Sweet sorghum (Sorghum bicolor L.) accumulates high concentration of sucrose along with the juice in their stem. Hence, the opportunity of extensive research is in the physiology, biochemistry and molecular aspect of sucrose accumulation. Instead of this, the relationship between leaf photosynthetic activity and sucrose accumulation in the culm is not well understood. Communication between source and sink may play very crucial role in regulating sucrose synthesis. Assimilates partitioning is the process by which the products of photosynthesis are exported from leaves through the minor veins to storage tissues. Key aspects of researchers are to improve carbohydrate partitioning by increasing source capacity or plant or by increasing sink strength. Increases of carbohydrate production may possible by increasing light interception either by increasing the number of leaves or total leaf area, stay-green traits, and by enhancing the capacity of photosynthesis of the plant to fix carbon. In this review, we discuss many aspect of sweet sorghum that make it an ideal crop for biofuel, ethanol and syrup production and also provide an overview of genetic diversity and resources available for engineering and marker-assisting breeding of sweet sorghum.
Sweet sorghum, Sugar metabolism, Photosynthesis.
Sorghum (Sorghum bicolor L. Moench) is a C4 herbaceous annual plant, and is popularly known by a particular names in particular country like great millet, kafir corn, durra, mtama, jowar, kaoliang and milo in West Africa, South Africa, Sudan, Eastern Africa, India, China, United States respectively (Purseglove, 1972). Being a C4 plant, sweet sorghum has a highly efficient photosynthetic pathway and is very efficient in the utilization of soil nutrients also. Hence, plant contains high concentrations of soluble sugars (10–15 %) in the stalk juice. The name ‘‘sweet sorghum’’ is used to identify those varieties of sorghum, which has juicy along with sugar content in the stalk. Partitioning of carbohydrate is the process by which the products of photosynthesis are exported from leaves through the minor veins to storage tissues. Key aspects of researchers are to improve carbohydrate partitioning by increasing source capacity or by increasing sink strength. Carbohydrate production may increase by increasing light interception either by increasing the number of leaves or total leaf area, stay-green traits, and by enhancing photosynthesis capacity of the plant to fix CO2 (Sakamoto et al., 2006; Hammer et al., 2009; Zheng et al., 2009; Zhu et al., 2010; Raines, 2011; Ruan et al., 2012a). The change in sugars content between the time period of anthesis and harvest is already established, while the content of reducing sugars decrease, and sucrose increases in the stem during maturation time in (Lingle et al., 2012). Grain sorghums are used for human food, while forage sorghums are used for animal feed, and sweet sorghums for edible syrup. It requires less water than sugarcane and is tolerant to drought and flooding and also it is capable of re-growth as ratoon crops. In this respect, sweet sorghum is one of the best alternatives option (Rooney et al., 2007; Zhang et al., 2010; Linton et al., 2011; Yu et al., 2012).
Photosynthetic capacity
Storage organs are the anatomical structures that accumulate sugar from photosynthesis. They include fruits, seeds, roots, and stems. Stem acting as a storage organ is found in grasses, such as sweet sorghum (Sorghum bicolor) and sugarcane (Saccharum officinarum) (Slewinski, 2012). Being a C4 plant, both species have the capacity to perform high speed of photosynthesis and accumulate high concentrations of fermentable sugars in the stem, mainly sucrose. Therefore both the plants are ideal plant for strong stem sinks, and being used for the production of ethanol as a biofuel (Waclawovsky et al., 2010; Calviño and Messing, 2012). In sweet sorghum, photo-assimilate is used for plant growth and development during initial vegetative phase. Thereafter, internodes elongated and stem initiate sugar accumulation where most of the photo-assimilate stored in the form of sucrose (Hoffmann-Thoma et al., 1996; Lingle, 1999; Almodares et al., 2008; Almodares and Hadi, 2009; Calviño and Messing, 2012; Slewinski, 2012). Mature Internodes of sweet sorghum stem accumulate higher concentration of sucrose content in comparison to younger one (Hoffmann-Thoma et al., 1996). Ali et al., (2008) reported that a little variation is found in terms of sugar composition among the cultivated sweet sorghum varieties and on the basis of simple sequence repeats (SSRs) marker study he grouped in to two types of sweet sorghum group IX high sugar content and group VII low sugar content. Moreover, on the basis of molecular analysis i.e. based on SSR and SNP marker, sweet sorghum varieties categories in to three major groups (Murray et al., 2009). While, on the basis of AFLP and SSR markers analysis, sweet sorghum classified into two types with various brix content (Pecina-Quintero et al., 2012).
Relationship between source and sink
The rate of sucrose accumulation often increases near anthesis in culm internodes, which is a period in which least demand appear in reproductive sinks (Vietor et al., 1989). However, photoassimilate can be remobilized to reproductive sink that accumulated in the culm tissue during anthesis if photoassimilate export from the source leaves is low [Vietor et al., 1989 and Sowder et al., 1997). A good relationship between source and sink tissue is essential because supply of photoassimilate from source is depending upon the consumption of the intermediate (Hofmeyr and Cornish-Bowden, 2000). Leaves are the major source of carbohydrate supply while sink demand includes growth, respiration and storage in plant. In sugarcane, culm parenchyma cell may be a strong additional demand component of sucrose. This kind of additional storage, contribute a lot to maintain a high sink demand of sucrose in the stem resulting a high yield of sucrose (Fig. 1). The frequency of end product formation is governed by feedback inhibition of the supply process by the concentration of the intermediate metabolite, which is determined by the balance between supply and demand (Fig. 1A). In plants, demand is likely to be the result of metabolic activities such as growth, respiration and storage, especially in high carbon accumulating species such as sugarcane. In this regard, Feedback inhibition is indicated as a sensitivity function of supply to the concentration of the intermediate, sucrose i.e. sucrose (Hofmeyr, 1998).
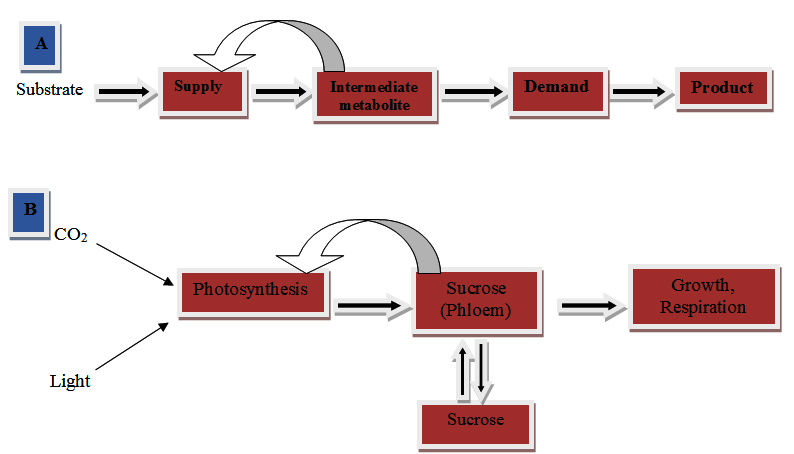
Fig. 1. A simplified model of metabolic supply and demand system modified from Hofmeyr (1998)
Sugar transport and storage in the stem
Sweet sorghum and sugarcanes may vary in respect of high levels of sucrose in their stems. In mature sugarcane stems, sucrose transport follows a predominantly symplastic movement when moving from the phloem into the storage parenchyma cells (Lingle, 1989; Rae et al., 2005; Patrick et al., 2013). This hypothesis is supported by evidence observed from the study of sap collected from the xylem of sugarcane stems, which was very close with the phloem (Welbaum et al., 1992), moreover, it was observed that a fluorescent dye loaded into leaves is unloaded in the phloem of stem and move symplastically via plasmodesmata into the stem parenchyma cells (Rae et al., 2005). During the sucrose accumulation in the stem, parenchyma cell apoplast facilitate as an additional storage compartment that increases sink strength. At maturity stage, when sucrose accumulates at higher concentration in the stem apoplast, reverse translocation of sucrose into the phloem is inhibited by the lignified and suberized cell walls (Rae et al., 2005; Walsh et al., 2005; Patrick et al., 2013). Biochemical studies shown that the sucrose must be store in parenchyma cells by crossing the plasma membrane cell in the stem of sweet sorghum, that itself indicate the support of sugar transporter (Hoffmann-Thoma et al., 1996). Sucrose transporters are the H+/sucrose symporter that is able to translocate sucrose across the membrane (Lalonde et al., 2004; Sauer, 2007; Braun and Slewinski, 2009). There are six class of popular sucrose transporter (SUTs) i.e. is SUT1, SUT2, SUT3, SUT4, SUT5, SUT6 have been proposed that encode protein located in the plasma membrane and tonoplast in sorghum genome (Braun and Slewinski, 2009). The transporters are involved in the high sucrose accumulation in sweet sorghum was tested by transcriptional analysis and concluded that SUT1 and SUT4 are highly correlated with sugar content in sweet sorghum stem (Qazi et al., 2012). Some other sugar transporter proteins i.e. SWEETs and TMTs are recently shown to transport sucrose across the membrane (Wingenter et al., 2010 and Chen et al., 2012). Even though, exact role of additional sucrose transporters in sucrose transfer is not yet established in the stem (Braun, 2012; Chen et al., 2012).
Sugar metabolism in the stem
Metabolism and storage processes of sucrose in the stem are important step of sink strength. During initial growth of sweet sorghum and sugarcane stems, INVs contribute to strengthen the sink and stem size. However, later on sucrose is stored in the vacuoles of stem parenchyma cells. Out of the total store sucrose in the stem, only least amount is hydrolyzed during its transfer into the ripening stem (Lingle, 1989; Tarpley et al., 1996; Tarpley and Vietor, 2007). Further, after the stems have matured and elongation has ceased, these tissues show low metabolic activity (Tarpley et al., 1996), prompting the question as to whether sucrose metabolism remains an important driver of sink strength in maturing sweet sorghum and sugarcane stem tissues. The enzymes involved in sucrose metabolism are INV, sucrose synthase (SUS), sucrose phosphate synthase (SPS), and sucrose phosphate phosphatase (SPP). INV enzymes catalyze the cleavage of sucrose into glucose and fructose. Different types of INV enzymes are found in different places in the plant system like cell wall, vacuole, and cytoplasm (Ruan et al., 2010; Vargas and Salerno, 2010; Patrick et al., 2013). Winter and Huber, (2000) also reported that sucrose synthase is another enzyme that split sucrose into fructose and UDP-glucose. Ultimately, sucrose phosphate synthase and sucrose phosphate phosphatase play crucial role jointly in irreversible synthesis of sucrose from UDP-glucose and fructose-6-phosphate (Lunn and Mac Rae, 2003). Qazi et al., 2012) reported that grain sorghum accumulates least amount of sucrose in the stem in comparison to sweet sorghum and the carbohydrates produced are moved toward storage in seeds.
Increased demand of energy resources in respect of biofuel throughout the world, sweet sorghum (Sorghum bicolor L.) became a most popular optional crop for the production of biofuel. Therefore, this crop compels researcher to do advanced research in this field. Because detailed study of physiological and biochemical analysis in addition to molecular investigation will help to understand or identify the novel characters in deep for better understanding of rate of photosynthesis in the leaves, source sink relationship between leaves and stem, sugar transport and storage in stem and sugar metabolism. The knowledge from above mentioned criteria shall also help to know how plant regulates the portioning of sugar inside the stem of sweet sorghum.
- Ali, M. L., Rajewski, J. F., Baenziger, P. S., Gill, K. S., Eskridge, K. M. and Dweikat, I. Assessment of genetic diversity and relationship among a collection of US sweet sorghum germplasm by SSR markers. Mol. Breed., 2008; 21: 497–509
- Almodares, A. and Hadi, M. R. Production of bioethanol from sweet sorghum: a review. Afr. J. Agric. Res., 2009; 47: 72–780
- Almodares, A., Taheri, R. and Adeli, S. Stalk yield and carbohydrate composition of sweet Sorghum (Sorghum bicolor (L.) Moench) cultivars and lines at different growth stages. Malays. Appl. Biol., 2008; 37: 31–36
- Braun, D. M. and Slewinski, T. L. Genetic control of carbon partitioning in grasses: roles of sucrose transporters and tie-dyed loci in phloem loading. Plant Physiol., 2009; 14: 971–81.
- Braun, D. M. SWEET! The pathway is complete. Science,2012; 335: 173–174
- Calviño, M. and Messing, J. Sweet sorghum as a model system for bioenergy crops. Curr. Opin. Biotechnol., 2012; 233: 23–329
- Chen, L. Q., Qu, X. Q., Hou, B. H., Sosso, D., Osorio, S. and Fernie A. R. Sucrose efflux mediated by SWEET proteins as a key step for phloem transport. Science,2012; 335: 207–211
- Hammer, G. L., Dong, Z., McLean, G., Doherty, A. I., Messina, C. and Schussler, J. Can changes in canopy and/or root system architecture explain historical maize yield trends in the U.S. Corn belt. Crop Sci., 2009; 49: 299–312.
- Hoffmann-Thoma, G., Hinkel, K., Nicolay, P. and Willenbrink, J. Sucrose accumulation in sweet sorghum stems internodes in relation to growth. Physiol. Plant., 1996; 97: 277–284.
- Hofmeyr, J. H. S. and Cornish-Bowden, A. Regulating the cellular economy of supply and demand, FEBS Letters, 2000; 476: 47-51.
- Hofmeyr, J. H. S. Anaerobic energy metabolism in yeast as a supply–demand system. In: Cornish-Bowden A, ed.New beer in an old bottle. Eduard Buchner and the growth of biochemical knowledge. Universitat de Valencia, 1998; 225–242.
- Lalonde, S., Wipf, D. and Frommer, W. B. Transport mechanisms for organic forms of carbon and nitrogen between source and sink. Annu. Rev. Plant Biol.,1998; 55: 341–372.
- Lingle, S. Evidence for the uptake of sucrose intact into sugarcane internodes. Plant Physiol., 1989; 90: 6–8
- Lingle, S. E. (1999). Sugar metabolism during growth and development in sugarcane internodes. Crop Sci., 1998; 39:480–486.
- Lingle, S. E., Tew, T. L., Rukavina, H. and Boykin, D. L. Post harvest changes in sweet sorghum I: brix and sugars. Bioenergy Res., 2012; 5: 158–67.
- Linton, J. A., Miller, J. C., Little, R. D., Petrolia, D. R. and Coble, K. H. Economic feasibility of producing sweet sorghum as an ethanol feed stock in the South eastern United States. Biomass Bioenergy, 2011; 35: 3050–3057.
- Lunn, J. E. and Mac Rae, E. New complexities in the synthesis of sucrose. Curr. Opin. Plant Biol., 2003; 6: 208-214.
- Murray, S. C., Rooney, W. L., Hamblin, M. T., Mitchell, S. E. and Kresovich, S. Sweet sorghum genetic diversity and association mapping for brix and height. Plant Genome, 2009; 2: 48–62.
- Patrick, J. W., Botha, F. C. and Birch, R. G. Metabolic engineering of sugars and simple sugar derivatives in plants. Plant Biotechnol. J.,2013; 11: 142–156.
- Pecina-Quintero, V., Anaya-López, J. L., Zamarripa-Colmenero, A., Montes-Garcia, N., Nuñez-Colín, C., Solis-Bonilla, J. L., Aguilar-Rangel, M. R. and Prom, L. Genetic diversity of sweet sorghum germplasm in Mexico using AFLP and SSR markers. Pesq Agropec Bras.,2012; 47: 1095–1102.
- Qazi, H. A., Paranjpe, S. and Bhargava, S. Stem sugar accumulation in sweet sorghum – activity and expression of sucrose metabolizing enzymes and sucrose transporters. J. Plant Physiol., 2012; 169: 605–613
- Rae, A. L., Perroux, J. M. and Grof, C. P. Sucrose partitioning between vascular bundles and storage parenchyma in the sugarcane stem: a potential role for the ShSUT1 sucrose transporter. Plant, 2005;220: 817–825
- Raines, C. A. Increasing photosynthetic carbon assimilation in C3 plants to improve crop yield: current and future strategies. Plant Physiol., 2011; 15: 536–42
- Rooney, W. L., Blumenthal J., Bean, B. and Mullet, J. E. Designing sorghum as a dedicated bioenergy feedstock. Biofuels Bioprod. Biorefin., 2007; 1147–157
- Ruan, Y. L., Jin, Y., Yang, Y. J., Li, G.J., and Boyer, J. S. Sugar input, metabolism, and signaling mediated by invertase: roles in development, yield potential, and response to drought and heat. Mol. Plant, 2010; 3: 942–955
- Ruan, C. J., Shao, H. B. and Teixeira da Silva, J. A. A critical review on the improvement of photosynthetic carbon assimilation in C3 plants using genetic engineering. Crit. Rev. Biotechnol., 2012a; 3: 21–21.
- Sakamoto, T., Morinaka, Y., Ohnishi, T., Sunohara, H., Fujioka, S. and Ueguchi-Tanaka, M. Erect leaves caused by brassinosteroid deficiency increase biomass production and grain yield in rice. Nat. Biotechnol., 2006; 24: 105–109.
- Sauer, N. Molecular physiology of higher plant sucrose transporters. FEBS Lett.,2007; 581: 2309–2317
- Slewinski, T. L. Diverse functional roles of monosaccharide transporters and their homologs in vascular plants: a physiological perspective. Mol. Plant, 2012; 46: 41–662.
- Sowder, C. M., Tarpley, L., Vietor, D. M. and Miller, F. R. Leaf photoassimilation and partitioning in stress-tolerant sorghum. Crop Science, 1997; 37: 833-838.
- Tarpley, L., Vietor, D. M., and Miller, F. R. Metabolism of sucrose during storage in intact sorghum stalk. Int. J. Plant Sci., 1996; 157: 159–163
- Tarpley L. and Vietor D. M. Compartmentation of sucrose during radial transfer in mature sorghum culm. BMC Plant Biology, 2007; 7: 1-10
- Vargas, W. A., and Salerno, G. L. The Cinderella story of sucrose hydrolysis: alkaline/neutral invertases, from cyanobacteria to unforeseen roles in plant cytosol and organelles. Plant Sci., 2010; 178: 1–8.
- Vietor, D. M., Cralle, H. T. and Miller, F. R. Partitioning of 14C-photosynthate and biomass in relation to senescence characteristics of sorghum. Crop Science, 1989; 29: 1049-1053
- Waclawovsky, A. J., Sato, P. M., Lembke, C. G., Moore, P. H. and Souza, G. M. Sugarcane for bio-energy production: an assessment of yield and regulation of sucrose content. Plant Biotechnol. J., 2010; 8: 263–276.
- Walsh, K. B., Sky, R. C. and Brown, S. M. The anatomy of the pathway of sucrose unloading within the sugarcane stalk. Funct. Plant Biol.,2005; 32: 367–374
- Welbaum, G., Meinzer, F., Grayson, R. and Thornham, K. Evidence for the consequences of a barrier to solute diffusion between the apoplast and vascular bundles in sugarcane stalk tissue. Funct. Plant Biol.,1992; 19: 611–623.
- Winter, H. and Huber, S. Regulation of sucrose metabolism in higher plants: localization and regulation of key enzymes. Crit Rev Plant Sci., 2000; 19: 31–67.
- Yu, J., Zhang, T., Zhong, J., Zhang, X. and Tan, T. Biorefinery of sweet sorghum stem. Biotechnol Adv., 2012; 30: 811–816
- Zhang, C., Xie, G. Li, S., Ge, L. and He, T. The productive potentials of sweet sorghum ethanol in China. Appl Energy, 2010; 87: 2360–8.
- Zheng, H. J., Wu, A. Z., Zheng, C. C., Wang, Y. F., Cai, R. and Shen, X. F. QTL mapping of maize (Zea mays) stay-green traits and their relationship to yield. Plant Breed., 2009; 12: 854–62
- Zhu, X. G., Long, S. P. and Ort, D. R. Improving photosynthetic efficiency for greater yield. Annu. Rev., Plant Biol., 2010; 61: 235–261.
© The Author(s) 2018. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


