ISSN: 0973-7510
E-ISSN: 2581-690X
The bio preservative effect of purified bacteriocin produced during lactic acid fermentation of wild Himalayan fig pickle was investigated in RTS-drink. A total number of seven isolates (SS1, SS2, SS3, SS4, SS5, SS6 and SS7) were isolated from fermented wild Himalayan fig pickle. SS7 isolate was identified as Lactobacillus genus according to Bergeys Manual of Systemic Bacteriology. SS7 isolate of Lactobacillus genus produced bacteriocin with maximum antagonistic activity against Escherichia coli, listeria monocytogenes and Bacillus cereus, though it was more effective against Bacillus cereus than others. Completely purified bacteriocin showed increased in zone of inhibition against food borne pathogens then partially purified bacteriocin. Bacteriocin was thermo stable as it retains its stability up to 121 °C. The maximum antimicrobial activity was retained within the pH range of 6–7, but it was adversely affected by the addition of proteolytic enzyme papain. The bio preservative efficacy of bacteriocin of SS7 isolate was found in RTS-drink. In RTS-drink, Minimal Inhibitory Concentration of bacteriocin was found to be 4.0 mL for Bacillus cereus, E. coli and 4.5 mL for L. monocytogenes.
Bacteriocin, Natural lactic acid, Fig Fruit
Lactic acid microbes (LAB) have been utilized for a considerable length of time in the fermentation of food, for flavor and texture, as well as because of their capacity to prevent the development of pathogenic microorganisms (McAulife et al., 2001). The interest for the utilization of microorganisms and their metabolites for the avoidance of food spoilage and the expansion of shelf-life of food have expanded amid the most recent decade (Sanlibaba et al., 2009). The utilization of more food with chemical preservatives has additionally expanded customer concern and made an interest for more “natural” and “minimal processed” food. Accordingly, there has been an extraordinary interest for naturally produced antimicrobial agents. Further, bacteriocins are inhibitory against foodborne pathogens, for example, L. monocytogenes and are not antibiotics. They can be distinguishing from clinical antibiotics on the basis of synthesis and method of activity (Cleveland et al., 2001).
Bacteriocins synthesized ribosomally and are antibacterial polypeptides that are typically inhibitory to strains firmly identified with the producing microorganisms. (Nes and Johnsb org, 2004). Bacteriocin production from Gram-positive microorganisms, specifically, the lactic acid microorganism show genuinely wide inhibitory spectra with preservation of food (Galvez et al., 2008) and therapeutic (Jack et al., 1995) potentials. Since, protection of food has turned into an undeniably essential global concern, the use of antimicrobial peptides from lactic acid bacteria (LAB) that objective food pathogens without creating toxic metabolites or other unfavorable impacts has gotten incredible consideration. Late flare-ups of emerging pathogens, for example, L. monocytogenes have incited the food industry, people in general, and the administration to scrutinize the sufficiency of current strategies for food preservation (Cleveland et al., 2001). The inhibitory range of some bacteriocins incorporates food deterioration and food borne pathogenic microorganisms (Todorov and Dicks, 2005). Production of bacteriocins by lactic acid bacteria (LAB) have gotten significant consideration as of late on account of their conceivable use as food additives, because of lessening in the utilization of chemical preservatives. The bacteriocin produced by the strains isolated from naturally fermented wild Himalayan fig fruit has neither been characterized nor did its efficacy checked in RTS-drink. Therefore, the present investigations were carried out and the results obtained are discussed here.
Fermented wild Himalayan fig fruit
The mature fruits were collected from mid-hill region of Himachal Pradesh. Unripened wild Himalayan fig fruits were collected and fermented to prepare lactic acid fermented pickle. The pickle was prepared using 2 % salt and 1 % mustard seeds. Natural fermentation of the mixture was monitored till the lactic acid concentration reached 0.75 % after completion of natural fermentation isolation was done. Isolation of predominant microflora was done from this sample.
Food borne pathogens
Food borne pathogens i.e. E.coli 0165, Listeria monocytogenes MTCC 2729 and Bacillus cereus MTCC 5521 were procured from IMTECH, Chandigarh. These standard test pathogens were used in screening of bacteriocin producing bacterial isolates and to check the efficacy. The procured test strains were sub cultured twice on nutrient agar at 37ºC, and later cultures were preserved in 40 % glycerol at -20ºC.
Isolation and identification of bacteriocin producing bacteria
The bacteriocin producing bacteria from naturally fermented wild Himalayan fig pickle was isolated by spread plate method using MRS agar. After incubation for 48 h at 37°C, typical colonies were isolated. Further, streaked on MRS agar and the pure lines were obtained. The isolates were differentiated on the basis of their morphological, biochemical characteristics such as oxidase and catalase test and motility test were tentatively identified up to the genus level (Baird-Parker, 1975).
Screening of isolates for antagonistic activity
Antimicrobial activity of the bacterial isolates against all the pathogenic microorganisms was determined by well diffusion method (Kimura et al., 1998). MRS agar plates were inoculated with 100 mL of each target microorganism after growing them in a broth and diluting appropriately. In the well diffusion method wells of 7 mm diameter were cut out of agar plates containing growth of food borne pathogens. 100 µl cultures of each isolated bacteria was poured in the wells of each indicator lawned petri plate. The plates were then incubated at 37 ºC for 24 h and zones of inhibition against maximum number of inhibitor were observed. The isolates which showed the maximum zone of inhibition against indicator microorganisms were selected for further studies.
Partial purification with ammonium sulphate
Isolated bacteria showing maximum zone of inhibition was grown in MRS broth at 37 °C for 48 h. After incubation, the broth was centrifuged at 5000 rpm for 10 min and the cells were separated out. Supernatant was used as a crude bacteriocin. The crude bacteriocin sample produced was treated with solid ammonium sulphate to 0, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75 and 80 per cent saturation. The mixtures were stirred for 2 h at 4 ºC and later centrifuged at 20,000 rpm for 1 h (4ºC). The precipitates were re-suspended in 25 ml of 0.05 M potassium phosphate buffer (pH 7.0). Inhibition zone of different fractions was recorded in comparison with the crude bacteriocin.
Complete purification with Gel filtration chromatography (Gautam and Sharma, 2015)
3 gm of sephadex G-75 was suspended in 500 ml of buffer (0.5 M phosphate buffer) for 48 h. When sephadex G-75 swelled it was packed in the column having dimensions of (31×2.5 cm). To avoid the air bubbling in column continuous flow of buffer was there. 3 ml of Fraction 1 was loaded on the sephadex G-75 column. Elution of the column was done with help of 0.5 M phosphate buffer and 3 ml fractions were collected. A flow rate of 3 ml in 7 min was maintained throughout the process. The protein content of the collected fractions was measured at 280 nm. Further, these fractions were observed for the bacteriocin activity. The active fractions having maximum bacteriocin activity were pooled together and stored at 4º C. To estimate the molecular weight and purity of the protein the sample was checked out by SDS-PAGE (Sambrook and Russel, 2001).
Characterization of bacteriocin
Thermostability (Joshi et al., 2006)
5 ml of bacteriocin was taken in test tubes. Each test tube then overlaid with paraffin oil to prevent evaporation and then treated at different temperature at 40, 70, 90 and 121oC for 15 min. The heat treated samples was then be assayed for antagonistic activity.
Effect of pH (Todorov and Dicks, 2005)
5 ml bacteriocin was taken in test tubes. The sample in the test tubes adjusted at 2-9 pH range, each test tube with dilute NaOH or HCl (1 N NaOH or 1 per cent HCl solution) and allowed to stand at room temperature for 2 hrs. Further, the treated bacteriocin was assayed using well diffusion method.
Effect of proteolytic enzyme (papain) (Joshi et al., 2006)
A 5 mL sample of bacteriocin was taken in test tubes and treated with enzyme papain (100 TU) 1 mg/mL at pH 7.0. The test tubes with and without enzyme (control) were incubated for 2h at 37°C and heated for 3 min at 100°C to denature the enzyme. Both the control and the sample were assayed for antagonistic activity.
Determination of preservative effect of bacteriocin in RTS-drink (Joshi et al., 2006)
The RTS-drink was sterilized and inoculated with test organism Bacillus cereus, Escherichia coli and Listeria monocytogenes. Initial count of inoculated sample was recorded and then bacteriocin was added. After 24 and 72 hrs the plate count was observed and compared with the control (without bacteriocin).
Based on morphological characters i.e. Colour, form, margins, elevation, texture and biochemical test such as catalase and oxidase test of bacterial isolates were noted down. The colour of colonies varied cream, white to dirty white. Majority of isolates were white in colour and very less were dirty white. The forms of bacterial colonies were varied from circular to irregular. Among all the isolates only SS6 and SS2 have undulate margins. The textures of 5 isolates were same i.e. mucoid except two isolates SS4 and SS7. Gram staining was performed with all the isolated bacteria. All the isolates were found to be rod shaped and gram negative in gram staining reaction except two isolate i.e. SS4 and SS7. Motility and catalase test were positive in case of all the isolates except SS4 and SS7. Isolate SS7 from wild Himalayan fig pickle produced the maximum zone of inhibition against all the food borne pathogens and was maximum against Bacillus cereus. Mondal and Aruna (2014) studied that the bacteriocin produced by Lactobacillus casie and Streptococcus species inhibited the growth of Pseudomonas aeruginosa, Staphylococcus aureus, Escherichia coli, Klebsiella species.
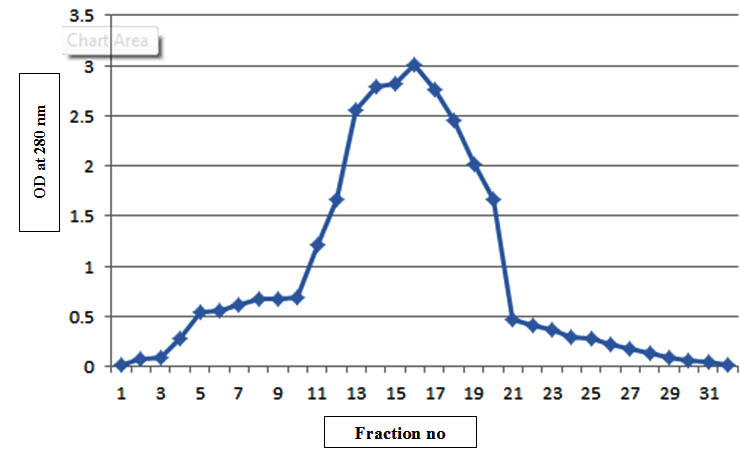
Fig. 1. Elution profile of completely purified bacteriocin
There was increase in the antagonistic activity of bacteriocin from partial purification to complete purification (Table 1). The purification fold of bacteriocin by complete purification was increased upto 4.10 from 1.6. The fraction number 13 to 19 having highest antagonistic activity. The zone of diameter increased from 12 mm to 24 mm after complete purification of bacteriocin against Bacillus cereus. This indicates that the protein purified by gel filtration chromatography showed the good results. The antagonistic activity of purified protein ascertained the genuine status of bacteriocin. Completely purified bacteriocin was then lyophilized and was loaded in the wells 15 % gel with different concentration of 10 and 15 µl along with marker. The single band of approx 7.5 kDa was observed (Plate 1) indicating the homogenity of protein. Bacteriocin synthesized from L. fermentum MF belongs to Class II <10 kDa. Since Class I bacteriocin having molecular weight <5 kDa and Class III having >10 kDa (Rajaram et al., 2010).
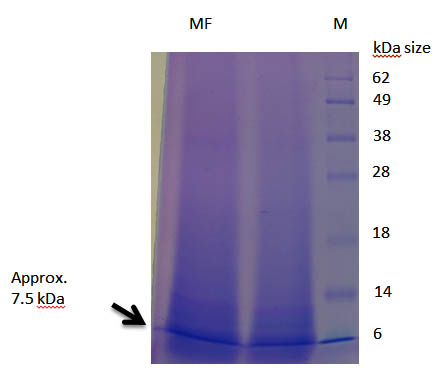
Plate 1. SDS page of completely purified bacteriocin
Table (1):
Purification and recovery of bacteriocin produced by SS7 isolate
Purification Steps |
Crude Bacteriocin |
Partial Purification (Ammonium Sulphate Precipitation) |
Complete Purification (Gel Filtration Chromatography) |
|---|---|---|---|
Volume (Ml) |
100 |
20 |
2 |
Activity Unit (Au/Ml) |
3×103 |
7×103 |
9×103 |
Total Activity (Au) |
3×105 |
1.4×105 |
1.8×104 |
Protein (Mg/Ml) |
2.012 |
2.981 |
1.536 |
Specific Activity (Au/Mg) |
1427.21 |
2348.20 |
5859.37 |
Purification Fold |
1 |
1.6 |
4.10 |
Recovery |
100 |
141 |
73 |
Bacteriocin retains its stablility at 40 ºC, 70 ºC, 90 ºC and 121 ºC against test pathogens. The zone size at 40 ºC was 22 mm, 18 mm and 15 mm for E. coli, Bacillus cereus and listeria monocytogenes respectively. The purified bacteriocin is stable at wide ranges of temperature (Figure 2). Similar result was observed by Mojgini et al., (2009) they compared the heat stability of bacteriocins of L. brevis and L. fermentum. The observation was that the bacteriocin produced by L. brevis NM 24 retained its activity after heating at 121 p C for 60 minutes. The thermo stability of this bacteriocin discussed here demonstrates the bio preservative property in combination with thermal processing for food products preservation.
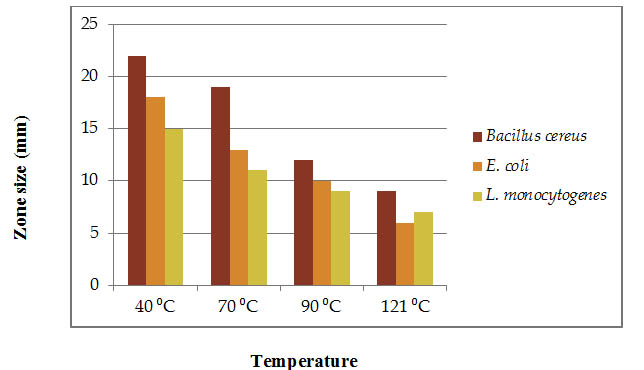
Fig. 2. Effect of temperature on completely purified bacteriocin
It was observed that completely purified bacteriocin was most active against all the three test indicators within the pH range of 6.0 to 7.0. At pH 7.0 completely purified bacteriocin inhibited E. coli, B. cereus and L. monocytogenes by forming zones of 13 mm, 21 mm and 7 mm showed in Figure 3. When pH shifted to slight acidic side i.e. at pH 5.0 the zone size decreased to 3 mm against B. cereus, E. coli (2 mm) and L. monocytogenes (3 mm). Beyond pH 5.0 to 2.0 the completely purified bacteriocin lost its activity. On alkaline side after pH 7.0 the bacteriocin become partially sensitive to B. cereus (3 mm), E. coli (2 mm) and L. monocytogenes (3.4 mm). Oh et al., (2000) studied that the bacteriocin was completely stable at pH 6.0 and 7.0 and 50 % of activity remained after subjection to the pH values between 3.0 and 10.0. Bacteriocin loses its activity completely after treatment with proteolytic enzyme. The bacteriocin Pediocin ACH from Pedicoccus acidilacti was sensitive to proteolytic enzymes and was completely inactivated by several proteolytic enzymes (Singhal and Kulkarni, 1999). The steadiness of bacteriocin to various conditions reveals that such compounds can withstand the conditions typically experienced in food handling, so it would sustain during processing.
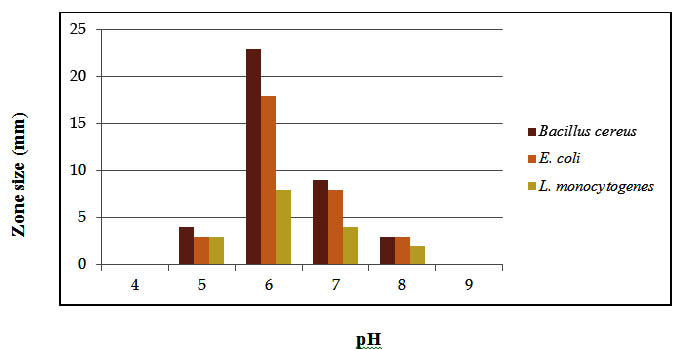
Fig. 3. Effect of ph on completely purified bacteriocin
The completely purified bacteriocin from isolate SS7 was tested for preservative effect against B. cereus, E. coli and L. monoctogenes. The preservative effect in RTS-drink increased with the increase in concentration of bacteriocin. The highest percent reduction was observed 4.0 ml for B. cereus and 4.5 ml for E. coli and L. monoctogenes as shown in Table 2. The recommended units for nisin (widely accepted Bio preservative) the commercial bacteriocin is 10-50 ppm. Nisin must be used in dosages of less than 0.5g/kg (500 mg/1000 gm) in meat and dairy products. Apply as 5 % aqueous solution using sterile water (Jones, 2005). Briefly, the outcomes show that bacteriocin had a few alluring attributes of a biopreservative.
Table (2):
Efficacy/Preservative effect of completely purified bacteriocin in RTS drink against Bacillus cereus, E.coli and L. monocytogenes
| Bacteriocin Concentration (Ml) | Percent Reduction | Total Plate Count (Cfu/Ml) | ||||
|---|---|---|---|---|---|---|
| Bacillus cereus | E.Coli | L. monocytogenes | Bacillus Cereus | E.coli (A=98) | L. monocytogenes (A=134) | |
| Control Of Culture | Nil | Nil | Nil | 120 | 98 | 134 |
| 0.5 | 18.3 | 15.3 | 21.6 | 98 | 83 | 105 |
| 1.0 | 29.1 | 37.7 | 35.0 | 85 | 61 | 87 |
| 1.5 | 42.5 | 50.0 | 41.0 | 69 | 49 | 79 |
| 2.0 | 60.8 | 61.2 | 51.4 | 47 | 38 | 65 |
| 2.5 | 73.3 | 77.5 | 64.1 | 32 | 22 | 48 |
| 3.0 | 86.6 | 88.7 | 80.5 | 16 | 11 | 26 |
| 3.5 | 94.1 | 95.9 | 85.8 | 7 | 4 | 19 |
| 4.0 | 100 | 100 | 92.5 | 0 | 0 | 10 |
| 4.5 | – | – | 100 | – | – | 0 |
OD=600 nm, Absorbance= 0.589 For Bacillus cereus
OD=600 nm, Absorbance= 0.521 For E. coli
OD=600 nm, Absorbance= 0.603 For L. monocytogenes
*Percent Reduction =(A-B) X 100/A
Where,
A is the number of viable microorganisms before treatment
B is the number of viable microorganism after treatment (Total plate count of viable microorganism)
- McAulife O, Ross R P, Hill C L. Antibiotics: structure, biosynthesis and mode of action. FEMS Microbiology Reviews. 2001; 25: 285–308.
- Sanlibaba P1nar, Nefiseakkoc, Mustafaakcelik. Identification and characterisation of antimicrobial activity of nisin a produced by lactococcus lactis subsp. Lactis ll27. Czech journal of Food science. 2009; 27(1): 55-64.
- Cleveland J, Montville T J, Nes F I, Chikindas M L. Bacteriocins: safe, natural antimicrobials for food preservation. International Journal of Food Microbiology. 2001; 71:1–20.
- Nes I F, Johnsborg O. Exploration of antimicrobial potential in LAB by genomics. Curr. Opin. Biotech. 2004; 15:100–104.
- Galvez A, Lopez R L, Abriouel H, Valdivia E, Omar N B. Application of bacteriocins in the control of food borne pathogenic and spoilage bacteria. Crit. Rev. Biotechnol. 2008; 28:125-152.
- Jack R W, Tagg J R, Ray B. Bacteriocins of gram-positive bacteria, Microbiol. Rev. 1995; 59:171-200.
- Todorov S M, Dicks L M T. Lactobacillus plantarum isolated from molasses produces bacteriocins active against Gram-negative bacteria. Enzyme and microbial techonology, 2005; 36:318-326.
- Baird-Parker A C. Gram-Positive Bacillus. In: Bergey’s Manual of Determinative Bacteriology, R.E. Buchanan, N.E. Gibbons (Eds.), Williams and Wilkins Co., Baltimore, USA (1975) pp. 492–515.
- Kimura H, Sashihara T, Matsusaki H, Sonomoto K, Ishizaki A. Novel bacteriocin of Pediococcus sp. ISK-1 isolated from well – aged bed of fermented rice bran. Annals of New York Academy of Sciences, 1998; 864: 345-348.
- Gautam N and Sharma N. Purification and characterization of purified bacteriocin of Lactobacillus brevis isolated from traditional fermented food of H.P. Indian Journal of Biochemistry and Biophysics 2009; 46: 337-341.
- Joshi V, Sharma S and, Rana N. Production, Purification, Stability and Efficacy of Bacteriocin from Isolates of Natural Lactic Acid Fermentation of Vegetables. Food Technology and Biotechnology. 2006; 44(3): 435–439.
- Sambrook J and Russell R W. Molecular cloning: A laboratory manual, 3rd ed. Cold spring harbor laboratory press, cold spring harbor, N.Y. (2001).
- Mondal R and Aruna B. Biopreservative activity of bacteriocin-producing lactic acid bacteria isolated from fermented green gram batter. Intl. J. Food. Ferment. Technol. 2014; 4(2): 121-128.
- Rajaram G, Manivasagan P, Thilagavathi B and Saravanakumar A. Purification and Characterization of a Bacteriocin Produced by Lactobacillus lactis Isolated from Marine Environment. Advance Journal of Food Science and Technology. 2010; 2(2): 138-144.
- Mojgani N, Sabiri G, Ashtiani M P and Torshizi M A K. Characterization of Bacteriocins produced by Lactobacillus brevis NM 24 and L. fermentum NM 332 Isolated from Green Olives in Iran. The International Journal of Microbiology 2009; 6.
- Oh S, Kim S H and Worobo R W. Characterization and purification of bacteriocin produced by a potential probiotic culture L. acidophilus 30SC. Journal of Dairy Science 2000; 83(12): 2747-2752.
- Singhal R S and Kulkarni P R. Production of food additives by fermentation. In: Biotechnology: Food Fermentation, Microbiology, Biochemistry and Technology. Vol II. (eds.) V.K. Joshi and Ashok Pandey, Educational Publishers and Distributors, New Delhi (1999) pp. 1159.
- Jones E, Salin V, Gary W and Williams. Nisin and the market for commercial bacteriocins. TAMRC Consumer and Product, Research Report No. CP-01-05.146. 2005.
© The Author(s) 2018. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


