ISSN: 0973-7510
E-ISSN: 2581-690X
Hepatitis is the inflammation of the liver cause by viral infections which effect cells in the tissue of the liver. Periodontal disease is a bacterial inflammatory disease that affects the gingiva and bone supporting the teeth. This study aimed to evaluate and compare the periodontal clinical parameters of patients with hepatitis B virus and hepatitis B and C super-infection. Sample population consisted of 23 patients had hepatitis B and 20 patients with both hepatitis B and C viruses infection. While the control group consisted of 20 healthy individuals without hepatitis. Males and females were included with age range (40-50) years old. The periodontal clinical parameters were examined and recorded for each group. The results showed highly significant difference between the study and control groups for all the clinical parameters (plaque and gingival index, pocket depth and clinical attachment level. Also high significant differences were reported for all the clinical periodontal parameters when comparing between patients with virus B infection and patients with both virus B and virus C infection. The patients with hepatitis had worse periodontal health than the healthy persons, and in comparison, the patients with hepatitis B and C viruses had worse periodontal health than hepatitis B patients.
Periodontal, patients, hepatitis B and C, viruses.
Periodontal diseases are one of the most widely spread diseases of humanity , no nation and no country being free from periodontal diseases1. These are infectious diseases causing inflammation of the supporting tissues of the teeth2. Despite the fact that the bacterial components of dental plaque are the etiological factors, but periodontal diseases have a complex interaction between microbial and host tissues factors modified by many risk factors3.
Systemic diseases are among the factors that affect periodontal diseases. Hepatitis, which is inflammation of the liver, is important systemic disease because it has the ability to disturb the normal function of the liver4. A number of viruses causes most cases of hepatitis but it could be due to other infections or autoimmune disease. Viral hepatitis may include: hepatitis A virus(HAV), hepatitis B virus(HBV), hepatitis C virus (HCV), hepatitis D virus which requires infection with HBV virus at the same time, hepatitis E virus and hepatitis G virus which is a distant relative of HCV10.
Viral hepatitis mostly present in acute phase, but HBV, HCV, or HDV can progress to the chronic form. The common symptoms include weakness, nausea and vomiting, vague pain or discomfort in upper right abdomen, fever, jaundice, and the chronic signs of viral hepatitis may include fullness of the abdomen, recurrent fatigue and sometimes accumulation of fluid (ascites)11, 12.
Hepatitis B is a serious infection which can cause a wide range of symptoms. It is caused by hepatitis B virus and may lead to premature death from liver cirrhosis, liver failure or cancer5.
Hepatitis C is produced by hepatitis C virus infection, which is an important health problem with public concern because of an estimated 160 million persons infected over the world6.
HCV represents the causative agent of chronic hepatitis, cirrhosis and hepatic carcinoma. Thus, hepatitis C causes 27% and 25% of all cirrhosis and hepatocellular carcinoma cases respectively7. Hepatitis B virus and hepatitis C virus are infections are very serios public health problem which have many consequences concerning psychological problems. HBV and HCV are the most common causes of occupational diseases transmitted from patients to the health care workers and vice versa, also to the workers families8. It was estimated that 14.4% and 1.4% of the health workers in hospitals are infected with HBV and HCV respectively. Physicians, dentists, nurses lab staff and dialysis centers workers are at high risk of this infection9. Therefore, the present study aspects such common oral manifestations of hepatitis B and C infections to be able to diagnose, prevent and manage appropriately the transmission and progression of this fetal disease.
Hepatitis C virus infection can involve a variety of extra-hepatic conditions including the oral cavity, some oral manifestation are oral diseases like lichen planus and sjogren-like sialoadenitis19.
Sometimes patients may have two possible patterns of infections with hepatitis viruses, either co-infection, when the viruses infect the patient at the same time, or super-infection, when the infected patient exposed later to a second pattern of hepatitis virus, such as HBC and HDV and HBV and
HCV 13.
Human Samples: Sample population consisted of (63) individuals, all with age range 45-55 years old . The test or study group consisted of 43 patients who subdivided into two subgroups , hepatitis B positive 23 patients which is the first subgroup, and hepatitis B and C positive20 patients which is the second subgroup. The sample recruited here were patients attending in Al-Kadhemia Teaching Hospital and Al-Yarmook Teaching Hospital. The control group consisted of 20m adults with the same age range and are healthy individuals with no history of any systemic disease or drugs intake for at least one month.
Clinical examination
All the individuals in both study and control groups were carefully examined clinically. For each patient, all the teeth were examined with the exclusion of the third molar. The collected data involved the assessment of plaque index, gingival index, probing pocket depth and the assessment of clinical attachment level . Both probing pocket depth and the clinical attachment level were divided into three categories according to severity (1-2mm), (3-5mm) and (>=6 mm).
Statistical analysis: The data analysis were used in order to determine and analyze the results of the study under application of SPSS (Statistical Process for Social Science) version 14 application
- Descriptive data analysis : Mean value, standard deviation, standard error, and two extreme values (minimum and maximum) of the calculated men values.
- Inferential data analysis: These were used to accept or reject the statistical hypothesis, which include the one way ANOVA procedure produce a one way analysis of variance for quantitative dependent variable, and least significant difference (LSD) test.
For plaque index and gingival index, the statistical analysis showed that mean values were higher for study group than control group, with highest responding towards hepatitis (B and C) subgroup. By applying one way ANOVA and LSD tests, highly significant differences were revealed in testing means between the study and control groups, with the high levels at hepatitis (B and C) subgroup then decreased for hepatitis B subgroup and finally the lowest readings levels for control group.
| Parameter | Groups | No. | Mean | S.D. | S.E. | Min. | Max. | ANOVA- Testing equality of means
Sig. (*) |
|---|---|---|---|---|---|---|---|---|
| Plaque Index | Hep. (B) | 23 | 1.49 | 0.26 | 0.06 | 0.95 | 2.27 | 0.000 (HS) |
| Hep. (B+C) | 20 | 2.08 | 0.29 | 0.07 | 1.41 | 2.68 | ||
| Control Neg. | 20 | 1.21 | 0.17 | 0.04 | 0.89 | 1.57 | ||
| Gingival Index | Hep. (B) | 23 | 1.51 | 0.26 | 0.05 | 1.10 | 2.00 | 0.000 (HS) |
| Hep. (B+C) | 20 | 2.01 | 0.28 | 0.06 | 1.50 | 2.51 | ||
| Control Neg. | 20 | 1.26 | 0.24 | 0.05 | 0.78 | 1.73 |
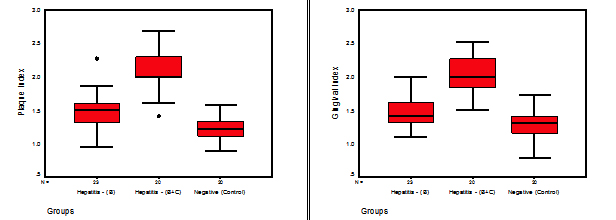 Fig. 1. Stem-Leaf plots for the studied readings of (Plaque & Gingival Index) parameters in different groups
Fig. 1. Stem-Leaf plots for the studied readings of (Plaque & Gingival Index) parameters in different groupsTable (1-3):
Multiple Comparison for (Plaque & Gingival) parameters by (LSD) method for all pairs of the studied groups.
| Parameter | Group (I) | Group (J) | Sig. | C.S. (*) |
|---|---|---|---|---|
| Plaque Index | Hep. – (B) | Hep. – (B+C) | 0.000 | HS |
| Control | 0.000 | HS | ||
| Hep. – (B+C) | Control | 0.000 | HS | |
| Gingival Index | Hep. – (B) | Hep. – (B+C) | 0.000 | HS |
| Control | 0.002 | HS | ||
| Hep. – (B+C) | Control | 0.000 | HS |
For probing pocket depth for (0-2), (3-5), (>=6) categories the mean of number of surfaces that had deeper pocket depth was higher for the study group than the control group, and higher for hepatitis B and C subgroup than hepatitis B subgroup. By applying ANOVA test highly significant differences were revealed between the control group and hepatitis B and hepatitis B and C subgroups. When using LSD test, except for comparison between hepatitis B and B and C subgroups for (0-2) and (3-5) categories non- significant differences were observed, however, the hepatitis B and C subgroup had higher number of surfaces having (>=6) category of pocket depth than the control and hepatitis B groups.
Table (2-1):
Summary Statistics of the studied readings of (Pocket Depth) parameter in different groups.
| Pocket Depth Parameter | Groups | No. | Mean | S.D. | S.E. | Min. | Max. | ANOVA- Testing equality of means Sig. (*) |
|---|---|---|---|---|---|---|---|---|
| (0 – 2) | Hep. (B) | 23 | 40.22 | 17.16 | 3.58 | 6 | 84 | 0.000 (HS) |
| Hep. (B+C) | 20 | 32.90 | 17.72 | 3.96 | 5 | 88 | ||
| Control Neg. | 20 | 70.45 | 10.61 | 2.37 | 45 | 90 | ||
| (3 – 5) | Hep. (B) | 23 | 40.61 | 12.68 | 2.64 | 6 | 69 | 0.001 (HS) |
| Hep. (B+C) | 20 | 43.15 | 16.63 | 3.72 | 0 | 73 | ||
| Control Neg. | 20 | 27.50 | 7.92 | 1.77 | 17 | 48 | ||
| ≥ 6 | Hep. (B) | 23 | 3.96 | 2.31 | 0.48 | 0 | 8 | 0.000 (HS) |
| Hep. (B+C) | 20 | 9.45 | 5.64 | 1.26 | 0 | 24 | ||
| Control Neg. | 20 | 1.25 | 1.80 | 0.40 | 0 | 6 |
Figures 2 represents graphically studied readings of different classes of (Pocket Depth) parameter in different groups.
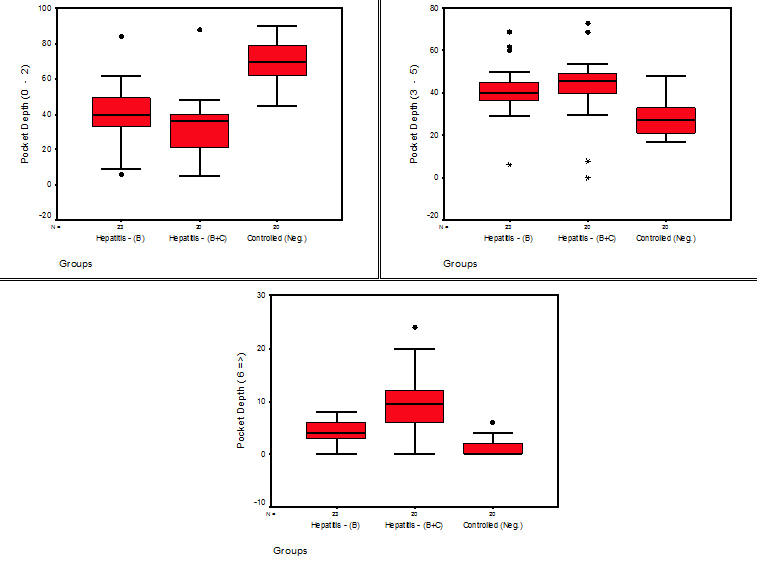 Fig. 2. Stem-Leaf plots for the studied readings of (Plaque & Gingival Index) parameters in different groups
Fig. 2. Stem-Leaf plots for the studied readings of (Plaque & Gingival Index) parameters in different groupsTable (2-3):
Multiple Comparison for (Pocket Depth) parameter in each class among all pairs of the studied groups.
| Parameter | Group (I) | Group (J) | Sig. | C.S. (*) |
|---|---|---|---|---|
| (0 – 2) | Hep. – (B) | Hep. – (B+C) | 0.130 | NS |
| Control | 0.000 | HS | ||
| Hep. – (B+C) | Control | 0.000 | HS | |
| (3 – 5) | Hep. – (B) | Hep. – (B+C) | 0.366 | NS |
| Control | 0.000 | HS | ||
| Hep. – (B+C) | Control | 0.000 | HS | |
| ( ≥ 6 ) | Hep. – (B) | Hep. – (B+C) | 0.000 | HS |
| Control | 0.017 | S | ||
| Hep. – (B+C) | Control | 0.000 | HS |
Regarding the clinical attachment level, the mean of number of surfaces for (0-2) category was higher for the control group than the study groups.
While for (3-5) and (>=6) categories the study groups had higher number of surfaces than the control group. By applying ANOVA test, highly significant difference were revealed between the groups for (0-2) and (>=6) categories and significant difference the groups for the (3-5) category. When using LSD tests, significant and highly significant differences were obtained for all comparisons between the control and hepatitis B and hepatitis B and C subgroups except when comparing between hepatitis B and hepatitis B and C subgroups for (3-5) category and hepatitis B and control group for (>=6) category were non-significant differences .
Table (3-1):
Summary Statistics of the studied readings of (Attachment) parameter in different groups.
| Clinical attachment Parameter | Groups | No. | Mean | S.D. | S.E. | Min. | Max. | ANOVA- Testing equality of meansSig. (*) |
|---|---|---|---|---|---|---|---|---|
| (0 – 2) | Hep. (B) | 23 | 39.04 | 15.48 | 3.23 | 0 | 78 | 0.000 (HS) |
| Hep. (B+C) | 20 | 30.30 | 10.20 | 2.28 | 9 | 54 | ||
| Control Neg. | 20 | 64.70 | 10.04 | 2.24 | 50 | 90 | ||
| (3 – 5) | Hep. (B) | 23 | 43.13 | 16.96 | 3.54 | 16 | 82 | 0.012 (S) |
| Hep. (B+C) | 20 | 44.00 | 9.29 | 2.08 | 27 | 63 | ||
| Control Neg. | 20 | 33.30 | 7.33 | 1.64 | 14 | 44 | ||
| ≥ 6 | Hep. (B) | 23 | 2.61 | 2.57 | 0.54 | 0 | 6 | 0.000 (HS) |
| Hep. (B+C) | 20 | 10.25 | 4.51 | 1.01 | 0 | 18 | ||
| Control Neg. | 20 | 1.20 | 1.74 | 0.39 | 0 | 5 |
Table (3-3):
Multiple Comparison for (Attachment) parameter in each class among all pairs of the studied groups.
| Parameter | Group (I) | Group (J) | Sig. | C.S. (*) |
|---|---|---|---|---|
| (0 – 2) | Hep. – (B) | Hep. – (B+C) | 0.024 | S |
| Control | 0.000 | HS | ||
| Hep. – (B+C) | Control | 0.000 | HS | |
| (3 – 5) | Hep. – (B) | Hep. – (B+C) | 0.976 | NS |
| Control | 0.001 | HS | ||
| Hep. – (B+C) | Control | 0.001 | HS | |
| ( ≥ 6 ) | Hep. – (B) | Hep. – (B+C) | 0.000 | HS |
| Control | 0.146 | NS | ||
| Hep. – (B+C) | Control | 0.000 | HS |
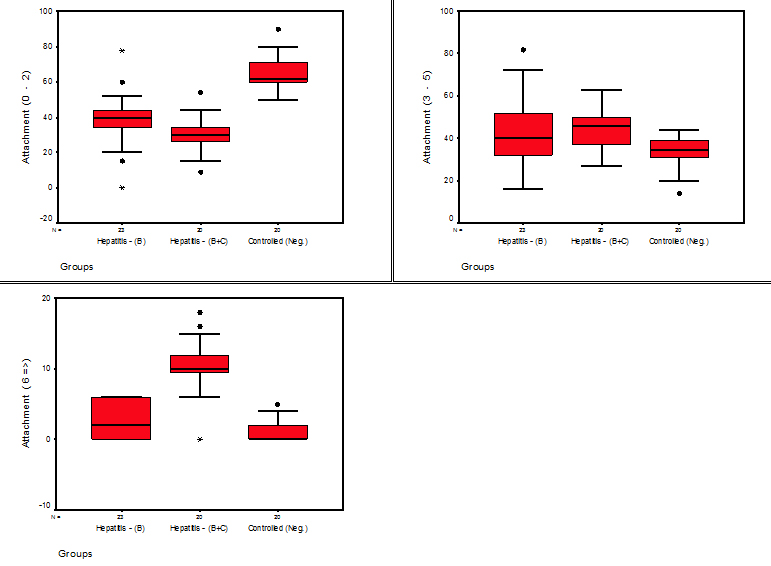 Fig. 3. Stem-Leaf plots for the studied readings of (Attachment) parameters in different groups
Fig. 3. Stem-Leaf plots for the studied readings of (Attachment) parameters in different groupsThere was a highly significant difference between the study and control groups regarding the plaque index results, this could be related to the psychological and physiological condition of the hepatitis patients14,15, when the super infection with either hepatitis B on hepatitis C viruses or vice versa, this super infection could be fetal and life threatening, so the patient had less interest about oral hygiene. This agreed with Grossmann et al 2009 who found many patients with hepatitis C infection present with poor dental health which contributed to worsen their quality of life16.
For the gingival index, there was a highly significant difference between the study and control groups. Also, highly significant difference was revealed when comparing between hepatitis B and hepatitis B and C subgroups. This because of the fact that liver dysfunction can be presented by many oral manifestations like mucosal membrane jaundice, bleeding disorder, petechiae, bruising, gingivitis, gingival bleeding (even in response to minimal trauma), chelitis, xerostomia and oral soreness17. On the other hand, the severity of bleeding tendency, periodontal disease and bad oral hygiene were associated with the risk of hepatitis infection and the discovery of hepatitis markers in the whole saliva18,19. Many oral side effects after interferon and ribavirin (hepatitis C virus infection treatment) were reported. These involve bleeding and swelling of gingiva, toothache, gingivitis, periodontitis, dental caries, taste disorders, mucosal damage, oral lichen planus, oral haemorrhage and dry lips20.
For pocket depth, it was found that hepatitis B and C subgroup had higher number of tooth surfaces with deeper pocket depths than hepatitis B subgroup which had larger number of surfaces with deeper pocket depths than the control groups. Well, these results agreed with what was stated in a small number of studies had been conducted in Japan. These studies established detection of high level of serum alanine aminotransferase (ALT) and ”- glutamyl transferase (GGT), these are usually used for screening of liver disease in asymptomatic patients21. The researcher revealed that the levels of ALT and GGT were increased in patients with periodontal pockets (e”4 mm) as compared to healthy individuals, also, a significant association between periodontal pockets and GGT levels even after adjustment for age, gender, cigarette smoking and alcohol consumption22.
Lastly, we found the hepatitis B and C subgroup had highest number of surfaces suffering from clinical attachment loss than hepatitis B subgroups, and both had larger number of surfaces having clinical attachment loss than the control group. The association between liver cirrhosis and periodontal disease were discussed in many studies. It was determined that the persons with liver cirrhosis showed a tendency to have more clinical attachment loss than the healthy individuals. Notably, significant differences between healthy controls and patients with cirrhosis were found in each age group23. It had also been found that cirrhosis patients exhibited a worse periodontal status compared with healthy volunteers24,25. Furthermore, it was found that patients with cirrhosis for more than 3 years showed greater clinical attachment loss, dental plaque and calculus when compared with less than 3 years cirrhosis patients25. These effects on periodontium by liver diseases and cirrhosis may be due to decreased blood flow of the mucogingival junction and increased level of serum alkaline phosphatase26.
- Buatongsri V, Songpaison Y, Hong prasong N, Phantumvanti P, Clark N The distribution of severe periodontitis in urban (Bankok) and rural (payao province) high risk to stress group of this population. Cu Dent J, 2002; 25:1-4.
- Newman MG, Takei H , Klokkevold PR, Carranza FA. Carranza’s clinical periodontology. 11th ed. Philadelphia : Saunders, 2012;. P.160.
- March PD, Moter A, Devine DA. Dental plaque biofilms Communities conflict and control. Periodontology, 2000-2001; 55(1):16-35.
- Ryder S and Bechingham I. ABC of diseases of liver, pancreas and biliary system. BMJ, 2001; 322(7279): 151-153.
- Koff RS : Hepatitis viruses and vaccines. Infect Dis Clin North Am, 2001; 15(1): 83-95.
- Merk E Merk Manual of Diagnosis and Therapy . Chronic hepatitis. 2002; Home edition.
- Bain VG and Ma M. First principle of gastroenterology, acute hepatitis, 6th Edition, 2006; Ch 14, 138-139.
- Ramsay DB, Friedman M, Borum ML. Does the race or gender of hepatitis C infected patients influence physicians, assessment of hepatitis A and hepatitis B serologic status. South Med J, 2007; 100: 683-5.
- Lavomchy D. Evolving epidemiology of hepatitis C virus. Clin Microbiol Infect, 2011; 17(2): 107-115.
- Pertz JF, Armstrong GL, Famington LA, Hutin YJ, Bell BP. The contributions of hepatitis B virus and hepatitis C virus to cirrhosis and primary liver cancer worldwide. J Hepatol, 2006; 45(4): 529-538.
- Askarian M, Yadollah M, Kuochak F, Danaei M, Vakili V, Momeni M. Precautions for health care workers to avoid hepatitis B and C virus infection. Int J Occup Environ Med, 2011; 2: 191-8.
- Polish LB, Tong MJ, Co RL, Coleman PJ, Alter MJ. Risk factors for hepatitis C virus infection among health care personnel in a community hospital. Am J Infect Control. 1993; 21: 196-200.
- Panov VL. Oral Manifestation of hepatitis C virus. J of IMAB Annual Proceeding (Scientific Paper), 2013; 19(4).
- Hsieh TH, Liu CJ, Chen DS, Chen PJ. Natural course and treatment of hepatitis D virus infection. J formos Med Assoc, 2006; 105(11):869-81.
- Davidovich E, Schwarts Z, Davidovich M, Eidelman E, Bimstein E. Oral findings and periodontal status in children, adolescents and young adults suffering from renal failure. J Clin Periodont, 2005; 32:1076-1082.
- Alsahaf HF. Periodontal health status among patients with chronic renal failure . (clinical and biochemical study). A M.Sc. thesis submitted to the University of Baghdad, 2006; p:72.
- Grossmann Sde M, Teixeira R, de Aguiar MC, de Moura MD, do Carmo MA. Oral mucosal conditions in chronic hepatitis C Brazilian patients: A cross-sectional study. J Public Health Dent, 2009; 69:168-75.
- Panov Vl. Oral cavity – Biosystems and possible source of infectious material. Dissertation, 2010; 189. [in Bulgarian].
- Farghaly AG, Mansour GA, Mahdy NH, Yousri A. Hepatitis B and C virus infections among patients with gingivitis and adult periodontitis: Seroprevalence and public health importance. J Egypt Public Health Assoc, 1998; 73:707-35.
- Carrozzo M. Oral diseases associated with hepatitis C virus infection. Part 1. Sialadenitis and salivary glands lymphoma. Oral Dis, 2008; 14:123-30.
- Oh HJ, Kim TH, Sohn YW, Kim YS, Oh YR, Cho EY, Shim SY, Shin SR, Han AL, Yoon SJ, et al. Association of serum alanine aminotransferase and ã-glutamyltransferase levels within the reference range with metabolic syndrome and nonalcoholic fatty liver disease. Korean J Hepatol. 2011; 17:27–36.
- Morita T, Yamazaki Y, Fujiharu C, Ishii T, Seto M, Nishinoue N, Sasaki Y, Kawato T, Motohashi M, Maeno M. Serum ã-glutamyltransferase level is associated with periodontal disease independent of drinking habits in Japanese adults. Med Sci Monit. 2014; 20:2109–2116.
- Novacek G, Plachetzky U, Pötzi R, Lentner S, Slavicek R, Gangl A, Ferenci P. Dental and periodontal disease in patients with cirrhosis – role of etiology of liver disease. J Hepatol. 1995; 22:576–582.
- Movin S. Relationship between periodontal disease and cirrhosis of the liver in humans. J Clin Periodontol. 1981; 8:450–458.
- Funatsu K, Yamada M, Kawishima Y, Nishida J, Ueno M, Ebihara Y, Mizuno Y, Oda M, Tsuchiya M. Microcirculatory disturbances of oral mucosa and periodontal disease in patients with liver cirrhosis. J Gastroenterol Hepatol. 1989; 4(Suppl 1):99–102.
- Jaiswal G, Deo V, Bhongade M, Jaiswal S. Serum alkaline phosphatase: A potential marker in the progression of periodontal disease in cirrhosis patients. Quintessence Int. 2011; 42:345–348.
© The Author(s) 2018. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


