ISSN: 0973-7510
E-ISSN: 2581-690X
Halophilic micro-organisms often synthesize and produce extracellular polysaccharides (EPS), whose physical, chemical properties and material properties vary greatly from each other. The extracellular polysaccharide (EPS) development of Halomonas sp. MN795630 strain type halophilic bacterium (NASH) was investigated and whether biotechnological applications were feasible. After 168 hours of incubation, 4 g/L of EPS was produced and all elements from the medium were completely used during the growth. Sucrose has been identified as the most favorable carbon source for production of EPS and maximum production (6 g/l). Beef extract level was shown to be the best for EPS production among different nitrogen sources. Optimum production of EPS (10 g/L) were achieved by supplementing the medium with 4M NaCl, pH adjusted at 9 and the medium was inoculated with 7% initial inoculum. The purified EPS were characterized chemically. Fourier transform infrared (FTIR) spectrophotometer was observed in several functional groups. EPS also demonstrated an significant inhibitor of Candida albicans ATCC 10231 and Pseudomonas aeruginosa ATCC 9027 (20.4 and 14.7 mm), respectively. EPS show satisfactory results when applied as anti-oxidant, anti-inflammatory and emulsifier.
Exopolysaccharides, Halomonas sp. NASH, Optimum conditions, Antimicrobial activity, Antioxidant activity, Anti-inflammatory
Exopolysaccharides microbial are polysaccharides that are extracellularly formed as capsules or slimes. Such microbial EPS are typically categorized into 2 broader classes: homopolysaccharide consisting of a single monosaccharide unit and a heteropolysaccharide with two or more monosaccharide units. In nature, microbial EPS is non-toxic , biodegradable and renewable1. They play a major role in the defense of desiccation2 and are also useful in the formation of biofilms and also useful in forming biofilms3. Use as gelling agents, biosurfactants, emulsifiers, viscosities4,5, biosorbants6,7 Antimicrobials, Anti-Cancer Agents and Antioxidants8-10.
EPS is comparatively less reported from extremophilic microorganisms, especially halophilic ones. The generation of EPS from halophilic bacteria in intense marine ecosystems and its biological activities have been investigated11. Several workers have recorded the extracellular polysaccharides formed by halophilic archaea and bacteria, and the members of the Halomonas genus were classified as the most potential producers12,13. The literature survey has shown clearly that the knowledge on the extracellular polysaccharides development and characterization by various halophilic micro-organisms isolated from hypersalin environments is not adequate12,14. However, the demand for extracellular polysaccharides of halophiles with better properties than existing ones is increasing. The present study was carried out with the goal of producing EPS by Halomonas sp. NASH, optimizing some environmental parameters for the production of EPS by Halomonas sp. NASH, characterizing the EPS produced and finally evaluating its efficacy as an antimicrobial, antioxidant and as a biosurfactant.
Microorganisms and cultural conditions
Halomonas sp. NASH was provided by Dr. Asmaa M. Youssif (Botany and Microbiology Department, Faculty of Science, Alexandria University) and isolated from sediment sample in Wadi El-Natroun lakes, and set in the Genbank as Halomonas sp. NASH with accession number MN795630. This halophilic isolate was maintained on IRAM media15 with the following composition (g/L); Magnesium sulfate, 20; Potassium chloride, 5; Calcium chloride, 0.2; Yeast extract, 4; Peptone, 5; Sodium chloride 223.3 (4M). The pH of the media was changed to 8 before sodium bicarbonate sterilization. The medium was sterilized immediately after preparation at 121°C for 20 minutes. For solid media, 20g agar was added. Strain was incubated at 35-37 ℃ for 7 days. The isolate was stored in 20% (v/v) glycerol at -20°C for subsequent investigation.
EPS production
Inoculum was prepared for the production of EPS by growing the culture in IRAM medium. 250 ml Erlenmeyer flask containing 50ml of medium consisting of (g/l): Magnesium sulfate, 20; Potassium chloride, 5; Calcium chloride, 0.2; Yeast extract, 4; Peptone, 5; Sodium chloride 223.3 (4M). The pH of the media had been set at 8 before sodium bicarbonate sterilization. This flask was inoculated on an IRAM medium grown with old stock culture. The flask was incubated in a 35-37°C, 200 rpm rotator incubator shaker for 7 days and then was centrifuged to obtain filtrate and extract EPS from it.
Correlation between growth & EPS production at different growth phases
A seed culture was prepared for the not each strain by inoculating 250ml conical flask containing a 50 ml IRAM medium with a loopful of the strain and rotary shaker (200 rpm) incubation, until the growth was OD600 = (0.8-1). Inoculate flasks each containing 50 ml of the sterilized optimized medium in regular 0,5 ml inoculum from the seed culture previously prepared. Flasks were incubated shacked at 200 rpm at 37°C for 7 days. Samples were taken at time intervals to measure growth at wave length 600nm. Flasks were centrifugal for 10 minutes at 10,000 rpm and EPS was removed with supernatant.
One-time variable (OVAT) method for optimizing EPS production
The effect of different parameters including carbon and nitrogen sources, concentrations of inoculum, pH and NaCl were the parameters investigated by Halomonas sp. NASH for optimum production of EPS. The NASH strain was grown in medium IRAM and incubated at 37°C, 120 rpm for 7 days. Both experiments were performed in 250 ml Erlenmeyer flask using a 50 ml IRAM medium. EPS was measured at the end of each trial, and gravimetrically calculated. The effect of carbon sources and nitrogen on the development of EPS was assessed, organic and inorganic sources. Glucose, sucrose, lactose, maltose, fructose, galactose, glycerol and sodium citrate, respectively, which are organic and inorganic sources of carbon. Ammonium chloride, ammonium sulphate, sodium nitrate, potassium nitrate, yeast extract, tryptone, beef extract and casein hydrolysate, which were representative of inorganic and organic nitrogen sources. The effect of pH on the output of EPS was achieved by changing the medium pH at 6, 7, 8, 9 and 10. The results of different NaCl (0, 1, 2, 3, 4 and 5 M) concentrations have also been studied. Halomonas sp. NASH tested the effect of inoculum size on EPS production by inoculating the production medium with various inoculum sizes from 1 to 10 percent (v/v) and shaking at 37°C for 7 days16.
EPS extraction and Purification
The fermented cells were harvested after seven days, and the cell suspension was heated to 100°C for 10 min to inactivate the enzymes. The suspension has been cooled to room temperature and centrifuged for 20 minutes to extract biomass at 4,000 rpm Sevage reagent (chloroform: n-butanol at 5:1 v/v) was further treatment of the crude solution three times for removal of protein. EPS was precipitated with cold ethanol (three times volume) and left at 4°C overnight. Centrifugation was used for the precipitation at 10,000 rpm for 15 min and was dissolved in Milli Q water. It was subsequently enveloped into a dialysis bag (12-14 KDa) and dialysed with Milli Q water at 4°C for 48 hours for partial purification17.
Characterization of partial purified EPS
Carbohydrate and protein contents
According to the Dubois method the total carbon content was measured18. Adding 25 μl of 85 percent phenol to one ml of the sample solution, accompanied by adding 2.5 ml of concentrated sulphuric acid, shaking the mixture after each addition. The mixture was put in a boiling water path (100°C) for 10 min, then cooled to room temperature before reading spectrophotometrically at 488 nm against a blank of one ml distilled water The regular solution of glucose was used to produce a calibration curve which was used to measure the sample carbohydrate content. Using the method described by19, the total soluble protein was determined quantitatively. The 100 μl sample was thoroughly mixed with 3 ml of the alkaline solution and allowed to stand in room temperature for at least ten minutes. An amount of 0.25 ml of the diluted Folin-Ciocalteau reagent (2:1, v/v) was added rapidly to the mixture and mixed immediately. The mixture was left to stand for 30 min. Thereafter, the extraction was measured at 760 nm against the blank. Using a calibration curve, constructed with Bovine Serum Albumin as a reference, the protein concentration of the unknown samples was estimated.
FT-IR analysis
In transmission mode, The FT- IR (BRUKER, Vertex 70) and OPUS pellets in the 4000 400 cm-1 range were used to obtain IR transmission spectroms using potassium bromide (KBr)20.
Biotechnological applications of EPS
Antimicrobial activity
For EPS antimicrobial potential determination, two tested Gram-positive bacteria Bacillus subtilis ATCC 6633, Staphylococcus aureus ATCC 6538, as well as two Gram negative bacteria; Escherichia coli ATCC 19404, Pseudomonas aeruginosa ATCC 9027 and one yeast strain Candida albicans ATCC 10231 were selected as indicator strains. These strains were cultivated in the medium Lauria Broth (LB) overnight at
30 °C. The culture was then diluted by LB media and incubated at an acceptable temperature at 5 hours under a 200-rpm agitation to an initial OD 600 of 106 CFU/mL indicator strain. The measurement inoculum for the indicator strain was based on the cell counts ‘growth curve and optical diameter. By the agar diffusion test, the antimicrobial activity was detected21. 50 mm of agar nutrient are poured on all plates inoculated by a predictor microorganism. Upon solidification wells are punched and two drops of sterile water agar have been applied to each of their bodies with a 055 cm corkborer. 100 ul of filtered EPS were transferred to a well after sterilization by using
0.22 μl filters. All the plates were incubated for 24-48 h at the appropriate temperature. The clear zone radius around each well (Y) and the well radius (X) are measured linearly in mm after incubation, whereby Y2 is divided by X2 and determines an absolute unit (AU) for clear zone. According to the following equation: AU= Y2/X2, The absolute unit of each EPS which implies a positive antibiotic action result22.
Antioxidant activity
Free radical 1,1-diphenyl 2-picrylhydrazyl scavenging activity (DPPH) was assessed as a measure of antioxidant activity using the23 approach of partially purified exopolysaccharide. Generic polysaccharides were used as a reference compound, such as ascorbic acid. Scavenging effect (%) = (Ac –As)/Ac× 100. Ac is control absorption, As is the sample absorption.
Anti-inflammatory assay
The Mizushima technique was tested in albumin denaturation inhibition24,25 to test the anti-inflammatory function.
Biosurfactant
Applying the same amount of paraffin oil to an EPS sample (v/v), then vortexing with high speed (2 min) and permitted to stand at 24 hours, the EPS emulation index (E24) was calculated. The percentage of E24 was calculated using the following equation: E24 = Height of emulsion formed (cm) ×100 / Total height of solution (cm)26.
Correlation between growth & EPS production at different growth phases
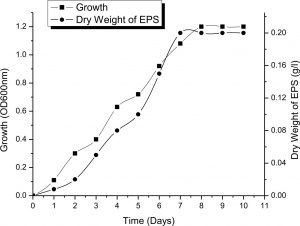
The correlation between growth and EPS production was planned to be investigated at different growth phases. In this experiment the growth of the bacterium was controlled in incubated at 37°C in batch cultures in IRAM medium shacked. Standard inoculum (1%) is taken from seed cultures previously prepared (OD600~ 0.8-1) and used to inoculate 50 ml of media in 100 ml flasks. Then, the flasks were shaken at 120 rpm at 37°C. Timely samples were used to measure spectro-metrical growth at wavelength 600 nm and output of EPS at regular intervals. Data in Fig. 1 reveal that cells of Halomonas sp. NASH entered the exponential phase of growth after 2 days of incubation and the stationary phase of growth after 9 days of incubation. EPS was growth phase dependent, low production was detected at the beginning of exponential growth, and increased exponentially with bacterial growth till it become constant at stationary phase. The maximum EPS production was recorded in the middle of exponential phase recording 4g/l.
Fig. 1. Correlation between growth of Halomonas sp. NASH grown in IRAM and incubated at 37°C under shaken conditions for 10 days and EPS production
One-time variable (OVAT) method for optimizing EPS production
Effect of different carbon sources
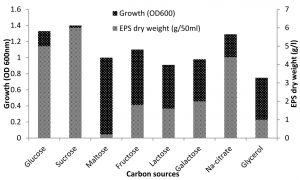
Halomonas sp. NASH has shown that the addition of sucrose, glucose, and sodium citrate at the average level of 1% has resulted in different bacterium rates of growth and EPS production. Sucrose was most effective in EPS production (6g/l) followed by glucose (5g/l) and sodium citrate (4.4g/1). However, compared to sucrose, glucose and sodium citrate exerted maximum influence on the growth of the isolate. Carbon sources like maltose, glycerol, fructose, lactose along with galactose produced poor to moderate EPS
(Fig. 2). Similar results were obtained by Biswas and paul27 who mentioned that glucose was one of the carbon sources which affected on the growth and production of EPS by Halomonas xianhensis SUR308. Also, Llamas13 proved that EPS production and maximum growth by Halomonas almeriensis was obtained by using 1% glucose as a carbon source. Glucose has been reported to influence EPS production in a number of bacterial species including Halomonas spp, glucose at >1.0% level retarded the EPS production28-30.
Fig. 2. Effect of various sources of carbon on growth and EPS production by Halomonas sp. NASH. Fermentations were carried out in IRAM medium supplemented with 1% carbon source under continuous shaking (200 rpm) at 37°C and pH 8 with 1% (v/v) initial inoculum for 7 days.
Effect of different nitrogen sources
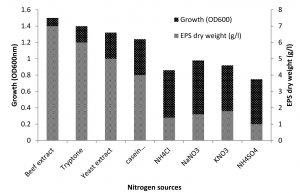
Ammonium sulfate, peptone, sodium nitrate, urea and yeast extract, however, are known to encourage both a rate of growth and the EPS production due to the presence of organic nitrogen sources31. From the results as illustrated in Fig. 3, it was evident that all the organic nitrogen sources like peptone, yeast extract, casein hydrolysate, beef extract, tryptone have positive influence on the EPS synthesis. In presence of organic nitrogen sources, the isolate was capable of accumulating remarkable amounts of EPS (5–7g/L). However, amongst the different organic nitrogen source, beef extract was most preferred one which led to the production of 7 g/L of EPS. This may be due to vitamins and cofactors present in organic nitrogen sources which could have played the key role in inducing growth and EPS production32. Biswas and paul27, proved that organic nitrogen sources promote both growth rate of Halomonas xianhensis SUR308 and the EPS production than inorganic nitrogen sources especially casein hydrolysate gave maximum EPS production reached 6.5g/l. Gu and his team also said that the production of EPS from halophilic Kocuriarosea ZJUQH was influenced by organic nitrogen such as peptone, yeast extract and casein hydrolysate33.
Fig. 3. Effect of different sources of nitrogen on growth and EPS production by Halomonas sp. NASH. Fermentations were carried out in IRAM medium supplemented with 9g/l nitrogen source under continuous shaking (200 rpm) at 37°C and pH 8 with 1% (v/v) initial inoculum for 7 days.
Effect of pH
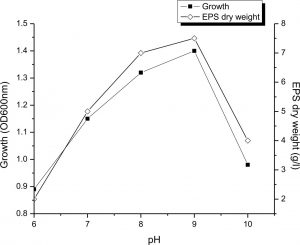
The figure 4 showed that both growth and output of EPS were significantly increased by growing the medium pH until 9. The maximum output of EPS at the pH 9 was reached, reaching 7.5g/L and gradually decreased by a pH rise of over 9. This is means that EPS production by Halomonas sp. NASH preferred alkaline condition. On the other hand, Biswas and paul27 mentioned that maximum EPS production by Halomonas xianhensis SUR308 was 2.99g/l at pH 7.5. Also, Gu et al.33 stated that maximum EPS production by halophilic Kocuria rosea was obtained at neutral pH. While16, proved that pH 6 was the suitable pH for the maximum production of EPS (23g/l) by Halomonas smyrnensis SVD III.
Fig. 4. Effect of pH on growth and EPS production by Halomonas sp. NASH. Fermentations were carried out in IRAM medium under continuous shaking (200 rpm) at different initial pH ranging from 6-10 and temperature at 37°C with 1% (v/v) initial inoculum.
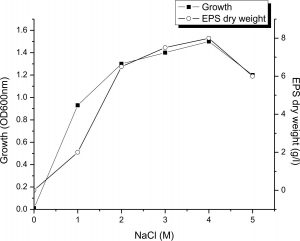
Effect of NaCl concentrations
As a halophilic organism, the isolate NASH showed a wide degree of tolerance to NaCl for growth and EPS production was more or less constant in the range of 2 to 4M NaCl in the medium, where by EPS production varied from 6.5 to 8 g/L. It was evident from the results (Fig. 5) that the production of EPS was maximum (8 g/L) at 4M NaCl. Similarly16, mentioned that maximum production of EPS (23g/l) by Halomonas smyrnensis SVD III obtained by adding 20% NaCl in the medium. Arias28 reported that optimum salt concentrations for EPS production by Halomonas maura was 2.5%, While the best for maximum production of EPS was 7.5 percent salt concentration by Halomonas eurihalina, Halomonas ventosae and Halomonas anticariensis.
Fig. 5. Influence of NaCl concentration on growth and EPS production by Halomonas sp. NASH. Fermentations were carried out in IRAM medium under continuous shaking (200 rpm) at NaCl concentrations ranging from 0–5M, pH and temperature of 9 and 37°C with 1% (v/v) initial inoculum
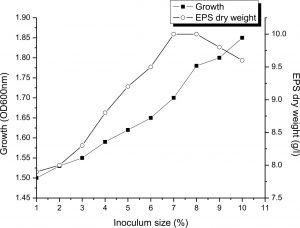
Influence of initial inoculum dose
Since the initial inoculum added to this medium is known to affect the development of the EPS, the IRAM medium added to 4M NaCl, 1% sucrose and 9g of beef at pH 9 were inoculated in a newly cultivated isolate level at 1–10 percent (v/v) and incubated in a continuous shaking (200rpm), at 37°C. As shown in Fig. 6, 10 g/L of EPS was produced by inoculating the medium with an initial inoculum of 7%. At this stage, the culture density (OD) turned so thick and prevented normal shaking of the medium. To extract the EPS, the culture medium was initially diluted to separate the cell mass by centrifugation. Our results are similar to Biswas27, who mentioned that 7% of Halomonas xianhensis SUR308’s inoculum size was optimal for 7.87 g/L EPS. While16, mentioned that maximum production of EPS (23g/l) by using 10% inoculum size of Halomonas smyrnensis SVD III.
Fig. 6. Influence of initial inoculum dose on growth and EPS production by Halomonas sp. NASH. Fermentations were carried out in IRAM medium 4M NaCl at different inoculum dose of 1–10% (v/v) under continuous shaking (200 rpm) at pH and temperature of 9 and 37°C.
Characterization of EPS produced by Halomonas sp. NASH
Carbohydrate and protein contents
The study showed that the EPS from Halomonas sp. NASH are acidic in nature, with a total content of 80,5±4 mg/g of carbohydrates and a total protein content of 9±2 mg/g. In similar results, the Bacillus subtilis basal medium and the malt medium respectively, had total carbohydrate of 0.91 mg/100 ml and 0. 43 mg/100 ml, which indicates his presence significantly in the extract34. In all samples examined, a low protein content (around 1%) makes the methods used to differentiate proteins against polysaccharides to be more effective35. Maalej ‘s tests for EPS extracted from Pseudomonas stutzeri AS22 (1 percent of protein) were identical36.
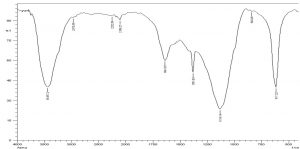
The FTIR Spectrum
The exopolysaccharide spectrum FTIR (Fig. 7) displays different bands concentrations. The FTIR of exopolysaccharide spectrum (Fig. 7) shows different rates of bands. The spectrum shows characteristic absorption peaks at 3448.72, 2970.38, 2252.86, 1643.35, 1381.03, 1134.14, 640.96 and 617.22 cm-1; at 3448.72 cm-1 the large vibration of the O-H stretch suggested that carbohydrates were in free hydroxy classes. The band at 2970.38 cm-1 approves the stretching vibration of C–H stretching of alkane group.
A very spectral peak of 2252,86 cm-1 was reached, which suggested –C≡C– stretched alkyne vibration. At 1643.35 cm-1, the sharp band indicated C=C deepening vibration, indicating the ring of phenyl or the presence of conjugated carbonyl groups. The peak at 1381.03 cm-1 identifies the vibration bending of O-H alcohol group. The band is 1134.14 cm-1 in which the vibration (CO, alcohol, ester, ether and phenol) groups are extended. Alkyne in the exopolysaccharide can occur at bands 640,96 and 617,22 cm-1. As a Fourier infrared spectroscopy transform, a fast and sensitive analysis technique was used to qualitative microbial and cell components such as EPS37. The broad FTIR peaks (Fig. 7), 3448,72, 2970,38, 2252,86, and 1381,03 cm-1 of EPS were obtained from Halomonas NASH have confirmed the carbohydrate presence. Orsod and his team stated that marine bacteria had extracted EPS from which alkenes, ketones and isocyanate, alcohols and ethers, carboxylic acid ester and phenol groups were indicated for absorption of the EPS38.
Biotechnological applications of EPS
Antimicrobial activity
EPS revealed a wide spectrum of antimicrobials against measured Gram positives Bacillus subtilis ATCC 6633, Staphylococcus aureus ATCC 6538, Gram negative bacteria; Escherichia coli ATCC 19404, Pseudomonas aeruginosa ATCC 9027 and yeast strain Candida albicans ATCC 10231. Table 1 data shows that EPS has specific antibacterial activity levels [Table 1].
Table (1):
Antimicrobial activity of the EPS produced by Halomonas sp. NASH expressed as absolute unit (AU).
Pathogens |
AU |
|---|---|
Bacillus subtilis ATCC 6633 |
10.5 |
Staphylococcus aureus ATCC 6538 |
12.6 |
Escherichia coli ATCC 19404 |
8.5 |
Pseudomonas aeruginosa ATCC 9027 |
14.7 |
Candida albicans ATCC 10231 |
20.4 |
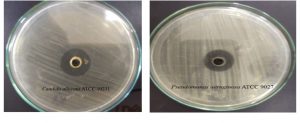
Fig. 8. Illustration of the antimicrobial activity of the EPS production by Halomonas sp. NASH against Candida albicans ATCC 10231 and Pseudomonas aeruginosa ATCC 9027.
The highest antibacterial activity (14.7 and 12.6 AU) was recorded against Pseudomonas aeruginosa ATCC 9027 and Staphylococcus aureus ATCC 6538 respectively (Fig. 8). On another hand, antifungal activity of EPS produced by Halomonas sp. NASH against Candida albicans ATCC 10231 was (20.4 AU). Nwodo states that surface active EPSs contain molecules with amphiphilic behaviour, have various chemical and surface structures and can include bio-film formation and/or have antibacterial oranti-fungal activity at times39. Several studies have shown that EPS from microbial organisms have high antibiotic activity and several antibacterial mechanisms for EPS have been suggested, including cell division, cell wall destruction, cytoplasmic membrane decomposition and DNA decomposition40,41. While the antagonistic function of polysaccharides had not been identified in these studies, protocols of these studies may lead to new areas where antibacterial activity of polysaccharide’s can be studied42.
Antioxidant activity
The obtained data indicate that EPS from Halomonas sp. NASH has good antioxidant activity. Exopolysaccharides demonstrated 61.38±0,22% antioxidant activity at a 2 mg/ml concentration, while ascorbic acid had 83.08±0,30% antioxidant activity at a 2 mg/ml concentration. The findings show that EPS can be used as a natural antioxidant alternative to synthetic antioxidants. EPS also exhibited free radical scavenging activity for DPPH from various micro-organisms43-46. Free radicals, which lead to chronic conditions, such as atherosclerosis, diabetes, rheumatoid arthritis, post-infarction, heart disease and cancer, stroke and septic shocks, aging, and other human degenerative diseases can damage bio-molecules such as lipids, protein and DNA47. The antioxidant and free radical scaving behavior of exopolysaccharide isolated from the Pseudomonas AB1 was observed by Abdrabo and his team22. Challouf and his team was found to have moderate antioxidant activity through the use of the Trolox Equivalent Antioxidants activity check for exopolysaccharide extract from Cyanobacterium Arthrospira platensis48.
Anti-inflammatory Assay
Anti-inflammatory agent EPS from Halomonas sp. NASH was tested with an albumin denaturation assay inhibition. Tests showed that EPS displayed anti-inflammatory activity (66.04%) compared with ascorbic acid as control. Exopolysaccharides which produced by Cordyceps sinensis Cs-HK1 have significant anti-inflammatory activities49. Several EPSs have been identified as being anti-inflammatory50. Normal EPS can be isolated from Bacillus circulants with anti-inflammatory activity51. In addition, the marine bacterium Bacillus amyloliquefaciens 3MS2017 can generate an acidic EPS with anti-inflammatory, antioxidant and antitumor activity52. An EPS provided by Lactobacillus paraplantarum BGCG11 also showed anti-inflammatory action in rats, by decreasing regulations for the IL-1β and iNOS mRNA and increasing levels of IL-6 and IL-10 anti-inflammatory cytokines53.
Biosurfactant activity
Biosurfactant activity of EPS which production from Halomonas sp. NASH was tested by emulsifying capacity and calculation of emulsification percentage. The findings suggest high levels of biosurfactant activity for Halomonas sp. NASH with 74.74 percent emulsification index, 67.47 and 54.96 percent respectively for paraffin oil, xylene and benzene. The biotechnological potential of microbial polysaccharides is demonstrated as immunomodulators and healers in Pharmaceutical and Gelling and Thickening Industries in food processing industries. In detoxifying areas contaminated with petrochemical oil, some EPS are used as biosurfactants54. EPSs are also used as surfactants and emulsifiers noted for the biodegradability of these agents55.
Halomonas sp. NASH synthesized large quantities of exopolysaccharide when grown under the best growth conditions, and this EPS is suitable as an antimicrobial, antioxidant , anti-inflammatory and emulsifier.
ACKNOWLEDGMENTS
None.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
LSA carried out the statistical experiment analysis. NMF wrote the manuscript. All authors read and approved the manuscript for publication.
FUNDING
None.
ETHICS STATEMENT
This article does not contain any studies with human participants or animals performed by any of the authors.
AVAILABILITY OF DATA
The datasets generated during and/or analysed the current study are available from the corresponding author on reasonable request.
- Freitas F, Alves VD, Reis MAM. Advances in bacterial exopolysaccharides: from production to biotechnological applications. Trends in Biotechnology. 2011;29(8):388-398.
Crossref - Bhaskar PV, Bhosle NB. Bacterial extracellular polymeric substance (EPS): a carrier of heavy metals in the marine food-chain. Environment International. 2006;32(2):191-198.
Crossref - Sungur EI, TuretgenI, Javaherdashti R, Cotuk A. Monitoring and disinfection of biofilm-associated sulfate reducing bacteria on different substrata in a simulated recirculating cooling tower system. Turk J Biol. 2010;34(4):389-397.
Crossref - Poli A, Anzelmo G, Nicolaus B. Bacterial exopolysaccharides from extreme marine habitats: production, characterization and biological activities. Mar Drugs. 2010;8:1779-1802.
Crossref - Satpute SK, Banat IM, Dhakephalkar PK, Banpurkar AG, Chopade BA. Biosurfactants, bioemulsifiers and exopolysaccharides from marine microorganisms. Biotechnol Adv. 2010;28(4):436-450.
Crossref - Martins PSdeO, Almeida NFde, Leite SGF, Application of a bacterial extracellular polymeric substance in heavy metal adsorption in a co-contaminated aqueous system. Braz J Microbiol. 2008;39(4):780-786.
Crossref - Moppert X, Costaouec TL, Raguenes G, et al. Investigations into the uptake of copper, iron and selenium by a highly sulphated bacterial exopolysaccharide isolated from microbial mats. J Ind Microbiol Biotechnol. 2009;36(4):599-604.
Crossref - Liu J, Luo J, Ye H, Sun Y, Lu Z, Zeng X. In vitro and in vivo antioxidant activity of exopolysaccharides from endophytic bacterium Paenibacillus polymyxa EJS-3. Carbohydr Polym. 2010;82(4):1278-1283.
Crossref - Kocharin K, , Rachathewee P, Sanglier J-J, Prathumpai W. Exobiopolymer production of Ophiocordyceps dipterigena BCC 2073: optimization, production in bioreactor and characterization. BMC Biotechnology. 2010;10:51.
Crossref - Liu C-T, Chu F-J, Chou C-C, Yu R.C. Antiproliferative and anticytotoxic effects of cell fractions and exopolysaccharides from Lactobacillus casei 01. Mutat Res Genet Toxicol Environ Mutagen. 2011;721(2):157-162.
Crossref - Oren A. Microscopic examination of microbial communities along a salinity gradient in saltern evaporation ponds: a ‘halophilic safari’, in Adaptation to Life at High Salt Concentrations in Archaea, Bacteria, and Eukarya. Springer. 2005;41-57.
Crossref - Bouchotroch S, Quesada E, Moral AD, Llamas I, Bejar V. Halomonas maura sp. nov., a novel moderately halophilic, exopolysaccharide-producing bacterium. Int J Syst Evol Microbiol. 2001;51(5):1625-1632.
Crossref - Llamas I, Amjres H, Mata JA, Quesada E, Bejar V. The potential biotechnological applications of the exopolysaccharide produced by the halophilic bacterium Halomonas almeriensis. Molecules. 2012;17(6):7103-7120.
Crossref - Garcia MT, et al. Halomonas organivorans sp. nov., a moderate halophile able to degrade aromatic compounds. Int J Syst Evol Microbiol. 2004;54(5):1723-1728.
Crossref - Yeannes MI, Ameztoy IM, Ramirez EE, FELIX, Monica MC. Culture alternative medium for the growth of extreme halophilic bacteria in fish products. Food Science and Technology. 2011;31(3): 561-566.
Crossref - Deshmukh S, Kanekar S, Bhadekar R. Production and characterization of exopolysaccharide from marine moderately halophilic bacterium halomonas smyrnensis svd iii. Int J Pharm Pharm Sci. 2017;9(10);146-151.
Crossref - Saravanan C, Kavitake D, Kandasamy S, Devi PB, Shetty PH. Production, partial characterization and antioxidant properties of exopolysaccharide α-d-glucan produced by Leuconostoc lactis KC117496 isolated from an idli batter. Journal of Food Science and Technology. 2019;56(1):159-166.
Crossref - Dubois M, KAA Gilles, JK Hamilton, PAJ Rebers. Colorimetric method for determination of sugars and related substances. Analytical Chemistry. 1956;28(3):350-356.
Crossref - Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951;193(1):265-275.
- Ventorino V, Nicolaus B, Donato PD. Bioprospecting of exopolysaccharide-producing bacteria from different natural ecosystems for biopolymer synthesis from vinasse. Chem Biol Technol Agric. 2019;6(1):18.
Crossref - Nehal F, Sahnoun M, Smaoui S, Jaouadi B, Bejar S, Mohammed S. Characterization, high production and antimicrobial activity of exopolysaccharides from Lactococcus lactis F-mou. Microbial Pathogenesis. 2019;132:10-19.
Crossref - Abdrabo MA-RA, Hassan SWM, Ibrahim HAH, Abdul-Raouf UM. Optimization of ExopolySaccharides Production from Marine Pseudomonas mendocina AB1 with Emphasis on Different Valuable Applications. J Eco Heal Env. 2019;7(1):7-20.
Crossref - Chandran H, Sharma K. Characterization and Biological Activities of an Alkali Soluble Exopolysaccharide from Acetobacter. Microbiology Research Journal International. 2019:29;1-10.
Crossref - Mizushima Y, Kobayashi M. Interaction of anti-inflammatory drugs with serum proteins, especially with some biologically active proteins. J Pharm Pharmacol. 1968;20(3):169-173.
Crossref - Sakat S, Tupe P, Juvekar A. Gastroprotective effect of methanol extract of Oxalis corniculata Linn (whole plant) experimental animals. Planta Medica. 2010;76(12):P090.
Crossref - Barakat KM, Hassan SW, Darwesh OM. Biosurfactant production by haloalkaliphilic Bacillus strains isolated from Red Sea, Egypt. Egypt J Aquat Res. 2017;43(3):205-211.
Crossref - Biswas J, Paul AK. Optimization of factors influencing exopolysaccharide production by Halomonas xianhensis SUR308 under batch culture. AIMS Microbiology. 2017;3(3):564-579.
Crossref - Arias S, Moral Ade, Ferrer MR, Tallon R, Quesada E, Bejar V. Mauran, an exopolysaccharide produced by the halophilic bacterium Halomonas maura, with a novel composition and interesting properties for biotechnology. Extremophiles. 2003;7(4):319-326.
Crossref - Martinez-Checa F, Bejar V, Martinez-Canovas MJ, Llamas I, Quesada E. Halomonas almeriensis sp. nov., a moderately halophilic, exopolysaccharide-producing bacterium from Cabo de Gata, Almeria, south-east Spain. Int J Syst Evol Microbiol. 2005;55(5):2007-2011.
Crossref - Mata JA, Bejar V, Llamas I, et al. Exopolysaccharides produced by the recently described halophilic bacteria Halomonas ventosae and Halomonas anticariensis. Research in Microbiology. 2006;157(9):827-835.
Crossref - Farres J, Caminal G, Lopez-Santin J. Influence of phosphate on rhamnose-containing exopolysaccharide rheology and production by Klebsiella I-714. Appl Microbiol Biotechnol. 1997;48(4):522-527.
Crossref - Abe K, Hayashi H, Maloney PC. Exchange of aspartate and alanine mechanism for development of a proton-motive force in bacteria. J Biol Chem. 1996;271(6):3079-3084.
Crossref - Gu D, Jio Y, Wu J, Liu Z, Chen Q. Optimization of EPS production and characterization by a halophilic bacterium, Kocuria rosea ZJUQH from Chaka Salt Lake with response surface methodology. Molecules. 2017;22(5):814.
Crossref - Vijayabaskar P, Babinastarlin S, Shankar T, Sivakumar T, Anandapandian KTK. Quantification and characterization of exopolysaccharides from Bacillus subtilis (MTCC 121). Adv Biol Res. 2011;5(2):71-76.
- Ziadi M, Bouzaiene T, M’hir S, et al. Evaluation of the efficiency of ethanol precipitation and ultrafiltration on the purification and characteristics of exopolysaccharides produced by three lactic acid bacteria. BioMed Res Int. 2018;2018.
Crossref - Maalej H, Boisset C, Hmidet N, Buon L, Heyraud A, nasri M. Purification and structural data of a highly substituted exopolysaccharide from Pseudomonas stutzeri AS22. Carbohydr Polym. 2014;112:404-411.
Crossref - Pal A, Biswas A, Chatterjee S, Kaul AK. Optimization of cultural conditions for production of exopolysaccaride by Halomonas marina HMA 103 under Batch-Culture. American Journal of Microbiology. 2015;6(2):31-39.
Crossref - Orsod M, Joseph M, Huyop F. Characterization of exopolysaccharides produced by Bacillus cereus and Brachybacterium sp. isolated from Asian sea bass (Lates calcarifer). Malays J Microbiol. 2012;8(3):170-174.
Crossref - Nwodo UU, Green E, Okoh AI. Bacterial exopolysaccharides: functionality and prospects. Int J Mol Sci. 2012;13(11):14002-14015.
Crossref - Wu M-H, Pan T-M, Wu Y-J, Chang S-J, Chang M-S, Hu C-Y. Exopolysaccharide activities from probiotic bifidobacterium: Immunomodulatory effects (on J774A. 1 macrophages) and antimicrobial properties. Int J Food Microbiol. 2010;144(1): 104-110.
Crossref - He F, Yang Y, Yang G, Yu L. Studies on antibacterial activity and antibacterial mechanism of a novel polysaccharide from Streptomyces virginia H03. Food Control. 2010;21(9):1257-1262.
Crossref - Li S, Huang R, Shah NP, Tao X, Xiong Y, Wei H. Antioxidant and antibacterial activities of exopolysaccharides from Bifidobacterium bifidum WBIN03 and Lactobacillus plantarum R315. Journal of Dairy Science. 2014;97(12):7334-7343.
Crossref - Lin SM, Baek CY, Jung J-H, et al. Antioxidant Activities of an exopolysaccharide (Deinopol) produced by the extreme Radiation-Resistant Bacterium Deinococcus radiodurans. Sci Rep. 2020;10(1):55.
Crossref - Mohamed SS, Selim MS, Mahmoud MG, Ibrahim AY, Ghazy EA. Production, characterization, and antioxidant activities of bacterial exopolysaccharides extracted from petroleum oil water. Egyptian Pharmaceutical Journal. 2019;18(1):42-52.
- Liang T-W, Tseng S-C, Wang S-L. Production and characterization of antioxidant properties of exopolysaccharide (s) from Peanibacillus mucilaginosus TKU032. Marine Drugs. 2016;14(2):40.
Crossref - Ghalem BR. Antioxidant and antimicrobial activities of exopolysaccharides from yoghurt starter. Adv Biochem. 2017;5(5):97-101.
Crossref - Uttara B, Singh AV, Zamboni P, Mahajan RT. Oxidative stress and neurodegenerative diseases: a review of upstream and downstream antioxidant therapeutic options. Curr Neuropharmacol. 2009;7(1):65-74.
Crossref - Challouf R, et al. Evaluation of cytotoxicity and biological activities in extracellular polysaccharides released by cyanobacterium Arthrospira platensis. Brazilian Archives of Biology and Technology. 2011;54(4):831-838.
- Li L-Q, Song A-X, Yin J-Y, Siu K-C, Wong W-T, Wu J-Y. Anti-inflammation activity of exopolysaccharides produced by a medicinal fungus Cordyceps sinensis Cs-HK1 in cell and animal models. Int J Biol Macromol. 2020;149:1042-1050.
Crossref - Ibrahim AY, Mahmoud M, Asker MM. Anti-inflammatory and antioxidant activities of polysaccharide from Adansonia digitata: An in vitro study. Int J Pharm Sci Rev Res. 2014;25(2):174-182.
- Vidhyalakshmi R, Valli R, Nachiyar C, Kumar N, Sunkar G, Swetha. Bacillus circulans exopolysaccharide: Production, characterization and bioactivities. Int J Biol Macromol. 2016;87:405-414.
Crossref - El-Newary SA, Ibrahim AY, Asker MS, Mahmoud MG, El-Awady ME. Production, characterization and biological activities of acidic exopolysaccharide from marine Bacillus amyloliquefaciens 3MS 2017. Asian Pac J Trop Dis. 2017;10(7):652-662.
Crossref - Dinic M, Pecikoza U, Djokic J, et al. Exopolysaccharide produced by probiotic strain Lactobacillus paraplantarum BGCG11 reduces inflammatory hyperalgesia in rats. Front Pharmacol. 2018;9:1-12.
Crossref - Mishra A, Jha B. Microbial exopolysaccharides. The Prokaryotes. 2013:179-192.
Crossref - Rosenberg E, Ron EZ. Bioemulsans: microbial polymeric emulsifiers. Curr Opin Biotechnol. 1997;8(3):313-316.
Crossref
© The Author(s) 2020. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.