ISSN: 0973-7510
E-ISSN: 2581-690X
A study in the Neonatal Department of Maternity Hospital in HCM, Vietnam, in 2019 reported that the prevalence of Carbapenemase-producing Enterobacteriaceae (CPE) was 42.20% (n = 83). However, risk factors of CPE colonization and transmission were still an unsolved question. Hence, we implemented this study. A prospective study was conducted from April to July 2020 at the Childbirth Ward of Hung Vuong Hospital, where 359 pairs of mothers and their neonates participated in our research. We applied laboratory methods to confirm CPE colonization and its antibiotic resistance, including rectal swab tests, chromo-carba plates, MALDI-TOF method, antibiograms, and rep-PCR method. The 23.0 version of SPSS was a software to analyze personal characteristics, prevalence, and risk factors for CPE colonization. Adjusted odds ratio and 95% confidence interval were considered significant at P < 0.05. The results showed that the prevalence of CP E. coli transmission between mothers and neonates was 0.28% (1/359), confirmed by the rep-PCR method. The characteristics that reduced the CPE-colonization risks in mothers were the mother’s age (19-23 years old), vaginal delivery, mothers caring for neonates, skin-to-skin contact time, and breastfeeding. However, the risk factors that increased the CPE colonization in neonates were the NICU admission before fecal sampling and the number of vaginal examinations performed on mothers before delivery. Although the prevalence of mother-to-neonate CPE transmission was low, screening for CPE colonization at hospital admission, adhering to hand hygiene, and implementing aseptic medical practices are crucial standards for preventing and controlling CPE colonization in the healthcare sector.
CPE, Neonates, Mothers, Risk Factors, Mother-to-Neonate CPE Transmission
The third-generation cephalosporins, in the early 1980s, were the best choice against lactamase-producing bacteria. However, their effectiveness was reduced by plasmid-encoded lactamases, as shown in the first report in 1983.1 From 1989 to 2004, extended-spectrum β-lactamases (ESBLs) spread in Europe, North America, South and Central America, Africa, the Middle East, Australia, and Asia.2 In early 1990, IMP carbapenemases, called the first plasmid-mediated transferable carbapenemases, emerged in Japan.3 Continuously, carbapenem-resistant Enterobacteriaceae (CRE) or carbapenemase-producing Enterobacteriaceae (CPE) existed in many countries worldwide, including high-income countries,4,5 and, secondly, in lower and middle-income countries.6 All spreading drug-resistant clones detected in carriers are in the commensal microflora.7 CRE colonization is most often in the gastrointestinal tract of carriers.8 A recent systematic review reported 1,806 patients colonized with CRE at the time of admission. It implied that CRE existed in the community, and this study also found that 299 (16.5%) acquired the clinical infection during hospital stay.9
In 49 Asian countries that participated in the CRE study, three countries reported the highest resistance rates to imipenem: Indonesia (5.8%), Vietnam (3.0%), and the Philippines (3.7%).9-13 A study of “Prevalence and risk factors of carbapenemase-producing organisms in a Neonatal Department of Maternity Hospital in the South of Vietnam” from January to March 2019, with 83 neonates chosen randomly, showed the prevalence of CPO was 42.20% (P470).14 The questions are whether CPO may be transmitted from mothers to neonates in Hung Vuong hospital, and what are the risk factors of CPO colonization in mothers and their neonates? It is the reason we conducted this study.
Method
Definition of carbapenemase-producing bacteria
Carbapenemase-producing Enterobacteriaceae (CPE) are microorganisms belonging to the group of Enterobacteriaceae that are unsusceptible to carbapenems through producing a carbapenemase enzyme.12 It is usually, but not always, through the production of carbapenemase.
CRE are Enterobacteriaceae resistant to any carbapenem antimicrobial whose minimum inhibitory concentrations are ≥4 mcg/ml for doripenem, meropenem, or imipenem, or ≥2 mcg/ml for ertapenem; or documented to produce carbapenemase (CPE).8 Clinically, CRE are screened with MELAB Chromogenic CARBA and then confirmed with the rapid BD Phoenix system.
Design and data collection
Study design
This prospective cohort study was conducted from April to July 2020 at Hung Vuong Hospital, specifically in the Delivery Department, where we collected fecal samples from mothers approximately 24 hours after admission to the hospital. Additionally, we also collected neonate fecal samples in the Post-Delivery or Neonatal Department of neonates. The length of time for the fecal sample collection in neonates was about 24 to 48 hours after delivery. There are approximately 3,600 pregnant women admitted to the Delivery Department every month. It means the number of neonates born here was approximately 3600 for one month. Mothers admitted into the Delivery Department were chosen randomly in our study. The children of these mothers were subjects invited to participate in our research. The sample size of mothers can be calculated with the Yamane formula15 as follows:
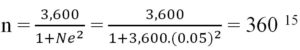 and the sample size for neonates was also 360
and the sample size for neonates was also 360
Where: n = the sample size, N = the population size, e = the acceptable sampling error (95% confidence level and p = 0.05 are assumed).
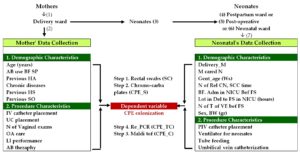
Conceptual Framework and Flow Diagram of the Study is shown in Figure 1.
Figure 1. Conceptual Framework and Process of Conducting the study of Prevalence and risk factors of CPE in mothers and neonates and transmission of carbapenemase producing organisms in Women Hospital
ABU: Antibiotic use, AB: Antibiotic, Adm in NICU Bef FS: Admission in NICU before fecal sampling, BF: Breast feeding, BW (gr): Birth Weight (gr), CPEC: CPE confirmation, CPES: CPE screening, CPE_TC: CPE transmission, confirmation, Del M: Delivery method, GA (Ws): Gestational age (Ws), HA: Hospital admission, HS: Hospital stay, LI: Labor induction, LoT fr Del to FS in NICU (hrs.): Length of time from delivery to fecal sampling in NICU (hours), MCN: Mother cared Neonates, N: Number, N of Rel CN: Number of Relatives cared Neonates, N of T of VE Bef FS: Number of times of Vaginal examination before fecal sampling, OA: Obstetric Analgesia, PIV: Peripheral intravenous catheter, SC: Sample collection, Sex, SO: Surgical operation, SP: Study participation, SSC time: Skin to skin contact time. UC: Urinary catheters
Inclusion criteria
All mothers who were admitted and delivered their babies at the Delivery department in Hung Vuong Hospital from April to July 2020 were subjects in this study.
Exclusion criteria
If mothers did not wish to participate in this study, they and their babies would not be the subjects in this study, and neonates had an abnormal anus or did not have an anus.
One neonate did not continue participating in our study because this neonate was transferred to the Children’s Hospital on the first day after birth to be treated by a neonatal specialist. Hence, the number of mothers and neonates participating in our study was 359 for each group.
Material
Data collection
We collected the study data from the medical records,
- Independent variables collected for mothers included characteristics: (a) demographics such as age (years), prior antibiotic treatment, prior hospital admission, chronic disease, prior hospital stay, prior surgical intervention1,16; and (b) procedures performed on mothers,16,17 such as peripheral intravenous catheter and urinary catheter use;
- Independent variables collected for neonates included characteristics: (a) demographics1,16 such as sex, gestational age (weeks), age (days), birth weight (kgs), length of stay (days), prior exposure to antibiotics, Cesarean section/Vaginal delivery; and procedure characteristics performed on neonates,1,16 such as invasive device: peripheral intravenous catheter, Mechanical ventilation,2,16 feeding tube and umbilical vein catheterization use.
- The dependent variable was CPE for mothers and neonates.
Conceptual Framework and Flow Diagram of Study
Microbiology methods
Tools to screen and detect CPE include: First, we utilized the rectal swabs (Nantong Renon Laboratory Equipment Co., Ltd., No. 128, Xiaohai Road, Sanhe Town, Haimen, Jiangsu) to collect the fecal samples. Then, we transported these samples to the Lab Unit of OUCRU, Oxford University Clinical Research Unit, in HCM City, to screen CPE by ChromID carba plates produced with the selective medium called Chromagar (29 Av. George Sand, 93210 Saint-Denis, France). Next, we used the MALDI TOF method (Bruker Daltonics GmbH & Co. KG, Fahrenheitstraße 428359 Bremen, Germany) for species identification of colonies on blood agar. At the end of step one, we use antimicrobial susceptibility testing to confirm the antibiotic resistance of the species.
In step two, we determine the association between mothers and their children by first displaying the genome patterns of mothers and neonates with the rep-PCR DNA fingerprinting technique (CD Genomics, SUITE 111, 17 Ramsey Road, Shirley, NY 11967, USA). Secondly, we perform whole-genome sequencing to investigate this association, if possible.
Statistical analysis
23.0 version of SPSS software was a tool used to analyze data. Descriptive statistics analyzes characteristics associated with demographics, procedures performed on mothers and neonates, and the prevalence of CPE. Multivariate logistic regression analyzes the risk factors related to CPE and non-CPE status, and the adjusted odds ratio with a 95% confidence interval was considered significant. The level of statistical significance was P < 0.05.
Prevalence of CPE colonization in mothers and neonates
The study result showed twenty-one mothers colonized by CPE, including nineteen E. coli and two K. pneumoniae, in whom four mothers colonized by two CPE included three mothers colonized by E. coli and (K. pneumoniae or Ent. cloacae), and one colonized by K. pneumoniae and E. coli. Hence, the prevalence of CPE colonization in mothers and neonates was 5.85% (21/359), and 3.62% (13/359, respectively, as shown in Table 1, while carbapenem-producing K. pneumoniae (KPC) appeared in neonates.
Table (1):
Prevalence of CPE in mothers and neonates
| CPE 1 in Mother | Freq. (%) | CPE 2 in Mother | Freq. (%) | % Mother CPE | CPE in Neonates | Freq. (%) | % Neonate CPE | % CP. E. coli transmission between M&C |
|---|---|---|---|---|---|---|---|---|
| E. coli | 16 (4.45) | 5.85 (21/359) | E. coli | 5 (1.39) | 3.62 (13/359) | 0.28 (1/359) | ||
| 3 (0.84) | K. pneumoniae | 1 (0.28) | K. pneumoniae | 5 (1.39) | ||||
| K. pneumoniae | 1 (0.28) | Ent. cloacae | 2 (0.56) | |||||
| Ent. cloacae | 1 (0.28) | Ent. kobei | 1 (0.28) | |||||
| K. pneumoniae | 2 (0.56) | E. coli | 1 (0.28) | |||||
| Non-CPE | 338 (94.15) | Non-CPE | 355 (98.88) | Non-CPE | 346 (96.38) | |||
| Total | 359 (100) | 359 (100) | Total | 359 (100) |
CPE transmission between mothers and neonates
Our study findings showed that the CPE of a child was the same as that of a mother, a 32-year-old teacher, who was admitted to the Maternity Hospital approximately 86 hours before study participation, and the Delivery department for 6 hours before collecting feces by rectal swab. This 32-year-old mother had no risk of CPE colonization before our study participation, including the length of stay in hospital, the past year, antibiotic use, and nosocomial infections due to CPE or non-CPE before participating in this study. This mother gave birth vaginally and breastfed her 37 week-gestational age and 2350 gram-weight male baby, who spent skin-to-skin contact with her mother for two hours, did not take antibiotics before taking fecal sampling, was not admitted to the Neonatal Department, and was taken care of by his mother and father.
Based on the method of rep-PCR, we confirmed that the CPE of the mother was identical to that of her neonate, as shown in Figure 2. It means that the mother transmitted CP. E. coli to her child. Hence, the prevalence of CP E. coli transmission between a mother and her child was 0.28 (1/359) (Table 1).
Risk factors of CPE colonization in mothers
Univariate binary logistic regression of risk factors of CPE colonization in mothers
Our study research also showed that the total doses of antibiotics consumed by mothers before fecal sample collection were risk factors for CPE colonization, compared with mothers who did not consume antibiotics. For example, the groups of mothers with total antibiotic doses less than two or more than three doses had the risk of CPE colonization more than 7 or 5 times compared with the group of mothers not using antibiotics, with P = 0.023, OR = 7.022, 95% CI: 1.306-37.755, and P = 0.007, OR = 5.267, 95% CI: 1.568-17.695, respectively. The length of time from labor induction to sample collection, from 36 to 48 hours, and over 48 hours, was a risk factor associated with CPE colonization more than 18 and 12 times, compared with the group of mothers with the length of time from labor induction to sample collection less than 24 hours, as described in Table 2, with P = 0.042, OR = 18.444, 95% CI: 1.108-307.005 and P = 0.008, OR = 12.296, 95% CI: 1.932-78.280, respectively. The length of time from labor induction to sample collection was over 48 hours, which meant mothers spent more than 48 hours in the hospital.
Table (2):
Univariate Binary Logistic Regression of Risk Factors of CPE Colonization in mothers in the Delivery Department of Maternity Hospital, April to July 2020
| Cell | Code | B | S.E. | Wald | df | Sig. | Exp(B) | 95% CI for EXP(B) | ||
|---|---|---|---|---|---|---|---|---|---|---|
| Lower | Upper | |||||||||
| 1 | ABU before study participation | Non-ABU: (0), ABU: (1) | -.115 | .083 | 1.936 | 1 | .164 | .891 | .757 | 1.048 |
| Constant | -1.831 | .695 | 6.937 | 1 | .008 | .160 | ||||
| 2 | Total AB doses used by Mother before fecal sample collection | T of ABD = 0 dose | 10.961 | 2 | .004 | |||||
| T of ABD ≤2 doses (1) | 1.949 | .858 | 5.158 | 1 | .023 | 7.022 | 1.306 | 37.755 | ||
| T of ABD ≥3 doses (2) | 1.661 | .618 | 7.220 | 1 | .007 | 5.267 | 1.568 | 17.695 | ||
| Constant | -3.048 | .264 | 133.013 | 1 | .000 | .047 | ||||
| 3 | ADM before study participation about one year | Non-ADM: (0), ADM:(1) | .396 | 1.070 | .137 | 1 | .711 | 1.486 | .183 | 12.093 |
| Constant | -2.794 | .230 | 147.154 | 1 | .000 | .061 | 2 | |||
| 4 | Mother’s age (years) | ≤18 | 4.955 | 3 | .175 | |||||
| 19 to 23 | -2.702 | 1.266 | 4.554 | 1 | .033 | .067 | .006 | .802 | ||
| 24 to 28 | -1.030 | .835 | 1.520 | 1 | .218 | .357 | .070 | 1.835 | ||
| ≥29 | -.727 | .853 | .726 | 1 | .394 | .484 | .091 | 2.571 | ||
| Constant | -1.705 | .769 | 4.918 | 1 | .027 | .182 | ||||
| 5 | Number of days for hospital stay in the last ADM | Non-Hospital stay (0) | .844 | 2 | .656 | |||||
| <2 weeks (1) | 1.015 | 1.104 | .844 | 1 | .358 | 2.758 | .317 | 24.028 | ||
| 1 to 2 months | -18.397 | 40192.970 | .000 | 1 | 1.000 | .000 | 0.000 | |||
| Constant | -2.806 | .230 | 148.541 | 1 | .000 | .060 | ||||
| 6 | Duration of Hospital stay before sample collection (hours) | ≤24 | 6.390 | 2 | .041 | |||||
| 24 to 48 | 1.036 | .602 | 2.961 | 1 | .085 | 2.819 | .866 | 9.177 | ||
| >48 | 1.335 | .611 | 4.765 | 1 | .029 | 3.799 | 1.146 | 12.595 | ||
| Constant | -3.084 | .284 | 118.234 | 1 | .000 | .046 | ||||
| 7 | Chronic diseases | Non- Chronic diseases (0), Chronic diseases (1) | -18.504 | 7882.490 | .000 | 1 | .998 | .000 | 0.000 | |
| Constant | -2.698 | .225 | 143.274 | 1 | .000 | .067 | ||||
| 8 | Past surgical operation Before study participation | Non PSO (0) PSO (1) | -.511 | 1.045 | .239 | 1 | .625 | .600 | .077 | 4.651 |
| Constant | -2.747 | .231 | 141.857 | 1 | .000 | .064 | ||||
| 9 | Professional | HCW (0) | .311 | 4 | .989 | |||||
| State employee (1) | 18.513 | 14210.307 | .000 | 1 | .999 | 109692359.845 | 0.000 | |||
| Businessperson (2) | 18.430 | 14210.307 | .000 | 1 | .999 | 100966831.221 | 0.000 | |||
| Self-employed (3) | 18.677 | 14210.307 | .000 | 1 | .999 | 129237543.962 | 0.000 | |||
| Stay-at-home spouse (4) | 18.258 | 14210.307 | .000 | 1 | .999 | 85024699.975 | 0.000 | |||
| Constant | -21.203 | 14210.307 | .000 | 1 | .999 | .000 | ||||
| 10 | Duration of performance of profession (years) | Duration of work | .069 | .048 | 2.040 | 1 | .153 | 1.071 | .975 | 1.177 |
| Constant | -3.074 | .326 | 88.970 | 1 | .000 | .046 | ||||
| 11 | Obstetric Analgesia | OA: Y (1), N (0) | .111 | .464 | .058 | 1 | .810 | 1.118 | .451 | 2.773 |
| Constant | -2.820 | .285 | 97.532 | 1 | .000 | .060 | ||||
| 12 | Length of time from Obstetric Analgesia to Sample collection | Obstetric Analgesia (1), Non-Obstetric Analgesia (0) | .028 | .109 | .066 | 1 | .797 | 1.029 | .830 | 1.275 |
| Constant | -2.803 | .247 | 128.934 | 1 | .000 | .061 | ||||
| 13 | Labor induction | Yes (1), No (0) | 2.064 | .731 | 7.970 | 1 | .005 | 7.881 | 1.880 | 33.040 |
| Constant | -2.912 | .242 | 144.738 | 1 | .000 | .054 | ||||
| 14 | Length of time from Labor induction to Sample collection (hours) | LoT fr LI to SC ≤ 24 hrs. | 10.740 | 3 | .013 | |||||
| LoT_fr_LI_to_SC from >24 to 36 hrs. (1) | -18.288 | 28420.722 | .000 | 1 | .999 | .000 | 0.000 | |||
| LoT fr LI to Sc from 36 to 48 hrs. (2) | 2.915 | 1.435 | 4.127 | 1 | .042 | 18.444 | 1.108 | 307.005 | ||
| LoT fr LI to SC >48 hrs. (3) | 2.509 | .944 | 7.060 | 1 | .008 | 12.296 | 1.932 | 78.280 | ||
| Constant | -2.915 | .242 | 145.060 | 1 | .000 | .054 | ||||
Multivariate binary logistic regression of risk factors of CPE colonization in mothers
When analyzing the multivariate binary logistic regression of risk factors associated with CPE colonization in mothers, we determined that the total antibiotic dose from 1 to 2 doses, consumed by mothers before sample collection was a principal risk factor of CPE colonization, and the group of mothers consuming the antibiotic doses from 1 to 2 doses before sample collection, was colonized by CPE colonization with more than 5.3 times, with P = 0.025, OR = 5.336, 95% CI: 1.230-23.153, compared with the rest group of mothers (Table 3).
Table (3):
Multivariate Binary Logistic Regression of Risk Factors of CPE Colonization in mothers in the Delivery Department of Maternity Hospital, April to July 2020
| Characteristics | Code | B | S.E. | Wald | df | Sig. | Exp(B) | 95% CI for EXP(B) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lower | Upper | ||||||||||||||
| Age (years) | ≤18.00 | 3.761 | 3 | .288 | |||||||||||
| 19.00 to 23.00 | -2.159 | 1.353 | 2.548 | 1 | .110 | .115 | .008 | 1.635 | |||||||
| 24.00 to 28.00 | -.572 | .983 | .339 | 1 | .561 | .564 | .082 | 3.877 | |||||||
| ≥29.00 | -.164 | 1.005 | .027 | 1 | .870 | .849 | .118 | 6.086 | |||||||
| Delivery method (C) | Vaginal delivery (1), Cesarian section (0) | 1.398 | .767 | 3.317 | 1 | .069 | 4.045 | .899 | 18.201 | ||||||
| Mother care Child (C) | M cared C (1), M_n0 Cared C (0) | -1.031 | .777 | 1.758 | 1 | .185 | .357 | .078 | 1.637 | ||||||
| Length of time of Hospital stay before sample collection (hours) | t ≤ 24.00 | 2.880 | 2 | .237 | |||||||||||
| 24 < t ≤ 48 | 1.111 | .656 | 2.869 | 1 | .090 | 3.038 | .840 | 10.993 | |||||||
| t > 48 | .152 | 1.117 | .018 | 1 | .892 | 1.164 | .130 | 10.394 | |||||||
| Length of time from Labor induction to sample collection (hours) | LoT fr LI to SC: t ≤ 24 | 2.922 | 3 | .404 | |||||||||||
| LoT fr LI to SC: 24 < t ≤ 36 | -18.390 | 27704.599 | .000 | 1 | .999 | .000 | 0.000 | ||||||||
| LoT fr LI to SC: 36 < t ≤ 48 | 3.469 | 2.038 | 2.897 | 1 | .089 | 32.119 | .591 | 1744.745 | |||||||
| LoT fr LI to SC: t > 48 | 1.136 | 1.597 | .506 | 1 | .477 | 3.115 | .136 | 71.308 | |||||||
| Total of Antibiotic doses consumed by mother before sample collection | Zero dose | 5.000 | 2 | .082 | |||||||||||
| 1 to 2 doses | 1.674 | .749 | 5.000 | 1 | .025 | 5.336 | 1.230 | 23.153 | |||||||
| ≥3 doses | .186 | 1.560 | .014 | 1 | .905 | 1.205 | .057 | 25.634 | |||||||
| Constant | -2.830 | 1.230 | 5.296 | 1 | .021 | .059 | |||||||||
Univariate binary logistic regression of risk factors of CPE colonization in neonates
By the univariate binary logistic regression of risk factors of CPE colonization in the neonates at the Post Delivery Department of Maternity, we detected that the vaginal delivery, mother caring for her neonate, and the interaction of these two factors, one positive factor, reduced the risks of CPE colonization in neonates, with P = 0.014, OR = 0.240, 95% CI: 0.077-0.752, in the Cell 1; P = 0.001, OR = 0.148, 95% CI: 0.047-0.464, in the Cell 2; and P = 0.022, OR = 0.056, 95% CI: 0.005-0.657, in the Cell 2* of the Table 4, respectively. The other beneficial factors reducing the CPE colonization in neonates included neonates’ gestational age (weeks), with P = 0.046, OR = 0.309, 95% CI: 0.097 -0.979, in The Cell 3 of Table 4, and especially, the number of relatives cared neonates less than two relatives, with P = 0.000, OR = 0.020, 95% CI: 0.003-0.134, in the Cell 4 of Table 4. One advantageous factor associated with decreasing the risk of CPE colonization was the skin-to-skin contact time between mothers and neonates for two hours, with P = 0.002, OR = 0.151, 95% CI: 0.046-0.492, in the Cell 5 of Table 4 and mothers breastfeeding their neonates prevented and reduced CPE colonization in her neonates, with P = 0.007, OR = 0.183, 95% CI: 0.053-0.634, In the Cell 6 of Table 4.
Along with positive factors in our study, we also found negative features associated with CPE colonization in neonates. For instance, neonates admitted to the NICU before fecal sampling had a risk of CPE colonization compared with neonates not admitted to the NICU, with P = 0.000, OR = 21.524, 95% CI: 5.332-86.888, as shown in Cell 7 of Table 4.
In addition, we recognized the lengths of time from delivery to fecal sampling in NICU from 17-36.00 hours, and over 36.00 hours were the risk factors of CPE colonization, compared with the group of neonates collected with fecal samples at the beginning of NICU admission, with P = 0.001, OR = 25.111, 95% CI: 3.727-169.190, and P = 0.002, OR = 18.833, 95% CI: 3.046-116.456, respectively, as shown in Cell 8 of Table 4.
Finally, vaginal examination before fecal sampling was a risk factor for CPE colonization when the number of vaginal examinations was from 6 to 10 times compared to that from 1 to 5 times, with P = 0.016, OR = 12.571, 95% CI: 1.601 -98.737, as shown in Cell 9 of Table 4.
Table (4):
Univariate Binary Logistic Regression of Risk Factors of CPE Colonization in Neonates at the Post Delivery Department of Maternity Hospital, April to July 2020, April to July 2020
| Cell | Characteristics | Code | B | S.E. | Wald | df | Sig. | Exp(B) | 95% CI for EXP(B) | ||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Lower | Upper | ||||||||||
| 1 | Delivery method | Caesarean section: (0), Vaginal delivery: (1), | -1.427 | .583 | 6.000 | 1 | .014 | .240 | .077 | .752 | |
| Constant | -2.485 | .368 | 45.598 | 1 | .000 | .083 | |||||
| 2 | Mother cared Child | Mother no cared child: (0), Mother cared Child: (1) | -1.909 | .582 | 10.770 | 1 | .001 | .148 | .047 | .464 | |
| Constant | -1.872 | .439 | 18.219 | 1 | .000 | .154 | |||||
| 2* | Delivery method by mother cared Child | Delivery method by mother cared Child | -2.886 | 1.258 | 5.260 | 1 | .022 | .056 | .005 | .657 | |
| Constant | -2.313 | .332 | 48.660 | 1 | .000 | .099 | |||||
| 3 | Gestational age (Weeks) | Gest age <37 Ws: (0) Gest age ≥37 Ws: (1), | -1.175 | .588 | 3.984 | 1 | .046 | .309 | .097 | .979 | |
| Constant | -2.416 | .467 | 26.791 | 1 | .000 | .089 | |||||
| 4 | Number of Relative cared child | N of Rel car Child: (0) | 16.311 | 2 | .000 | ||||||
| N of Rel car Child ≤2 (1) | -3.908 | .968 | 16.311 | 1 | .000 | .020 | .003 | .134 | |||
| N of Rel car Child ≥3 (2) | -21.608 | 11602.711 | .000 | 1 | .999 | .000 | 0.000 | ||||
| Constant | .405 | .913 | .197 | 1 | .657 | 1.500 | |||||
| 5 | Skin to skin contact time (hours) | SSC time = (0) | 9.830 | 2 | .007 | ||||||
| SSC time = (1) | -.847 | 1.101 | .592 | 1 | .442 | .429 | .049 | 3.711 | |||
| SSC time = (2) | -1.892 | .603 | 9.830 | 1 | .002 | .151 | .046 | .492 | |||
| Constant | -2.097 | .401 | 27.419 | 1 | .000 | .123 | |||||
| 6 | Breastfeeding | Breast Feed Yes: (1), Breast Feed No: (0) | -1.699 | .635 | 7.171 | 1 | .007 | .183 | .053 | .634 | |
| Constant | -1.872 | .537 | 12.146 | 1 | .000 | .154 | |||||
| 7 | Admission in NICU before fecal sampling | Adm in NICU B fecal sample: (1), No Adm in NICU B fecal sample: (0) | 3.069 | .712 | 18.582 | 1 | .000 | 21.524 | 5.332 | 86.888 | |
| Constant | -3.629 | .338 | 115.447 | 1 | .000 | .027 | |||||
| 8 | Length of time from delivery to fecal sampling in NICU (hours) | L of T fr del to Fecal sample in NICU: 0 hr. (1), | 18.599 | 2 | .000 | ||||||
| L of T fr del to Fecal sample in NICU 17.00 to 36.00 hrs (2) | 3.223 | .973 | 10.967 | 1 | .001 | 25.111 | 3.727 | 169.190 | |||
| L of T fr del to Fecal sample in NICU: >36 hrs (3) | 2.936 | .930 | 9.974 | 1 | .002 | 18.833 | 3.046 | 116.456 | |||
| Constant | -3.629 | .338 | 115.447 | 1 | .000 | .027 | |||||
| 9 | Number of times of Vaginal examination before fecal sampling | No of Vag exam from 1 to 5 times: (0) | 8.311 | 2 | .016 | ||||||
| No of Vag exam: from 6 to 9 times: (1) | 2.531 | 1.052 | 5.795 | 1 | .016 | 12.571 | 1.601 | 98.737 | |||
| No of Vag exam: from ≥10 times: (2) | .639 | 1.421 | .202 | 1 | .653 | 1.895 | .117 | 30.716 | |||
| Constant | -4.970 | 1.003 | 24.529 | 1 | .000 | .007 | |||||
| 10 | Sex | Male: (1), female: (2), | .258 | .567 | .208 | 1 | .649 | 1.295 | .426 | 3.931 | |
| Constant | -3.412 | .415 | 67.631 | 1 | .000 | .033 | |||||
| 11 | Weight (grams) | ≤2500 grs: (1), >2500 grs: (2) | -1.322 | .810 | 2.661 | 1 | .103 | .267 | .054 | 1.305 | |
| Constant | -.758 | 1.531 | .245 | 1 | .621 | .469 | |||||
| 12 | Delivery method | Caesarean section: (0), Vaginal delivery: (1), | 1.224 | .990 | 1.529 | 1 | .216 | 3.400 | .489 | 23.652 | |
| Mother cared Child | Mother no cared child: (0), Mother cared Child: (1) | -.601 | .739 | .662 | 1 | .416 | .548 | .129 | 2.332 | ||
| Delivery method by Mother care Child | Delivery method by Mother care Child | -2.886 | 1.258 | 5.260 | 1 | .022 | .056 | .005 | .657 | ||
Multivariate binary logistic regression of risk factors of CPE colonization in neonates
The result of multivariate binary logistic regression of risk factors of CPE colonization in neonates showed the number of vaginal examination times before fecal sampling from 6-10 times was the principal risk factor of CPE colonization, which influenced all other risk factors detected in mothers that had vaginal birth, compared with that of from 1-5 times, with P = 0.041, OR = 14.487, 95% CI: 1.115-188.237, as shown in the Characteristic 9 of Table 5.
Table (5):
Multivariate Binary Logistic Regression of Risk Factors of CPE Colonization in neonates in the Delivery Department of Maternity Hospital, April to July 2020
| Characteristics | Codes | B | S.E. | Wald | df | Sig. | Exp(B) | 95% C.I.for EXP(B) | |
|---|---|---|---|---|---|---|---|---|---|
| Lower | Upper | ||||||||
| 1. Delivery method | Caesarean section: (0), Vaginal delivery: (1), | -20.583 | 15563.029 | .000 | 1 | .999 | .000 | 0.000 | |
| 2. Mother cared child | Mother no cared child: (0), Mother cared Child: (1) | .176 | .997 | .031 | 1 | .860 | 1.193 | .169 | 8.412 |
| 3. Delivery method by Mother care Child | Delivery method by Mother cared Child | 20.362 | 15563.029 | .000 | 1 | .999 | 696910049.542 | 0.000 | |
| 4. Number of relative cared child | N of Rel cared Child: (0), N of Rel cared Child: ≤2 (1), N of Rel cared Child: ≥3 (2) | -22.926 | 15563.029 | .000 | 1 | .999 | .000 | 0.000 | |
| 5. Gestational age (Weeks) | Gest age <37 Ws: (1) Gest age ≥37 Ws: (2), | -.692 | .955 | .525 | 1 | .469 | .501 | .077 | 3.254 |
| 6. Skin to skin contact time (hours) | SSC time = (0) | 1.763 | 2 | .414 | |||||
| SSC time = (1) (1) | -.575 | 1.353 | .181 | 1 | .671 | .562 | .040 | 7.978 | |
| SSC time = (2) (2) | -1.988 | 1.498 | 1.763 | 1 | .184 | .137 | .007 | 2.578 | |
| 7. Breastfeeding | Breast Feed Yes: (1), Breast Feed No: (0) | -.016 | 1.142 | .000 | 1 | .988 | .984 | .105 | 9.223 |
| 8. Length of time from delivery to fecal sampling in NICU (hours) | LoT from NICU ADM to SC: 0 (hour) | .865 | 2 | .649 | |||||
| LoT from NICU ADM to SC ≤36 (1) | 2.667 | 3.131 | .726 | 1 | .394 | 14.392 | .031 | 6649.807 | |
| LoT_from NICU ADM to SC: >36 (2) | 1.747 | 2.144 | .664 | 1 | .415 | 5.738 | .086 | 383.637 | |
| 9. Number of times of Vaginal examination before fecal sampling | No of Vag exam from 1 to 5 times: (0) | 5.466 | 2 | .065 | |||||
| No of Vag exam: from 6 to 10 times: (1) | 2.673 | 1.308 | 4.174 | 1 | .041 | 14.487 | 1.115 | 188.237 | |
| No of Vag exam: from ≥11 times: (2) | 1.128 | 1.620 | .484 | 1 | .486 | 3.088 | .129 | 73.966 | |
| 10. Neonatal Birth weight (gram) | Neonatal Birth weight: ≤2500 grs (0), Neonatal Birth weight >2500 grs (1) | -1.072 | 1.342 | .638 | 1 | .424 | .342 | .025 | 4.748 |
| 11. Sex | Male (1), Female (0) | .384 | .721 | .284 | 1 | .594 | 1.469 | .358 | 6.035 |
| Constant | 20.590 | 15563.029 | .000 | 1 | .999 | 874888019.484 | |||
Source of CPE colonization in mothers
Based on the results of prevalence and risk factors associated with CRE colonization, displayed in Tables 1, 2, and 3. Table 6 lists participants’ professions and shows that the prevalence of CPE was 0% (n = 8), 6.40% (n = 173), 5.90% (n = 51), 7.40% (n = 27), 5.00% (n = 100) in the group of HCWs, State employees, Businesspersons, Self-employed persons, and Stay-at-home spouses, respectively. The result of Table 6 was surprising, as CPE did not appear in HCWs. However, we detected CPE in state employees and Housewives, who had the potential to contact hospital environments and healthcare, fewer than the HCWs, a key characteristic of all CRE-colonized mothers, especially CR. E. coli colonized mothers did not acquire any hospital infections, any chronic disease, or any surgical operation before participating in this study (Table 6).
Table (6):
Characteristics associated with CPE in mothers and neonates
| OCCUP (1) | Study Participants (2) | CPE_1 (3) | CPE_2 (4) | M&C had CPE ID (5) | CPE_HI (6) | Non-CPE HI (7) (Case) | Non-HI (8) | ABU Bef Part Study (9) (Case) |
|---|---|---|---|---|---|---|---|---|
| HCWs (n=8) | M | 0 | 0 | No | 0 | 0 | 0 | 0 |
| Nn | 0 | 0 | 0 | 0 | 0 | |||
| State Employee (n=173) | M | E. coli: ten | E. coli: one | Yes: E. coli | 0 | SSI = 1 | 0 | 11 |
| K. pneumoniae: one | Ent. cloacae: one | PWI = 1 | ||||||
| Nn | E. coli: one | 0 | 0 | NI | ||||
| K. pneumoniae: two | ||||||||
| Ent. cloacae: one | ||||||||
| Ent. kobei: one | ||||||||
| Businessperson (n=51) | M | E. coli: two | K. pneumoniae: one | No | 0 | 0 | 0 | 4 |
| K. pneumoniae: one | ||||||||
| Nn | E. coli: two | 0 | 0 | 0 | ||||
| Ent. cloacae: one | ||||||||
| Self-employed (n=27) | M | E. coli: two | No | 0 | 0 | 0 | 2 | |
| Nn | E. coli: one | 0 | 0 | 0 | ||||
| Stay-at-home spouse (n=100) | M | E. coli: five | K. pneumoniae: one | No | 0 | 0 | 0 | 7 |
| Nn | E. coli: one | 0 | 0 | 0 | ||||
| K. pneumoniae: three |
C: Child, CPE ID: CPE Infectious disease, HI: Hospital infection, M: Mother, NI: Neonatal infection, Nn: Neonate, OCCUP: Occupation, PWI: Perineal wound infection, SSI: Surgical site infection
Table (6):
Characteristics associated with CPE in mothers and neonates (Cont.1)
| OCCUP (1) | Study Participants (2) | Last ABU Bef Adm, HL Ago fr Adm (10) (Month) | M cared C (11) (%) | Rel cared C (12) (%) | GA (13) (Weeks) | NICU adm Bef SC (14) (Case) | Reas for NICU ADM (15) (Case) | Deliv M – VAG Del (16) (%) |
|---|---|---|---|---|---|---|---|---|
| HCWs (n=8) | M | 87.50% | ||||||
| Nn | 100% | 36 to 40 | 0 | 0 | 75% | |||
| State Employee (n=173) | M | ≤3mos (4/173) | 86.71% | |||||
| ≥6 mos (6/173) | ||||||||
| Nn | 98.84% | 35 to 41 | 4 | NeoJ: seven, RF: two | 72.30% | |||
| PTB: one | ||||||||
| NI: two. | ||||||||
| Businessperson (n=51) | M | ≥9 mos (4/51) | 86.30% | |||||
| Nn | 96.10% | 33 to 40 | 3 | NeoJ: two, RF: two | 60.80% | |||
| PTB: one, | ||||||||
| BLDS: one, | ||||||||
| Self-employee (n=27) | M | ≤3 mos (1/27) | 81.50% | |||||
| ≥ 9 mos (1/27) | ||||||||
| Nn | 100% | 32 to 41 | 1 | PTB: one, | 66.66% | |||
| Stay-at-home spouse (n=100) | M | ≤3 mos (1/100) | 91.00% | |||||
| ≥9 mots (5/100) | ||||||||
| Nn | 99% | 33 to 41 | 3 | NeoJ: six, RF:2, | 75% |
Adm: Admission, HL Ago fr Adm: How long ago from admission, M cared C: Mother cared child, Rel cared C: Relative cared child, GA (Ws): Gestational age (Ws), NI: Neonatal infection, NICU adm Bef FS: NICU Admission before fecal sampling, Reas for NICU ADM: Reasons for NICU admission, Del M – VAG Del: Delivery method- Vaginal delivery, NeoJ: Neonatal Jaundice, PTB: Premature birth, RF: Respiratory failure, BLDS: Bloody stool
Table (6):
Characteristics associated with CPE in mothers and neonates (Cont.2)
OCCUP (1) |
Study Participants (2) |
M: Chronic diseases (cases) (17) |
M: Sx Op Bef P in CPE study (cases) (18) |
Characteristics of M: Dur of T from HV ADM to SC (hours) (19) |
Characteristics of M with CPE colonization: Dur of T from HV ADM to SC (hours), n=21 (20) |
Characteristics of N with CPE colonization: Dur of T from Del to SC (hours), n = 13, (21) |
|---|---|---|---|---|---|---|
HCWs (n=8) |
Mother |
No |
1 |
Mean = 20.37 hrs, |
Mean =33.76 hrs, |
Mean =40.07 hrs, |
State Employee (n=173) |
Mother |
18 |
10 |
Std. Deviation = 45.77 hrs, Min = 0 hrs, |
Std. Deviation = 46.35 hrs, Min = 0 hrs, |
Std. Deviation = 4.462 hrs, Min = 31 hrs, |
Businessperson (n=51) |
Mother |
2 |
6 |
Max = 549 hrs (22.87 days), |
Max =159.00 hrs (6.63 days), |
Max =47.00 hrs (1.95 days), |
Self emloyee (n=27) |
Mother |
1 |
3 |
86.60% (from 0 to 28.00 hrs) |
76.20% (from 0 to 28.00 hrs) |
100%
|
Stay-at-home spouse (n=100) |
Mother |
5 |
7 |
OCCUP: Occupation, M: Mother, M: Sx Op Bef P in CPE study: Mother: surgery operation before participating in CPE study, M: Dur of T from HV ADM to SC (hour)s: Mother: duration of time from HV Hospital admission to sampling collection, Characteristics of M: Dur of T from HVADM to SC (hours): Characteristics of Mother: Duration of time from HV admission to sampling collection, Characteristics of M with CPE colonization: Dur of T from HV ADM to SC (hours): Characteristics of Mothe with CPE colonizationr: Duration of time from HV admission to sampling collection, Characteristics of N with CPE colonization: Dur of T from Del to SC (hours): Characteristics of Neonatal with CPE colonizationr: Duration of time from HV admission to sampling collection (hours)
Moreover, one impressive character was that the duration from hospital admission at the study site to the fecal sample collection in mothers with CPE colonization was 76.20% (from 0-28.00 hrs) (Min: 0 hours, and Max = 159.00 hrs (equal to 6.63 days).
In neonatals
Table 6 showed that the duration of time from delivery to sample collection in the neonatal colonized with CPE was 31.00-47.00 hours. The result in Table 6 confirmed one case of non-hospital-acquired and non-CPE caused infection determined in one neonate.
Duration of time from labor induction to fecal sample collection
Table 6 showed that the duration of time from delivery to sample collection in the neonatal colonized with CPE was 31.00-47.00 hours, and only one case of non-hospital-acquired and a non-CPE caused infection was a neonatal infection found in one neonate.
Association between the duration of time from labor induction and fecal sample collection over 36 hours
Table 2 showed that labor induction and the duration of time from labor induction to sample collection (more than 36 hours) were risk factors for CPE colonization. Specifically, the group of pregnant women (n = 7), who spent time from labor induction to sample collection (over 36 hours, had a CPE colonization prevalence at 42.90% (3/7). Three of six pregnant women who consumed antibiotics had CPE colonization, with 50% (3/6).
The results described in Table 1 suggested how to determine the origin of KPC in neonates and distinguish K. pneumoniae-producing carbapenemase in mothers and children. It was whether Carbapenemase-producing E. coli found in mothers was the same source in neonates. Could the origin of K. pneumoniae and E. coli in mothers and neonates be from the community, the hospital environment, healthcare workers, or other sources that did not belong to the hospital?
Source of CPE colonization in mothers
As shown above, the duration from hospital admission at the study site to the fecal sample collection in mothers with CPE colonization was 76.20% (from 0-28.00 hrs), and characteristics of mothers with CPE colonization: The duration of time from admission to sample collection (hours) in pregnant mother acquired CPE colonization, was 76.20% (from 0-28.00 hrs), Table 6, (Cont.2). Thus, was this length of time enough for the CPE to exist in the gastrointestinal tract of the mothers? A case-control study reported that duration of hospital stay over 28 days (OR 23.6, 95% CI 4.9-113.3) in the last 12 months was a risk factor for acquiring K. pneumoniae producing carbapenemase, detected with rectal swabs.18 Moreover, another case-control study reported that hospital stays exceeding twenty days were a risk factor for acquiring carbapenemase-producing Enterobacteriaceae, as determined similarly with rectal swabs (AOR: 4.9, 95% CI: 1.4-15.5; P < 0.001).19 The duration of CPE detection in the two studies above showed that CPE found in mothers in our research could appear before admission to Hung Vuong Hospital or be due to cross-contamination happening at the study site.
We recognized that, for example, the hospital admission before study participation about one year, the number of days for hospital stay in the last admission, chronic diseases, past surgical operation before study participation, professional, and duration of performance of professional (years) weren’t risk factors of CPE colonization in our study. Hence, the CPE detected in mothers in our study could belong to the community where they have lived. For example, multidrug-resistant organisms can spread to members of the same household. Researchers also reported that ESBL carriers can transmit genetically related ESBL-producing bacteria to their family members, with a prevalence from 17%-32%.20-25
Source of CPE colonization in neonatals
In Table 6, fecal sample collection in the neonatal colonized with CPE was from 31.00 to 47.00 hours after delivery. This result showed that CPE colonization in neonates could originate from potential contamination between the hospital environment, healthcare practices, and the neonates.
CPE transmission between mothers and neonates
The result of evidence of CPE transmission, shown in Figure 2, suggested that CPE transmission between this mother and her baby might be associated with the vaginal canal, which supplied an environment suitable for developing the colonization of pathogenic bacteria, which might move ascendingly into the uterus of mothers after the event of the premature rupture of the amniotic membrane. The result of this event was the contamination of amniotic fluid. Consequently, if the neonate inhaled or swallowed the infected amniotic fluid, pathogenic bacteria would colonize in their gut. Then, they would cause intrapartum sepsis, as studies reported K. pneumoniae found in the neonate’s gut is the principal pathogen of intrapartum sepsis.26-28 Although the CPE transmission prevalence was very low in our study, it confirmed that the CPE transmission issue should be a concern in the future due to the pressure of antibiotic resistance caused by CPE as a problem in developing countries.29 For example, a study in Algeria conducted on all mothers and their newborns in two maternity units, including Bejaia (Maternity A) and northern Algeria (Maternity B) in Tizi Ouzou, was prospective and randomly recruited from 357 mothers and 365 newborns from January 1 to April 30, 2017. This study reported a mother-newborn pair carrying asymptomatic OXA-48-producing E. coli and K. pneumoniae isolates.30 The researchers in Algeria also suggested that the CPE of mothers could contaminate their neonates, or neonates’ CPE could be from other sources.30
Risk factors of CPE colonization in mothers
The results of the univariate binary logistic regression associated with Risk factors of CPE colonization in mothers suggested the question was whether the time from labor induction to fecal sample collection with the rectal swab over 48 hours influenced CPE contamination in mothers. This question is essential and valuable in controlling and preventing CPE transmission or contamination in mothers because it will be a new direction in determining the factors related to CPE contamination or transmission for mothers spending hospital stays for delivery because in our study, one positive factor influencing CPE colonization was the mothers’ age from 19-23 years old, which reduced the CPE colonization compared with the age of mothers, less than or equal to 18 years old, with P = 0.033, OR = 0.067, 95% CI: 0.006-0.802 (Table 2).
One significant detail we analyzed above, the associations between the duration of time from labor induction to fecal sample collection over 36 hours was the risk factor of CPE colonization at 42.90% (n = 7), and one impressing finding is that mothers, who consumed antibiotics (less than two doses or more than three doses), and stayed in the hospital for over 48 hours before fecal samples were collected, they would acquire CPE colonization (Table 7).
Table (7):
Characteristics associated between labor induction in mothers and the length of time from labor induction to sample collection over 36 hours
| Characteristics | Freq. | Percent |
|---|---|---|
|
||
| no dose | 1 | 14.3 |
| ≤2 doses | 5 | 71.4 |
| ≥3 doses | 1 | 14.3 |
| 2. Prevalence of CPE | ||
| Non-CPE | 4 | 57.1 |
| CPE | 3 | 42.9 |
| 3. Length of time from labor induction to sample collection | ||
| >36-48 hrs | 2 | 28.6 |
| >48 hrs | 5 | 71.4 |
| 4. Association between 2 and 3 | ||
| Length of time from labor induction to sample collection | Freq of CPE | |
| >36-48 hrs | 1 | 100 |
| >48 hrs | 2 | 100 |
| 5. Association between 1 and 2 | ||
| Antibiotic use | Freq of CPE | |
| Yes (≤2 or ≥3 doses) | 3 | 100 |
| 6. Length of time of Hospital stay Before Sample collection | ||
| >48 hrs | 7 | 100 |
The result of the multivariate binary logistic regression of risk factors of CPE colonization in mothers noted that the mothers had a history of antibiotic use before study participation, with the total number of antibiotic doses (1-2 doses) in the period of hospital stay being the principal risk factor of CPE colonization compared with mothers without antibiotic use.31 This result showed that maternal CPE colonization could happen before hospital admission. CPE colonization could appear in the community before pregnant women’s admission to Hung Vuong Hospital. The origin of maternal CPE colonization could be the antibiotic overuse in the community, where over-the-counter antibiotics can be bought at pharmacies without a doctor’s prescription, as some research reported in Vietnam.32
The overuse of antibiotics in livestock has been a significant challenge in controlling multidrug-resistant organisms in Vietnam, as evidenced by the presence of antibiotic residues in food, such as pork and chicken meat.33-35
Antibiotic residue could be one of the causes of bacterial antibiotic resistance in the human gastrointestinal tract, leading to the development of multi-resistant organisms.36 Hence, maternal CPE colonization could be present in the maternal gastrointestinal tract before the time of pregnant women’s hospital admission.
Risk factors of CPE colonization in neonates
The results of the univariate binary logistic regression of risk factors of CPE colonization in neonates showed that admission to the Neonatal Intensive Care Unit before the fecal sample collection performed on neonates was a risk factor for CPE colonization, as shown above (Table 4). The Neonatal Intensive Care Unit was a potential source of CPE contamination. At a Portuguese NICU, a prospective, observational, longitudinal, and cohort study reported that the prevalence of CPE isolates detected in 173 admitted neonates was 5.8%, including the most frequent carbapenemase-producing Klebsiella pneumoniae isolates.37 Next, another study in a neonatal intensive care unit in Morocco reported that during hospitalization, 12.5% (26/207) of them acquired CPE that exclusively expressed the blaOXA-48 gene.38 The study results, as described above, including our findings, showed neonates might acquire CPE colonization through the cross-infection route in which CPE-colonization source can present in HCWs, neonates’ relatives, or in hospital environments, and not in mothers because the from-mother-to-neonate transmission prevalence of 0.28% (1/359) (E. coli) was very low. It meant that NICU-admitted neonates, before the fecal sample collection, were at risk of being colonized by CPE more than other neonates without a NICU admission history. Hence, the NICU admission was a potential risk factor for CPE colonization in our study.
Impressively, our study result also showed that CPE colonization detected in neonates had a history of NICU admission for over 17 hours, while A retrospective case-control study conducted by matching pairs based on the date of birth and gestational age to determine the ESBL-colonized infants at the NICU of Soroka University Medical Center, Israel from 2013-2014 proved the mean time from admission to ESBL-producing-bacteria colonization was 15 days.39 Our study results suggested a potential CPE contamination occurred between HCW, or the hospital environment, and neonates in the NICU. The former studies reported that the areas used for CPE patients to stay in the hospital were significantly contaminated.37,38,40 Hence, we should collect neonates’ fecal samples at the beginning of NICU admission to detect early CPE colonization in neonates admitted to NICU and have a plan to prevent and control CPE colonization in neonates during hospital stay. Significantly, as reported in our results (Table 4), vaginal examination was associated with CPE colonization in neonates. Our study showed that the number of vaginal examinations from 6-9 was a risk factor for CPE colonization, as shown in Table 4. The risk of bacterial colonization in the first vaginal examination in pregnant women was 0.34 times lower than that in more than one vaginal examination in women (OR 0.34; 95% CI 0.241, 0.481).41
This result implied that good medical practices have become a crucial standard in preventing CPE colonization/transmission from HCWs or the hospital environment to mothers in healthcare settings. For example, some studies reported that the transmission from HCWs was more significant in the areas where the nosocomial outbreak appeared.42 Two prospective studies determined the HCW-to-patient transmission route of the unique MRSA clones. Another study demonstrated that the HCWs’ hands were the source of S. aureus transmission from patient to patient, reported in three US academic medical centers.43,44
Next, A significant and impressive result is that the benefit of breastfeeding in our study was to reduce CPE colonization in neonates. A few studies showed this benefit. For example, trials reported that breastfeeding is beneficial for children because breastfed children had a lower frequency of antibiotic use than unbreastfed children. There was a strong relationship between the duration of breastfeeding and the frequency of antibiotic use in children.45,46 Moreover, breastfeeding was a beneficial factor in preventing acquisition of ESBL-producing Enterobacteriaceae, with an adjusted hazard ratio of 0.29 (95% CI, 0.11, 0.80).47
Another question is how the risk factors influenced each other. It is impressive that we analyzed the result of multivariate binary logistic regression of risk factors related to CPE colonization in neonates (Table 5). We found that the number of vaginal examinations from 6-10 times was the principal risk of CPE colonization. Another study reported that the risk of bacterial colonization from the first vaginal examination in pregnant women was 0.34 times lower than that from more than one vaginal examination (OR 0.34; 95% CI 0.241, 0.481).43 This result suggested that limiting the number of vaginal examinations is the best solution to reduce CPE colonization in neonates.
Antibiotic consumption before fecal sample collection and the duration from labor induction to sample collection were risk factors for CPE colonization in pregnant women. At the same time, the beneficial characteristics for reducing CPE colonization in neonates included the vaginal delivery, mother caring for neonates, gestational age over 37 weeks, less than two relatives caring for neonates, two hours for skin-to-skin contact between mothers and breastfeeding, Especially, the interaction between two characteristics, including vaginal delivery, mother caring for neonates reduced the CPE colonization in neonates.
The findings of mother-to-neonate CPE transmission were significantly low. However, the trend of CPE transmission in the group of mothers and neonates, and CPE-colonized mothers targeting interventions with screening CPE colonization at the beginning of hospital admission is the best solution to reduce this cross-transmission in endemic settings, and hand hygiene compliance and aseptic medical practices are crucial standards to prevent and control CPE in the healthcare sector.
Ultimately, the use of over-the-counter antibiotics in the community and the misuse of antibiotics in livestock farming need to be closely monitored by government regulations. The Government opens communication channels to guide and educate the community on when to use antibiotics and how to use them when prescribed by a doctor. It shouldn’t use antibiotics as a means to promote livestock growth. These procedures will reduce bacterial antibiotic resistance, including multidrug-resistant organisms, CPE.
Limitation of study
As described, we did not collect the fecal samples of HCWs and relatives of neonates and environmental samples to determine the transmission between neonatal CPE colonization and HCWs, or neonates’ relatives or environment.
ACKNOWLEDGMENTS
The authors would like to thank the OUCRU in HCM, all doctors and registered nurses working at the Delivery, Post Delivery, Neonatal Department, all staff of the Microbiology Unit of the Laboratory Department, and especially Dr. Hoang Thi Diem Tuyet, Ph.D. Director of Hung Vuong Hospital for support and encouragement.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
NVK conceptualized the study. PTH contributed to preparing the questionnaires. TTTH, NTKN and HNP collected data. CTT, NVMH, JIC and TTBC performed the experiments. NMN and NVK analyzed the data. NVK wrote the manuscript. SB and PTH reviewed and revised the manuscript. All authors read and approved the final manuscript for publication.
FUNDING
This study was funded by the OUCRU in HCM and Hung Vuong Hospital to perform the microbiological tests associated with this study.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
This study was approved by the Board of Hospital Directors and the Ethics Committee of Hung Vuong Hospital.
INFORMED CONSENT
Written informed consent was obtained from the participants before enrolling in the study.
- Knothe H, Shah P, Krcmery V, Antal M, Mitsuhashi S. Transferable resistance to cefotaxime, cefoxitin, cefamandole and cefuroxime in clinical isolates of Klebsiella pneumoniae and Serratia marcescens. Infection. Nov-Dec 1983;11(6):315-317.
Crossref - Paterson DL, Bonomo RA. Extended-spectrum β-lactamases: a clinical update. Clin Microbiol Rev. Oct 2005;18(4):657-86.
Crossref - Meletis G. Carbapenem resistance: overview of the problem and future perspectives. Ther Adv Infect Dis. 2016;3(1):15-21.
Crossref - Temkin E, Adler A, Lerner A, Carmeli Y. Carbapenem-resistant Enterobacteriaceae: biology, epidemiology, and management. Ann N Y Acad Sci. 2014;1323:22-42.
Crossref - Lerner A, Adler A, Abu-Hanna J, Cohen Percia S, Kazma Matalon M, Carmeli Y. Spread of KPC-producing carbapenem-resistant Enterobacteriaceae: the importance of super-spreaders and rectal KPC concentration. Clin Microbiol Infect. 2015;21(5):470.e1-7.
Crossref - Stewardson AJ, Marimuthu K, Sengupta S, et al. Effect of carbapenem resistance on outcomes of bloodstream infection caused by Enterobacteriaceae in low-income and middle-income countries (PANORAMA): a multinational prospective cohort study. Lancet Infect Dis. 2019;19(6):601-610.
Crossref - Kumarasamy KK, Toleman MA, Walsh TR, et al. Emergence of a new antibiotic resistance mechanism in India, Pakistan, and the UK: a molecular, biological, and epidemiological study. Lancet Infect Dis. 2010;10(9):597-602.
Crossref - McGann P, Hang J, Clifford RJ, et al. Complete sequence of a novel 178-kilobase plasmid carrying blaNDM-1 in a Providencia stuartii strain isolated in Afghanistan. Antimicrob Agents Chemother. 2012;56(4):1673-9.
Crossref - Tischendorf J, de Avila RA, Safdar N. Risk of infection following colonization with carbapenem-resistant Enterobactericeae: A systematic review. Am J Infect Control. 2016;44(5):539-43.
Crossref - Navon-Venezia S, Chmelnitsky I, Leavitt A, Schwaber MJ, Schwartz D, Carmeli Y. Plasmid-mediated imipenem-hydrolyzing enzyme KPC-2 among multiple carbapenem-resistant Escherichia coli clones in Israel. Antimicrob Agents Chemother. 2006;50(9):3098-3101.
Crosref - Poirel L, Al Maskari Z, Al Rashdi F, Bernabeu S, Nordmann P. NDM-1-producing Klebsiella pneumoniae isolated in the Sultanate of Oman. J Antimicrob Chemother. 2011;66(2):304-6.
Crossref - Cho SH, Shin HH, Choi YH, Park MS, Lee BK. Enteric bacteria isolated from acute diarrheal patients in the Republic of Korea between the year 2004 and 2006. J Microbiol. 2008;46(3):325-330.
Crossref - Islam MA, Talukdar PK, Hoque A, et al. Emergence of multidrug-resistant NDM-1-producing Gram-negative bacteria in Bangladesh. Eur J Clin Microbiol Infect Dis. 2012;31(10):2593-600.
Crossref - Abstracts from the 5th International Conference on Prevention & Infection Control (ICPIC 2019). Antimicrob Resist Infect Control. 2019;8(1):148.
Crossref - Yamane T. Statistics: An Introductory Analysis. New York: Harper & Row. 1967.
- Zingg W, Soulake I, Baud D, et al. Management and investigation of a Serratia marcescens outbreak in a neonatal unit in Switzerland – the role of hand hygiene and whole genome sequencing. Antimicrob Resist Infect Control. 2017;6(1):125.
Crossref - Li X, Xu X, Yang X, et al. Risk factors for infection and/or colonisation with extended-spectrum β-lactamase-producing bacteria in the neonatal intensive care unit: a meta-analysis. Int J Antimicrob Agents. 2017;50(5)::622-628.
Crossref - Rock C, Thom KA, Masnick M, Johnson JK, Harris AD, Morgan DJ. Frequency of Klebsiella pneumoniae carbapenemase (KPC)-producing and non-KPC-producing Klebsiella species contamination of healthcare workers and the environment. Infect Control Hosp Epidemiol. 2014;35(4):426-429.
Crossref - Segagni Lusignani L, Presterl E, Zatorska B, Van den Nest M, Diab-Elschahawi M. Infection control and risk factors for acquisition of carbapenemase-producing Enterobacteriaceae. A 5 year (2011-2016) case-control study. Antimicrob Resist Infect Control. 2020;9(1):18.
Crossref - Hilty M, Betsch BY, Bogli-Stuber K, et al. Transmission dynamics of extended-spectrum β-lactamase-producing Enterobacteriaceae in the tertiary care hospital and the household setting. Clin Infect Dis. Oct 2012;55(7):967-75.
Crossref - Lohr IH, Rettedal S, Natוs OB, Naseer U, Oymar K, Sundsfjord A. Long-term faecal carriage in infants and intra-household transmission of CTX-M-15-producing Klebsiella pneumoniae following a nosocomial outbreak. J Antimicrob Chemother. 2013;68(5):1043-1048.
Crossref - Tande D, Boisrame-Gastrin S, M nck MR, et al. Intrafamilial transmission of extended-spectrum-β-lactamase-producing Escherichia coli and Salmonella enterica Babelsberg among the families of internationally adopted children. J Antimicrob Chemother. 2010;65(5):859-865.
Crossref - Valverde A, Grill F, Coque TM, et al. High rate of intestinal colonization with extended-spectrum-beta-lactamase-producing organisms in household contacts of infected community patients. J Clin Microbiol. 2008;46(8):2796-2799.
Crossref - Rodriguez-Bano J, Lopez-Cerero L, Navarro MD, Diaz de Alba P, Pascual A. Faecal carriage of extended-spectrum beta-lactamase-producing Escherichia coli: prevalence, risk factors and molecular epidemiology. J Antimicrob Chemother. 2008;62(5):1142-9.
Crossref - Lo WU, Ho PL, Chow KH, Lai EL, Yeung F, Chiu SS. Fecal carriage of CTXM type extended-spectrum beta-lactamase-producing organisms by children and their household contacts. J Infect. 2010;60(4):286-92.
Crossref - Das P, Singh AK, Pal T, Dasgupta S, Ramamurthy T, Basu S. Colonization of the gut with Gram-negative bacilli, its association with neonatal sepsis and its clinical relevance in a developing country. J Med Microbiol. 2011;60(11):1651-1660.
Crossref - Simonsen KA, Anderson-Berry AL, Delair SF, Davies HD. Early-onset neonatal sepsis. Clin Microbiol Rev. 2014;27(1):21-47.
Crossref - Chereau F, Herindrainy P, Garin B, et al. Colonization of extended-spectrum-β-lactamase- and NDM-1-producing Enterobacteriaceae among pregnant women in the community in a low-income country: a potential reservoir for transmission of multiresistant Enterobacteriaceae to neonates. Antimicrob Agents Chemother. 2015;59(6):3652-3655.
Crossref - Suwantarat N, Carroll KC. Epidemiology and molecular characterization of multidrug-resistant Gram-negative bacteria in Southeast Asia. Antimicrob Resist Infect Control. 2016;5:15.
Crossref - Mairi A, Touati A, Ait Bessai S, et al. Carbapenemase-producing Enterobacteriaceae among pregnant women and newborns in Algeria: Prevalence, molecular characterization, maternal-neonatal transmission, and risk factors for carriage. Am J Infect Control. 2019;47(1):105-108.
Crossref - Pano Pardo JR, Serrano Villar S, Ramos Ramos JC, Pintado V. Infections caused by carbapenemase-producing Enterobacteriaceae: risk factors, clinical features and prognosis. Enferm Infecc Microbiol Clin. 2014;32(Suppl 4):41-48.
Crossref - Do TA, Quan PB, Le TT, et al. Antibiotic dispensing without a prescription across community pharmacies: A simulated patient study. Explor Res Clin Soc Pharm. 2025;18:100590.
Crossref - Ngoc Do MH, Yamaguchi T, Okihashi M, et al. Screening of antibiotic residues in pork meat in Ho Chi Minh City, Vietnam, using a microbiological test kit and liquid chromatography/tandem mass spectrometry. Food Control. 2016;69:262-266.
Crossref - Hedges S, Pelligand L, Chen L, et al. Antimicrobial residues in meat from chickens in Northeast Vietnam: analytical validation and pilot study for sampling optimisation. J Consum Protect Food Saf. 2024;19(2):225-234.
Crossref - Luu QH, Nguyen TBT, Nguyen TLA, Do TTT, Dao THT, Padungtod P. Antibiotics use in fish and shrimp farms in Vietnam. Aquac Rep. 2021;20:100711.
Crossref - Van TTH, Moutafis G, Tran LT, Coloe PJ. Antibiotic resistance in food-borne bacterial contaminants in Vietnam. Appl Environ Microbiol. 2007;73(24):7906-11.
Crossref - De Geyter D, Blommaert L, Verbraeken N, et al. The sink as a potential source of transmission of carbapenemase-producing Enterobacteriaceae in the intensive care unit. Antimicrob Resist Infect Control. 2017;6:24.
Crossref - Yan Z, Zhou Y, Du M, et al. Prospective investigation of carbapenem-resistant Klebsiella pneumonia transmission among the staff, environment and patients in five major intensive care units, Beijing. J Hosp Infect. 2019;101(2):150-157.
Crossref - Leikin-Zach V, Shany E, Yitshak-Sade M, et al. Neonatal Risk Factors for Colonization with Extended-Spectrum Beta-Lactamase-Producing Bacteria in the Neonatal Intensive Care Unit. Isr Med Assoc J. 2018;20(5):286-290.
- Lerner A, Adler A, Abu-Hanna J, Meitus I, Navon-Venezia S, Carmeli Y. Environmental contamination by carbapenem-resistant Enterobacteriaceae. J Clin Microbiol. 2013;51(1):177-81.
Crossref - Elliyas S, Gaind R, Kanwal SK, Singh S, Arya S. Bacterial Colonization of Vagina in Indian Women During Labor and Its Association With Puerperal and Neonatal Sepsis: A Tertiary Hospital Study. Cureus. 2021;13(3):e13943.
Crossref - Danzmann L, Gastmeier P, Schwab F, Vonberg R-P. Health care workers causing large nosocomial outbreaks: a systematic review. BMC Infect Dis. 2013;13(1):98.
Crossref - Wilson AP, Hayman S, Whitehouse T, et al. Importance of the environment for patient acquisition of methicillin-resistant Staphylococcus aureus in the intensive care unit: a baseline study. Crit Care Med. 2007;35(10):2275-2279.
Crossref - Ben-David D, Mermel LA, Parenteau S. Methicillin-resistant Staphylococcus aureus transmission: the possible importance of unrecognized health care worker carriage. Am J Infect Control. 2008;36(2):93-97.
Crossref - Davisse-Paturet C, Adel-Patient K, Divaret-Chauveau A, et al. Breastfeeding Status and Duration and Infections, Hospitalizations for Infections, and Antibiotic Use in the First Two Years of Life in the ELFE Cohort. Nutrients. 2019;11(7)
Crossref - Korpela K, Salonen A, Virta LJ, Kekkonen RA, de Vos WM. Association of Early-Life Antibiotic Use and Protective Effects of Breastfeeding: Role of the Intestinal Microbiota. JAMA Pediatr. 2016;170(8):750-757.
Crossref - Rodriguez-Revuelta MJ, Lopez-Cerero L, Serrano L, Luna-Lagares S, Pascual A, Rodriguez-Bano J. Incidence and Risk Factors for Acquisition of Extended-Spectrum β-Lactamase-Producing Enterobacteriaceae in Newborns in Seville, Spain: A Prospective Cohort Study. Int J Antimicrob Agents. 2018;52(6):835-841.
Crossref
© The Author(s) 2026. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.