ISSN: 0973-7510
E-ISSN: 2581-690X
Enterococci infections, have drawn attention of clinicians due to rapid increase in high level aminoglycoside resistance (HLAR). This resistance predicts failure of β-lactam antibiotic and aminoglycoside combination, which is the current treatment of choice for serious enterococci infections. This led us to investigate about the prevalence of HLAR enterococci in our hospital setting in Shimoga, Karnataka. All the clinical samples received at Microbiology laboratory, McGan hospital, Shivamogga between June 2016 and December 2016 was considered for the study. Enterococci were identified and further speciated by following conventional method. Antibiotic susceptibility pattern of all the isolates was determined according to CLSI guidelines. High level gentamicin resistance was detected by disc diffusion method using 120µg gentamicin disc. A total of 64 Enterococci species were isolated from the clinical specimens. On speciation, Enterococcus faecalis (E. faecalis) accounted for 72% (46) of the total isolates, 24%(16) of the isolates were Enterococcus faecium (E. faecium)and 4% (2) isolates were Enterococcus durans (E. durans). High level gentamicin resistance was noted in 42% (27) among the total isolates. E. faecium was found to be more resistant compared to E. faecalis. E. durans was a sensitive strain. Our study reconfirms the high prevalence of HLAR in the clinical setting. The study stresses on the need for regular screening of HLAR in all Enterococci infections and revise the battery of drugs for its treatment according to the report.
Enterococcus, Enterococcus faecalis, Enterococcus faecium, high level aminoglycoside resistance, Gentamicin
Enterococci, previously classified as group D Streptococci is now placed under a new family; Enterococcaceae. These Gram positive cocci reside in human intestine and are considered as normal bowel flora. Though they are considered to be less virulent than Streptococcus pyogenes, they are one of the most feared nosocomial pathogens isolated. Common nosocomial infections caused by this genus are urinary tract infections, intra- abdominal infections, peritonitis, bacteremia and endocarditis.1,2
Compared to other clinically important Gram positive cocci, this genus is intrinsically resistant to the antimicrobial agents commonly used in hospitals. In addition, Antibiotic resistance has been acquired, and disseminated via horizontal transfer of mobile genetic elements mediated mainly by conjugative plasmids. Though all acquired resistance of this organism is not significant clinically, high level aminoglycoside resistance greatly affects Enteroccoci therapy. This is because; severe enterococci infections are treated with combination of cell wall acting agent like vancomycin and an aminoglycoside. And high level aminoglycoside resistance predicts resistance to this combination therapy.1,3,4
This resistance pattern seen in genus Enterococci has become an issue of concern since decades but remains so even now. Hence this study was conducted to investigate the same so as to design the guidelines regarding measures to prevent spread of the resistant strains in the hospital and also to prevent the emergence of vancomycin resistance which is the treatment of choice for HLAR Enterococci.
This cross-sectional study was carried out in the Microbiology laboratory, McGan hospital, Shivamogga, between June 2016 and December 2016. The study was approved by the Institutional Ethical Committee of Shimoga Institute of Medical Sciences, Shivamogga.
All the pus, urine, blood and sterile body fluid samples received in our laboratory during the study period was included for the study. Faecal samples, and samples which were leaked were excluded.
The pathogen isolated by growth on blood agar after 24 hour aerobic incubation at 37°C was identified as enterococci species by Gram’s staining, catalase test and bile esculin hydrolysis test. However, for blood samples, subculture for 7 days was done before reporting negative. Further, speciation was done by arginine decarboxylation, pyruvate utilization, and fermentation of arabinose, raffinose, mannitol and ribose tests.5,6
Susceptibility pattern of the isolated enterococci species were carried out according to CLSI guidelines using disc diffusion method. Detection of High level gentamicin resistance was performed using120µg gentamicin antibiotic disc (Himedia).7
In HLGR detection by disc diffusion method, zone of inhibition of 6 mm was considered as resistant, 7–9 mm as inconclusive and ≥ 10 mm as Susceptible.7
A total of 64 enterococci species were isolated during our seven month study period. Out of these 64 isolates, 27 were from pus, 21 from urine and 16 from blood samples.
On speciation, Enterococcus faecalis accounted for 72% (46) of the total isolates, 24% (16) of the isolates were Enterococcus faecium and 4% (2) isolates were Enterococcus durans. Distribution of these three enterococci species in various clinical samples is documented in Table 1.
Table (1):
Sample wise distribution of different Enterococci species.
Sample |
E. faecalis |
E.faecium |
E.durans |
Total |
|---|---|---|---|---|
Pus |
16 |
09 |
02 |
27 |
Urine |
18 |
03 |
00 |
21 |
Blood |
12 |
04 |
00 |
16 |
Total |
46 |
16 |
02 |
64 |
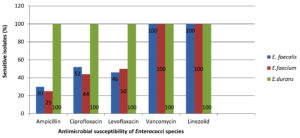
The antibiotic sensitivity pattern of the isolates is documented in Table 2. Species wise resistance pattern to various antibiotics is depicted in Fig. 1. For urinary isolates, apart from the antibiotics listed in the table, nitrofurantoin (300µg) and norfloxacin (10µg) antibiotic discs were used. And out of 21 urinary isolates, 9 (43%) were resistant to nitrofurantoin and 13 (62%) were resistant to norfloxacin.
Table (2):
Antibiotic sensitivity pattern of Enterococci isolates.
Antibiotics |
Sensitivity (n=64) |
|---|---|
Ampicillin |
20 (31%) |
Ciprofloxacin |
33(52%) |
Levofloxacin |
31(48%) |
Vancomycin |
64(100%) |
Linezolid |
64(100%) |
High level gentamicin |
37(58%) |
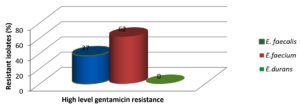
High level gentamicin resistance was noted in 27 (42%) isolates. Number of HLGR noted in different enterococci species is depicted in Fig. 2.
Genus Enterococci, once neglected group; considering them as commensals have gained great attention these days due to high rate of severe nosocomial infections caused by them. Two most common species isolated from clinical samples are E. faecalis and E. faecium. These are the organisms of low pathogenicity but with a property of inborn resistance to various antibiotics like relative resistance to most of the β-lactam antibiotics because of low affinity penicillin binding proteins (PBP). Enterococci also shows tolerance to penicillin i.e., penicillin acts only as bacteriostatic. Enterococci also show intrinsic low level resistance to aminoglycosides due to lack of oxidative uptake of the drug.8,9
However, serious enterococci infections like bacterial endocarditis are treated with combination of β-lactam antibiotic and an aminoglycoside. To a bacteriostatic beta lactam, the addition of aminoglycoside results in synergistic, bactericidal effect and beta lactam antibiotic indeed enhances the uptake of aminoglycoside resulting in synergistic killing of the organism. Aminoglycoside warrantied for usage in this combination are only gentamicin and streptomycin.
But the current detriment for the effective treatment is emergence of High level aminoglycoside resistance. It is defined by growth at concentration of 2000mg/L and 500 mg/L of streptomycin and gentamicin respectively on brain heart infusion agar. Presence of this high level resistance abolishes the synergistic effect of these agents. HLR to gentamicin is mainly due to aminoglycoside modifying enzyme and that to streptomycin is because of mutation in 30S ribosomal subunit and presence of streptomycin adenyltransferase.8,9
In our study, E. faecalis was recovered the most, followed by E. faecium. Both these species have shown highest resistance to ampicillin (69%) followed by levofloxacin (52%). Many studies in India have reported resistance to penicillin in the range of 40-80 per cent.10-12
It is glad to note that no vancomycin resistant enterococci (VRE) were isolated in our setting. And similarly, all the isolates were sensitive to linezolid which was consistent with other studies from India.13-15 However, the prevalence of vancomycin resistant Enterococci (VRE) in India is reported to be between 0% and 30%.16-18
High level gentamicin resistance was noted in 42% of the isolates and E. faecium was found to be more resistant (62% of the total E. faecium isolated) compared to E. faecalis ( 37% of the total E. faecalis isolated). E. durans was found to be a sensitive strain.
Our study report is almost in line with that of various studies carried out in different parts of our country like study by Dadfarna N et al.,19 who have reported 43.7% HLAR, Barman et al.,20 who have reported 56.7% HLAR, Jain S et al.,15 and Fernandes et al.,18 who have noted 54% and 53% HLAR in their studies respectively.
Alternative treatment options for serious HLAR Enterococci infections
Vancomycin is the drug of choice in these cases of HLAR Enterococci infections. But however resistance to vancomycin has also been noticed since 1986. The best available evidence suggests that the emergence and spread of these pathogens are promoted by poor infection control techniques and by antibiotic selective pressure. Antibiotic selective pressure favouring the emergence and spread of Vancomycin Resistant Enterococci(VRE) may involve more than simply the extent of vancomycin use. Specifically, extended-spectrum cephalosporins and similarly active beta-lactams and drugs with potent activity against anaerobes appear to predispose to Vancomycin resistant Enterococci colonization and infection. Hence this should be taken care of by following strict antibiogram policy in all medical setup.21
Daptomycin is the drug of choice in VRE. Daptomycin, a cyclic lipopeptide has a unique mechanism of action that results in destruction of the membrane potential.22 Daptomycin – Ceftarolin combination is opted in daptomycin non susceptible strains as ceftaroline is proved to restore daptomycin activity by promoting its surface binding.23
Quinupristin-Dalfopristin, a protein synthesis inhibitor is the drug of choice in HLAR, VR E. faecium infections. However, it has no action in E. faecalis infections due to intrinsic resistance.21
Linezolid, an oxazolidinone antibiotic with broad spectrum activity against Gram-positive bacteria, that inhibits protein synthesis is not used in serious HLAR Enterococcal infections like endocarditis inspite of its low resistance rate due to its bacteriostatic nature.21
Increased prevalence of HLAR has become a problem of reality. Combination of β- lactam antibiotics and an aminoglycoside, which stood first among the choice for the treatment of serious enterococci infection, can no more help in killing these pathogens. This is a grave development and the fact that enterococci have great potential for the dissemination of acquired resistant gene is a matter to be considered seriously. A must screening for HLAR using gentamicin and/or streptomycin by disk diffusion/agar dilution /broth microdilution should be practiced for all serious enterococci infections. Judicious use of Vancomycin, the drug of choice for HLAR enterococci infections is also advisable. Along with these, strict containment measures, adherence to hospital infection control policy to prevent their dissemination should be taken up in all clinical settings.
ACKNOWLEDGMENTS
None.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
None.
ETHICS STATEMENT
This article does not contain any studies with human participants or animals performed by any of the authors.
AVAILABILITY OF DATA
All datasets generated or analyzed during this study are included in the manuscript.
- Title M. Streptococcus, Enterococcus and other similar organisms. In. “Bailey and Scott’s Diagnostic Microbiology”. 13th Edition. New York. Elsevier publisher. 2014; 251-52.
- Emaneini M, Khoramian B, Jabalameli F, et al. Prevalence of high-level gentamicin-resistant in Enterococcus faecalis and Enterococcus faecium in an Iranian hospital. J Prev Med Hyg. 2016;57(4):E197-E200. PMCID: PMC5289030
- Gladwin M, Trattler W, Mahan SC. Streptococcus. In. “Clinical Microbiology made ridiculously simple.” 6th Edition. Jaypee Brothers Medical Publishers. 2014;27-28.
- Forwad KR, Kennedy J, Degagne PA, Bartlett K, Harding GKM. The rapid emergence of high level gentamicin resistance in enterococci. Can J Infect Dis. 1990;1(3):97-100.
Crossref - Colee JG, Fraser AG, Marmion BP, Simmons A. Mackie and McCartney Practical Medical Microbiology. 14th ed. New York. Elsevier Publishers. 1999.
- Winn W, Allen S, Janda W, Koneman E, Procop G, Schreckenberger, et al. Koneman’s colour Atlas and Textbook of Diagnostic Microbiology. 6th ed. Philadelphia: Lippincott Williams and Wilkins publishers. 2006.
- Clinical and Laboratory Standard Institute. Performance Standard for Antimicrobial Susceptibility Testing. Twenty Fifth Informational Supplement. M 100- S 26 .Wanye, PA, USA. CLSI. 2015.
- Arias CA and Murray BE. Enterococcus species, Streptococcus gallolyticus and Leuconostoc species. In. Mandell, Douglas and Bennett’s “Principle and practice of infectious diseases”. 8th Edition. Philadelphia. Elsevier publishers. 2015;2328-2339.
Crossref - Marothi YA, Agnihotri H, Dubey D. Enterococcal resistance- An overview. Indian J Med Microbiol. 2005;23(4):214-219.
Crossref - Jada S, Jayakumar K. Prevalence of Enterococcus species from various clinical specimens in Shri Sathya Sai Medical College and Research institute with special reference to speciation & their resistance to vancomycin. Int J Med Sci Clin Res. 2012;3(4):154-160.
Crossref - Mulla S, Patel KG, Panwala T, Rewadiwala S. Prevalence of enterococci with higher resistance level in a tertiary care hospital: a matter of concern. Natl J Med Res. 2012;2(1):25-27.
- Sreeja S, Babu PRS, Prathab AG. The prevalence and the characterization of the Enterococcus species from various clinical samples in a tertiary care hospital. J Clin Diagn Res. 2012;6(9):1486-1488.
Crossref - Sekar R, Srivani R, Vignesh R, Kownhar H, Shankar EM. Low recovery rates of high-level aminoglycoside-resistant Enterococci could be attributable to restricted usage of aminoglycosides in Indian settings. J Med Microbiol. 2008;57(3):397-398.
Crossref - Chakraborty A, Pal NK, Sarkar S, Gupta MS. Antibiotic resistance pattern of Enterococci isolates from nosocomial infections in a tertiary care hospital in Eastern India. J Nat Sci Biol Med. 2015;6(2):394-397.
Crossref - Jain S, Kumar A, Kashyap B, Kaur IR. Clinico-epidemiological profile and high-level aminoglycoside resistance in Enterococcal septicemia from a tertiary care hospital in east Delhi. Int J Appl Basic Med Res. 2011;1(2):80-83.
Crossref - Agarwal J, Kalyan R, Singh M. High-level aminoglycoside resistance and beta-lactamase production in Enterococci at a tertiary care hospital in India. Jpn J Infect Dis. 2009;62:158-159.
- Taneja N, Rani P, Emmanuel R, Sharma M. Significance of vancomycin resistant Enterococci from urinary specimens at a tertiary care centre in northern India. Indian J Med Res. 2004;119:72-74.
- Fernandes SC, Dhanashree B. Drug resistance & virulence determinants in clinical isolates of Enterococcus species. Indian J Med Res. 2013;137:981-985.
- Dadfarma N, Imani Fooladi AA, Oskoui M, Hosseini HM. High level of gentamicin resistance (HLGR) among enterococcus strains isolated from clinical specimens J Infect Public Health. 2013;6(3):202-208.
Crossref - Barman J, Nath R, Saikia L. Drug resistance in Enterococcus species in a tertiary level hospital in Assam, India. Indian J Med Res. 2016;143(1):107-110.
Crossref - Nigo M, Munita JM, Arias CA, Murray BE. What’s New in the Treatment of Enterococcal Endocarditis? Curr Infect Dis Rep. 2014;16(10):431-447.
Crossref - Steenberg JN, Alder J, Thorne GM, Tally FP. Daptomycin: a lipopeptide antibiotic for the treatment of serious Gram-positive infections. J Antimicrob Chemother. 2005;55(3):283-288.
Crossref - Sakoulas G, Rose W, Nonejuie P, et al. Ceftaroline restores daptomycin activity against daptomycin- nonsusceptible vancomycin resistant Enterococcus faecium. Antimicrob Agents Chemother. 2014;58(3):1494-1500.
Crossref
© The Author(s) 2022. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.