ISSN: 0973-7510
E-ISSN: 2581-690X
Fowl cholera (FC) caused by serotypes of Pasteurella multocida includes A:1, A:3, A:4 is a highly fatal septicemic disease. Preliminary trials of P. multocida A:1 bacteriophage lysate vaccine against FC was evaluated. Lytic phage and P. multocida ratio was standardized to obtain stable lysate batches. Consequently, three batches of lytic phage preparation were produced; estimation of protein and carbohydrate content amongst batches did not shown any significant variation indicating same batches can be produced by standardized procedure. Protective response trials in poultry with P. multocida A:1, A:3, A:4 against plain lysate and alum adsorbed lysate (1% alum) on vaccinated group showed both homologous and heterologous protection compared with inactivated whole cell group provided only homologous protection. Assessment of antibody response towards P. multocida A:1, A:3, A:4 antigen evaluated by Indirect Haemagglutination test (IHA) reveals presence of protective antibody titer.
P. multocida, Bacteriophage lysate, Fowl cholera, Immune response, Indirect Haemagglutination test (IHA).
Fowl cholera (FC) is a primary pasteurellosis caused by multiple serotypes of Pasteurella multocida (Watt et al., 2003, Wilson and Ho, 2013). Vaccination is one of the most important preventive measures against this economically important disease. Currently available vaccines against FC are preparations of inactivated whole cells of one or more serotypes of the organism. But none of these preparations possesses all of the desirable traits of an ideal vaccine. Since P. multocida is a poor immunogen, not only a large amount of antigenic biomass is required (Rani et al., 2006) for a FC vaccine, addition of an adjuvant also becomes necessary for enhancing its efficacy up to a desirable level. Further, considering the multiplicity of and serotypes of the organism associated with the diseases, it becomes desirable that the immunizing agent must at least be capable of imparting an across serotype, if not an across-serogroup immunity. But, the conventional inactivated bacterins against FC lack the ability to induce an across-serotype or serogroup response and all the prevalent serotypes are needed to be incorporated in the vaccines (Marchart et al., 2003). It therefore becomes necessary to incorporate all the prevalent serotypes of P. multocida in inactivated vaccines. A high antigenic mass is also needed to induce a satisfactory clinical protection against the serotypes. This makes production and testing of vaccines very cumbersome process. An improved preparation capable of inducing an across-serotype protective response is therefore desirable. All attempts made during the last 30 years towards developing an improved vaccine against FC have met with varying degree of success. Many different approaches to develop better vaccine candidates capable of inducing an across-serotype protective response have been tried (Shivchandra et al., 2011). Though variable successes with protective efficacies have been claimed, none found the results equivalent to or better than existing inactivated, multi-serotype whole cell vaccines.
The immunological explanation for mono-specific immune responses of inactivated vaccines relates to the conformational alteration of antigens during the process of chemical & physical inactivation, or protein extraction. Parry et al. (1977) showed that live E. coli were superior to heat-killed or formalinized organisms at eliciting specific immunity. The reasons were thought to be structural alterations in the antigenic moieties by chemical or physical inactivation of bacteria. Similar observations have also been made with P. multocida, specially with the FC strains, where it has been demonstrated that the vaccines prepared from in vivo grown strains elicit a cross protective response (Rimler and Rhoades, 1889, Wang and Glisson, 1994 and Diallo et al., 2000). Therefore, a method of inactivation or antigen extraction that does not or minimally alter the antigenic moieties of the organism appears to be the key to developing a cross-protective immunizing preparation. For this purpose, use of bacterial phage-lysates as acellular vaccines provides a viable and technologically feasible option.
Phage lysates comprise a means of effectively killing bacteria while minimally altering their antigenicity (Pasternack et al., 2009). Phage lysate bacterins contain bacteriophage particles (primarily responsible for the first phase of protection mediated by phage itself) released by the burst and bacterial antigenic components in a solubilized phase (associated the second phase of protective response mediated by antibodies or effecter cells) produced by the burst caused by lytic bacteriophage. The P. multocida phage-lysates therefore, become possible candidates as novel class of immunotherapeutic agents against FC (and HS) and hold great potential to be used as improved vaccines against primary and secondary pasteurellosis. It appears to be a methodology that is expected to provide an almost ideal immunizing preparation that may be capable of inducing an “across-serotype” and preferably, an “across capsular serogroup” protective response. Phage-lysate can circumvent the necessity of large amounts of “antigenic biomass” of multiple serotypes for the production of FC vaccines. This investigation reports the efficacy of P. multocida phage-lysate in inducing across-serotype protective response in chickens.
Bacterial Strains
Lyophilized cultures P. multocida FC serotype A:1 was obtained from the Type Culture Lab of the Division of Biological Standardization, Indian Veterinary Research Institute. P. multocida A:3 and A:4 were procured from the Division of Bacteriology and Mycology, IVRI. All isolates of P. multocida were revived on 5% Sheep Blood agar plates. Each culture was transferred to several Blood agar slants after confirmation of its purity and morphological identity by Gram’s staining. The identity of each strain was revalidated by usual biochemical methods. Target host (bird) passage was given to each FC strain before preparing their Master Lots in phase I. Eighty percent lethal dose for susceptible chicken was determined for each serotype. In the protective efficacy trials, these 3 serotypes were used for challenge of birds. The A:1 strain was used for preparation of phage lysates.
Bacteriophage
A partially characterized bacteriophage against the A:1 strain of P. multocida (Durairajan, 2012) was selected for the present investigations. The phage (provisionally designated as IVRI/Pm-1) was revived and propagated by agar overlay and liquid culture methods (Jain et al., 2015) and maintained under lyophilized condition.
Generation of Phage Lysate
About 500 ml of log phase NZCYM broth (Difco, India) culture of P. multocida (A:1) was centrifuged at 5000 rpm/15 min (Remi, India) and the sediment was re-suspended in fresh pre-sterilized NZCYM broth medium to match with Brown’s Opacity Scale 3. The total viable count of this suspension was approximately 1.5×108/ml. To this suspension, phage IVRI/Pm-1 was aseptically added to obtain 1 MOI (Phage-bacteria ratio approximately 1:50). The phage-bacteria mixture was incubated at 37p C with intermittent shaking in an incubator. The viable count of the suspension were monitored by samples collected at 0, 30 min, 1h, 2h, 3h, 4h, 5h, 6h intervals by usual method. When more than 90 percent reduction in the turbidity was visually observed, the lysate was centrifuged to remove residual bacteria (if needed) and filtered through pre-sterilized 0.22µ filtration assembly (Genetix). The filtrate was collected in strict aseptic conditions and stored at 4p C.
Composition of Phage lysates
Three batches (500 ml each) of Phage-lysates were produced through separate cycles. Protein (Lowry’s et al., 1951) and Carbohydrate (Dubois et al, 1956) concentrations as well as Phage count 3 of each batch were determined (Table.1). Cell wall protein profile of concentrated preparations of each batch was determined by SDS PAGE and Western Blot Analysis.
Evaluation of protective response against direct challenge of birds
Test preparations
Preparation of inactivated Whole cell (WC) vaccine
Pure suspension of P. multocida A:1 in phase I grown on BHI Agar (Difco, India) was adjusted to Brown’s opacity scale 3 (so as to contain approximately 108 bacterial cells/ml) and inactivated by a suitable quantity of formaldehyde. Sterility test on the suspension was carried out by inoculating Blood agar plates, Fluid Thioglycollate Broth and Soybean Casein Digest Broth before adjuvantation (Aluminium hydroxide gel).
Alum gel adsorbed (APL) and Plain (PL) phage lysates
For adjuvantation, sterilized 10 percent aluminium hydroxide gel suspension in saline was mixed with inactivated whole cell (WC) and pooled phage lysate preparation in ratio of 1:10 (final aluminium concentration 1 percent) (APL) and incubated at 37°C for 24 hours. The amount of protein adsorbed was determined by calculating the residual protein in supernatant after centrifugation at 11000g for 10 minutes.
Immunization and challenge
The objective of this trial was to compare cross-protective responses of un-adjuvanted (PL) and alum adsorbed (AL) phage-lysates with that of a monovalent inactivated whole cell preparation of P. multocida A:1 containing 108 organisms per ml along with 1 percent alum gel. The procedure recommended by Indian Pharmacopoeia (2014) for determining the potency of Fowl Cholera Vaccines was followed.
Sixty apparently healthy chickens aging 4-8 week were divided in 4 groups of 15 birds each. Each bird of the first group was administered with 1 ml of the whole cell preparation through subcutaneous route. Similarly, each bird of the second and third groups received 0.5 ml of plain un-adjuvanted (PL) and alum-adsorbed (AL) lysate respectively. The birds of the fourth group were maintained along with the immunized groups as un-vaccinated controls. A booster immunization with the same dose of the respective preparation was given to each bird after 14 days of primary inoculation. Pre-vaccination, 7th, 14th, 21st and pre-challenge (28th) day blood sample from each bird was collected to obtain serum samples (Table-2).
After 14 days of booster inoculation, all groups were divided into 3 sub-groups having 5 birds. The first sub-groups of each group were challenged by virulent P. multocida A:1 through subcutaneous route with a dose sufficient to kill 80 percent un-immunized healthy birds. Similarly, each bird of 2nd and 3rd sub-groups of each group were challenged with virulent doses of A:3 and A:4 serotypes respectively. Birds were observed for 14 days before termination of experiment. Post-mortem was conducted on the dead birds to confirm the cause of death by isolation of P. multocida from heart blood (Table: 3).
Determination of antibody response
Humoral immune response of birds was monitored on various days post vaccination in terms of serum antibody tires against each of the 3 P. multocida serotypes by Indirect Haemagglutination Test as per the methodology of Sawada et. al., 1985. Sonicated antigen (Ames, 1974) with slight modification of P. multocida A:1, A:3, A:4 and P52 were used for sensitizing glutaraldehyde fixed (GA) and Tannic acid treated (TG) sheep RBC stocks.
Safety tests in Mice and Chickens
Safety test on the test preparations was conducted in mice and chickens as per the accepted methodology of Indian Pharmacopoeia (2014).
Since bacteriophage lysates against P. multocida are expected to induce an across-serotype protective response in susceptible bird species, the preparations provide an opportunity to improve upon the currently available vaccines against FC and HS. The present investigation was undertaken to compare the protective and antibody responses of chickens to immunization with phage lysate of a virulent P. multocida A:1 isolate from FC and inactivated whole cell vaccine prepared from the same organism.
A consistently lytic bacteriophage, designated as IVRI/Pm-1 (Durairajan, 2012) against a virulent strain of P. multocida A:1 was used for generation of phage lysates. Three batches of phage lysates were produced and characterized. The first essential requirement for a new vaccine is standardization of a process through which consistently stable batches of the finished product can be generated through several optimized production cycles. Since, it was found difficult to quantify the antigenic contents of the lysate in terms of protein or carbohydrate concentrations per dose; the initial total count of the P. multocida strain was fixed before adding phage. It was found that an NZCYM broth suspension of 14-16 hour incubated growth of the indicator P. multocida, adjusted to a viable count of approximately 1- 2×108 cfu/ml, was lysed by IVRI Pm-1 at 1: 50 phage/bacteria ratio, within 3-4 h with complete clearance of turbidity. The phage counts of the lysates were found to be within the limits of 108-109/ml. The immunobiological principles or the active antigenic ingredients of the lysates viz., total proteins and polysaccharide contents and phage counts were not found to vary significantly between the batches (Table:1) indicating that antigenically stable lysate preparations can be prepared by adopting an optimized process.
Table (1):
Composition of lysate batches content in batches.
Preparations |
Protein content (µg/ml) |
CHO content (µg/ml) |
Phage (Pfu/ml) |
|---|---|---|---|
PL-I |
1.6 |
444 |
2.1 x 108 |
PL-II |
1.4 |
467 |
1.8 x 108 |
PL-III |
1.3 |
459 |
1.5 x 108 |
Test PL |
1.2 |
450 |
108 |
To compare the immunoreactive polypeptides present in the P. multocida A:1 phage lysate with sonicated cell wall polypeptides of A:1, A:3, A:4 by SDS-PAGE and immunoblot analysis were done using chicken serum raised against phage lysate. About 30X concentrated phage lysate revealed the presence of 5 dominant polypeptide bands of 27 kDa, 35 kDa, 55kDa and 70 kDa and 100 kDa in electrophoretograms (Fig-1 and 2). In contrast, the cell wall preparations of indicator strain and A:3, A:4 and P52 revealed 16 and 12 major polypeptide bands respectively in the region between 130 kDa to 25 kDa. No identifiable difference between cell wall protein profiles of A:1, A:3, A:4 and P52 could be observed. Our observations are in agreement with the earlier observations of Johnson et al (1991), who have reported that the cell membrane proteins profiles do not vary among the P. multocida serotypes. No polypeptide band could be visualized in unconcentrated lysate preparations (Figure: 1). Several attempts have been made to identify specific immunogenic proteins of P. multocida isolates from HS and FC. The general conclusion of all these attempts is that no single component of P. multocida is entirely responsible for protective immunity. All cell wall fractions, LPS, proteins and polysaccharide contribute towards protective immunity. It can be concluded form the present investigation that 27 kDa, 35 kDa, 55 kDa and 70 kDa proteins detected in phage lysates may be associated with a protective response in bird against virulent challenge but, their specific role remains to be elucidated.

Fig. 1. Electrophoretogram showing protein profile of P. multocida A:1 phage lysate (Silver stained) 1-Unconcentrated phage lysate; M-Protien marker; 2 & 3-Concentrated phage lysate
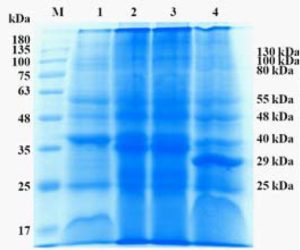
Fig. 2. Electrophoretogram showing cell wall protein profile of sonicated P. multocida (CBB Stained) M-Protein marker; 1- P. multocida A:1, 2-P. multocida A:3, 3- P. multocida A:4, 4- P. multocida P52 strain
The antibody titers detected by IHA (Table-4) using sonicated (Ames, 1974) P. multocida antigens A:1, A:3, A:4 and P52 showed the classical pattern of response against each serotype (Fig. 3, 4 and 5) with initial rise over the pre-vaccination levels, achievement of a peak and then a gradual decline over the period. However, some significant differences were observed with the immunizing preparation amongst plain and alum adsorbed phage lysate.
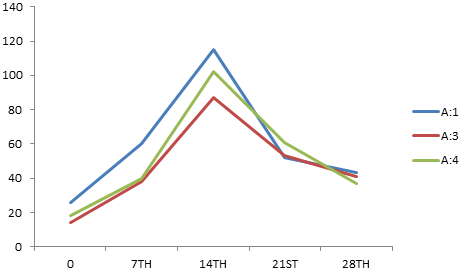
Fig. 3. Mean IHA titer against A: 1, A: 3, A: 4 on various days post immunization with WC vaccine
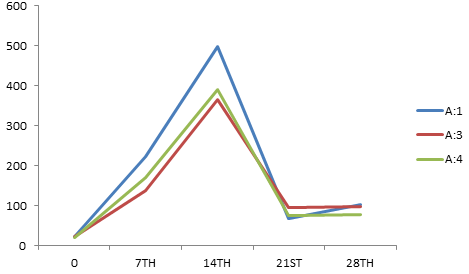 Fig. 4. Mean IHA titer against A:1, A:3, A:4 on various days post immunization with APL vaccine
Fig. 4. Mean IHA titer against A:1, A:3, A:4 on various days post immunization with APL vaccine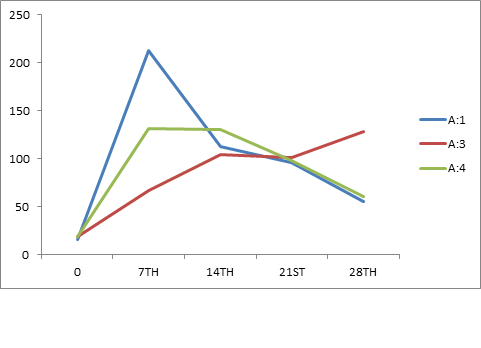 Fig. 5. Mean IHA titer against A:1, A:3, A:4 on various days post immunization with PL vaccine
Fig. 5. Mean IHA titer against A:1, A:3, A:4 on various days post immunization with PL vaccineTable (2):
Immunization and challenge schedule of birds.
| Immunization | Challenge (80% lethal dose) (Sub-groups) (N) |
||||
|---|---|---|---|---|---|
| Group (N) | Test preparation (Dose, route) |
Bleeding schedule (days) | A:1 | A:3 | A:4 |
| I (15) | WC; 1 ml; S/C Booster on 14th day |
Pre-immunization, 7th, 14th, 21st and pre-challenge (28th) |
5 | 5 | 5 |
| II (15) | APL; 0.5 ml; S/C Booster on 14th day | -do- | 5 | 5 | 5 |
| II (15) | PL; 0.5 ml; S/C Booster on 14th day |
-do- | 5 | 5 | 5 |
Table (3):
Protective response of birds against 3 serotypes of P. multocida following immunization with whole cell and phage-lysate vaccines.
| Vaccine group | Challenge Serotype |
No. Survived/ challenged |
Percent survival against challenge serotype |
Overall percent protection afforded against all serotypes |
|---|---|---|---|---|
| Whole cell | A:1 | 4/5 | 80 | 33 |
| A:3 | 0/5 | – | ||
| A:4 | 1/5 | 20 | ||
| PL | A:1 | 3/4 | 75 | 58 |
| A:3 | 3/5 | 60 | ||
| A:4 | 2/5 | 40 | ||
| APL | A:1 | 5/5 | 100 | 80 |
| A:3 | 3/5 | 60 | ||
| A:4 | 4/5 | 80 | ||
| Controls | A:1 | 1/5 | 20 | 7 |
| A:3 | 0/5 | – | ||
| A:4 | 0/5 | – |
Table (4):
Mean Indirect Haemaggultination (IHA) titer on various DPI amongst un-immunized, WC, APL, PL against A:1, A:3, A:4.
| A:1 | A:3 | A:4 | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pre-imm | 7th | 14th | 21st | 28th | Pre-imm | 7th | 14th | 21th | 28th | Pre-imm | 7th | 14th | 21st | 28th | |
| UI | 15 | 23 | 43 | 25 | 30 | 7 | 19 | 39 | 32 | 43 | 10 | 23 | 33 | 34 | 35 |
| WC | 26 | 60 | 115 | 52 | 43 | 14 | 38 | 87 | 53 | 41 | 18 | 40 | 102 | 61 | 37 |
| APL | 22 | 222 | 497 | 68 | 140 | 22 | 137 | 365 | 95 | 98 | 21 | 171 | 391 | 76 | 79 |
| PL | 16 | 213 | 113 | 96 | 55 | 19 | 67 | 104 | 101 | 128 | 19 | 131 | 130 | 98 | 61 |
Protective response trials on groups of chickens immunized with whole-cell and phage-lysate preparations against challenge with virulent doses of A:1, A:3 and A:4 serotypes revealed that the inactivated whole cell preparation provided a satisfactory protection only against homologous A:1 challenge (80 percent) but not against heterologous A:3 and A:4 serotypes. The phage-lysate preparations on the other hand were found capable in imparting protection against all serotypes. The un-adjuvanted preparation was found to induce partial protection against challenge whereas, the alum gel adsorbed preparation provided satisfactory cross-protective immunity (Table:3).
Since, the phage-lysates were found to induce a satisfactory across-serotype protective response in birds at lower dose, it can be concluded that the phage-lysate of P. multocida A:1 is a better immunizing preparation than the whole-cell inactivated preparations.
ACKNOWLEDGMENTS
Authors are thankful to Director, Indian Veterinary Research Institute, Izatnagar-243122 for providing the necessary facilities to carry out this research work.
- Wilson, B. A., Ho, M. Pasteurella multocida: from zoonosis to cellular microbiology. Clin. Microbio. Rev. 2013; 26(3):631–655.
- Watt, J. M., Swiatlo, E., Wade, M. M., Champlin, F. R. Regulation of capsule biosynthesis in serotype A strains of Pasteurella multocida. FEMS Microbiol. Lett., 2003; 225:9-14.
- Rani, S. S., Chaturvedi, V. K., Gupta, P. K., Pandey, K. D., Kumar, B., Kumar, S., Effect of various growth conditions on the biomass of P. multocida (P52) in fermenter. Indian J. Comp. Microbiol. Immunol. Infect. Dis., 2006; 27(2):116-117.
- Marchart, J., Dropmann, G., Lechleitner, S., Schlapp, T., Wanner, G., Szostak, M.P., Lubitz W. Pasteurella multocida– and Pasteurella haemolytica– ghosts: new vaccine candidates, Vaccine, 2003; 21:3988-3997.
- Shivachandra, S. B., Viswas, K. N., Kumar, A. A. A review of Hemorrhagic Septicemia in cattle and buffalo. Ani. Health Res. Rev. 2011; 12(1); 67-82.
- Parry, H., Allen, W. D., & Porter, P. Intestinal Immune Response To E. coli Antigens In The Germ-Free Chicken. Immunol., 1997; 32:731-741.
- Rimler, R. B. and Rhoades, K. R. Solubilisation of membrane-associated cross-protection factor(s) of Pasteurella multocida. Avian Dis., 1989; 33:258-263.
- Wang, C. and Gilsson, J. R. Passive cross-protection provided by antisera directed against in-vivo expressed antigens of Pasteurella multocida. Avian Dis., 38(3):506-14.
- Diallo, I. S., Frost, A. J. Characteristics of a haemolytic extract from avian Pasteurella multocida Vety. Microbiol. 2000; 72:37-45.
- Pasterneck, Method for vaccination of poultry by bacteriophage lysate bacterin. 2009; US 2009/ 0297561 A1.
- Jain, L., Rawat, M., Prajapati, A., Tiwari, A. K., Kumar, B., Chaturvedi, V. K., Saxena, H. M., Ramakrishnan, S., Kumar J., Kerketta, P. Protective immune-response of aluminium hydroxide gel adjuvanted phage lysate of Brucella abortus S19 in mice against direct virulent challenge with B. abortus 544. Biologicals., 2015; 43:369-376
- Lowry, O. H., Rosbrough N. J., Farr A. L., and Randall R. J., Protein measurement with the Folin- Phenol reagent. J. Biol. Chem., 1951; 193:265-275.
- Dubois M., Gilles, K. A., Hamilton J. K., Rebers P. A., Smith F. Colorimetric method for determination of sugars and related substances Anal. Chem., 1956; 28(3):350-356.
- Indian pharmacopoeia Vol. III: section 2.2.11., 2014, Government of India, Ministry of Health and Family Welfare., Ghaziabad: Indian Pharmacopoeia Commission.
- Ames, G. F. Resolution of bacterial protein by polyacrylamide gel electrophoresis on slabs. J. Biol. Chem. 1974; 249:634-644.
- Sawada T, Rimler R, Rhodes K. Indirect Haemaggultination test that uses glutaraldehyde-fixed sheep erythrocytes sensitized with extract antigens for detection of Pasteurella multocida. J. Clin. Microbio., 1982; 15(5):752-756.
- Durairajan, R., Development and characterization of bacteriophage-lysed bacterin of Pasteurella multocida., 2012, PhD Thesis submitted to (Deemed University) Indian Veterinary Research Institute, Izatnagar (UP), India.
- Johnson, R. B., Dawkins, H. J., Spencer, T. L., Electrophoretic profiles of Pasteurella multocida isolates from animals with Hemorrhagic Septicaemia Am. J. Vety. Res., 1991; 52(10):1644-1648.
© The Author(s) 2017. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


