ISSN: 0973-7510
E-ISSN: 2581-690X
Two Bacillus strains isolated from Ntoba mbodi : Bacillus megaterium (B. me NM 02) Bacillus licheniformis (B. li NM01), has shown a significant caseinolytic enzyme activity. We set optimization of growth and enzyme production conditions. Several parameters have been optimized: temperature, pH, various types of media, carbon and nitrogen sources. In both strains, growth is possible from 25 to 60°C with an optimum temperature at 37°C for B.li and at 35°C for B. me. Enzyme production was observed from 25 to 55°C with an optimum temperature of 30°C for B. li. Enzyme production was observed from 25 to 50°C with an optimum temperature of 35°C in B. me. For pH, growth and enzyme production can be at 5, 7 and 9 with an optimum at 7 for both strains. The LB medium is better for growth and enzyme production than TSB for B. li and B. me. Among the carbon sources used, fructose is better for growth after 48 hours of incubation in both strains (B. li: 0,93±0,001, B.me: 0,928±0,002), but for enzyme production fructose remains the best carbon source for B. li (14,33±1,24) , while starch is the best for B. me (14.66±1,24). Concerning nitrogen sources, in both strains the best source of growth is the yeast extracts (B. li: 0,969±0,015, B.me: 0,952±0,01). For enzyme production, the two sources can be used for B. li. (14,333±1,247) but for B. me (14,333±0,471) only the yeast extracts is the best as well as for growth and enzyme production. Furthermore, in both strains the produced enzyme was partially purified by using ammonium sulfate precipitation, and SDS-PAGE has been hold, profiles of specific bands are useful to give more information and differentiate the two strains.
Optimization, caseinolytic enzyme, Ntoba mbodi, Bacillus licheniformis, Bacillus megaterium.
Enzymes are macromolecules involved in many cellular reactions. Proteases are groups of proteins included in the subclass hydrolases, within the main class enzymes. Serine alkaline proteases (SAP) are one of the most important groups of industrial enzymes. They account for approximately 35% of the total microbial enzyme sales1.
Proteases or proteolytic enzymes find many applications in many domains as : pharmacy, food, biotechnology, and detergents2, 3. Commercially used proteases are produced by bacteria, plants , animals and fungi4, 5. Proteases are produced by many groups of bacteria: Bacillus, Pseudomonas and others. The genus Bacillus is recognize as one of the most important producer of exocellular proteases1,5. Proteases production by micro-organisms is a natural phenomenon. Bacillus species produce proteases during the exponential and stationary phases during growth. Extracellular protease production is strongly influenced by media composition, the ration of C/N, metabolizable sugars6 and metallic ions1,7. More over protease production is also affected by rapidly metabolizable source, like amino acids in the medium and by some physic factors (aeration, inoculum density , pH and temperature). Proteases production by microorganisms is connected to the growth and that is characteristic of each microorganism1. Many studies have been focused on proteases production by microorganisms, these studies have stated strategies of growth and protease production7, 8, 9. Some studies have given information in the relation between growth and protease production. Those studies corroborate with the hypothesis that : proteases production is controled by number of mechanisms that occur in the transition between the end of the exponential phase and the begening of the stationary phase10, 11, 12. By any way all studies are strict about the variability and make an amphasis on the optimal conditions of protease production that, depend strictly on each microorganism13, 14,15.
Caseinolytic proteases P(ClpPs) are complex of oligoméric proteines with contibute on cellular homeostasy and virulence regulation to bacteria. Whatever most of organisms possesse only one ClpP protein, in some can be encoded two or more ClpP isoforms16. The caséinolytic protease P(ClpP) is an highly conserved enzyme in bactéria and higher organisms17, 18, 19.
In the republic of Congo fermented manioc leaves are used as a food by Congolese in some area. They are differently called : Ntoba mubodi in the Bouenza, Lilleyuka in the Kouilou and Ntoba mbodi in the Pool. Microbiological study of the Ntoba Mbodi has been hold and has shown a diversity of bacteria, among them the species of genus Bacillus20.
Many species of genus Bacillus isolated from Ntoba Mbodi has been molecularly characterized by their ARNr 16S and ARNr 16S-23S . Many of them have shown proteolytic activities, as the fibrinolytic enzyme producer Ba NM76 described in15. A new strain named Lysinibacillus louembei sp. with a strong protéolytic activity has been isolated from Ntoba Mbodi.21.
Two Bacillus strains isolated from Ntoba mbodi : Bacillus megaterium (B. me NM 02) Bacillus licheniformis(B.li NM01), have shown a strong caseinolytic enzyme activity. In the present work we optimize growth and enzyme production conditions (temperature, pH, nitrogen and carbon sources), produce the enzyme. In both strains the produced enzyme was partially purified by using ammonium sulfate precipitation. SDS-PAGE has been hold and profils used to differenciate the strains.
Bacterial strains and media
Strains used in this study were Bacillus megaterium(B. me NM 02) and Bacillus licheniformis (B. li NM 01) available in our laboratory were isolated from Ntoba mbodi.
The media used were : Luria Bertani (0,5%NaCl, 1% Tryptone, 0,5% Extraits de levure); Tryptone soya broth TSB); Amidon (0, 5%;) Galactose (0, 5% ); Fructose (0, 5%) ; and mineral solution (MgSO4.5H2O: 0, 06%, KH2PO4: 0, 1%, Cacl2.6H2O: 0.2%, K2HPO 4.0, 2%).
Culture conditions
250ml Erlenmeyer containing 100ml of LB was inoculated with 1ml of overnight culture and incubated at 37°C for 48 hours. 2ml of bacterial suspension were used to compute the optical density (OD) at 600 nm with a type vis spectrophotometer (722 Ningb SI Instrument CO, LTD).12,15
Enzyme assay
Puri modified techniques [9]. was used to test caseinolytic enzyme. The 48h culture was collected in 1,5 eppendorf and centrifuged for 5minutes at 6000rpm in a type microcentrifuge( ).The supernatant was collected for enzyme activity.
1g of agarose was dissolved in 250ml erlenmeyer containing 100 ml de PBS at 0,1N, after boiling, we waited untill à 55-60°C, 10ml of skimmed milk were added. The mixture were poured in petri dishes. Wholes were made on the plate. 50¼l of sample were added in each whole, and the plate was incubated at 37°C for 12 hours. Caseinolytic activity was measured by lytic area through the diameter of the clear zone22, 23.
Influence of temperature on the growth and caseinolytic enzyme production
To study the effect of temperature on growth and caceinolytic enzyme production, we made culture on LB medium at different temperatures (25, 30, 35,37, 40 45, 50,55 et 60°C) for 48 h. Each two hours optical density at 600nm and enzyme activity were computed. Testing caseinolytic activity plates were incubated at 37°C, for 12h. Lytic area through the diameter of the clear zone was measured to show the enzyme activity12, 15, 22, 23.
Influence of pH on the growth and caseinolytic enzyme production
To study the effect of pH on growth and caceinolytic enzyme production, we made culture on LB medium at different pH :5, 7, 9 for 48h. Each two hours optical density at 600nm and enzyme activity were computed. Testing caseinolytic activity plates were incubated at 37°C, for 12h. Lytic area through the diameter of the clear zone was measured to show the enzyme activity15, 22.
Influence of different kinds od media on the growth an enzyme production
Two kinds of media were used to study their effects of on growth and caceinolytic enzyme production, we made culture on LB and TSB at pH : 7 for 48h. Each two hours optical density at 600nm and enzyme activity were computed. Testing caseinolytic activity plates were incubated at 37°C, for 12h. Lytic area through the diameter of the clear zone was measured to show the enzyme activity22, 23, 24.
Influence of carbon sources on the growth and caseinolytic enzyme production
Three kinds of carbon sources (galactose, Fructose, amidon) were used to study their effects on growth and caceinolytic enzyme production, we made culture on three different media which compositions are given in table 1. Each two hours optical density at 600nm and enzyme activity were computed. Testing caseinolytic activity plates were incubated at 37°C, for 12 h. Lytic area through the diameter of the clear zone was measured to show the enzyme activity23, 25,26.
Table (1):
Different media with different carbon sources.
Media |
Carbon sources |
Nitrogen source |
Mineral solution |
|---|---|---|---|
Medium 1 |
Galactose |
Yeast extracts |
MgSO4.5H2O: 0, 06%, KH2PO4 :0,1%, ,2 Cacl2.6H2O :0.2%, K2HPO4 .0, 2% |
Medium 2 |
Fructose |
Yeast extracts |
MgSO4.5H2O: 0, 06%, KH2PO4 : 0,1%, Cacl2.6H2O:0.2%, K2HPO4 .0, 2% |
Medium 3 |
starch |
Yeast extracts |
MgSO4.5H2O: 0, 06%, KH2PO4 : 0,1%, Cacl2.6H2O :0.2%, K2HPO4 .0, 2% |
Influence of nitrogen sources on the growth and caseinolytic enzyme production
Two kinds of nitrogen sources (yeast extracts, Bactopeptone) were used to study their effects on growth and caceinolytic enzyme production, we made culture on two different media which compositions are given in table 2. Each two hours optical density at 600nm and enzyme activity were computed. Testing caseinolytic activity plates were incubated at 37°C, for 12h. Lytic area through the diameter of the clear zone was measured to show the enzyme activity23, 23,26
Table (2):
Different media with different nitrogen sources.
Media |
Nitrogen sources |
Carbon source |
Mineral solution |
|---|---|---|---|
Medium 1 |
Yeast extracts |
starch |
MgSO4.5H2O: 0, 06%, KH2PO4 : 0,1%, Cacl2.6H2O :0.2%, K2HPO4 .0, 2% |
Medium 2 |
Bactopeptone |
starch |
MgSO4.5H2O: 0, 06%, KH2PO4 : 0,1%, Cacl2.6H2O : 0.2%, K2HPO4 .0, 2% |
Proteins SDS-PAGE
Protein SDS- PAGE has been hold. A 48h culture was centrifuged and the supernatant was precipitated with ammonium sulfate. The centrifugation of the solution was at 4°C, 10000t/min for 1hour with the centriguge type MICRO STAR 17R from VWR.. The precipitate was diluted in PBS and 100 µl was used as a sample
The reagents were: Acrylamids 30% / biacrylamids 0.8 %; Tris-HCL 1.5 M pH 8.8; Tris-HCL 0.5 M pH 6.8 ; Electrophoresis buffer : Tris 25mM, Glycine0.192 M, SDS 0.1%; Staining solution : Coomassi bleu 2.5g, méthanol 450ml, glacial acétic acid 100 ml, eau 400 ml, ajusted at 1 l; Dying solution : méthanol, glacial acétic acid 100 ml, eau 400ml, ajusted at 1 l.; Protein molecular markers protéines (Protein Page Ruler prestained, Fermentas)
Caseinolytic enzyme activity
The Figure 1 shows the caseinolytic activity in three bacillus strains isolated from Ntoba Mbodi : B. me., B. li., B. amy. The B. amy. is used as a positive control. The halo of B. me is more important than of the positive control. The halo of B.li is as important as af a positive control. The diameter of the halo is the expression of the enzyme production of the strain. E. coli is used as a negative control.
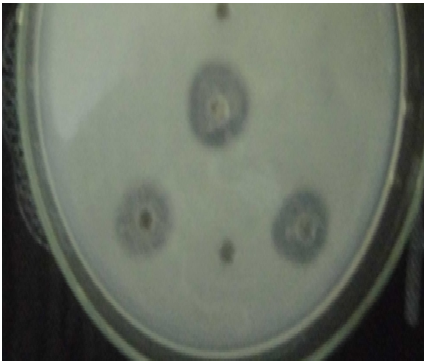 B. me : Bacillus megaterium, B. li : Bacillus licheniformis, B. amy: Bacillus amyloliquefaciens
B. me : Bacillus megaterium, B. li : Bacillus licheniformis, B. amy: Bacillus amyloliquefaciensFig. 1. Halo of caseinolytic enzyme activity of different Bacillus strains isolated from Ntoba Mbodi
Influence of température on growth and caseinolytic enzyme production
Fig-2 shows the influence of temperature on growth profiles for Bacillus licheniformis with the time. For the temperatures from 25 to 40°C, the increase of temperature is proportional with growth profiles. In contrast, after 40°C the increase of temperature become inversely proportional to the growth profiles. Growth is possible from 25 to 60°C, with the optimal temperature at 37°C.
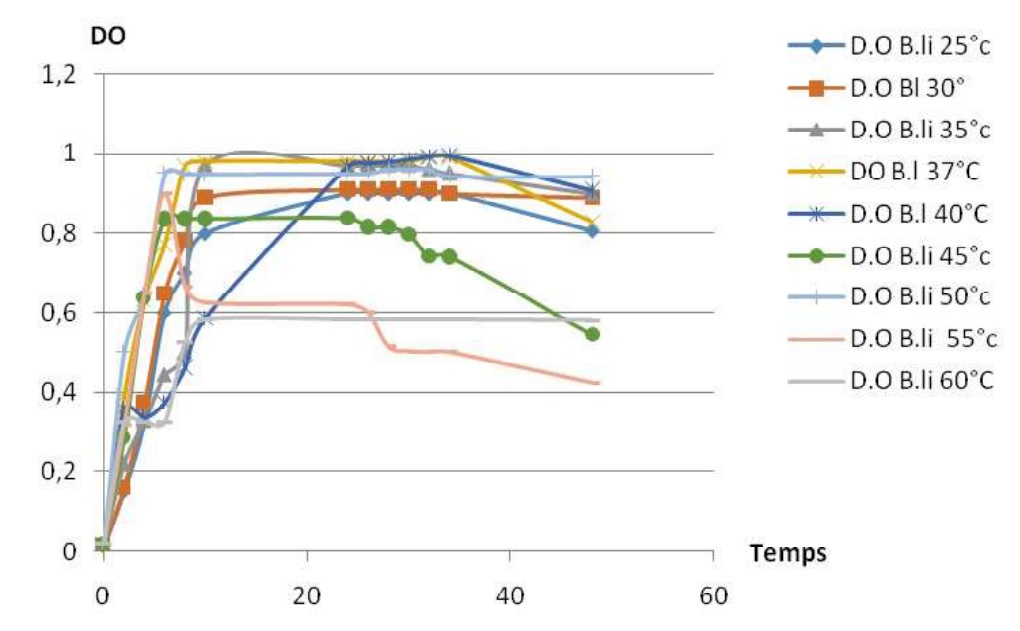 Fig. 2. Growth profiles of Bacillus licheniformis at different temperatures
Fig. 2. Growth profiles of Bacillus licheniformis at different temperatures All growth profiles have the sames phases; exponentially phase, stationary phase and the decreasing phase, but the different amplitudes.
Fig-3 shows the influence of temperature on caseinolytic enzyme production for Bacillus licheniformis. Indeed, the enzyme activity here is used to express the enzyme production with the incubation time. For the temperatures from 25 to 30°C the amplitude of caseinolytic enzyme production profils are proportional to increase of the temperature. For the temperatures from 30 to 55°C the amplitude caseinolytic enzyme production profiles is inversely proportional the increase of temperature. courbes de la production enzymatique augmentent, cela indique que la production enzymatique augmente. Caseinolytic enzyme production is possible for the temperature from 25 to 55°C, with optimal temperature of 30°C.
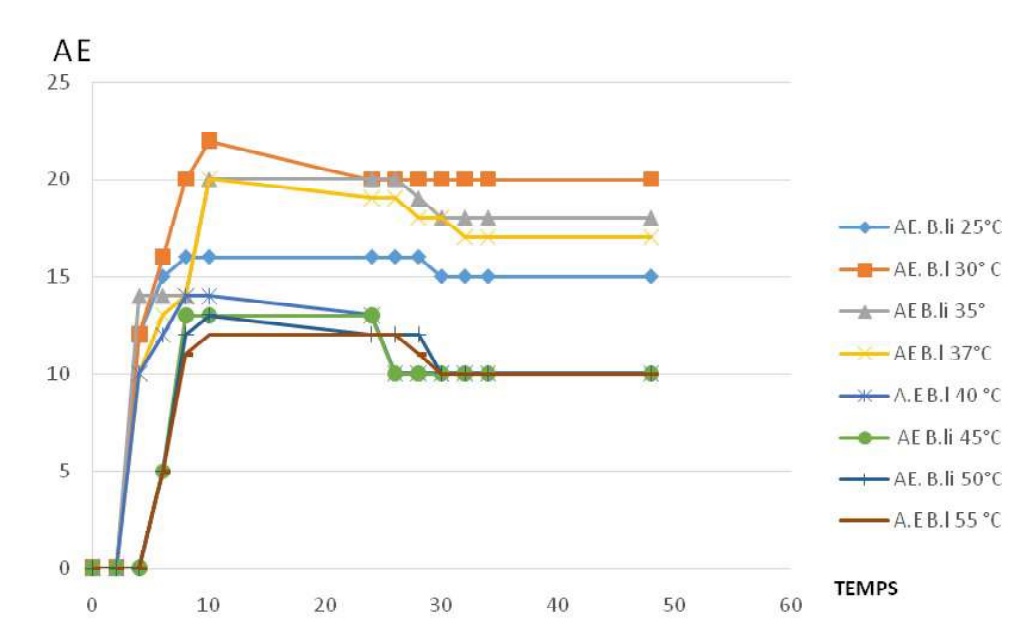 Fig. 3. Profiles of caseinolytic enzyme production by Bacillus licheniformis at different temperatures.
Fig. 3. Profiles of caseinolytic enzyme production by Bacillus licheniformis at different temperatures. All profiles of caseinolytic enzyme production have the sames phases; latency phase , exponentially phase of enzyme production, stationary phase of enzyme production and the decreasing phase, but the different amplitudes.
Fig-4 shows the influence of temperature on growth for Bacillus megaterium. For the temperatures from 25 to 45°C, the increase of temperature is proportional to the increase of the amplitudes of growth profiles. For the temperatures from 40 to 60°C, the increase of temperature is inversely proportional to the amplitudes of growth profiles. The growth of the strain is possible from 25 to 60°C, with the optimal temperature at [35°C-37°C]
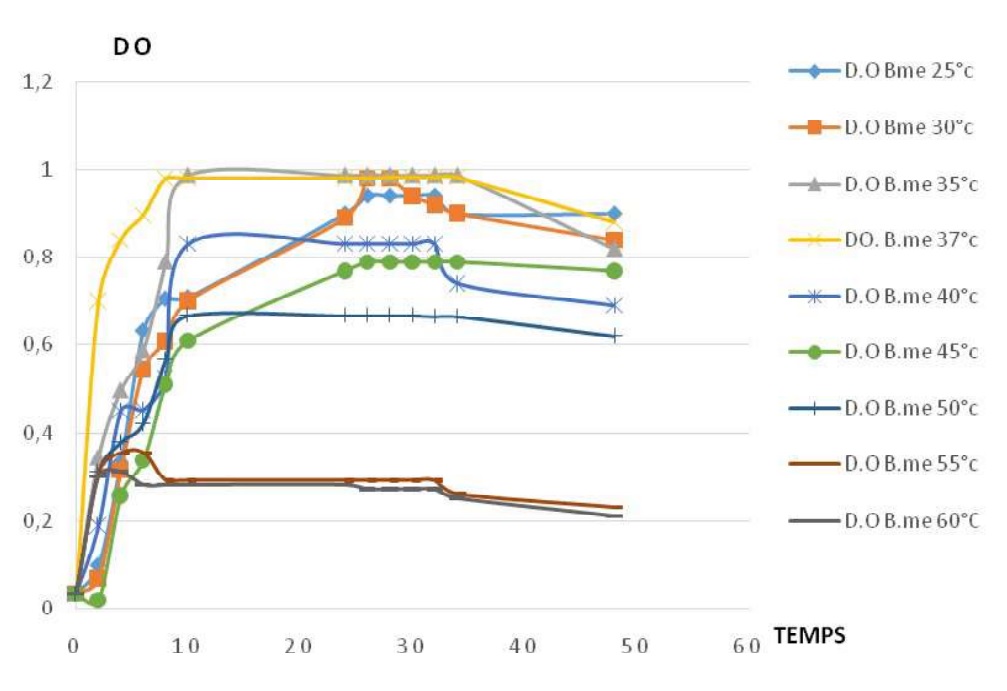 Fig. 4. Growth profiles of Bacillus megaterium at different temperatures
Fig. 4. Growth profiles of Bacillus megaterium at different temperaturesAll growth profiles have the sames phases; exponentially phase, stationary phase and the decreasing phase, but the different amplitudes.
Fig-5 shows the influence of temperature on caseinolytic enzyme production for Bacillus megaterium. For the température from 25 et 35°C, the increase of temperature is proportional to the increase of the amplitude of the enzyme production. For the temperature from 35 to 50°C, caseinolytic enzyme production decrease when the temperature increase. the caseinolytic enzyme production is possible from 25 to 50°C with the optimum at 35°C.
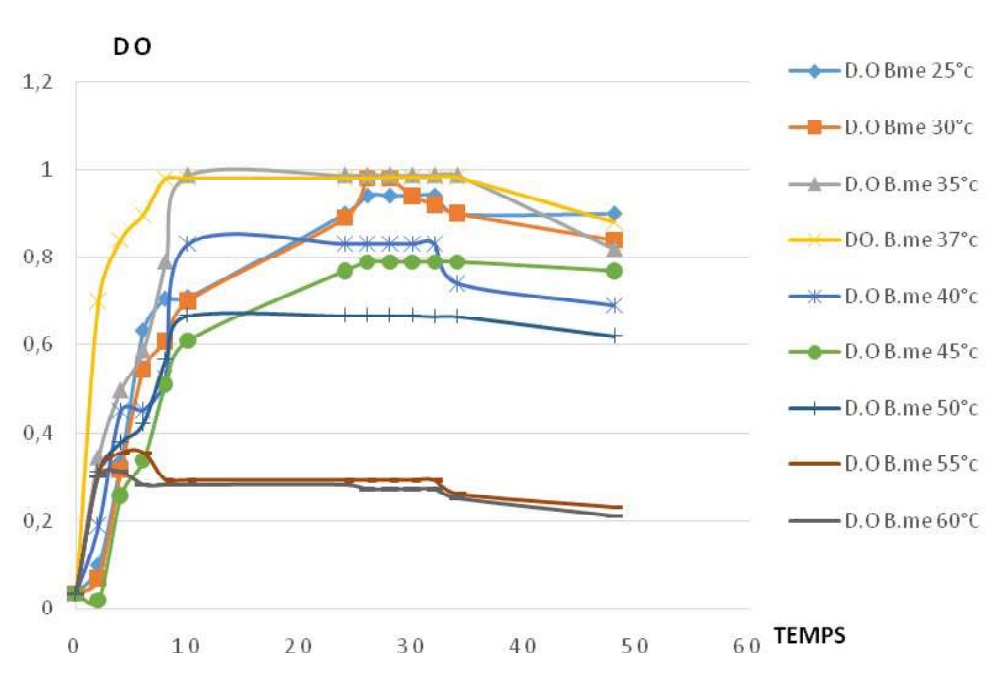 Fig-5. Profiles of caseinolytic enzyme production for Bacillus megaterium at different températures.
Fig-5. Profiles of caseinolytic enzyme production for Bacillus megaterium at different températures.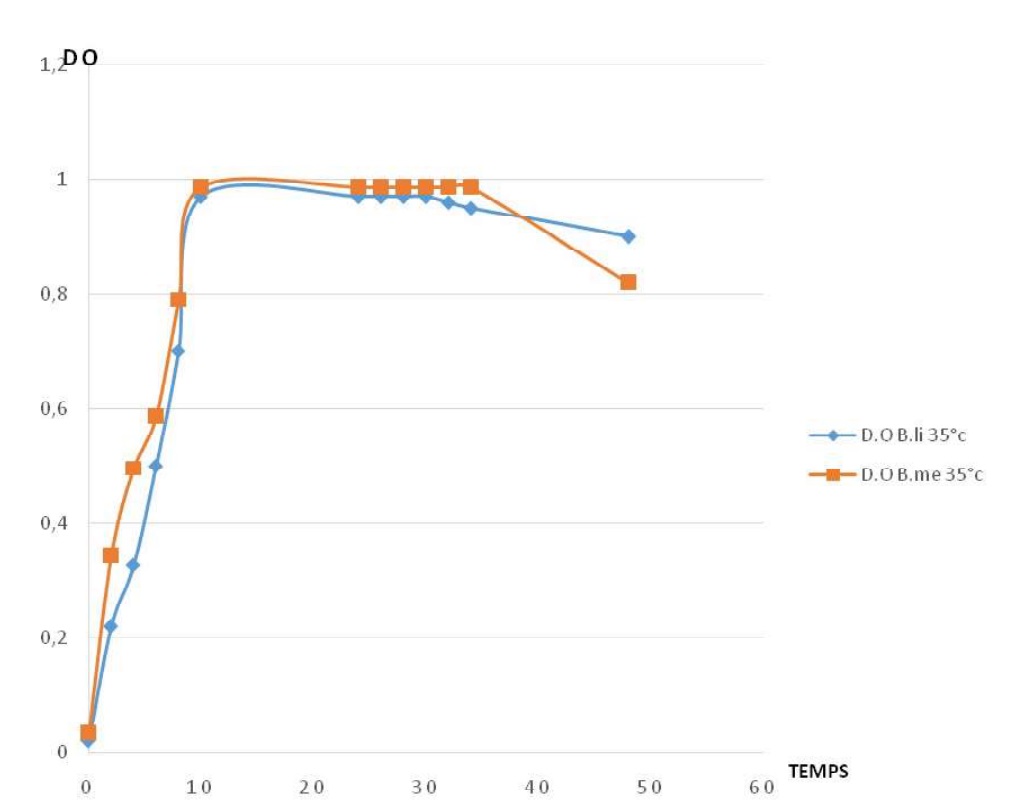 Fig. 6. Comparing growth profiles of Bacillus licheniformis (B. li) and Bacillus megaterium (B. me).
Fig. 6. Comparing growth profiles of Bacillus licheniformis (B. li) and Bacillus megaterium (B. me). Both profiles have the sames phases which are in correlation with the normal growth profile of bacteria. For both profiles from 0 to 10h, we have the exponentially phase. Stationary phase is from de 10 to 30 hours, for B. li, while from 10 to 34 hours. The decrease phase is from 30 to 48 hours for B. li, from 34 to 48 hours for B. me.
Fig-7. shows different phases of the caseinolytic enzyme in the two strains. In the both strains , the latency phase is observed between 0 to 2h.
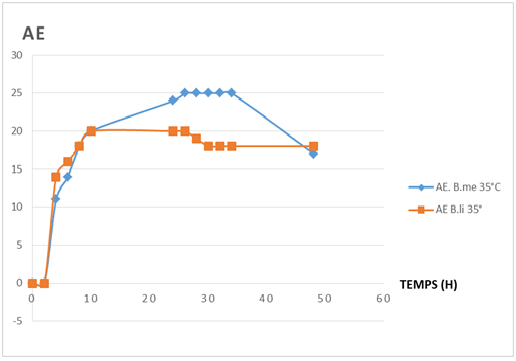 Fig-7. Comparing profiles of caseinolytic enzyme production of Bacillus licheniformis (B. li) and Bacillus megaterium (B. me).
Fig-7. Comparing profiles of caseinolytic enzyme production of Bacillus licheniformis (B. li) and Bacillus megaterium (B. me).For B. licheniformis, the profile comprise:
From 2 to 10h,an exponentially phase of enzyme production ;
From 10 to 26h, a stationary phase of enzyme production ;
From 26 to 30h, a decreasing phase of enzyme production ;
From 30 to 48h, a second stationary phase of enzyme production:
For B. megaterium, the profile comprise:
From 2 to 8 h, an exponentially phase of enzyme production ;
From 8 to 26 h, a stationary phase of enzyme production;
From 26 to 48h, a decreasing phase of enzyme production ;
From 34 to 48 h, une phase de diminution de production d’enzyme.
Fig-8 shows how evolve the enzyme production comparing to the growth in B. li. It is shown that the enzyme production is maximal during the stationary phase of the growth. This parallelism is the same for B. me.
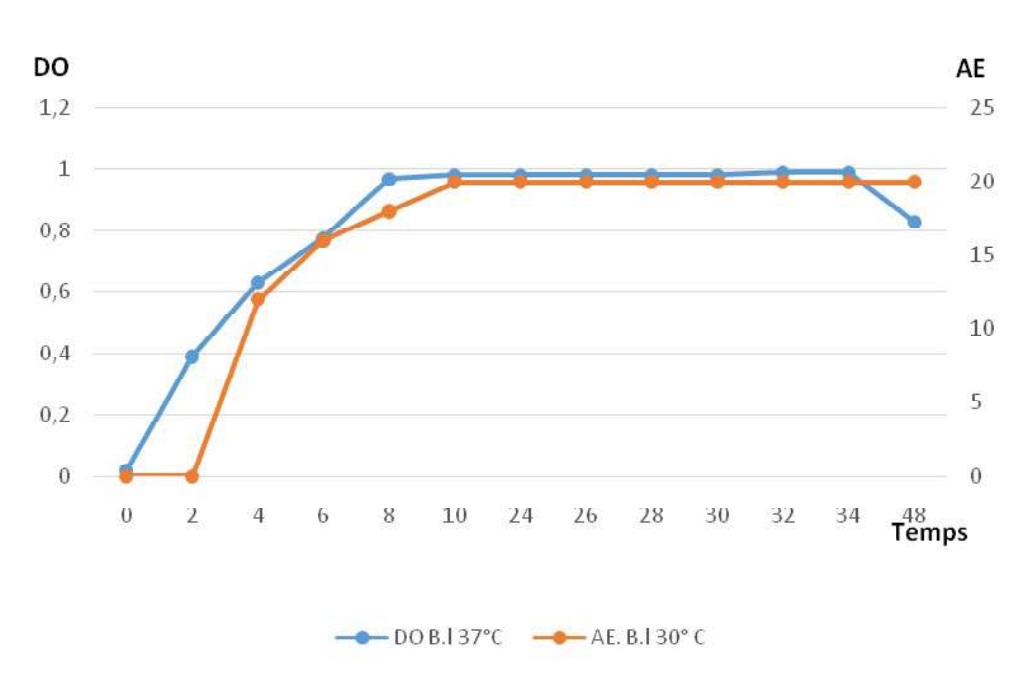
Fig. 8. Profiles of growth and enzyme production in Bacillus licheniformis.
Fig-9: shows that the pH increase rapidely at the end of the enzyme production phase, from 7 to 9 in the two studied strains.
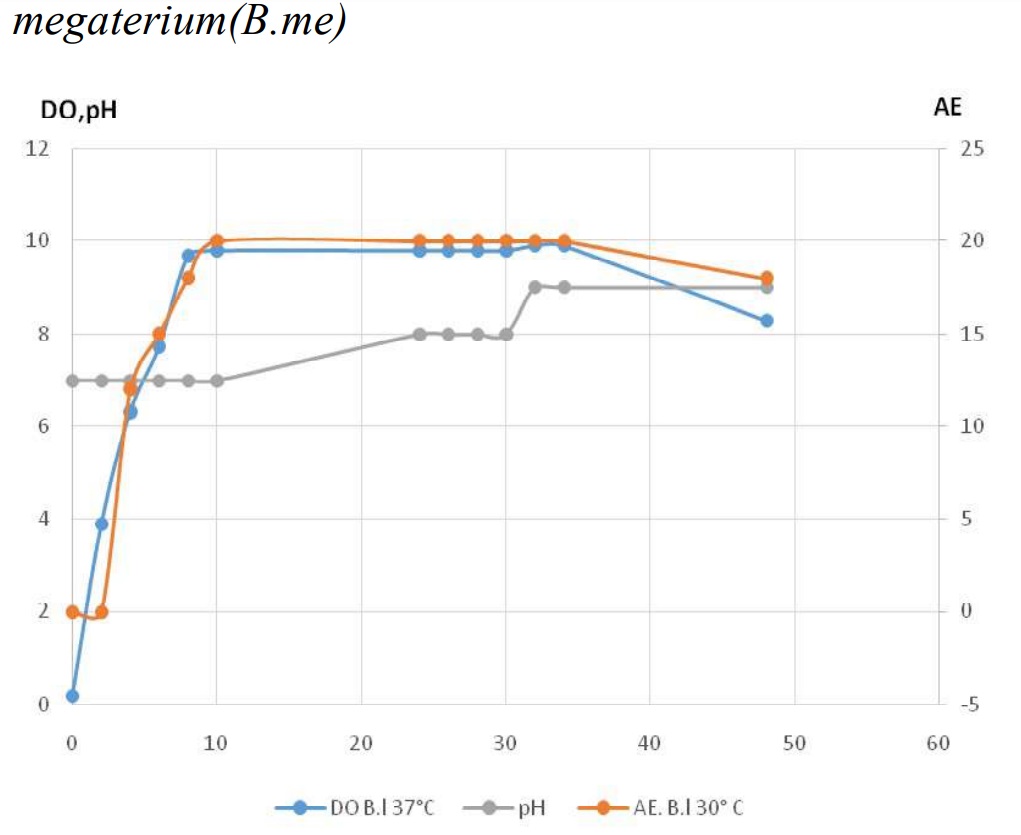 Fig. 9. Profiles of growth, enzyme production and pH variation during culture in the two studied strains.
Fig. 9. Profiles of growth, enzyme production and pH variation during culture in the two studied strains.Influence of pH of the medium in the growth and the enzyme production
Fig-10 shows the influencce of pH of the medium on growth in the two strains. It is shown that growth is possible at acidic pH. The phases are the same in the two strains.
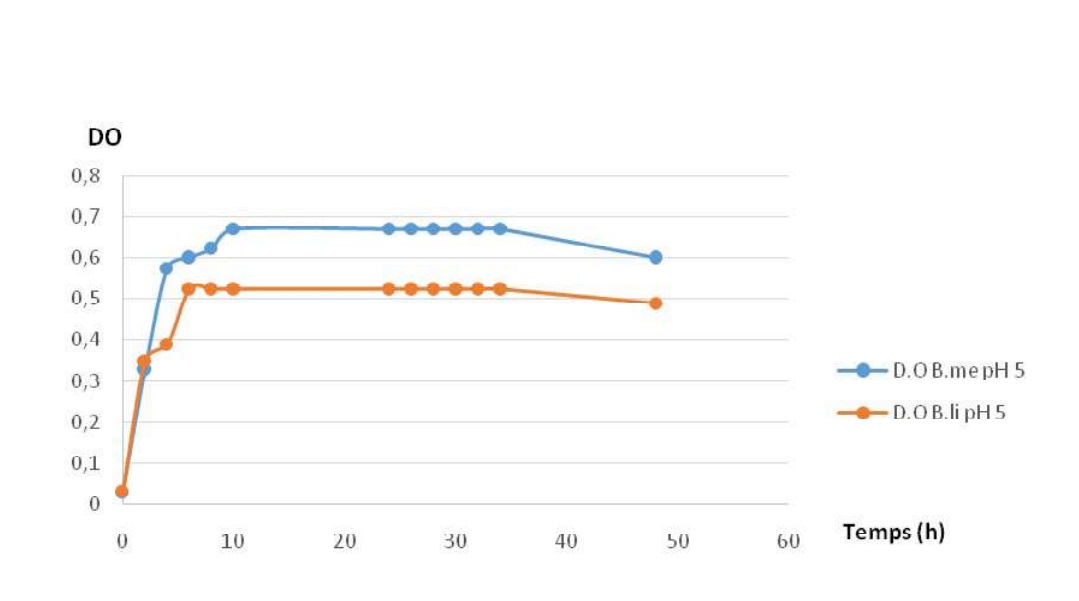
Fig-10. Growth profiles of Bacillus megaterium and B. licheniformis at pH 5
Fig-11 shows the influence of pH on growth in Bacillus megaterium and Bacillus licheniformis. It is shown that growth is possible in basic pH at 9, and the profiles comprises the sames phases.
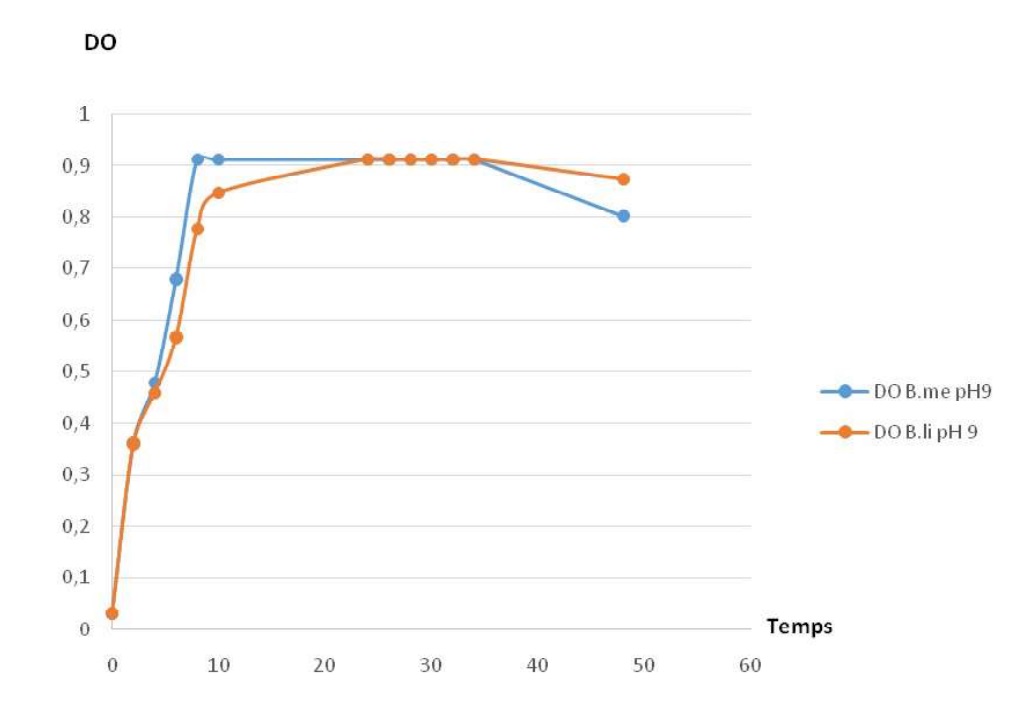 Fig. 11. Growth profiles at pH 9 in Bacillus megaterium et B. licheniformis
Fig. 11. Growth profiles at pH 9 in Bacillus megaterium et B. licheniformisFig-12 Shpows how caseinolytic enzyme production is possible in different pH of the cultured media The optimum is at pH 7.
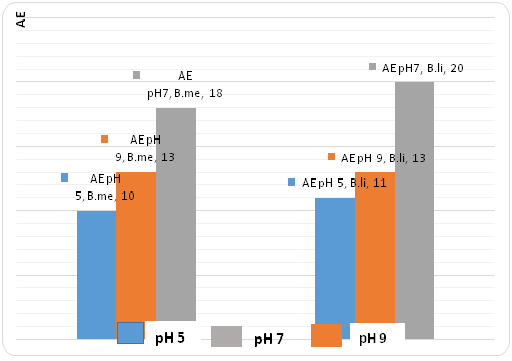 Fig. 12. Diagrams of caséinolytic enzyme production of B. li and B. me at different pH.
Fig. 12. Diagrams of caséinolytic enzyme production of B. li and B. me at different pH. Influence of different kinds of media on growth and the enzyme production
Fig-13 shows for the two strains growth profiles with the same phases. Growth is possible for the two used media, the LB medium is better than TSB.
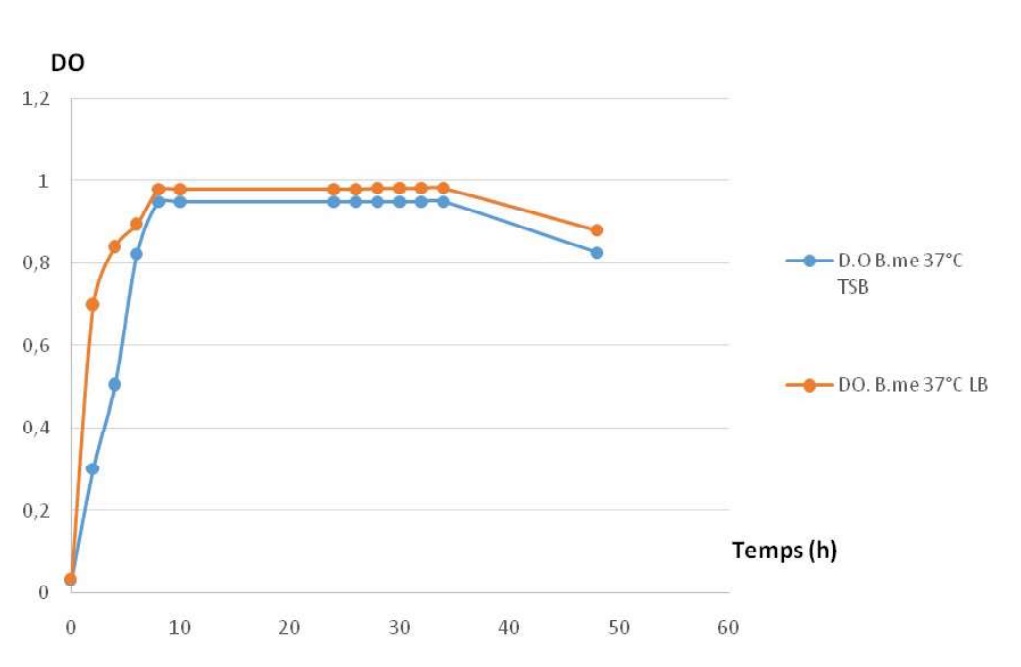 Fig. 13. Comparing growth profiles in different kinds of media for the two studied strains.
Fig. 13. Comparing growth profiles in different kinds of media for the two studied strains. Fig-14: shows the sames phases in the two caseinolytic enzyme production profiles.
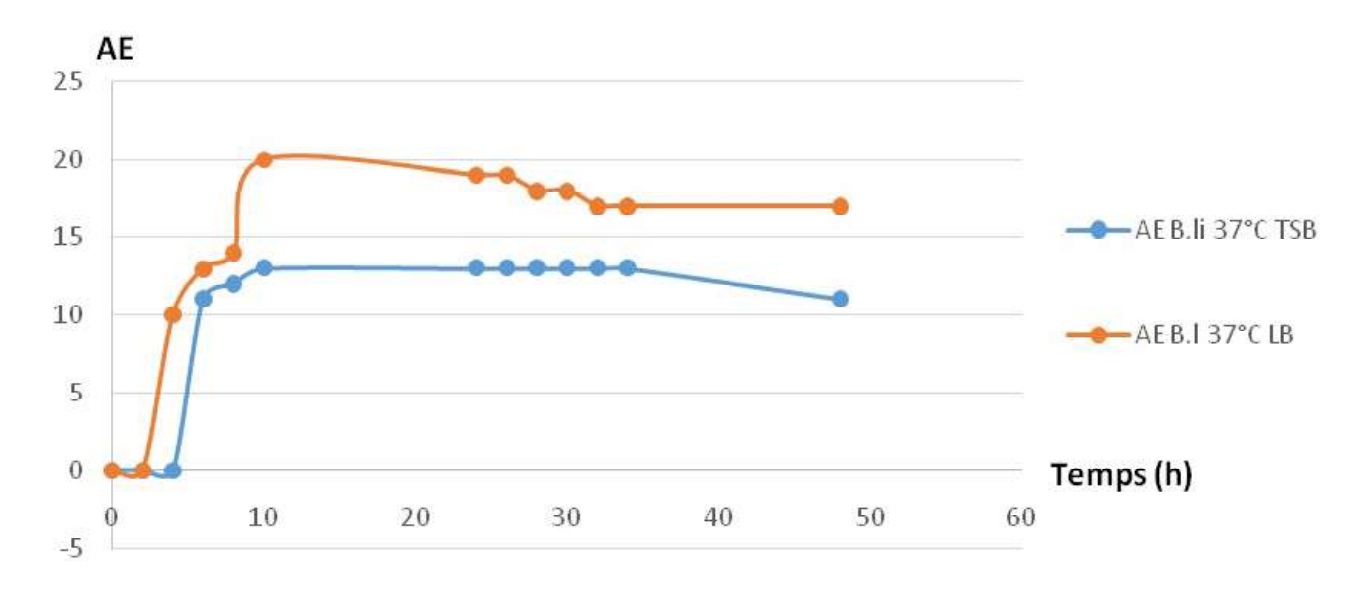 Fig. 14. Comparing profiles of caseinolytic enzyme production in two different media for the two studied strains.
Fig. 14. Comparing profiles of caseinolytic enzyme production in two different media for the two studied strains.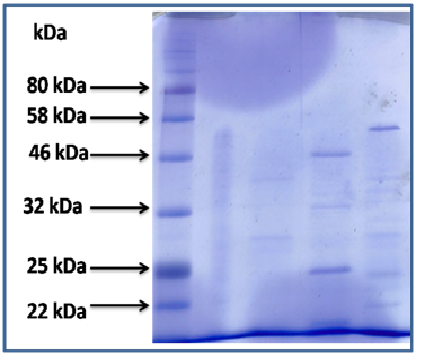 1: marker in KDa,
1: marker in KDa,2:supernatant after culture of B. li,
3:supernatant after culture of B. Me
4:ammonium sulfate precipitate of B. li supernatant after culture
5: ammonium sulfate precipitate of B. me supernatant after culture
Lane 4, two important bands are displayed at 46kDa and 25kDa respectively.
Lane 5 one important band is displayed on 56kDa.
Fig. 15: Proteins profiles on SDS-PAGE of two strains.
For LB medium : a latency phase in short and the amplitude of enzyme production is important. For the TSB medium : a latency phase is important and the amplitude of enzyme production is less important than in LB.
Influence of different carbon sources on the growth and caseinolytic enzyme production
In [Table-3] are shown how growth and caseinolytic enzyme production are influenced by different carbon sources in the two studied strains. Bacillus licheniformis and Bacillus megaterium. Sugars are important for growth and enzyme production in the two strains. Fructose is used to be the best carbon source.
Table (3):
Effect of three carbon sources on the growth and the enzyme production
| Growth (DO at 600nm) 48h | Enzyme production (diameter of halo in mm) 48h | |||
|---|---|---|---|---|
| Carbon sources | B. li | B. me | B. li | B. me |
| Galactose | 0,903±0,002 | 0,849±0,001 | 13,33±1,24 | 11,33±0,47 |
| Fructose | 0,93±0,001 | 0,928±0,002 | 14,33±1,24 | 14,33±1,24 |
| Amidon | 0,711±0,001 | 0,845±0,004 | 10,00±0,81 | 16,66±1,24 |
Influence of different nitrogen sources on the growth and the caseinolytic enzyme production
Among the two nitrogen sources used, yeast extracts is the best for growth in two strains B. licheniformis and B. megaterium). For enzyme production both sources can be used in Bacillus licheniformis, in contrast only the yeast extract is efficient in Bacillus megaterium. The results are shown in Table 4
Table (4):
Effect of two different nitrogen sources on the growth and the caseinolytic enzyme production in B. li and B. me.
| Growth (DO at 600nm) 48h | Enzyme production (diameter of halo in mm) 48h | |||
|---|---|---|---|---|
| Nitrogen sources | B. li | B. me | B. li | B. me |
| Yeast extracts | 0,969±0,015 | 0,952±0,01 | 14,333±1,247 | 14,333±0,471 |
| Bactopeptone | 0.514±0.003 | 0.739±0.002 | 14,333±1,247 | 11.666±0.942 |
7-Differentiation of strains used by SDS-PAGE
Figure 17 is showing the differentiation of the two bacillus strains by SDS-PAGE, this figure is showing clearly differences between the two strains.
The diameter of the halo testifying the caseinolytic enzyme activity is used in this work to express the enzyme production. The Temperature is an important factor for growth and the enzyme production. All growth profiles in the two strains (B. licheniformis and B. megaterium), with the temperature have the same phases, including exponentially phase, stationary phase and the decreasing phase. Figures 4 and 6. All profiles related to caseinolytic enzyme production, have also the sames phases, including the latence phase, an exponentially enzyme production phase, a stationnary production phase, and the decreasing production phase, Figures 5 and 7. For B. li, the optimal temperature of growth is 37ºC and the optimal temperature of enzyme production is 30ºC. For B. me the optimal temperature of growth is between [35-37] ºC, and the optimal temperature of enzyme production is 35ºC. These results are similar to those already published [15, 12, 27].
The growth profiles have not shown a latency phase. The absence of latency phase is related to the used method. The OD measure is done just to hours after the inoculation of an overnight culture. These results are different to those published by [24] which presents a latency phase for different method of inoculation.
The growth and the enzyme production are different phenomens, this is shown on the figure 10. At all temperatures and for the two studied strains, the enzyme production is optimal durind the stationnary phase of the growth, these results are similar to those published in [12, 15, 27].
During the exponnentially phase of growth, bacterial cells are dividing untill the nutriments are still in the medium, when the nutriments goes smaller and smaller in the amount, toxics products accumulate. This brings the changes in the culture conditions. The Figure 11 is illustrating how the pH of the medium has passed from 7 to 9. These results are identical to standard of bacterial culture and those published by [28].
For the two studied strains, the decreasing phases is followed by a stationary phase on which the OD value remained stable , and the caseinolytic enzyme production is still important. This phase is assimilate to the sporulation, these results are in harmony with those related to B. Licheniformis and B. megaterium, which gradually go on sporulation , according to [29 ,30]
All the two strains can be cultured at different pH (5,7 et 9), with the optimum at 7. These results are different than those published by [31], which stipulate that these strains could not growth and can not produce the enzyme when the starting pH of the medium is at 5.
In the two used media, the LB is better rather for growth and fore enzyme production, on the two studied strains.
Among different carbon sources used. The best growth is observed with fructose and galactose which are monosaccharides. For polysacchrides growth and enzyme production are low.
When using the two sources of nitrogen , yeast extracts have been revealed to be better than bactopepton for growth in the two strains (B.li 0,969±0,015 B.me 0,952±0,01).In B.li the two nitrogen sources can be used for enzyme production (Yeast extracts:14,333±1,247, bactopepton: 14,333±1,247), but in B.me only the yeast extract yield the important enzyme production. In deed in our laboratory, we have set the standard of the enzyme production, the reference strains B. amy yield on the fructose medium the standard growth of 0.954±0.001, and at 48h of culture the standard enzyme production is13.333±0.001. It is according to these standard values, the strain is declared a caseinolytic producer (non published results).
We used SDS PAGE to differentiate the two strains through partial purification of protein in the supernatant after culture, and ammonium sulfate precipitation. The Figure 16 is giving the protein profiles of the two studied strains. Specific bands can allow discrimination of the strains.
This article have helpful to our laboratory, for we have set new optimal conditions of two new strains in growth and caseinolytic enzyme production. We have discover the specificity of the two strains. Our methods has allowed to discriminate the caseinolytic producers by SDS-PGE. This article has shown the importance of setting standard measure for enzymatic production. In a near future we will do the total purification and sequencing these caseinolytic enzymes.
ACKNOWLEDGMENTS
We give thanks to Professor Goma Tchimbakala for providing material support .
- Biswanath Bhunia, Bikram Basak, Apurba Dey. A review on production of serine alkaline protease by Bacillus spp. J Biochem Tech 2012, 3(4).
- Moon, S.H. and S.J. Parulekar. A parametric study of protease production in batch and fed-batch cultures of Bacillus firmus. Biotechnol. Bioengg., 1991, 37: 467-483.
- Gupta R, Beg QK, Lorenz P. Bacterial alkaline proteases: molecular approaches and industrial applications. ApplMicrobiolBiotechnol 2002, 59:15-32.
- Patel, P.R.,. Enzyme isolation and purification. In: Biotechnology: Applications and Research. Technomic Publishing Co. Inc., USA, 1985, pp: 534-564.
- Udandi Boominadhan, Rajendran Rajakumar, Palanivel et al.. Optimization of Protease Enzyme Production Using Bacillus Sp. Isolated from Different Wastes. Botany Research International 2009, 2(2): 83-87.
- Beg QK, Saxena RK, Gupta R. De-repression and subsequent induction of protease synthesis by Bacillus mojavensisunder fed-batch operations. Process Biochem 2002, 37: 1103-1109.
- Varela H, Ferrari MD, Belobradjic L et al Effect of medium composition on the production by a new Bacillus subtilis isolate of protease with promising unhairing activity. World J MicrobiolBiotechnol 1996, 12: 643–645.
- De Coninck J, Bouquelet S, Dumortier V et al Industrial media and fermentation processes for improved growth and protease production by Tetrahymenathermophila. J IndMicrobiolBiotechnol 2000, 24: 285–290.
- Puri S, Beg QK, Gupta R . Optimization of alkaline protease production from Bacillus sp. by response surface methodology CurrMicrobiol 2002, 44: 286-290.
- Priest FG () Extracellular enzyme synthesis in the genus Bacillus. Bacteriol Rev 1977, 41:711-753.
- Strauch MA, Hoch JA. Transition-state regulators: sentinels of Bacillus subtilis post-exponential phase gene expression.Molecular Microbiol 1993, 77:337-342.
- Nguimbi Etienne, Wu ZhiRong . Production of a New Fibrinolytic Enzyme, Bacterium Growth and Enzyme Production Conditions, Purification and characterization of the New Enzyme.Biotechnology 2002, 12(2).
- Chu IM, Lee C, Li TS . Production and degradation of alkaline protease in batch cultures of Bacillus subtilis ATCC14416. Enzyme MicrobTechnol 1992, 14: 755-761.
- Power SD, Adams RM. Bacillus. in: Molecular Genetics and Biotechnology Applications, (Eds.) A. Ganesain, J.A. Hoch, Academic Press. New York, 1986, pp. 101-108.
- Nguimbi Etienne, Ahombo Gabriel, Moyen Rachel, et al . Optimization of Growth, Fibrinolytic Enzyme Production and PCR Amplification of Encoding Fibrinolytic Enzyme Gene in Bacillus amyloliquefaciens Isolated from Ntoba mbodi at Brazzaville. International Journal of Science and Research (IJSR), 2014, 3(11), 2319-7064.
- Evelyn Zeilera, AnjaListb, Ferdinand Alteb, et al (July 9, 2013). Structural and functional insights into caseinolytic proteases reveal an unprecedented regulation principle of their catalytic triad. PNAS, 110(28), 11302–11307
- Gottesman S, Roche E, Zhou Y, Sauer RT, The ClpXP and ClpAP proteases degrade proteins with carboxy-terminal peptide tails added by the SsrA-tagging system. Genes Dev, 1998; 12(9):1338–1347.
- Gottesman S, Maurizi MR, Wickner S, Regulatory subunits of energy-dependent proteases. Cell, 1997; 91(4):435–438.
- Gottesman S, Maurizi MR, Regulation by proteolysis: Energy-dependent proteases and their targets. Microbiol Rev, 1992; 56(4):592–621.
- Louembé D., Kobawila S.C, Bouanga Kalou G.et al. Etude microbiologique des feuilles fermentées de manioc ‘’Ntoba mbodi’’. Tropicultura,. 2003; 21(3); 106-111
- Labia Irène I. Ouoba, Alain B. Vouidibio Mbozo, Line Thorsen, Amarachukwu Anyogu, Dennis S. Nielsen, Simon C. Kobawila and Jane P. Sutherland. Lysinibacillus louembei sp. nov., a spore-forming bacterium isolated from Ntoba Mbodi, alkaline fermented leaves of cassava from the Republic of the Congo. International Journal of Systematic and Evolutionary Microbiology, 2015, 65, 4256–4262.
- Astrup T., Mullertz S. The fibrin plate method for estimating fibrinolytic activity. Arch. Biochem. Biophys., 1952; 40, 346-351.
- Kim S. and Choi N.S. Electrophoresis analysis of protease inhibitors in fibrin zygmography.Anal.Bochem., 1999; 270: 179-181.
- Kannan N., Aravindan R. and Viruthagiri T. Effect of culture conditions and kinetic studies on extracellular tannase production by Lactobacillus plantarumMTCC1407.). Indian Journal of Biotechnology, 2011; 10, pp321-328
- Cooney (N.L.), Levine (D.W.) Microbial utilization of methanol. –Adv. Appl. Microbiol., 1972; 15, 337-365.
- Mohamed Lagzouli (1), RedaCharouf (2) et al -optimisation de la croissance et de la production de glucoamylase extracellulaire par Candida guilliermondII II. Bull. Soc. Pharm. Bordeaux, 2007; 253: 251-270
- Choi N. S., Chung D.M., Ryu C.H, K.-S. Yong P., Maeng J, and Kim S. H. Identification of three extracellular proteases from Bacillus subtilis KCTC 3014.J Microbiol.Biotechnol., 2006; 16: 457-464.
- Chenel, I.P. Production d’enzymes protéolytiques par des bactéries non conventionnelles isolées de boues d’épuration municipales. Institut National de la -Recherche Scientifique – Eau, Terre et Environnement. Québec, 2005; 59.
- Mathieu drouin, étude de production de protéases alcalines par bacill us licheniformisen utilisant des boues d’épuration municipales comme substrat, Mémoire présentée pour l’obtention du grade de Maître ès sciences (M.Sc.), Microbilogie, Institut National de la Recherche Scientifique, 2005; 142, pp 17-18
- Prescott L., Harley J.P. et Klein D.A., Microbiologie, 2e Édition. DeBoeck-Wesmael,Bruxelles, Belgique, 1995; 1014 pages.
- Inkyung Park1, Jaiesoon Cho2. Production of an extracellular protease by an Antarctic bacterial isolate (Bacillus sp. JSP1) as a potential feed additive. Revista Colombiana de CienciasPecuarias 2011.
© The Author(s) 2017. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


