Crossref - McKeown E, Bykerk VP, Deleon F, et al. Quality assurance study of the use of preventative therapies in glucocorticoid-induced osteoporosis in early inflammatory arthritis: Result from the CATCH cohort. Rheumatology [Oxford]. 2012;51(9):1662- 1669.
Crossref - Sundarraj S, Thangam R, Sreevani V, et al. ϒ-Sitosterol from acacia nilotica L. induces G2/M cell cycle arrest and apopyosis through c-Myc suppression in MCF-7 and A549 cells. J Ethnopharmcol. 2012;141(3):803-809.
Crossref - Maurya R, Handa SS. Tinocordifolin, a sesquiterpene from Tinospora cordifolia. Phytochem. 1998;49(5):1343-1345.
Crossref - Thippeswamy G, Sheela ML, Salimath BP. Octacosanol isolated from Tinospora cordifolia downregulates VEGF gene expression by inhibiting nucular translocation of NFB and its DNA binding activity. Eur J Pharmcol. 2008;588(2-3):141-150.
Crossref - Mishra LC, Singh BB, Dagenais S. Scientific basis for the therapeutic use of Withania somnifera [ashwagandha]: A review. Altern Med Rev. 2000;5(4):334-346.
- Murthy MRV, Ranjekar PK, Ramassamy C, Deshpande M. Scientific basis for the use of Indian ayurvedic medicinal plants in the treatment of neurodegenerative disorders: Ashwagandha. Cent Nerv Syst Agents Med Chem,2010;10(3):238-246.
Crossref - Kumari R, Agrawal A, Singh KP, Singh GPI, Dubey GP. Chapter 28 Regulation of Serotonin in Depression: Efficacy of Ayurvedic Plants- Taylor & Francis Group, 6000 Broken Sound Parkway NW, Suite 300, Boca Raton, FL 33487-2742 CRC Press. 2016;397-420.
- Gohari AR, Saeidnia S, Mahmoodabadi MK. An overview on saffron, phytochemicals, and medicinal properties. Pharmacogn Rev. 2013;7(13):61-66.
Crossref - Eslami M, Bayat M, Mozaffari Nejad ASM, Sabokbar A, Anvar AA. Effect of polymer/nanosilver composite packaging on long-term microbiological status of Iranian saffron [Crocus sativus L]. Saudi J Biol Sci. 2016;23(3):341-347.
Crossref - Soleymani S, Zabihollahi R, Shahbazi S, Bolhassani A. Antiviral Effects of Saffron and its Major Ingredients. Current Drug Delivery. 2018;15(5):698-704.
Crossref - Razavi BM, Hosseinzadeh H. Saffron as an antidote or a protective agent against natural or chemical toxicities. Daru. 2015;23:31.
Crossref - Zirak MZ, Rezaee SA, Karimi G, and Hosseinzadeh H. Immunoregulatory and anti-inflammatory properties of Crocus sativus [Saffron] and its main active constituents: A review. Iran J Basic Med Sci. 2019;22(4):334-344.
- Shang A, Cao SY, Xu XY, et al. Bioactive compounds and biological functions of garlic [Allium sativum L.]. Foods. 2019;8(7):246.
Crossref - Tsai Y, Cole LL, Davis LE, Lockwood SJ, Simmons V, Wild GC. Antiviral Properties of Garlic: In vitro Effects on Influenza B, Herpes Simplex and Coxsackie Viruses. Planta Med. 1985;51(5):460-461.
Crossref - Schafer G, Kaschula CH. The immunomodulation and anti-inflammatory effects of garlic organosulphur compounds in cancer chemoprevention. Anticancer Agents Med Chem. 2014;14(2):233-240.
Crossref - Hofbauer R, Frass M, Gmeiner B, Kaye AD, Frost EA. Effects of garlic extract [Allium sativum] on neutrophil migration at the cellular level. Heart Dis. 2001; 3(1):14-17.
- Saastamoinen M, Sarkijarvi S, Hyyppa S. Garlic [Allium sativum] supplementation improves respiratory health but has increased risk of lower hematologic values in horses. Animals [Basel]. 2019;9(1):13.
Crossref - Lee DY, Li H, Lim HJ, Lee HJ, Jeon R, Ryu JH. Anti-inflammatory activity of sulfur- containing compounds from garlic. J Med Food. 2012;15(11):992-999.
Crossref - Lissiman E, Bhasale AL, Cohen M. Garlic for the common cold. Cochrane Database Syst Rev. 2014;2014(11):CD006206.
- Hsieh CC, PengWH, Tseng HH, Liang SY, Chen LJ, Tsai JC. The protective role of garlic on allergen-induced airway inflammation in mice. Am J Chin Med. 2019;47(5):1099-1112.
Crossref - Young HY, Luo YL, Cheng HY, Hsieh WC, Liao JC, Peng WH. Analgesic and anti-inflammatory activities of [6]-gingerol. J Ethnopharmacol. 2005;96(1-2):207-210.
Crossref - Chang JS, Wang KC, Yeh CF, Shieh DE, Chiang LC. Fresh ginger [Zingiber officinale] has anti-viral activity against human respiratory syncytial virus in human respiratory tract cell lines. J Ethnopharmacol. 2013;145(1):146-151.
Crossref - Denyer CV, Jackson P, Loakes DM, Ellis MR, Young DA. Isolation of antirhinoviral sesquiterpenes from ginger [Zingiber officinale]. J Nat Prod. 1994;57(5):658-662.
Crossref - Townsend EA, Siviski ME, Zhang Y, Xu C, Hoonjan B, Emala CW. Effects of ginger and its constituents on airway smooth muscle relaxation and calcium regulation. Am J Respir Cell Mol Biol. 2013;48(2):157-163.
Crossref - Cifci A, Tayman C, Yakut Hi, et al. Ginger [Zingiber officinale] prevents severe damage to the lungs due to hyperoxia and inflammation. Turk J Med Sci. 2018;48(4):892-900.
Crossref - Khan AM, Shahzad M, Raza Asim MB, Imran M, Shabbir A. Zingiber officinale ameliorates allergic asthma via suppression of Th-2-mediated immune response. Pharm Biol. 2015;53(3):359-367.
Crossref - Barua CC, Gupta PP, Patnaik GK, et al. Immunomodulatory effect of albizzia lebbeck. Pharm Biol. 2000;38(3):161-166.
Crossref - Shashidhara S, Bhandarkar AV, Deepak M. Comparative evaluation of successive extracts of leaf and stem bark of Albizzia lebbeck for mast cell stabilization activity. Fitoterapia. 2008;79(4):301-302.
Crossref - Joycharat N, Thammavong S, Limsuwan S, et al. Antibacterial substances from Albizia myriophylla wood against cariogenic Streptococcus mutans. Arch Pharm Res. 2013;36:(6):723-730.
Crossref - Shaikh FK, Gadge PP, Shinde AA, et al. Novel isoforms of proteinaceous α-amylase inhibitor (α-AI) from seed extract of Albizia lebbeck. Acta Physiologiae Plantarum.2013;35:3:901-909.
Crossref - Aher V, Wahi AK. Immunomodulatory effect of alcoholic extract of Terminalia chebula ripe fruits. J Pharm Sci Res. 2010;2:539-544.
- Shivaprasad HN, Kharya MD, Rana AC, Mohan S. Preliminary immunomodulatory activities of aqueous extract of Terminalia chebula. Pharm Biol. 2006;44:32-34.
Crossref - Sehar I, Kaul A, Bani S, Pal HC, Saxena AK. Immune up regulatory response of a non-caloric natural sweetener, stevioside. Chemico-Biol Interac. 2008;173(2):115-121.
Crossref - Belapurkar P, Goyal P, Tiwari-Barua P. Immunomodulatory effects of triphala and its individual constituents: a review. Indian J Pharm Sci. 2014;76(6):467-475.
- Elizabeth KM. Antimicrobial activity of Terminalia bellerica. Indian J Clin Biochem. 2005;20(2):150-153
Crossref - Sunila ES, Kuttan G. Immunomodulatory and antitumor activity of Piper longum Linn. and piperine. J Ethnopharmacol. 2004;90(2-3):339-346.
Crossref - Shenoy PA, Nipate SS, Sonpetkar JM, Salvi NC, Waghmare AB, Chaudhari PD. Anti-snake venom activities of ethanolic extract of fruits of Piper longum L. (Piperaceae) against Russell’s viper venom: characterization of piperine as active principle. J Ethnopharmacol. 2013; 147(2):373-382.
Crossref - Manohar IN, Kumari ON, Kumar A; From the proceedings of Insight Ayurveda 2013, Coimbatore. 24th and 25th May 2013. PA03.16. Role of herbal immunomodulators in Paediatric practice. Anc Sci Life. 2013;32(Suppl 2):S85.
Crossref - Makare N, Bodhankar S, Rangari V. Immunomodulatory activity of alcoholic extract of Mangifera indica L. in mice. J Ethnopharmacol. 2001;78(2-3):133-137.
Crossref - Savant C, Kulkarni AR, Mannasaheb BA, Gajar R. Immunomostimulant phytoconstituents from Mangifera indica L. bark oil. The Journal of Phytopharmacology. 2014;3(2):139-148.
- Garcia D, Leiro J, Delgado R, Sanmartin ML, Ubeira FM. Mangifera indica L. extract (Vimang) and mangiferin modulate mouse humoral immune responses. Phytother Res. 2003;17(10):1182-1187. PMID: 14669253
Crossref - Rivera DG, Hernandez I, Merino N, et al. Mangifera indica L. extract (Vimang) and mangiferin reduce the airway inflammation and Th2 cytokines in murine model of allergic asthma. J Pharm Pharmacol. 2011;63(10):1336-1345.
Crossref - Garrido G, Delgado R, Lemus Y, Rodriguez J, Garcia D, Nunez-Selles AJ. Protection against septic shock and suppression of tumor necrosis factor alpha and nitric oxide production on macrophages and microglia by a standard aqueous extract of Mangifera indica L. (VIMANG). Role of mangiferin isolated from the extract. Pharmacol Res. 2004;50(2):165-172.
Crossref - Du Z, Fanshi F, Lai YH, Chen JR, Hao E, Deng J, Hsiao CD. Mechanism of anti-dementia effects of mangiferin in a senescence accelerated mouse (SAMP8) model. Biosci Rep. 2019;20;39(9):BSR20190488
- Mondal S, Varma S, Bamola VD, et al. Double-blinded randomized controlled trial for immunomodulatory effects of Tulsi (Ocimum sanctum Linn.) leaf extract on healthy volunteers. J Ethnopharmacol. 2011;14;136(3):452-456.
Crossref - Choudhury SS, Bashyam L, Manthapuram N, Bitla P, Kollipara P, Tetali SD. Ocimum sanctum leaf extracts attenuate human monocytic (THP-1) cell activation. J Ethnopharmacol. 2014; 154(1):148-155.
Crossref - Maraghehpour B, Khayamzadeh M, Najafi S, Kharazifard M. Traditionally used herbal medicines with antibacterial effect on Aggegatibacter actinomycetemcomitans: Boswellia serrata and Nigella sativa. J Indian Soc Periodontol. 2016;20(6):603-607.
Crossref - Punturee K, Wild CP, Kasinrerk W, Vinitketkumnuen U. Immunomodulatory activities of Centella asiatica and Rhinacanthus nasutus extracts. Asian Pac J Cancer Prev. 2005;6(3):396-400. PMID: 16236006.
- Krishnamurthy RG, Senut M, Zemke D, Min J, Frenkel MB, Greenberg EJ. Asiatic acid, a pentacyclic triterpene from centella asiatica, is neuroprotective in a mouse model of focal cerebral ischemia. J Neurosci Res. 2009;87(11):2541-2550.
Crossref - Azim KF, Ahmed SR, Banik A, Khan MMR, Deb A, Somana SR. Screening and druggability analysis of some plant metabolites against SARS-CoV-2: An integrative computational approach. Inform Med Unlocked. 2020;20:100367.
Crossref - Lin WC, Lin JY. Berberine down-regulates the Th1/Th2 cytokine gene expression ratio in mouse primary splenocytes in the absence or presence of lipopolysaccharide in a preventive manner. Int Immunopharmacol. 2011;11(12):1984-1990.
Crossref - Niu XF, Zhou P, Li WF, Xu HB. Effects of chelerythrine, a specific inhibitor of cyclooxygenase-2, on acute inflammation in mice. Fitoterapia. 2011;82(4):620-625.
- Vaibhav K, Shrivastava P, Javed H, et al. Piperine suppresses cerebral ischemia-reperfusion-induced inflammation through the repression of COX-2, NOS-2, and NF-κB in middle cerebral artery occlusion rat model. Mol Cell Biochem. 2012;367:73-84.
Crossref - Son DJ, Akiba S, Hong JT, et al.Piperine inhibits the activities of platelet cytosolic phospholipase A2 and thromboxane A2 synthase without affecting cyclooxygenase-1 activity: different mechanisms of action are involved in the inhibition of platelet aggregation and macrophage inflammatory response. Nutrients. 2014;6(8):3336-3352.
Crossref - Hougee S, Sanders A, Faber J, et al. Decreased pro-inflammatory cytokine production by LPS-stimulated PBMC upon in vitro incubation with the flavonoids apigenin, luteolin or chrysin, due to selective elimination of monocytes/macrophages. Biochem Pharmacol. 2005;69(2):241-248.
Crossref - Shin EK, Kwon HS, Kim YH, Shin HK, Kim JK. Chrysin, a natural flavone, improves murine inflammatory bowel diseases. Biochem. Biophys Res Commun.2009;381:502-507.
- Andujar I, Recio MC, Bacelli T, Giner RM, Rios JL. Shikonin reduces oedema induced by phorbol ester by interfering with IκBa degradation thus inhibiting translocation of NF-κB to the nucleus. Br J Pharmacol. 2010;160(2):376-388.
Crossref - Vaillancourt F, Silva P, Shi Q, Fahmi H, Fernandes JC, Benderdour M. Elucidation of molecular mechanisms underlying the protective effects of thymoquinone against rheumatoid arthritis. J Cell Biochem. 2011;112(1):107-117.
Crossref - Kumar RA, Sridevi K, Kumar NV, Nanduri S, Rajagopal S. Anticancer and immunostimulatory compounds from Andrographis paniculata. J Ethnopharmacol. 2004;92(2-3):291-295.
Crossref - Ramgolam V, Ang SG, Lai YH, Loh CS, Yap HK. Traditional Chinese medicines as immunosuppressive agents. Ann Acad. Med. Singapore.2000;29:11-16.
- Khajuria A, Gupta A, Garai S, Wakhloo BP. Immunomodulatory effects of two sapogenins 1 and 2 isolated from Luffa cylindrica in Balb/C mice. Bioorg Med Chem Lett. 2007;17(18):1608-1612.
Crossref - Brinker AM, Ma J, Lipsky PE, Raskin I. Medicinal chemistry and pharmacology of genus Tripterygium [Celastraceae]. Phytochemistry. 2007;68(6):732-766.
Crossref - Shen YC, Chen CF, Chiou WF. Andrographolide prevents oxygen radical production by human neutrophils: possible mechanism[s] involved in its anti-inflammatory effect. Br J Pharmacol. 2002;135(5):399-406.
Crossref - Kannaiyan R, Shanmugam MK, Sethi G. Molecular targets of celastrol derived from Thunder of God Vine: potential role in the treatment of inflammatory disorders and cancer. Cancer Lett. 2011;303(1):9-20.
Crossref
© The Author(s) 2021. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.