Carica papaya L. leaves contain potent secondary metabolites, such as alkaloids, phenolics, flavonoids, saponin, tannins, glycosides, and other crucial phytochemicals. The leaf is a source of several vitamins conferring it an anti-oxidant property. The leaf is also rich in dietary minerals and essential fatty acid. The fresh leaf extracts are used as a possible remedy for dengue viral infections in Asian countries. This review compiles the knowledge about the physiochemicals, phytochemicals, and biochemicals of Carica papaya L. leaves. This includes the identified chemical structures of alkaloids, phenolic, flavonoids, linoleic, and linolenicacids. Additionally, it covers up-to-date information on in-vitro anti-cancer and anti-gout and in-vivo anti-microbial properties, anti-oxidant, anti-sickling, hepatoprotective, hypolipidemic, hypoglycemic, and anti-gout effects.
Fresh Leaves, Phytochemicals, Dietary Minerals, Vitamins, Anti-oxidant and bio-activity
Carica papaya L. is commonly known as pawpaw tree. It is a nutraceutical plant. All plant parts have vital immunostimulative and antioxidant effects. The Indian traditional medicinal system prescribes its leaf extract to control dengue viral infections. In other Asian countries, it is also used as a remedy for the same1. The leaf contains a mixture of chemicals, which can be described broadly as physiochemical, phytochemicals and biochemicals.
Physiochemicals
The physiochemical nature of the dried leaf has following contents in % w/w: Total Ash (17.4), Acid insoluble ash (13.3), Water soluble ash (3.25), Sulfated ash (27.05), Ethanol soluble extractive (8.4), Water-soluble extractive (17.7), Moisture content (5.70), Crude fat (2.8+0.3), Crude fiber (23+0.1), Crude protein (29.5+0.1), and Carbohydrate (35.9+0.3)2.
Phytochemicals
It contains alkaloids, phenols, flavonoids, saponin, tannins, and glycosides. They could be described as follows:
Alkaloids
The significant phytochemicals of Carica papaya L. leave are bitter tasting alkaloids (1,300–1,500 ppm). It is in the form of carpaine (150–4,000 ppm) (Figure 1), pseudocarpaines, macrocyclic piperidine, dehydrocarpaine I and II (1000 ppm), and nicotine (102.8 ppm)3.
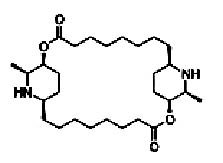
Fig. 1. Structure of carpaine (-13, 26-dimethyl-2, 15-dioxa-12, 25-diazatricyclotriacontane-3, 16-dione)
Carpaine
Its molecular formula is C28H50N2O4 (Figure 1), which accounts for a molecular mass of 478.70g/Mol. It is used to reduce cardiovascular problems, act as amebicide and was found to inhibit Mycobacterium tuberculosis under in-vitro assay condition4.
Phenolic acids
The leaf contains phenolic acids such as protocatechuic acid (Figure 2), coumaric acids, caffeic acid (Figur3), chlorogenic acid, and coumarin (Figure 4)5,2. These are known as esterified phenolic compounds6. These compounds are linked with malic acid by an ester linkage.
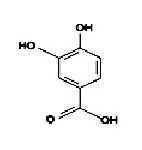
Fig. 2. Structure of protocatechuic acid5
Protocatechuic acid
It is 3,4-dihydroxybenzoic acid(C7H6O4) (Figure 2). It is a type of antioxidant polyphenol.
p, m, o-Coumaricacids and caffeicacid
Coumaric acids are hydroxyl derivatives of cinnamic acid (Figure 3). p-Coumaric acids possess anti-oxidant property, which results in the inhibition of the low density lipoprotein oxidation7.
p-Coumaric acid (3) R1=R3=R4=H, R2=OH
m-Coumaric acid (1a) R1=OH, R2=R3=R4=H
o-Coumaric acid (1b) R1=R2=R3=H, R4=OH
Caffeic acid (5) R1=R4=H, R2=R3=OH
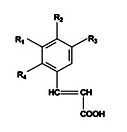
Fig. 3. Structure of coumaricandcaffeicacids5
Chlorogenic acid
It is a polyphenol compound, belongs to the family of esters of hydroxyl cinnamic acids, and has antioxidant effects. The molecular formula is C16H18O9 (Figure 4). Molecular mass is 354.31g mol–1. This can form an ester linkage with caffeic acid and quinic acid.
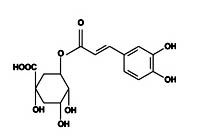
Fig. 4. Structure of chlorogenicacids5
Coumarin
Its molecular formula is C11H10O4 (Figure 5) and the molecular weight is 206.19gmol–1.
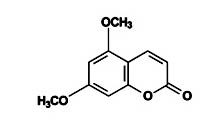
Fig. 5. Structure of 5, 7-dimethoxycoumarin5
Flavonoids
Flavonols (0–2,000 ppm) are the types of flavonoids. The leaf contains kaempferol and quercetin and other glycosylated flavonols such as manghaslin, clitorin, nicotiflorin, and rutin6.
Kaempferol and Quercetin
Kaempferol (C15H10O6) and quercetin (C15H10O7) (Figure 6) are main flavanols present in Carica papaya L. leaves.
Kaempferol (6) R1=R2=R3=R4=OH, R5=H
Quercetin (7) R1=R2=R3=R4=R5=OH

Fig. 6. Structure of kaempferol and Quercetin5
Other Phytochemicals
Tannins (5,000–6,000 ppm), phytosterol, cardiacglycosides, saponin glycosides, iridoids, anthraquinones, and organic acids such as malic acid, minor quantities of various malic acid derivatives and quinicacid are present in the leaves6.
Biochemicals
Vitamins
The extracts are very rich in the anti-oxidant vitamins such as vitamin A (7873.4 mg/kg), folic acid (2.4mg/kg), vitamin B12 (2.8 mg/kg), and vitamin C (89.0 mg/kg)8.
Minerals
The leaf contains Ca (8612.50 mg/Kg), Mg (67.75mg/Kg), Na (1782.00mg/Kg), K (2889.00 mg/Kg), Fe (90.50mg/Kg), Mn (9.50 mg/Kg)9 and calcium oxalate. Other essential growth-promoting biochemicals are aminoacids such as proline and glutamine, enzymatic proteins such as a-amylase, b-amylase, and carbohydrate such as starch and cellulose.
Fatty acids
Linoleic (Figure 7) acid is an unsaturated omega-6 fatty acid. Linolenic acid (Figure 8) is an omega-3 fatty acid. These two compounds were found to show anti-malarial property in in-vitro biological assay conditions10.
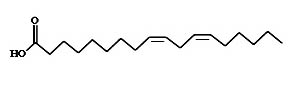
Fig. 7. Structure of linoleic acid (9, 12-octadecadienoic acid)
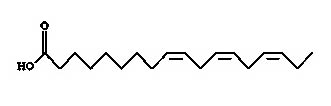
Fig. 8. Structure of linolenic acid (9, 12, 15-octadecatrienoic acid)
Pharmacognostics properties of the leaves
Antibacterial activity
The in-vitro activity against clinical pathogens such as Klebsiella pneumonia, K.oxytocin, Enterococcus faecalis, Proteus mirabilis11, Escherichia coli, Pseudomonas aeruginosa12, Salmonella typhi13, Salmonella paratyphi A, Shigella flexneri14, Streptococcus pyogenes, Staphylococcus aureus, Bacillus subtilis, and Micrococcus luteus has been detected in the leaf extract using agar and disk diffusion methods.
Anti-fungal activity
Carica papaya leave extracts have broad spectrum anti-fungal activity against Aspergillus flavus, Candida albicans, Trichophyton rubrum, T. mentagrophytes15,16, Cryptococcus neoformans, Candida tropicalis, and C. kefyr17. Colletotrichum gloeosporioides, Rhizopus stolonifer, and Fusarium spp.18 were also inhibited. The dry leaves can control the radial growth of mycelia of Fusarium verticillioides19. An aqueous extract controlled Alternaria sp., Fusarium sp., Pestalotiopsis sp., and Rhizopus sp.20
Anti-helminthic
The leaf extract at 5% concentration caused paralysis to the Pheretima posthuma, an Indian adult earthworm21.The healthy leaves were used to cure ascariasis infection caused by a pathogenic form of Ascaridia galli22and Ancylostoma caninum in an in-vivo mouse animal model23.
Antitumor activity
The leaf extracts can block the proliferative responses in cervical carcinoma, breast adenocarcinoma, hepatocellular carcinoma, lung adenocarcinoma, and pancreatic epithelial carcinoma. Apart from these, the leaf extracts also help in protecting the vascular systems and functional systems. It mediated Th1 type shift in human and acted as a potent anti-tumor agent in an in-vitro assay24.
Immunostimulant activity during DENV viral infection
In a study, a leaf extract was orally administered to the patients to reduce the dengue viral symptoms. It increased the thrombocyte25 and platelet count in a mouse model infected with dengue virus. During the dengue viral infection, a crushed leaf extract was found to improve the body immunity1.
Antioxidant effect
The leaf contains flavonoids (quercetin), phenols, tannins26 and vitamins as the source of natural anti-oxidants. It has a potent hydroxy radical scavenging activity as found in some in-vitro conditions. It reduced acrylamide induced oxidative stress in stomach, liver, and kidney in an in-vivo animal model27. In an in-vitro assay, it scavenged1,1-diphenylehydrazyl (DPPH), 2,2- azinobis-(3-ethyle benzothiazoline-6-sulfonate) (ABTS), nitric oxide, superoxide, and lipid peroxide in rat brain and liver28. It inhibited nitric oxide and TNF-a production in in-vitro conditions29. It reduced alcohol-induced acute gastric damage and blood oxidative stress in Sprague-Dawley rats30.
Hepatoprotective effect
The fresh leaves of the plant are traditionally used to cure liver associated problems.The leaf extracts help regularize liver enzyme during cirrhosis, hepatitis, and jaundice conditions. Carbon tetrachloride-induced liver damage was rectified by the aqueous extracts of C. papaya leave in in-vivo animal models31. The freeze-dried leaf extracts considerably protected hepatic cells from oxidative injury caused by tert-butyl hydroperoxide in in-vitro conditions. This chemical induced free radicals were scavenged by the anti-oxidants, phenolic and flavonoids, which are present in the leaves32.
Anti-inflammatory effects
The leaves also act as analgesic. It can reduce carrageenan induced paw oedema, cotton pellets induced granuloma, and formaldehyde induced arthritis in in-vivo models33. It could reduce Salmonella typhi infection induced inflammation in albino rats34.
Hypoglycemic effect
The extracts showed a significant hypoglycemic effect in in-vivo rat animal models35, where diabetes was induced by streptozotocin36.
Anti-sickling effect
The methanolic extracts of the leaves protected the membranes and reduced the sick cell formation under in-vitro conditions37.
Anti-gout effect
Xanthine oxidase enzyme is primarily involved in gout. The dry leaf extracts are the source of xanthine oxidase inhibitors, which can act as an anti-gout agent38.
Toxicity
The leaf extracts showed no pathological differentiation forms in the tissues and there were no contrary effects on the major functional organs like liver, kidney, and bone marrow by its aqueous extracts in in-vivo animal models.Thus the leaf extracts were found to be safe.
Carica papaya L. leaves contain chemicals that are multi-functional and of a great medicinal importance. Several in-vitro and in-vivo studies have approved its therapeutic efficacy and attainable mechanisms against newly emerging infectious diseases. However, further research is required for the development of potential anti-viral drugs from this leaf. The immunity development mechanisms during dengue viral infections have confirmed the anti-viral efficacy of Carica papaya L. leaves. The further exploration of dengue viral control mechanism is required and it may lead to the development of several anti-viral therapies. The toxicity level of pawpaw leaves is less and it doesnot hinder the medicinal beneficial effect. Therefore, the ethanomedicinal usage of these leaves is much safer than any other synthetic drugs.
ACKNOWLEDGMENTS
Our special thanks to Prof. Naoki Yamamoto, ID lab, Department of Microbiology, Yong Loo Lin School of Medicine, the National University of Singapore for anti-DENV studies.
- Nisar, A., Hina, F., Muhammad, A., Bilal, H.A., Ijaz, M., Lubna F. Dengue fever treatment with Carica papaya leaves extracts. Asia. Pacifi. J Trop. Biomed., 2011; 330–333.
- Zunjar, V., Mammen, D., Trivedi, B.M., Daniel, M. Pharmacognostic, Physicochemical and Phytochemical Studies on Carica papaya Linn. Leaves. Pharmacogno. J., 2011; 3(20):5–8.
- J Duke. 2007. CaricapapayaL.Caricaceae. (http://sun.ars-grin.gov:8080/npgspub/xsql/duke/plantdisp.xsql?taxon=209.)
- Burdick, E., Carpaine, M. An alkaloid of Carica papaya. Chemistry and pharmacology. Economic Bot., 1971; 25(4): 363–365.
- Canini, A., Daniela, A., Giuseppe, D’A., Pietro, T. Gas chromatography–mass spectrometry analysis of phenolic compounds from Carica papaya L. leaf. J. Food Composition Analy., 2007; 20: 584–590.
- Adlin, A., Noor, R.A., Siti, Z.H., Badrul, A.R., Raja, H.R.S., Noordini, A., Ibrahim, J., Hussin, M., Zakiah, I. Repeated Dose 28-Days Oral Toxicity Study of Carica papaya L. Leaf Extract in Sprague Dawley Rats. Molecule., 2012; 17: 4326–4342.
- Lun-Yi, Z., Greg, C., Henry, G., Xianglin, S., Vince, C., Val, V. Effect of antioxidant protection by p-coumaric acid on low-density lipoprotein cholesterol oxidation. Am. J. Physiol. Cell Physiol., 2000; 279: C954-C960.
- Imaga, N.A., George, O.G., Veronica, I.O., Sunday, A., Tomi, D-E., Bola, O., Patience, N.D., Mojisola, O., Alero, O., Felix, C.E. Phytochemical and antioxidant nutrient constituents of Carica papaya and Parquetinanigrescens extracts. Sci. Res. Essays.,2010;5(16): 2201–2205.
- Ayoola, P.B., Adeyeye, A. Phytochemical and nutrient evaluation of Carica papaya (pawpaw) leaves. Int. J. Res. Rev. Appl. Sci.,2010; 5(3).
- Paula M., William, C., Paschal, E., Peter, S. In vitro antiplasmodial activities of extracts from five plants used singly and in combination against Plasmodium falciparum parasites. J. Med. Plant. Res., 2012; 6(47): 5770–5779.
- Yusha’u, M., Onuorah, F. C., Murtala, Y. In-vitro sensitivity pattern of some urinary tract isolates to Carica papaya extracts. Bay J. Pure. Appl. Sci., 2009; 2(2):75 – 78.,
- Indranil, B., Soroj, K.C., Anupam, G., Goutam, C. Antibacterial activities of some plant extracts used in Indian traditional folk medicine, Asia. Pac. J. Trop. Biomed., 2011; S165-S169.
- Oluduro, A., Omoboye, O. In Vitro Antibacterial Potentials and Synergistic Effect of South-Western Nigerian Plant Parts Used in Folklore Remedy for Salmonella typhi infection. Nat. Sci., 2010; 8(9).
- Rahman, S., Imran, M., Muhammad, N., Hassan, N., Chisthi, A.K., Khan, A.F., Sadozai, K.S., Khan, S.M. Antibacetial screening of leaves and stem of Carica papaya. J. Med. Plant. Res., 2011; 5(20): 5167–5171.
- Khan, J.A., Yadav, J., Srivastava, Y., Pal, P.K. In Vitro Evaluation of Antimicrobial Properties of Carica papaya. Int. J. Biol. Pharm. Allied Sci., 2012; 1(7):933–945.
- Okunola, A. A., Muyideen, T. H., Chinedu, P. A., Tomisin, J., Harrison, Abia., Victor, U. O., Babatunde, E. E. Comparative studies on antimicrobial properties of extracts of fresh and dried leaves of Carica papaya (L) on clinical bacterial and fungal isolates. Advan. Appl. Sci. Res., 2012; 3(5):3107–3114.
- Baskaran, C., Rathabai, V., Velu, S., Kubendiran, K. The efficacy of Carica papaya leaf extract on some bacterial and a fungal strain by well diffusion method. Asia. Pac. J. Trop. Dis., 2012; S658-S662.
- Abirami, L.S.S., Pushkala, R., Srividya, N. Antimicrobial Activity of Selected Plant Extracts against Two Important fungal Pathogens Isolated from Papaya Fruit. Int. J. Res. Pharmaceut. Biomed. Sci., 2013; 4(1):234–238.
- Olahan, G.S., Amadi, J.E. Fungicidal effect of pawpaw (carica papaya L.) leaf extracts on Fusarium verticilliodes. African Sci., 2006; 7(1):189–193.
- Pedro Cha´vez-Q., Tania Gonza´lez-F., Ingrid Rodrý´guez-B., Santiago Gallegos-T. Antifungal Activity in Ethanolic Extracts of Carica papaya L. cv. Maradol Leaves and Seeds. Ind. J. Micro., 2011; 51(1):54–60.
- Saumendu, D.R., Rimlee, G., Sumit, D., Dibyendu, S., Ratnali, B., Sagnik, H. Pharmacognostic Evaluation and Anthelmintic Activity of Leaf and Stem Extract of Carica papaya. J. Pharmaceutica. Res., 2012; 25(9):4763–4766.
- Oparaochal, E.T, Okorie, C., Anosike, J., Obi, R. Vermifugal Effect Of Three Local Herbs (Carica Papaya, Spigilia Anthelmia And Phyllantus Niruri) On Broiler Chickens Infected With Ascaridia Galli. African J. Appl. Zool. Environmen. Bio., 2008; 10: 61 – 64.
- Shaziya, B., Goyal, P.K. Anthelmintic effect of Natural Plant ( Carica papaya ) extract against the Gastrointestinal nematode, Ancylostoma caninum in Mice. ISCA J. Biological Sci., 2012; 1(1): 2–6.
- Noriko, O., Nam, H. D., Emi, K., Akira, K., Satoshi, I., Chikao, M. Aqueous extract of Carica papaya leaves exhibits anti-tumor activity and immunomodulatory effects. J. Ethnopharmacol., 2010; 127: 760–767.
- Kathiresan, S., Surash, R., Sharif, M.M., Mas, R.M.., Walther, H. W. Thrombocyte counts in mice after the administration of papaya leaf suspension. Wie.Klinis. Wochensch., 2009; 121 (suppl 3): 19–22.
- Sushil, C.S., Nitin, S., Rawat, U., Sati, O.P. Medicinal plants as a source of antioxidants. Res. J. Phytochem., 2010; 4(4): 213–224.
- Kodry, M. S. Antioxidant and Immunostimulant Effect of Carica Papaya Linn. Aqueous Extract in Acrylamide Intoxicated Rats. Act. Informatic. Medic., 2012; 20(3): 180–185.
- Srikanth, G., Manohar, B.S., Kavitha, C.H.N., Bhanoji, R.M.E., Vijaykumar, N., Pradeep, C.H. Studies on in-vitro antioxidant activities of Carica papaya aqueous leaf extract. Res. J. Pharmaceutica. Biologi. Chem. Sci., 2010; 1(2):59- 65.
- Lee, K.H., Padzil, A.M., Syahida, A., Abdullah, N., Zuhainis, S.W., Maziah, M., Sulaiman, M.R., Israf, D.A., Shaari, K., Lajis, N.H. Evaluation of anti-inflammatory, antioxidant and antinociceptive activities of six Malaysian medicinal plants. J. Med. Plant. Res., 2011; 5(23): 5555–5563.
- Indran, M., Mahmood, A.A., Kuppusamy, U.R. Protective Effect of Carica papaya L Leaf Extract against Alcohol Induced Acute Gastric Damage and Blood Oxidative Stress in Rats. W. Ind. Med. J., 2008; 57(4): 323.
- Mohammed, A., Abubakar, S.A., Sule, M.S. Hepatoprotective effect of aqueous leaf extract of carica papaya linn. Against ccl4-induced hepatic damage in rats. Internat. J. Pharmaceutic. Sci. Rev. Res., 2011; 11(2).
- Sheri-Ann, T., Sonia, R., María, A. M., Raquel, M., Michael, H., Surash, R., Nazalan, N., Maqsudul, A., Laura, B., Luis, G. Protective effects of papaya extracts on tert-butyl hydroperoxide mediated oxidative injury to human liver cells (An in-vitro study). Free Rad. Antioxi., 2012; 2(3).
- Bamidele, V. O., Olubori, M. A., Adeoye, A. F., Ayodele, O. S. Anti-inflammatory activities of ethanolic extract of Carica papaya leaves. Inflammopharmacol., 2008; 16(4): 168–173.
- Oladunmoye and Osho. Anti-inflammatory activity of ethanolic leaf extract from Carica papaya in rats orogastrically dosed with Salmonella typhi and Staphylococcus aureus. J. Plant Sci., 2007; 2(4): 447–452.
- Yasmeen, M., Prabhu, B. Antihyperglycemic and hypolipidemic activities of aqueous extract of Carica papaya Linn. Leaves in alloxan-induced diabetic rats. J. Ayurv. Integ. Med., 2012; 3(2): 70 – 74.
- Isela, E. Juárez-R., Juan, C Díaz-Z., Jorge, L., Pedro, H. Miranda-O., Andrés, E. Castell-R., Carlos, A., Tovilla-Z., Arturo, Rodríguez-H., Hidemi, Aguilar-M., Teresa, Ramón-F., Deysi, Y. Bermúdez-O. Hypoglycemic effect of Carica papaya leaves in streptozotocin-induced diabetic rats. BMC Complement. Alternat. Med., 2012; 12:236.
- Imaga, N.O.A., Gbenle, G.O., Okochi, V.I., Akanbi, S.O., Edeoghon, S.O., Oigbochie, V., Kehinde, M.O., Bamiro, S.B. Antisickling property of Carica papaya leaf extract. Afr. J. Biochem. Res., 2009; 3(4): 102–106.
- Saiful, M.N.A., Parveen, J., Azura, A. Purification of Xanthine Oxidase Inhibitor from Carica papaya Leaves using Reversed Phase Flash Column Chromatography (RPFCC) – High Performance Thin Layer Chromatography (HPTLC). Aus. J. Bas. Appl. Sci., 2012; 6(1): 117–122.
© The Author(s) 2018. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


