ISSN: 0973-7510
E-ISSN: 2581-690X
Legionella pneumophila is one of the most severe causative agents responsible for lower respiratory tract infections. Pathogenesis of Legionella diseases is mainly occurred due to the activities of some latent virulence factors. The present investigation was carried out to study the prevalence rate of L. pneumophila and its putative virulence factors in the respiratory samples of patients. Three-hundred and fifty respiratory samples were taken from patients suffered from RTIs. All samples were cultured and their positive results were subjected to several PCR reaction. Prevalence in patients suffered from RTIs were 11.53%. lidA (70.58%), ralF (23.52%) and ivhB (17.64%) were the most commonly detected virulence factors in the L. pneumophila recovered from the respiratory samples. Considerable presence of virulent strains of L. pneumophila in patients showed that important public health issue facing hospitalized patients.
Legionella pneumophila, Respiratory tract infections, Prevalence, Virulence factors.
Respiratory Tract Infections (RTIs) refers to any of a number of infectious diseases involving the respiratory tract. An infection of this type is normally further classified as an upper respiratory tract infection (URTI) or a lower respiratory tract infection (LRTI). Lower respiratory infections, such as pneumonia, tend to be far more serious conditions than upper respiratory infections, such as the common cold. These are one of the most common infectious diseases al-around the world with high rates of morbidity and mortality.1,2
One of the main pathogenic agent which are responsible for causing LTRI is Legionella species (spp.)2-5. Among all known species of Legionella, Legionella pneumophila (L. pneumophila) has the highest clinical impact and is a causative agent of various clinical complications like human legionellosis and community-acquired and nosocomial pneumonia2-5. L. pneumophila is motile, gram-negative, fastidious and aerobic bacilli, non-fermentative, heterotrophic, catalase-positive, and urease and nitrate negative bacterium2-5. Confusion, headache, fever, abdominal pain, diarrhea, non-productive cough chills, and myalgia are the main clinical symptoms of RTIs and pneumonia caused by L. pneumophila2-5.
Pathogenesis of RTIs and also pneumonia caused by this bacterium are often derived from the presence of some putative virulence factors. In the other hand, to apprise the pathogenesis of diseases caused by L. pneumophila, study the latent virulence factors is required. Among all latent virulence factors which were attributed in pathogenesis of L. pneumophila infections, the poreformation protein rtxA, a number of effectors such as ralF, lidA and lepA, secretion system is prepilin peptidase pilD dependent and Legionella vir homolog (lvh) (a type IV secretion system involved in conjugation).6-8
The present investigation was carried out in order to study the prevalence rate and distribution of virulence factors in the L. pneumophila strains isolated from samples taken from Iranian patients suffered from RTIs.
Samples collection
From January to November 2015, a total of 350 respiratory samples including Broncho Alveolar Lavages (BAL) (n=50) and also respiratory secretions (n=300) were sent to our laboratory center from hospitalized patients suffering from RTIs. In this study, a total of 150 respiratory samples were randomly selected and analyzed for presence of L. pneumophila.
Bacterial isolation
Prior to culture, samples were centrifuged for 15 min at 2,500rpm, and the top 7.5ml of the resulting suspension was removed. The remaining cell concentrate was mixed and used for culture. Aliquots of 100 µL of prepared samples were spread on duplicate plates of aBCYE selective medium Agar (Difco Laboratories, Detroit, Mich., USA) and to plates containing L-cysteine (0.44mg mL-1), ferric pyrophosphate (0.250 mg mL-1), glycine (3.0 gL-1), vancomycin (0.0025 mg mL-1) and polymyxin B (0.006 mgmL-1), which are named áBCYE-GVP selective agar medium. Plates were incubated at 37ºC in a humidified atmosphere without CO2 during 5 days. Colonies with the typical ground glass appearance of Legionella were sub cultured on two nonselective media, sheep-blood agar and áBCYE agar without L-cysteine. Colonies that grew on áBCYEGVP but not on non-selective media were considered putative Legionella strains, and were Gram stained and subcultured on a selective medium. The identification of putative Legionella strains as L. pneumophila was carried out using Legionella specific latex reagents (Oxoid, Hampshire, England) and direct immunofluorescence assay with poly clonal rabbit sera (m-Tech Alpharetta, Ga., USA).
PCR confirmation
L. pneumophila isolates were submitted to DNA extraction using the DNA extraction kit (Fermentas, Germany), according to the manufacturer’s instructions. Set of primers for lepA gene of the L. pneumophila was designed by Khedri et al. (2015) (9). The extracted DNA of each sample was kept frozen at -20°C until used. Primer sequences used for PCR, Legionella-F: 5′- GCTAATACCGCATAATGTCTGAGG-3′ and Legionella-R: GGTGCTTCTGTGGGTAACG-3′ (354 bp) were used for ramification of 16S ribosomal RNA (16S rRNA) gene of the L. pneumophila. PCR reactions were performed in a total volume of 25 µL, including 1.5 mM MgCl2, 50 mM KCl, 10 mM Tris-HCl (pH 9.0), 0.1% Triton X-100, 200 µM dNTPs each (Fermentas, Germany), 25 pmoL of each primer, 1.5 U of Taq DNA polymerase (Fermentas, Germany), and 3 µL (40-260 ng/µL) of DNA. The samples were placed in a thermal cycler (Mastercycler gradient, Eppendorf, Germany) with an initial denaturation step at 95°C for 5 min, then amplified for 30 cycles of denaturation at 94°C for 1 min, annealing at 62°C for1 min, extension at 72°C for 1 min and final extension step at 72°C for 5 min.
Amplification of virulence factors
Table 1 represents list of primers, PCR conditions and volume of each reaction used from amplification of virulence factors Khedri et al. (2015) (9). All runs were done in a DNA thermo-cycler (Mastercycler gradient, Eppendorf, Germany). The PCR amplification products (10 ìl) were subjected to electrophoresis in a 1% agarose gel in 1X TBE buffer at 80 V for 30 min, stained with ethidium bromide, and images were obtained in a UVIdoc gel documentation system (UK). The PCR products were identified by 100 bp DNA size marker (Fermentas, Germany). A DNA of L. pneumophila ATCC 33152 was used as positive control and DNA of a laboratory isolate strain of E. coli as negative control.
Table (1):
List of primers and PCR conditions used for amplification of putative virulence factors of L. pneumophila strains isolated from patients suffered from RTIs (9).
| Target gene | Primer sequence (5′-3′) | Size of product (bp) | PCR program | Volume of reaction (50 µL) |
|---|---|---|---|---|
| lidA |
F: CACAGGCTAAGGAAGACAGAGGC R: TAATTTCCTTGACTTTTTCCGCAG |
270 | 1 cycle: 940C -6min |
5 µL PCR buffer 10X 2 mM Mgcl2 150 µM dNTP (Fermentas) |
| 30 cycle: 950C -30s 640C -1min 720C -55s |
0.75 µM of each primers F & R 1.5 U Taq DNA polymerase (Fermentas) | |||
| 1 cycle: 720C – 7min |
3 µL DNA template | |||
| lepA |
F: GTTGGGCACTACAGTTATCTCTTC R: GTTAGTTACTACGGTTTCAATACGAC |
354 | 1 cycle: 950C -5min |
5 µL PCR buffer 10X 2 mM Mgcl2 150 µM dNTP (Fermentas) |
| 30 cycle: 940C -50s 590C -1min 720C -1min |
0.75 µM of each primers F & R 1.5 U Taq DNA polymerase (Fermentas) | |||
| 1 cycle: 720C -5min |
3 µL DNA template | |||
| ralF |
F: ACCAGCCCAGGATATGAACTTAC R: ATAGTAGCTTGTGCGGATGTTTTG |
230 | 1 cycle: 950C -5 min |
0.75 µM of each primers F & R 1.5 U Taq DNA polymerase (Fermentas) |
| 30 cycle: 940C -1 min 620C -1min 720C -55s |
0.75 µM of each primers F & R 1.5 U Taq DNA polymerase (Fermentas) 3 µL DNA template | |||
| 1 cycle: 720C -10 min |
3 µL DNA template | |||
| ivhB |
F: GTCAAACAACTTCATTCAAACACC R: GGCAATAAATTCACAATCCAGAG |
272 | 1 cycle: 95 0C -5min |
5 µL PCR buffer 10X 2 mM Mgcl2 150 µM dNTP (Fermentas) |
| 30 cycle: 940C -1min 600C -1min 720C -55s |
0.75 µM of each primers F & R 1.5 U Taq DNA polymerase (Fermentas) | |||
| 1 cycle: 720C -5min |
3 µL DNA template | |||
| rtxA |
F: ATTGCTTTTTCAGGTATCACTAACG R: ATTCGTTGATGTACTAATAGGCTGG |
265 | 1 cycle: 950C -5min |
5 µL PCR buffer 10X 2 mM Mgcl2 150 µM dNTP (Fermentas) |
| 30 cycle: 940C -1min 620C -1min 720C -1min |
0.75 µM of each primers F & R 1.5 U Taq DNA polymerase (Fermentas) | |||
| 1 cycle: 720C – 6min |
3 µL DNA template |
Statistical analysis
The data were analyzed using SPSS (Statistical Package for the Social Sciences) software and P values were calculated using Chi-square and Fisher’s exact tests to identify statistically significant relationships for the distribution of L. pneumophila and virulence factors. A P value < 0.05 was considered statistically significant.
A total of 150 samples were tested for presence of L. pneumophila and its putative virulence factors. Prevalence of L. pneumophila in patients of our study were 11.53%.
Table (2):
Total prevalence of Legionella pneumophila in the respiratory samples taken from patients suffered from RTIs.
Types of samples |
No. samples collected |
Prevalence of L. pneumophila (%) |
PCR confirmation (%) |
|---|---|---|---|
Patients |
150 |
17 (11.33) |
17 (11.33) |
Figure 1-4 represent the results of the gel electrophoresis for putative virulence factors of the L. pneumophila recovered from samples of patients suffered from RTIs. Table 3 represents the distribution of putative virulence factors in the L. pneumophila strains isolated from the samples of patients suffered from RTIs. All of the studied genes had the considerable prevalence in patients suffered from RTIs. We found that the most commonly detected virulence factors in the L. pneumophila recovered from the samples of studied samples were lidA (70.58%), ralF (23.52%) and ivhB (17.64%).
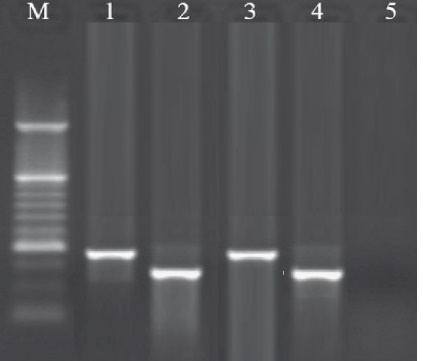
Fig. 1. Results of the gel electrophoresis for identification of ralF and lepA genes of the L. pneumophila strains. M: 100 bp DNA ladder (Fermentas, Germany), 1: Positive sample for lepA gene (354 bp). 2: Positive sample for ralF gene (230 bp), 3 and 4: Positive Control.
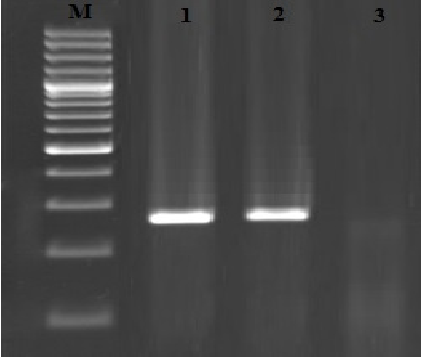
Fig. 2. Results of the gel electrophoresis for identification of lvhB gene of L. pneumophilain respiratory samples, M:100 bp DNA ladder (Fermentas, Germany), 1: Positive control, 2: Positive samples for lvhB gene (272 bp) and 3: Negative control.
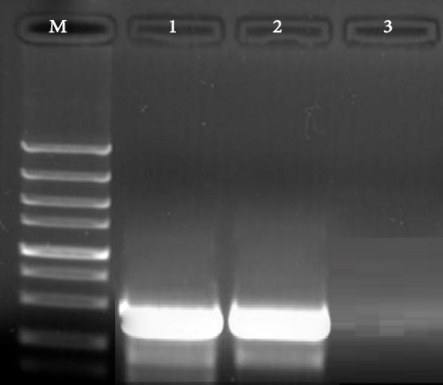
Fig. 3. Results of the gel electrophoresis for identification of lidA gene of L. pneumophila in respiratory samples, M:100 bp DNA ladder (Fermentas, Germany), 1: Positive control, 2: Positive samples for lidA gene (270 bp band) and 3: Negative control.
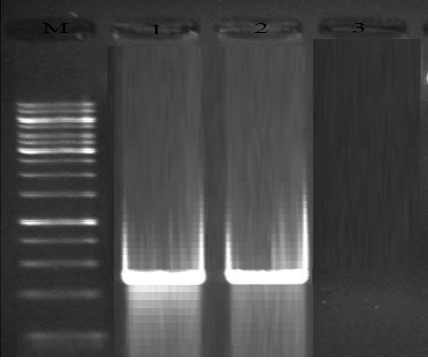
Fig. 4. Results of the gel electrophoresis for identification of rtxA gene of L. pneumophila in respiratory samples, M:100 bp DNA ladder (Fermentas, Germany), 1: Positive control, 2: Positive samples for rtxA gene (265 bp band) and 3: Negative control.
Table (3):
Distribution of putative virulence factors in the Legionella pneumophila strains isolated from the respiratory samples of patients suffered from RTIs.
| Types of samples (No. positive) | Distribution of putative virulence factors (%) | ||||
|---|---|---|---|---|---|
| lidA | lepA | ralF | ivhB | rtxA | |
| Patients (17) | 12 (70.58) | 2 (11.76) | 4 (23.52) | 3 (17.64) | 1 (5.88) |
This work was carried out to study the prevalence of L. pneumophila in the samples taken from patients suffered from RTIs. As far as we know, the present investigation is the first prevalence report of the L. pneumophila in samples taken from patients in Iran. As it showed, the prevalence of L. pneumophila in patients were 11.53%. Faradonbeh et al. (2015) (10) revealed that sex and age of patients and history of smoking were the main risk factors for occurrence of RTIs caused by L. pneumophila. They showed that the prevalence of L. pneumophila among samples was 12% which was higher than our results. Among all previous investigations which were conducted on the prevalence of L. pneumophila in clinical samples11-14, Ghotaslou et al. (2013) 11, Yu et al. (2008) (12) and Azara et al. (2006)13 reported that higher than 20% of samples were positive for L. pneumophila, while Chaudhry et al. (2000) 14 reported lower prevalence. Similar findings have been reported by Faradonbeh et al. (2015) (Iran)10, Ngeow et al. (2005) (Malaysia, Thailand, China, Philippines, Taiwan, South Korea, Singapore and Indonesia)15, Nagalingam et al. (2005) (Trinidad and Tobago)16 and Amemura-Maekawa et al. (2010) (Japan)17.
The second part of our investigation focused on the detection of putative virulence factors in the L. pneumophila strains of patients. As it showed, the most commonly detected virulence factors were lidA, ralF and ivhB. Similar findings were reported by Khedri et al. (2015)9. They showed that the prevalence of L. pneumophila was 12%. Total distribution of lepA, lidA, ralF, rtxA and ivhB virulence factors in the L. pneumophila strains of patients suffered from RTIs were 11.11%, 50%, 27.77%, 5.55% and 16.66%, respectively. Unfortunately, the numbers of investigations which were conducted on the distribution of putative virulence factors in L. pneumophila strains of clinical samples were low. In a study which was conducted on Australia by Huang et al. (2006)18, prevalence of rtxA and lvh genes in patients suffered from RTIs caused by L. pneumophila were 64.4% and 57.6%, respectively. High prevalence of lidA, ralF and lepA genes was also described previously by Newton et al. (2006)19, Gilmour. et al. (2007)20, Huang et al. (2006)18 and Khedri et al. (2015)9.
RtxA positive strains of L. pneumophila have a high ability to enter to monocytes and epithelial cells, and increased cytotoxicity and intracellular duplication21-23. Presence of lepA, lidA, ralF, rtxA and lvhB virulence genes is essential for the survival and growth of L. pneumophila in macrophages, for avoidance of phagosome acifidication and lysosome fusion and is necessary for instruction of apoptosis in human macrophages21-23.
In conclusion, we identified a large number of virulent strains of L. pneumophila in the samples of sick persons. Higher prevalence of bacteria in patients suffered from RTIs with respect to the high distribution of lepA, lidA, ralF, rtxA and lvhB virulence genes are two important findings of our study. Further studies need to be done to determine the exact roles of various virulence factors of L. pneumophila.
- Lieberman D, Lieberman D, Korsonsky I, Ben-Yaakov M, Lazarovich Z, Boldur I. Legionella species infection in adult febrile respiratory tract infections in the community. Scand J Infect Dis. 2002; 34(1):1-4.
- Maltezou HC, La-Scola B, Astra H, Constantopoulou I, Vlahou V, Kafetzis DA, Constantopoulos AG, Raoult D. Mycoplasma pneumoniae and Legionella pneumophila in community-acquired lower respiratory tract infections among hospitalized children: diagnosis by real time PCR. Scand J Infect Dis. 2004; 36(9):639-42.
- Viasus D, Di Yacovo S, Garcia-Vidal C, Verdaguer R, Manresa F, Dorca J, Gudiol F, Carratalà J. Community-acquired Legionella pneumophila pneumonia: a single-center experience with 214 hospitalized sporadic cases over 15 years. Medicine (Baltimore). 2013; 92(1):51-60.
- Darvishi M. Antibiotic Resistance Pattern of Uropathogenic Methicillin-resistant Staphylococcus aureus Isolated from Immunosuppressive Patients with Pyelonephritis. j pure appl microbio. 2016; 10(4):2663-2667.
- Carratalà J, Garcia-Vidal C. An update on Legionella. Curr Opin Infect Dis. 2010; 23(2):152-7.
- Newton HJ, Ang DK, van Driel IR, Hartland EL. Molecular pathogenesis of infections caused by Legionella pneumophila. Clin Microbiol Rev.2010; 23(2):274-98.
- Hacker J, Ott M, Wintermeyer E, Ludwig B, Fischer G. Analysis of virulence factors of Legionella pneumophila. Zentralbl Bakteriol. 1993 ; 278(2-3):348-58.
- Shevchuk O, Jäger J, Steinert M. Virulence properties of the legionella pneumophila cell envelope. Front Microbiol. 2011; 25; 2:74.
- Khedri F, Alaei Faradonbeh F, Eliyasi M, Barghi A, Doosti A, Emad P, Alaei Faradonbeh A. Molecular depiction of lepa, lida, ralf, rtxa and lvhb virulence factors of Legionella Pneumophila isolated from respiratory tract infections. InternatIonal archIves of MedIcIne 2015; 8: 1-11.
- Faradonbeh FA, Khedri F, Doosti A. Legionella pneumophila in Bronchoalveolar Lavage Samples of Patients Suffering from Severe Respiratory Infections: Role of Age, Sex and History of Smoking in the Prevalence of Bacterium. Srp Arh Celok Lek. 2015; 143(5-6):274-8.
- Ghotaslou R, Yeganeh Sefidan F, Akhi MT, Soroush MH, Hejazi MS. Detection of Legionella Contamination in Tabriz Hospitals by PCR Assay. Adv Pharm Bull. 2013; 3(1):131-4.
- Yu PY, Lin YE, Lin WR, Shih HY, Chuang YC, Ben RJ, et al. The high prevalence of Legionella pneumophila contamination in hospital potable water systems in Taiwan: implications for hospital infection control in Asia. Int J Infect Dis. 2008; 12(4):416-20.
- Azara A, Piana A, Sotgiu G, Dettori M, Grazia Deriu M, Masia MD, et al. Prevalence study of Legionella spp. contamination in ferries and cruise ships. BMC Publ Health. 2006; 6:100.
- Chaudhry R, Dhawan B, Dey AB. The incidence of Legionella pneumophila: a prospective study in a tertiary care hospital in India. Trop Doct. 2000; 30(4):197-200.
- Ngeow YF, Suwanjutha S, Chantarojanasriri T, Wang F, Saniel M, Alejandria M, Hsueh PR, Ping-Ing L, Park SC, Sohn JW, Aziah AM, Liu Y, Seto WH, Ngan CC, Hadiarto M, Hood A, Cheong YM. An Asian study on the prevalence of atypical respiratory pathogens in community-acquired pneumonia. Int J Infect Dis 2005, 9: 144-153.
- Nagalingam NA, Adesiyun AA, Swanston WH, Bartholomew M. Seroprevalence of Legionella pneumophila in Pneumonia Patients in Four Major Hospitals in Trinidad and Tobago. West Indian Med J 2005, 54: 375-378.
- Amemura-Maekawa J, Kura F, Helbig JH, Chang B, Kaneko A, Watanabe Y, Isobe J, Nukina M, Nakajima H, Kawano K,Tada Y, Watanabe H. Working Group for Legionella in Japan. Characterization of Legionella pneumophila isolates from patients in Japan according to serogroups, monoclonal antibody subgroups and sequence types. J Med Microbiol 2010, 59: 653659.
- Huang B, Yuan Z, Heron BA, Gray BR, Eglezos S, Bates JR, Savill J: Distribution of 19 major virulence genes in Legionella pneumophila serogroup 1 isolates from patients and water in Queensland, Australia. J Med Microbiol 2006, 55: 993-997.
- Newton HJ, Sansom FM, Bennett-Wood V, Hartland EL. Identification of Legionella pneumophila-specific genes by genomic subtractive hybridization with Legionella micdadei and identification of lpnE, a gene required for efficient host cell entry. Infect Immun. 2006; 74(3):1683-91.
- Gilmour MW, Bernard K, Tracz DM, Olson AB, Corbett CR, Burdz T, Ng B, Wiebe D, Broukhanski G, Boleszczuk P,Tang P, Jamieson F, Van Domselaar G, Plummer FA, Berry JD. Molecular typing of a Legionella pneumophila outbreak in Ontario, Canada. J Med Microbiol. 2007; 56(Pt 3):336-41.
- Cirillo SL, Bermudez LE, El-Etr SH, Duhamel GE, Cirillo JD: Legionella pneumophila entry gene rtxA is involved in virulence. Infect Immun 2001, 69: 508-517
- Ingmundson A, Delprato A, Lambright DG, Roy CR: Legionella pneumophila proteins that regulate Rab1 membrane cycling. Nature 2007, 450: 365-370.
- Bruggemann H, Cazalet C, Buchrieser C: Adaptation of Legionella pneumophila to the host environment: role of protein secretion, effectors and eukaryotic-like proteins. Curr Opin Microbiol 2006, 9: 86-94.
© The Author(s) 2017. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


