ISSN: 0973-7510
E-ISSN: 2581-690X
Acute Invasive Fungal Rhinosinusitis (AIFR) is a serious condition with aggressive course and high mortality rates. There is an upsurge in the incidence of invasive fungal rhinosinusitis in post COVID-19 patients. We have come across 20 AIFR cases in post COVID-19 patients. On retrospective exploration of the patient’s records, we found that 30% patients had received steroids and 90% had diabetes. All the patients were managed by administration of IV amphotericin B and local debridement of infected tissues. The mortality rate was as low as 15 %. We conclude that combined approach of Antifungal therapy with debridement of infected tissues improves the prognosis and survival of AIFR patients.
COVID-19, Fungal Sinusitis, Aspergillus, Mucor, Fungal Culture
The coronavirus disease 2019 (COVID-19) pandemic has caused several parallel challenges on a global scale. In the midst of this pandemic, there has been a new epidemic of acute invasive fungal rhinosinusitis (AIFR) infections.1
The recent spate of COVID-19–associated AIFR cases have earned global attention.
There has been a substantial increase in the incidence of AIFR of the maxillofacial region, namely mucormycosis and aspergillosis.2
In India, many states have reported more than 30,000 cases of AIFR, rendering it a notifiable disease.3
AIFR is defined as the presence of fungal hyphae within the sino-nasal mucosa, submucosa, vasculature, or bone with 1 month or less of sinusitis symptoms.4 AIFR is a serious infection and is considered the most aggressive subtype of fungal sinusitis with subsequent serious morbidity and mortality.5
The primary objective of the present study was to establish AIFR in relation to the COVID-19 pandemic and illustrate its characteristic clinical, microbiological, radiological, and histopathological profile and management at our institute, Sri Devaraj Urs Medical College (SDUMC), Kolar, Karnataka, India.
Study Design
This is a retrospective analytical study.
Setting
Department of Microbiology and Department of Otorhinolaryngology, R. L. Jalappa Hospital & Research Center, a tertiary care teaching hospital
Study period
3 months (May–July, 2021).
Participants
Twenty post-COVID-19 patients diagnosed with AIFR.
Ethical approval obtained
This study was approved by the Institutional Ethics Committee (IEC) of SDUMC/SDUAHER College [No. SDUMC/KLR/IEC/92/2021-22 dated 21/06/2021].
Statistical analysis
Data from 20 patients were collected and entered into Microsoft Excel, and descriptive data were analyzed.
Case series
This is a retrospective case series analysis of post-COVID-19 patients with AIFR admitted at our hospital and managed by the Department of Microbiology and the Department of Otorhinolaryngology from May to June, 2021.
Patients were diagnosed based on characteristic clinical manifestations, radio imaging, microbiological analysis (microscopy/culture), and histopathological examination of the sinonasal specimens.
We analyzed the patient demographic details, clinical presentations, comorbidities, laboratory results, imaging findings, and management and clinical outcomes.
In our case series, 20 cases of AIFR were observed. The age group ranged from 32 to 70 years, consisting of 14 (70%) male and six (30%) female patients. All cases were confirmed via Reverse transcription–polymerase chain reaction/antigen detection.
Symptoms predominantly seen in our patients were headache (50%), nasal discharge (40%), nasal obstruction (25%), fever (25%), facial pain (15%), and eye pain (20%), with proptosis in 5% of the patients as represented in Table 1.
Table (1):
Clinical presentation of patients.
Symptoms |
No. of patients (n = 20) |
|---|---|
Headache |
10 (50%) |
Nasal discharge |
8 (40%) |
Nasal obstruction |
5 (25%) |
Drooping of eyelids |
1 (5%) |
Watering of eyes |
2 (10%) |
Facial pain |
3 (15%) |
Eye pain |
4 (20%) |
Proptosis |
1 (5%) |
Eye swelling |
3 (15%) |
Facial swelling |
3 (15%) |
Fever |
5 (25%) |
Vomiting |
1 (5%) |
Dyspnea/breathlessness |
2 (10%) |
Cough |
1 (5%) |
H/O fall from bed |
1 (5%) |
In our study, only six (30%) patients received steroids during their COVID-19 treatment, and three (15%) patients received remdesivir as antiviral and antifungal treatment in 70% of patients. Oxygen was used in managing seven (35%) patients, and 90% and 30% of the patients had diabetes and hypertension as comorbidities, respectively; two patients had no comorbidity.
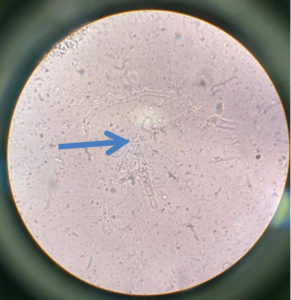
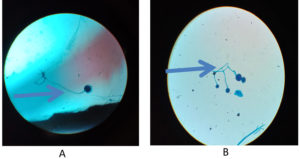
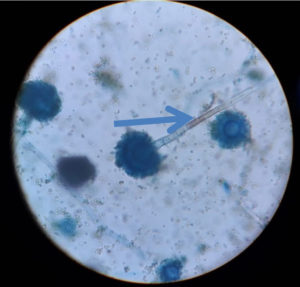
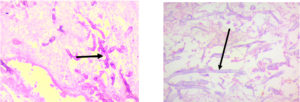
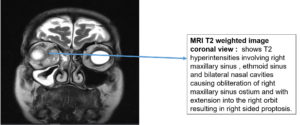
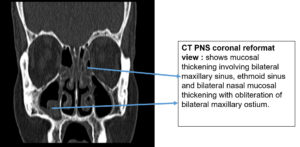
Regarding KOH reports, 11 cases were positive and fungal culture isolates with Rhizopus and Aspergillus species predominant; two cases were reported with co-infections as shown in Figure 1, 2 and 3 (Aspergillus niger and Rhizopus and A. niger and Candida species, respectively). Of the 20 cases, 14 were confirmed via histopathological examination as shown in Figure 4 and Table 2.
Table (2):
Microbiological and pathological findings.
Tests |
Negative |
Positive |
|---|---|---|
KOH (n = 20) |
9 (45%) |
11 (55%) |
Fungal culture (n = 20) |
0 |
20 (100%) |
Histopathological examination findings (n = 14) |
0 |
14 (100%) |
Figure 2. A & B – LPCB Mount showing
Rhizopus – Rhizoids seen in picture A, Bunch of Mucor (sporangiophores & sporangium ) seen in picture B.
In our study, we identified Rhizopus species in eight patients, Mucor in five patients, Aspergillus fumigatus in four patients, A. niger in three patients, and A. flavus in one patients; one patient was infected with Candida (co-infection with A. niger).
The clinical outcome with treatment was considered to be good because there was only 15% mortality (three cases); four patients were discharged against medical advice, and 13 patients were discharged after successful treatment.
There was compelling evidence of lymphopenia in the majority of patients, as seen in the CBC reports of 12 (60%) patients, consistent with the findings of a few previous studies. Nineteen patients underwent imaging tests (n = 20). Inflammatory marker (serum ferritin, lactate dehydrogenase, D-dimer (in some patients), and IL-6 marker (in one patient)) levels were examined as shown in Figure 5.
AIFR is a rapidly progressive and life-threatening infection of the nasal cavity and paranasal sinuses.6 AIFR is known to occur in patients with neutrophil dysfunction or neutropenia such as hematological malignancies, uncontrolled diabetes, aplastic anemia, and hemochromatosis and in patients receiving immunosuppressive drugs.7
Common clinical features noted in AIFR are fever, nasal congestion, acute onset of facial pain, and extension of pain into surrounding structures, such as the paranasal soft tissues, orbit, and cranial vault. Orbital involvement can result in attenuation of vision, whereas sinus or intracranial extension can be associated with proptosis or neurological impairment, respectively.8
There is extensive angioinvasion and hypercoagulability in COVID-19 patients, which act via ACE2 receptor inhibitors (ACE2 receptors are present in numerous organs, including the lungs, kidneys, blood vessels, and heart).9 The causal link between COVID-19 and AIFR remains a mystery.
Many factors contribute to causing this unholistic syndemic. SARS-CoV-2 infection causes impaired cell-mediated immunity, lymphopenia, and associated reduction in CD4 T and CD8 T cell counts.10 The use of steroids in COVID-19 treatment has further led to the inadvertent consequences of steroid-induced hyperglycemia and immunosuppression.5 To add to the existing problem, errant diabetes control leads to endothelial damage, and endotheliitis and thrombosis also predispose patients to fungal infections.11,12
The effects of corticosteroid microangiopathy on diabetes patients and the possible peripheral microthrombi in COVID-19 patients could contribute to the increase in AIFR infections in post-COVID-19 patients.13 SARS- CoV-2 causes upper and lower respiratory tract infections, similar to previous SARS-COV and MERS infections.14
Uncontrolled diabetes mellitus and extensive use of steroids are the two main factors exacerbating AIFR; both of these must be effectively managed.15 Therefore, the successful management of this fatal infection requires early identification of the disease and aggressive and prompt medical and surgical management to prevent fatality.16 In our study, 80% of the patients had diabetes with high HbA1c levels, which supported the findings.
In our case series, the possibility of a correlation between COVID-19 and AIFR infections was analyzed. Lymphopenia was noted in 12 of the 20 patients (60%) with COVID-19.
Viral replication stresses the role of inflammatory response and neutrophil and monocyte influx in the bloodstream and impaired neutrophil-lymphocyte ratio, thereby making the patient susceptible to systemic fungal infections.17 Phagocytic cells are the major host defense mechanisms against mucormycosis. Hyperglycemia and acidosis in diabetes patients are known to impair the ability of phagocytes to move toward and kill pathogens by both oxidative and nonoxidative mechanisms.18,19
Amphotericin-B is the treatment of choice for AIFR. It is available as amphotericin-B deoxycholate. Treatment should be started with liposomal amphotericin-B because it is less nephrotoxic.20
For refractory/intolerant cases for long-term maintenance, posaconazole/other azoles should be used as an alternative treatment
option.21,22
Our case series highlights the association of fungal sinusitis occurrence with post-COVID-19 in patients.
AIFR cases are continually increasing in the COVID-19 pandemic. This emphasizes the need for the early clinical diagnosis of AIFR in patients with COVID-19, which is essential for good treatment outcomes.23 A combination of medical management using antifungal treatment and surgery for the debridement of infected tissues improves the prognosis and survival of these patients.24
ACKNOWLEDGMENTS
We investigators acknowledge hospital MRD section for providing the patient`s files for extra data collection. Clinical samples in Microbiology department to be processed accordingly. We thank all the associated departments for all the necessary patient reports & data collection (Pathology, Radiology, ENT & Biochemistry department).
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
MSH, RB, RBM and AN helped in collection & processing of samples, data collection, analysis of data & writing. AN, SMAM, RK, AKS, BRN helped in design of study, analysis of data & manuscript editing. MSH, RBM, AN, HSB, SMAM, BRN and KNS helped in revision of intellectual content & manuscript editing.
FUNDING
None.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
This study was approved by the Institutional Ethics Committee, Sri Devaraj Urs Medical College, Tamaka, Kolar, India with reference number SDUMC/KLR//IEC/92/2021-22.
- Nagesh, Chinmay P. The “black fungus” through a gray lens, Indian J Ophthal. 2021;69(7): 1648-1649.
Crossref - Song G, Liang G, Liu W. Fungal Co-infections Associated with Global COVID-19 Pandemic: A Clinical and Diagnostic Perspective from China. Mycopathologia. 2020;185(4):599-606.
Crossref - Rocha ICN, Hasan MM, Goyal S, Patel T, Jain S, Ghosh A, Cedeño TDD. COVID-19 and mucormycosis syndemic: double health threat to a collapsing healthcare system in India. Trop Med Int Health. 2021.
Crossref - Deutsch PG, Whittaker J, Prasad S. Invasive and Non-Invasive Fungal Rhinosinusitis-A Review and Update of the Evidence. Medicina (Kaunas). 2019;28;55(7):319.
Crossref - Singh V. Fungal Rhinosinusitis: Unravelling the Disease Spectrum. J Maxillofac Oral Surg. 2019;18(2):164-179.
Crossref - Vadher PR, Mistry SN, Vasani A. Post Covid-19 Acute Invasive Fungal Rhinosinusitis (AIFR): A Study of Histopathological Findings After FESS in Radiologically Diagnosed AIFR. Indian J Otolaryngol Head Neck Surg. 2022:1-7.
Crossref - Chakrabarti A, Denning DW, Ferguson BJ, et al. Fungal rhinosinusitis: a categorization and definitional schema addressing current controversies. Laryngoscope. 2009;119(9):1809-18.
Crossref - El-Kholy NA, El-Fattah AMA, Khafagy YW. Invasive Fungal Sinusitis in Post COVID-19 Patients: A New Clinical Entity. Laryngoscope. 2021;131(12):2652-2658.
Crossref - Moorthy A, Gaikwad R, Krishna S, et al. SARS-CoV-2, Uncontrolled Diabetes and Corticosteroids-An Unholy Trinity in Invasive Fungal Infections of the Maxillofacial Region? A Retrospective, Multi-centric Analysis. J Maxillofac Oral Surg. 2021;20(3):418-425.
Crossref - Wang X, Gui J. Cell-mediated immunity to SARS-CoV-2. Pediatr Investig. 2020;4(4):281-291.
Crossref - Sosale A, Sosale B, Kesavadev J, Chawla M, Reddy S, Saboo B, Misra A. Steroid use during COVID-19 infection and hyperglycemia – What a physician should know. Diabetes Metab Syndr. 2021;15(4):102167.
Crossref - Otifi HM, Adiga BK. Endothelial Dysfunction in Covid-19 Infection. Am J Med Sci. 2022;363(4):281-287.
Crossref - Deng F, Gao D, Ma X, Guo Y, Wang R, Jiang W, Gong S. Corticosteroids in diabetes patients infected with COVID-19. Ir J Med Sci. 2021;190(1):29-31.
Crossref - Petrosillo N, Viceconte G, Ergonul O, Ippolito G, Petersen E. COVID-19, SARS and MERS: are they closely related? Clin Microbiol Infect. 2020;26(6):729-734.
Crossref - Guzmán-Castro, S, Chora-Hernandez, LD, Trujillo-Alonso, G, et al. COVID-19–associated mucormycosis, diabetes and steroid therapy: Experience in a single centre in Western Mexico. Mycoses. 2022; 65: 65–70.
- Akhondi H, Woldemariam B, Rajasurya V. Fungal Sinusitis. [Updated 2022 Jul 4]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK551496/
- Sekaran A, Patil N, Sabhapandit S, Sistla SK, Reddy DN. Rhino-orbito-cerebral mucormycosis: an epidemic in a pandemic. IJID Reg. 2022;2:99-106.
Crossref - Spellberg B, Edwards J Jr, Ibrahim A. Novel perspectives on mucormycosis: pathophysiology, presentation, and management. Clin Microbiol Rev. 2005;18(3):556-69.
Crossref - Ibrahim AS, Spellberg B, Walsh TJ, Kontoyiannis DP. Pathogenesis of mucormycosis. Clin Infect Dis. 2012;54 Suppl 1(Suppl 1):S16-22.
Crossref - Garg D, Muthu V, Sehgal IS, Ramachandran R, Kaur H, Bhalla A, Puri GD, Chakrabarti A, Agarwal R. Coronavirus Disease (Covid-19) Associated Mucormycosis (CAM): Case Report and Systematic Review of Literature. Mycopathologia. 2021;186(2):289-298.
Crossref - Kumari A, Rao NP, Patnaik U, Malik V, Tevatia MS, Thakur S, Jaydevan J, Saxena P. Management outcomes of mucormycosis in COVID-19 patients: A preliminary report from a tertiary care hospital. Med J Armed Forces India. 2021;77(Suppl 2):S289-S295.
Crossref - Madhavan, Y.; Sai, K.V.; Shanmugam, D.K.; Manimaran, A.; Guruviah, K.; Mohanta, Y.K.; Venugopal, D.C.; Mohanta, T.K.; Sharma, N.; Muthupandian, S. Current Treatment Options for COVID-19 Associated Mucormycosis: Present Status and Future Perspectives. J. Clin. Med.; 11:3620.
Crossref - Yasmin F, Najeeb H, Naeem A, Dapke K, Phadke R, Asghar MS, Shah SMI, De Berardis D, Ullah I. COVID-19 Associated Mucormycosis: A Systematic Review from Diagnostic Challenges to Management. Diseases. 2021; 9(4):65.
Crossref - Rao KM, Pooja N. Management of Mucormycosis in Post COVID-19 Patients and Its Outcome in a Tertiary Care Center: Our Experience. Indian J Otolaryngol Head Neck Surg. 2022:1-8.
Crossref
© The Author(s) 2022. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.