ISSN: 0973-7510
E-ISSN: 2581-690X
Urinary tract infection (UTI) in the community is a common infection in humans. The causative microorganisms for UTIs may vary in different places and also vary in their antibiotic sensitivity and resistant patterns. This study aimed to find out the incidence of gram-negative bacteria along with their antibiotic sensitivity pattern in community-acquired UTIs and to calculate the existence of Extended-spectrum beta-lactamase (ESBL) positivity among them. A Sum of 515 urine samples was tested and the causative organisms were identified. Antibiotic susceptibility tests were executed where it had shown growth for bacteria by both manual and automated methods. ESBL tests were performed to identify ESBL-producing isolates. From a total of 515 samples, 65 (12.65%) were culture positive for UTIs. The rate of infection was higher in females than in males. The most prevailing bacteria were Escherichia coli and Klebsiella pneumoniae. Out of total culture-positive isolates, only 43.08% were ESBL positives. The study found that the organisms displayed resistance to Ciprofloxacin and Cefixime and sensitivity to Gentamicin, Ertapenem, and Nitrofurantoin. Females have a higher prevalence of UTIs than males. Mostly, females suffer from UTIs in their reproductive years whereas males suffer in their old age. The prevalence of gram-negative bacteria is higher in community-acquired UTIs. These pathogens though present in the community, becoming resistant to commonly used antibiotics like Ciprofloxacin and Cefixime because of overuse and misuse of antibiotics at the community level.
ESBL, UTI, Gram-negative Bacteria, Antibiotic Sensitivity Pattern
The growth of microorganisms in the urinary tract leads to UTIs. The infection may be symptomatic or asymptomatic. Though it can be harmless in most cases, sometimes it can cause death in extreme conditions. Different studies have shown urinary tract infections to be the most regular infection caused by bacteria. Infections of the bladder area are termed cystitis and infections of the kidney are called pyelonephritis.1 Urinary tract infection rate is more in women, especially in pregnancy, and the elderly above the age of 60 years. Females are more susceptible to UTIs due to their reproductive physiology. The female urethra is shorter than the male which is 1.5-2 inches and 8 inches respectively. Sexually active women are the primary victim of this infection, especially after sexual intercourse.2 The cases of acute pyelonephritis occurrence are 30% in reproductive age women.3 It is also common in patients using catheters, which results in 40% of hospital-acquired infections.2 UTI is also the most usual form of Hospital Acquired Infection (HAI) consisting of up to 35% of nosocomial infections.4 Many factors are responsible for the prevalence of UTIs. Some of them like age, parity, immunosuppressants, geographical location, financial condition of the patient, etc. determine the occurrence and frequency of the infections.5 The transmission can occur through urine into hands, and possibly to others either through direct physical and sexual contact or via food or water.6 In most cases of community-acquired UTI, Escherichia coli is the prime cause of uncomplicated lower urinary tract infections. It is observed from epidemiologic studies that UTIs are majorly caused by E.coli, because of their heterogeneity at the genetic level.6 In addition, the other microorganisms, Klebsiella, Proteus, Pseudomonas, Enterobacter, and Gram-positive cocci like Staphylococcus, are associated with UTI.7 In the urinary tract, UPEC (Uropathogenic E.Coli) invasion is also more successful because of biofilm formation. There is not any test which can differentiate whether an Escherichia coli is Uropathogenic or not unless it is detected from urine. These pathogens’ multidrug resistance (MDR) genes make them easier to cause infections.8 UPEC are so adapted that they help them maintain colonization in the urinary tract and grow into large numbers exceeding 100,000 CFU/ml. In its treatment, bacterial quantification is used as a constituent of diagnosis and treatment. Normally, a count of >105 CFU is considered positive for UTI.9 Antibiotic resistance is one of the major problems, which is also a problem in UTIs, and is often a result of misuse of antibiotics and empirical treatments.10 UTIs can be overdiagnosed and over-treated because of a variety of reasons like specificity and sensitivity being low for prognosticating of bacteria in the urine. Also, the self-limiting behavior of uncomplicated UTIs is a problem because culture and sensitivity tests are not done for unsophisticated UTIs.11 Since 2000, the main cause of community-acquired UTI has been strains of bacteria producing extended-spectrum β-lactamase (ESBL), that inactivate common antibiotics like penicillins and cephalosporins.12,13 Since these strains are not detected in routine urine culture tests, it is important to know their antibiotics resistance patterns. The purpose of this study was to find out the frequency of gram-negative bacteria along with their antibiotic susceptibility pattern in community-acquired UTIs and to determine the prevalence of ESBL positivity among those bacteria.
The research work was performed over a time of 6 months from January 2021 to June 2021. During this period, 515 numbers of patients were included.
Sample Collection
Inclusion Criteria
All outpatients having symptoms of UTI.
Exclusion Criteria
- All indoor patients.
- Pregnant women.
- Neonates.
- Patients with Hospital-acquired UTI.
Mid-stream flow by clean catch technique was practiced for urine sample collection.
Culture of Urine Specimens
After collection, the urine was cultured in CLED (Cystine-Lactose electrolyte deficient) agar. The urine was inoculated using a calibrated loop of 0.01 ml of urine. The CLED plates were placed in an incubator overnight at 37°C. After 48hrs of aerobic incubation, it was reported as no growth.
The CLED plates which showed microbial growth colonies were observed morphologically and gram staining of the colonies were done.
Depending on the gram staining technique following Biochemical tests are done.
- Catalase test
- Coagulase test
- Oxidase test
- Nitrate reduction test
- Indole test
- Voges Proskauer test
- Urease test
- Mannitol motility agar
- Citrate test
- TSI agar
- Methyl red test
The Antibiotic susceptibility test was done by using The Kirby-Bauer test.
Kirby-Bauer Disk Diffusion Susceptibility Test
The diameter of the disk used is 6mm and the Clinical and Laboratory Standards Institute (CLSI) recommends this disk should be spaced on the agar plate for 24mm from the center of one disk to another to inhibit the overlapping of inhibition zones.
There should be a maximum of 15 mins between the inoculation of the agar with the colony and the application of the drug disk on the surface of the media. After that, the antimicrobial agent diffuses into the surrounding medium.
When a critical cell mass of bacteria is reached the inhibitory activity of the antimicrobial agents overcomes and bacterial growth occurs. The point at which the critical cell mass is reached appears as a sharp circle with a margin of bacterial growth.
The concentration of diffused antimicrobial agent at this interface growing an inhibitor bacterium is known as the critical concentration and approximates the Minimum Inhibitory Concentrations (MICs) obtained in the dilution test.20
VITEK 2 system was used for identification and Antibiotic sensitivity testing.
Vitek 2 System
The standard instrument contains 64 wells for cards that have 1 to 6 concentrations and for 9 to 20 antimicrobial compounds although the system runs both gram-positive and gram-negative identification panels at the same time when the susceptibility testing is performed. The initial method of formation of bacterium inoculum and dilution of the organism is done manually but all the other steps are automated Vitek 2 takes an average set time of 1.5 mins per isolate each smart carrier station has a barcode reader and holds up to 15 cards. A memory chip in the cassette captures and transfers the scan data to the reader incubator unit. Robotics of the instrument moves the loaded cassette through the successive station for barcode reading, test dilution preparation, and card inoculation. Each card is read by an optical station every 15mins and the amount of light transmitted is inversely proportional to the amount of growth achieved. It uses computer-assisted analysis of growth in plastic cards to calculate MIC.20
For ESBL confirmation the following methods were done.
Phenotypic Confirmatory Disk Diffusion Method
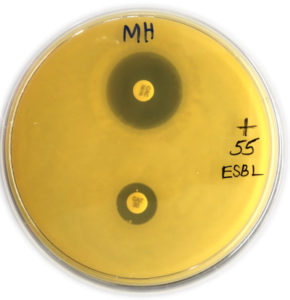
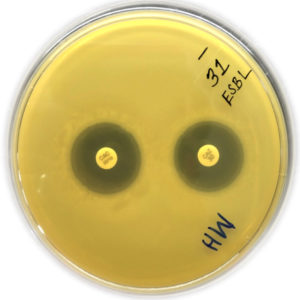
A combination of Ceftazidime (30µg) and Clavulanic acid (10 µg) with Ceftazidime (30 µg) was applied. After swabbing of culture over MHA plates these discs were placed on them and kept in an incubator. If there was a difference of 5 millimeters in the zone of inhibition of Ceftazidime and Ceftazidime-Clavulanic acid, it was observed as ESBL producing organism as shown in Figure 1 & 2.
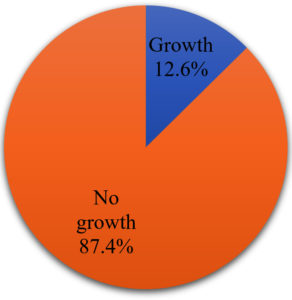
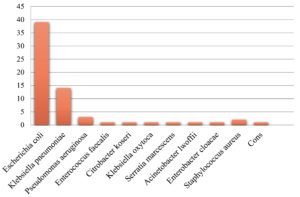
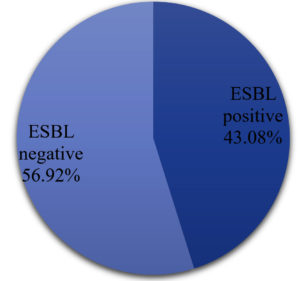
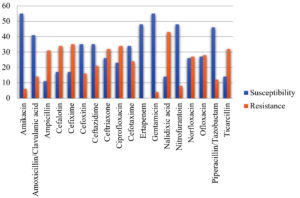
A Sum of 515 urine specimens was collected in the course of six months from January 2021 to June 2021. Out of 515 patients, 302 (58.6%) were females and 213 (41.4%) were males. Out of the total samples, 65 (12.6%) samples showed bacterial growth of which 40 (61.54%) were females and 25 (38.46%) were males. The remaining 450 (87.4%) samples showed no bacterial growth as shown in Figure 3. In females, the majority of UTI cases found were between the age group of 20 to 30 yrs (9, 22.5%) and 30-40 yrs, (9, 22.5%). In males, most UTI cases found were between the age group of 50-60 years, (5, 20%) and 60-70 years, (6, 24%) as listed in Table. Major common identified pathogens were Escherichia coli 39 (60.00%), Klebsiella pneumoniae 14 (21.54%), and Pseudomonas aeruginosa 3 (4.61%) as shown in Figure 4. Out of the 65 bacterial isolates, 28 (43.08%) isolates were ESBL positive and 37 (56.92%) were ESBL negative as shown in Figure 5. A high rate of resistance was observed against Nalidixic acid (69.35%), and ciprofloxacin followed by cefixime. High susceptibility was observed in Ertapenem (77.41%), Gentamicin followed by Nitrofurantoin as shown in Figure 6.
Table:
Age and Gender dispensation of UTI patients.
Age (In Years) |
Male |
Percentage % |
Female |
Percentage % |
|---|---|---|---|---|
0-10 |
2 |
8% |
1 |
2.5% |
10-20 |
0 |
0% |
0 |
0% |
20-30 |
1 |
4% |
9 |
22.5% |
30-40 |
6 |
24% |
9 |
22.5% |
40-50 |
1 |
4% |
7 |
17.5% |
50-60 |
5 |
20% |
6 |
15% |
60-70 |
6 |
24% |
6 |
15% |
70-80 |
4 |
16% |
2 |
5% |
Total |
25 |
100% |
40 |
100% |
The objective was to find out the incidence of gram-negative bacteria along with their antibiotic susceptibility pattern in community-acquired UTIs and to determine the prevalence of ESBL positivity among those bacteria. Female patients were major in age between 20-40yrs. In the case of males, patients were higher in the age above 60 years. Females suffer more from UTIs due to their shorter length of the urethra. Similar outcomes were found by S. Gupta et al. and SK Razak et al.14,15 Most predominant pathogen was found to be Escherichia coli and K.pneumoniae. Gram-negative bacteria were more prevalent than Gram-positive bacteria. The same kind of findings was observed in a study by Meena M et al. in which cases of E.coli, K.pneumoniae were higher at 37.2% and 10.2% respectively.16 The antibiogram chart of isolates revealed high resistance against nalidixic acid and ciprofloxacin followed by cefixime at the same time susceptible to ertapenem, gentamicin followed by nitrofurantoin. Higher Nitrofurantoin and amoxyclav sensitivity corresponded to a study by Vijay P Singh and Abhishek Mehta where nitrofurantoin was suggested for empirical treatment.17 Out of the total cases, ESBL producing isolates were 43.08% predominantly in E.coli and Kleb.pneumoniae. This was quite similar as observed through a research work by N.Kumar et al. where the presence of ESBL producers was 46.6% in uropathogens.18 These two organisms have been common pathogens for both community-acquired hospital-acquired (HA). The multidrug resistance (MDR) of ESBL-producing isolates has been a matter of concern. High resistance was observed against ceftazidime, ciprofloxacin, cefalotin and low resistance was observed against piperacillin/tazobactam, nitrofurantoin, amikacin. The results corresponded to a study by Chandrashekhar D. et al.19 Also factors like old age, previous antibiotics use, and pre-existing diseases like diabetes, lung diseases, maybe related and should be considered before empiric treatment.18 Resistance to common antibiotics hampers the antibiotic treatment against bacterial urinary tract infections. Therefore, it is important that early detection should be done so that suitable antibiotics can be prescribed. Misuse and overuse of wrong antibiotics further complicate the problems.
In this study period, out of 515 people tested for UTI, 65 were found positive for UTI, in which the ratio of the female patients was higher than the male patients. In females, the 20 – 40 years age group was most. In males, the greatest number of cases were observed above the age group of 50 years. The bacteria isolated from the urine were predominantly gram-negative and, in a few cases, gram-positive bacteria were isolated. Escherichia coli was the majority of UT infections accompanied by K.pneumoniae. Out of the total isolates, 43.08% showed ESBL positive. The antibiogram pattern disclosed the bacterial isolates were highly resistant to common antibiotics like ampicillin, ciprofloxacin, and nalidixic acid. The ESBL-positive bacterial isolates were resistant to most beta-lactam antibiotics like penicillins and cephalosporins. It showed resistance to cefalotin, cefixime, and ceftriaxone. Also, the bacterial isolates showed sensitivity to antibiotics like amikacin, ertapenem, and gentamicin. The ESBL-positive bacteria were sensitive to antibiotics like ceftazidime, clavulanic acid, and piperacillin/tazobactam. Antibiotic resistance in UTIs has been a problem for a long time and it still continues to be an issue. It leads to problems like higher medical costs, increased hospitalization durations, and higher mortality. Also, precautions can be taken to prevent UTIs through proper hygiene, safe sexual practices, and awareness. For the time being, it can be reduced by characterization of antibiotic resistance patterns in different areas, correct antibiotic prescription after culture and identification of bacteria in the urine, and proper diagnosis and treatments.
ACKNOWLEDGMENTS
The authors would like to thank Department of Microbiology, Institute of Medical Sciences & SUM Hospital, Bhubaneswar, Odisha, India for their support.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approve it for publication.
FUNDING
None.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
This article does not contain any studies with human participants or animals performed by any of the authors.
- Naboka YL, Mavzyiutov AR, Kogan MI, Gudima IA, Ivanov SN, Naber KG. Does Escherichia coli have pathogenic potential at a low level of bacteriuria in recurrent, uncomplicated urinary tract infection? Int J Antimicrob Agents. 2020;56(1):105983.
Crossref - Demilie T, Beyene G, Melaku S, Tsegaye W. Urinary bacterial profile and antibiotic susceptibility pattern among pregnant women in North West Ethiopia. Ethiop J Health Sci. 2012;22(2):121-128. PMCID: PMC3407835
- Smaill FM, Vazquez JC. Antibiotics for asymptomatic bacteriuria in pregnancy. Cochrane Database Syst Rev. 2019;(11):CD000490.
Crossref - Harding GK, Zhanel GG, Nicolle LE, Cheang M. Antimicrobial treatment in diabetic women with asymptomatic bacteriuria. N Engl J Med. 2002;347(20):1576-1583.
Crossref - Dimetry SR, El-Tokhy HM, Abdo NM, Ebrahim MA, Eissa M. Urinary tract infection and adverse outcome of pregnancy. J Egypt Public Health Assoc. 2007;82(3-4):203-218.
- Nerurkar A, Solanky P, Naik SS. Bacterial pathogens in urinary tract infection and antibiotic susceptibility pattern. Journal of Pharmaceutical and Biomedical Sciences. 2012;21(21).
Crossref - Kolawole AS, Kolawole OM, Kandaki-Olukemi YT, Babatunde SK, Durowade KA, Kolawole CF. Prevalence of urinary tract infections (UTI) among patients attending Dalhatu Araf Specialist Hospital, Lafia, Nasarawa state, Nigeria. International Journal of Medicine and Medical Sciences. 2010;1(5):163-167.
Crossref - Fatima N, Ishrat S. Frequency and risk factors of asymptomatic bacteriuria during pregnancy. J Coll Physicians Surg Pak. 2006;16(4):273-275. PMID: 16624191
- Nowicki B. Urinary tract infection in pregnant women: old dogmas and current concepts regarding pathogenesis. Curr Infect Dis Rep. 2002;4(6):529-535.
Crossref - Maki DG, Tambyah PA. Engineering out the risk for infection with urinary catheters. Emerg Infect Dis. 2001;7(2):342-347.
Crossref - Mulvey MA, Schilling JD, Martinez JJ, Hultgren SJ. Bad bugs and beleaguered bladders: interplay between uropathogenic Escherichia coli and innate host defenses. Proc Natl Acad Sci. 2000;97(16):8829-8835.
Crossref - Foxman B. The epidemiology of urinary tract infection. Nat Rev Urol. 2010;7(12):653-660.
Crossref - Peirano G, Richardson D, Nigrin J, et al. High prevalence of ST131 isolates producing CTX-M-15 and CTX-M-14 among extended-spectrum-β-lactamase-producing Escherichia coli isolates from Canada. Antimicrob Agents Chemother. 2010;54(3):1327-1330.
Crossref - Razak SK, Gurushantappa V. Bacteriology of urinary tract infection and antibiotic susceptibility pattern in a tertiary care hospital in South India. Int J Med Sci Public Health. 2012;1(2).
Crossref - Gupta K. Emerging antibiotic resistance in urinary tract pathogens. Infect Dis Clin. 2003;17(2):243-259.
Crossref - Meena M, Kishoria N, Meena DS, Sonwal VS. Bacteriological profile and antibiotic resistance in patients with urinary tract infection in tertiary care teaching hospital in western Rajasthan India. Infect Disord Drug Targets. 2021;21(2):257-261.
Crossref - Singh VP, Mehta A. Bacteriological profile of urinary tract infections at a tertiary care hospital in Western Uttar Pradesh, India. Int J Res Med Sci. 2017;5(5):2126-2129.
Crossref - Kumar N, Chatterjee K, Deka S, Shankar R, Kalita D. Increased Isolation of Extended-Spectrum Beta-Lactamase-Producing Escherichia coli From Community-Onset Urinary Tract Infection Cases in Uttarakhand, India. Cureus. 2021;13(3):13837.
Crossref - Chandrasekhar D, Dollychan A, Roy BM, Cholamughath S, Parambil JC. Prevalence and antibiotic utilization pattern of uropathogens causing community-acquired urinary tract infection in Kerala, India. J Basic Clin Physiol Pharmacol. 2018;29(6):671-677.
Crossref - Winn WC. Koneman’s color atlas and textbook of diagnostic microbiology. Lippincott williams & wilkins; 2006.
© The Author(s) 2022. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.