ISSN: 0973-7510
E-ISSN: 2581-690X
Necropsy was conducted for thirty-five dead bovines during the six months duration from February to July 2016 at post-mortem facility, IVRI. Detailed post-mortem examination was done by impression smear, gross and histopathological lesions examination. Out of thirty-five cases, eight showed the septicemic changes like petechial haemorrhages on serosal surface. The necropsy examination of these cases revealed the consolidation of lungs and haemorrhages on the epicardium, spleen, and small intestine. Gross lesions were correlated with the microscopic lesions. Microscopic examination showed diffuse congestion and haemorrhages along with the neutrophilic infiltration in different organs including lung, heart, and intestine. Impression smear from heart (8 samples) showed bipolar organism under microscope by Leishman’s stain. Based on the gross and microscopic examination along with further confirmation by bipolar staining in heart impression smear indicates septicaemia by Pasteurella multocida organism which leads to death of animals.
Septicaemia, Bovine, Bipolar, Histopathology, Pasteurella multocida.
Haemorrhagic septicaemia (HS) is the most important bacterial disease affecting the large ruminants in India, and resulting in huge economic loss (Dutta et al., 1990; Singh et al., 1996; Ranjan et al., 2011). It is highly fatal and acute diseases of large ruminants. It is caused by two specific serotypes of P. multocida designated B: 2 (Asian serotype) and E: 2 (African serotype) (De Alwis, 1999). Buffaloes have a shorter course of the disease when compared with cattle, which is believed to be a reason for more susceptibility to HS than cattle (De Alwis, 1999). Case fatality may approach 100% if treatment is not carried out at an early stage (Benkirane and De Alwis, 2002). Outbreaks of disease occur throughout the year irrespective of the season (Khera, 1979; Kumar et al., 2004). In India, the incidence of disease outbreaks varies considerably in different states and from year to year in each state (Khera, 1979). Epidemiological factors may play key role in the carrier-mediated rapid spread of infectious agent in the animals (Ataei et al., 2009).
The disease occurs in acute, sub-acute and chronic forms and is characterized by an initial phase of temperature elevation, a phase of respiratory involvement and a terminal phase of septicemia and recumbency leading to death (De Alwis, 1999). The incubation period is very short usually 1–3 days and the course of the disease may range from sudden death to a protracted course extending up to 5 days (Carter and De Alwis, 1989; De Alwis, 1999). The acute form of HS is manifested by sudden onset and death within 24 hours. The animals show a high rise in body temperature (41–42oC), weak pulse with rapid, shallow respiration and cyanosis of visible mucous membranes (De Alwis, 1999; Benkirane and De Alwis, 2002; OIE, 2009). Restlessness, mild colic pain, muscular tremors, lacrimation, nasal discharge and diarrhoea are also observed. In the sub-acute form, known as the subcutaneous edematous form, the animal survives for 2–3 days with edematous swelling of the throat and brisket region and bronchopneumonia. In the chronic form, the course of infection is longer with signs of short, rapid painful respiration coupled with mucopurulent or blood-stained nasal discharge (OIE, 2009). At post-mortem examination, the first obvious lesion is subcutaneous edema, particularly in the mandibular and brisket regions. Other lesions include petechial-to-ecchymotic haemorrhages, congestion and/or consolidation of the lungs, fibrinous pneumonia, pleurisy and pericarditis (De Alwis, 1999; OIE, 2009).
For diagnosis, specimens to be collected from infected live animals are blood samples and nasal secretions, and from dead animals are heart blood, liver, spleen, lung and bone marrow of long bones (OIE, 2009). Clinical diagnosis is usually done by characteristic signs, gross pathological lesions, herd history, morbidity and mortality pattern, species susceptibility and age group affected (OIE, 2009). Laboratory confirmation is based on demonstration of Gram-negative, bipolar, pleomorphic bacterial cells in a fresh blood smear by Gram’s, Leishman’s or Methylene Blue staining. Bacterial isolation is based on standard techniques using BHI broth and immunological methods using serum, such as the rapid slide agglutination test (Namioka and Murata, 1961), IHA (Carter, 1955), agar gel immunodiffusion test (Heddleston et al., 1972) and counter immune electrophoresis (Carter and Chengappa, 1981), Molecular approaches like Polymerase chain reaction are also employed for diagnosis (Biswas et al., 2004).
The recent outbreaks of HS were observed in many states (Kumar et al., 2011; Mondal et al., 2013; Mitra et al. (2013), even buffalo calves in organized farms found to be positive with heavy mortality (Vaid et al., 2012). Singh et al. (2007) reported the investigative cases of haemorrhagic septicaemia in villages of Uttarakhand, western Uttar Pradesh, and Delhi. The aim of the present study is to report and understand the disease pattern of HS by screening bovine necropsy samples.
Sample collection
This study was carried out on 35 dead bovine cases at post-mortem facility, IVRI from February to July 2016. During post-mortem examination of dead animals, gross pathological lesions were recorded systematically. The tissue samples of lungs, liver, spleen, kidneys, heart, intestine, lymph nodes and brain of dead animals were collected in 10% formalin for histopathological examination. The heart impression smears were collected on the clean sterile slide for the bacteriological examination.
Impression smears and Histopathology
The impression smears from heart were immediately fixed in methanol and stained with Leishman’s stain as per the standard protocol and examined under microscope. Tissues were processed and sections were cut at 4-5 microns thickness and stained with Haematoxylin and Eosin stains (Luna, 1968). The stained slides were examined under microscope and histopathological changes were recorded.
Post-mortem examination revealed the hemorrhages on the subcutaneous tissue and serosal layer of the peritoneum. Out of eight cases, only one case was showing accumulation of the edematous fluid at the subcutaneous tissue of the neck. On cut section, pre-scapular lymph nodes showed severe congestion (Fig. 1). The straw colored sero-sanguineous fluid was observed in the thoracic and peritoneal cavity. The petechial hemorrhages were observed in the serosal layer of the intestine, pericardium, spleen, and lung (Fig. 2). On cut section, the kidneys were showing congestion at the cortico-medullary junction. Out of eight cases, two cases were showing the severe hemorrhage on the endocardial surface. Small intestine revealed severe congestion and haemorrhages throughout its length. Out of eight cases, one case showed the meningeal congestion. The heart impression smears were stained with the Leishman’s stains showed the bipolar organisms (Fig. 3).
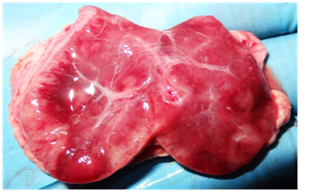 Fig. 1: Severely congested pre-scapular lymph node (Gross)
Fig. 1: Severely congested pre-scapular lymph node (Gross)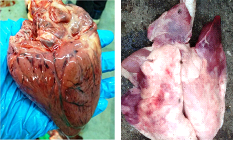 Fig. 2: Petechial to ecchymotic hemorrhages on the heart (A) and lung (B) (Gross)
Fig. 2: Petechial to ecchymotic hemorrhages on the heart (A) and lung (B) (Gross)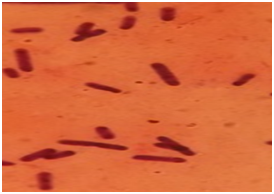 Fig. 3: Heart-impression smear showing bipolar organism stained with Leishman’s stain (x100)
Fig. 3: Heart-impression smear showing bipolar organism stained with Leishman’s stain (x100)Microscopic lesions were observed in the lung, heart, kidney, lymph nodes, spleen, brain and intestine by histopathology. Accumulation of fibrinous oedematous fluid was observed in the alveolar and septal spaces along with thickened alveolar septa in lungs (Fig. 4). The inflammatory cells mainly neutrophils were observed in interstitial spaces. In most of the cases severe congestion and haemorrhages in the lung parenchyma was observed. The bronchial epithelium showed degenerative changes along with denuded epithelium. Submucosa showed oedematous changes along with congestion and focal haemorrhagic lesions. Heart showed congestion and haemorrhages in epicedium and myocardium along with infiltration of inflammatory cells in the myocardium (Fig. 5). The small intestine showed vascular congestion and haemorrhages in mucosa and submucosa (Fig. 6). In the kidney hemorrhages and congestion at the cortex, medulla, and corticomedullary region has been observed. In lymphoid organs, germinal centres were replaced by red blood cells due to haemorrhages and lymphoid cell depletion at the subscapular and cortex of the lymph node has been seen. In the spleen increased red pulp area and lymphoid depletion were quite prominent. Other organs like liver showed degenerative and vascular changes suggestive of septicemic changes. In brain, the meningeal congestion was observed.
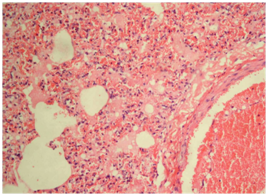 Fig.4: Lung showing fibrinous exudates in alveoli along with rupture of alveolar capillaries and infiltration of neutrophils (H&E, x400)
Fig.4: Lung showing fibrinous exudates in alveoli along with rupture of alveolar capillaries and infiltration of neutrophils (H&E, x400)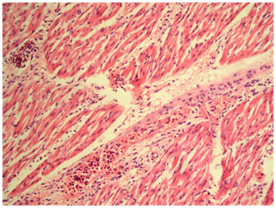 Fig. 5: Heart showing Infiltration of inflammatory cell in between myocardial fibers along with myocardial hemorrhages (H&E, x100)
Fig. 5: Heart showing Infiltration of inflammatory cell in between myocardial fibers along with myocardial hemorrhages (H&E, x100)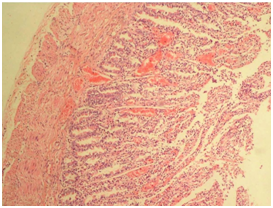 Fig. 6: Small intestine showing vascular congestion and haemorrhages in mucosa and sub mucosa (H&E, x100)
Fig. 6: Small intestine showing vascular congestion and haemorrhages in mucosa and sub mucosa (H&E, x100)The gross pathological lesions were correlated with histopathological lesions. The observed petechial hemorrhage on the serous membranes along with serosanguineous fluids in the thoracic and abdominal cavities were indicative of septicemic changes. The septicemic changes were correlated with the previous reports on septicemia (Buxton and Fraser, 1977 and Mitra et al., 2013). The gross lesions of lungs and intestine were more important in the present study as reported by Mitra et al. (2013) and Mondal et al. (2013). Generally in HS outbreaks septicemic changes on visceral organs and lungs has been reported. The reported histopathological lesions in the present study, correlated with the study done by Mondal et al. (2013). Based on the results of impression smear, gross and microscopic lesions, the animals of present study might be died because of septicaemia caused by P. multocida.
ACKNOWLEDGMENTS
The authors are thankful to the Director, ICAR-Indian Veterinary Research Institute, Izatnagar, U.P and Head, Division of Pathology, IVRI for providing all facilities to carry out histopathological work.
- Ataei S, Burchmore R, Christopher HJ, Finucane A, Parton R and Coote JG. Identification of immunogenic proteins associated with protection against haemorrhagic septicaemia after vaccination of calves with a live-attenuated aroA derivative of Pasteurella multocida B:2. Research in Veterinary Science 2009; 87: 207–210.
- Benkirane A and De Alwis MCL. Haemorrhagic septicaemia, its significance, prevention and control in Asia. Veterinary Medicine Czechoslovakia 2002; 47: 234–240.
- Buxton A and Fraser G., Animal Microbiology (Vol. l). Blackwell Scientific Publication. Oxford. London. Edinburg. 1977; 121-126.
- Carter GR and De Alwis MCL., Haemorrhagic septicaemia. In: Adlam C and Rutter JM (eds) Pasteurella and Pasteurellosis. London, UK: Academic Press, 1989; 131-160.
- De Alwis MCL., Haemorrhagic septicaemia. ACIAR Monograph No. 57. Canberra, Australia: Austrtalian Centre for International Agricultural Research. 1999; 11-24
- Dutta, T.K., Rajeev Gautam, Senthil Kumar V.S. and S.K. 4. Kotwal., Diagnosis of haemorrhagic septicaemia: past, present and future. Journal of Research, SKUAST- J, 2005; 4(1): 13- 24.
- Holt JG, Krieg NR, Sneath PHA, Staley JT and Williams ST., Bergey’s manual of determinative bacteriology. 9th edn. p. 196. Published by Springer, New York, USA, 1994.
- Khera, S.S., The incidence and distribution of epizootic diseases in India: haemorrhagic septicaemia. Bulletin Office International des Epizooties, 1979; 91: 331-347.
- Kumar Amit, Shekhar P, Chaudhury M K and Kumar Ajit., An outbreak of HS in bovines and its management. Intus Polivet. 2011; 12(2): 292.
- Kumar, A.A., Shivachandra, S.B., Biswas, A., Singh, V.P., Singh, V.P. and Srivastava, S.K., Prevalent serotypes of Pasteurella multocida isolated from different animal and avian species in India. Veterinary research communications, 2004; 28(8): 657-667.
- Luna LG., Manual of Histologic staining Methods the Armed Forces institute of Pathology. 3rd Edn. McGrawHill Book Co. New york, 1968.
- Mitra J, Chaudhury M, Bhattacharya C., Outbreak of Hemorrhagic Septicemia in free range buffalo and cattle grazing at riverside grassland in Murshidabad district, West Bengal, India. Explor. Anim. Med .Res. 2013; 3(2): 179-182.
- Mitra, J., Chowdhury, M. and Bhattacharya, C., Outbreak of hemorrhagic septicemia in free range buffalo and cattle grazing at riverside grassland in Murshidabad District, West Bengal, India. Explor Anim Med Res, 2013; 3(2): 178-182.
- Mondal, M., Maiti, S.K. and Ghosh, R.C., An outbreak of Haemorrhagic Septicaemia in bovines and its management. Intas Polivet,2013; 14(1): 76-78.
- OIE., Haemorrhagic septicaemia, Chapter 2.4.12. Terrestrial Manual. pp. 739–750. Office International Des Epizooties (OIE), Paris, France, 2009.
- Ranjan, R., Panda, S., Acharya, A., Singh, A. and Gupta, M., Molecular diagnosis of haemorrhagic septicaemia-A review. Veterinary World, 2011; 4(4): 189-192.
- Singh, K.P., Chandra, D., Rathore, R., Pandey, A.B., Telang, A.G. and Chauhan, R.S., Investigation of outbreaks of pasteurellosis in cattle and buffaloes. Indian J. Vet. Pathol. 2007; 31: 40-44.
- Singh, V.P., Kumar, A.A., Srivastava, S.K. and Rathore, B.S., Significance of HS in Asia: India. International workshop on diagnosis and control of HS. Bali, Indonesia, Indonesian Department of Agriculture, 1996; 1999, p.16.
© The Author(s) 2016. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


