ISSN: 0973-7510
E-ISSN: 2581-690X
Swine pasteurellosis is an acute infectious disease and is considered to be one of the most economically important diseases in Asia particularly in South and South East Asia leading to huge economic loss in livestock industry. A study was conducted to analyze the histopathological alterations caused by cytotoxins released by Pasteurella multocida serotype B, which caused Swine pasteurellosis in descript as well as nondescript pigs of Chhattisgarh, India. Affected animals exhibited pyrexia, dullness, staggering gait, anorexia, serous nasal discharge and dyspnoea. Typical lesions of oedematous swellings noted in the pharyngeal region. Case fatality rate was 95% in adult animals and 100% in piglets. The causative organism, Pasteurella multocida was isolated and characterized by biochemical tests and the detection of the KMT1, CapB, toxA, and ompH genes was performed using Polymerase Chain Reaction (PCR). Gross lesions included severe pneumonia and haemorrhages in lungs, petechial haemorrhages on serous membranes and other visceral organs. Lymph nodes were enlarged, oedematous and haemorrhagic. The histopathological lesions developed in lungs were typical fibrinous bronchopneumonia, multifocal suppuration and severe thickening of pleura. Heart showed presence of thrombi, haemorrhages and necrosis of myocardium. Hepatic architecture showed server haemorrhages and necrosis while kidneys and spleen showed degenerative and necrotic changes.
Swine, Pathology, Swine Pasteurellosis, Pneumonic pasteurellosis.
Pasteurella multocida is of substantial economic significance in the livestock industry. It is an important principal animal pathogen for over a century and is becoming crucial as human pathogen1 leading to a disease process termed Pasteurellosis. Pasteurella multocida B:2, which causes haemorrhagic septicaemia (HS) of ruminants, is believed to enter the host via respiratory and oral routes. The present study describes the pathological alterations following natural infection in pigs. It is a peracute disease and is considered to be one of the most economically important diseases in Asia particularly in South and South East Asia leading to huge economic loss in livestock industry. The clinical indication of this disease is often characterised by rapid course of high fever, respiratory distress, dullness, depression and followed by death generally within a day of infection2. Pathogenesis of P. multocida is a complex interaction between host specific factors and specific bacterial virulence factors; therefore, understanding the disease pathogenesis is complex and depends on the bacterial strain, the animal model and their interactions3. The key virulence factors identified in Pasteurella multocida include capsule, lipopolysaccharides, surface adhesions, iron regulated and iron acquisition proteins4. The protein toxin from Pasteurella multocida cause necrotic changes in vascular endothelium and induce all of the major symptoms in domestic livestock5.
Studies on outbreaks of swine pasteurellosisin in descript and non-descript pigs was attended in Raipur, Durg and Rajnandgaon district in Chhattisgarh state, India, during rainy season of 2012. The affected animals were off fed and had high fever (41-42°C). About 95% mortality in affected pigs occurred. The infected pigs were examined clinically. Detailed necropsy was conducted and gross lesions were recorded. Blood and tissue samples were collected aseptically for bacterial isolation. Further the isolates were confirmed by biochemical tests and molecular characterization by PCR by using following sets of primers targeting KMT1, CapB, toxA, and ompH genes. The pairs of Pasteurella multocida specific primers were synthesized from Sigma Aldrich Chemicals, USA. Primer sequences are as follows:
F KMT1SP6 5’- GCT GTA AAC GAA CTC GCC AC – 3’
R KMT1T7 5’- ATC CGC TAT TTA CCC AGT GG – 3’
F CAPB 5’-CAT-TTA-TCC-AAG-CTC-CAC-C-3’
R CAPB 5’-GCC-CGA-GAG-TTT-CAA-TCC-3’
F toxA 5’- CGT GAA CTG CGT ACT CAA-3’
R toxA 5’-AAG AGG AGG CAT GAA GAG-3’
F OmpH 5’-ACTATGAAAAAGACAATCGTAG-3
R OmpH 5’-GATCCATTCCTTGCAACATATT-3’
Further, the tissue samples from heart, lungs, liver, spleen, kidneys and Lymph nodes were collected in 10% Formal saline solution for histopathological studies. Morbid tissue samples then processed by routine histopathological techniques and stained with haematoxylin and eosin stains
A total of 13075 samples were collected during the study period. Typical cases of oedematous swellings noted in the pharyngeal region, these swellings spread to the ventral cervical region and brisket of adult pigs. On post-mortem examination, Petechiae and frothy exudates were present in trachea and bronchi. Petechiae were seen all over the serous membranes and peritoneum. There was hydrothorax with accumulation of straw coloured fluid (Fig.1). Endocarditis was observed in the heart, Lungs showed severe consolidation along with the presence of multiple focal haemorrhages. In chronic cases marbled appearance of lungs were seen (Fig.2). Spleens from most of the dead pigs were very much enlarged. Liver and kidneys were swollen and contained petechial haemorrhages. There was presence of haemorrhagic enteritis. Lymph nodes were enlarged, oedematous and haemorrhagic. Microscopically, Lungs showed typical fibrinous bronchopneumonia, multifocal suppuration, Septae were thickened with fibrin, combined with cellular infiltration and edema (Fig.3). Alveoli were filled with fibrinous exudate, erythrocytes and polymorphonuclear cells. Pleura was severely thickened. There were sub-pleural haemorrahages. Heart of some pigs showed presence of thrombi, Haemorrhages and necrosis of myocardium. Glomeruli and kidney tubules showed haemorrhages and necrotic changes. Severe congestion and haemorrhages were observed both in cortex and medulla of lymphnodes. There were haemorrhages and necrosis in Liver. The bacteriological isolation yielded bipolar Gram negative coccobacillary organisms. The isolates of Pasteurella multocida tested by Pasteurella multocida species specific PCR (PM-PCR) were found to give an amplified product of 460 bp size using primer KMT1SP6 and KMT1T7 (Fig.4). Three pairs of primer sets (CAPA, CAPB and CAPD) were used in the capsular PCR analysis of different isolates of Pasteurella multocida. However, only primer pair of CAPB resulted in the amplification of a single band of 590 bp representing the capsular type B of Pasteurella multocida (Fig.5). All the isolates in present study were associated with pneumonia were of capsular type B and yielded same amplicon size of 590 bp. On amplifification using ompH-specifific primer, a PCR product of the expected size of 1.2 kb was obtained.
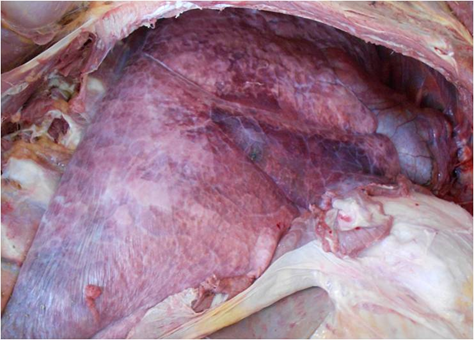 Fig.1. Photograph showing petechial haemorrhages on serous membranes and hydrothorax with presence of straw coloured fluid
Fig.1. Photograph showing petechial haemorrhages on serous membranes and hydrothorax with presence of straw coloured fluid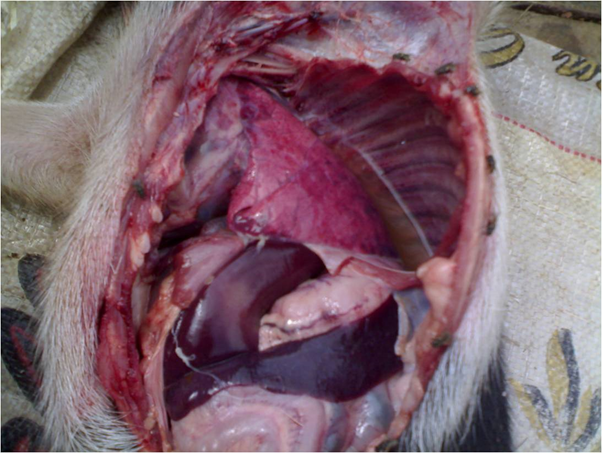 Fig. 2. Photograph showing marbled appearance of lungs, rubbery consistency, thickening of pleura and sub-pleural haemorrhages
Fig. 2. Photograph showing marbled appearance of lungs, rubbery consistency, thickening of pleura and sub-pleural haemorrhages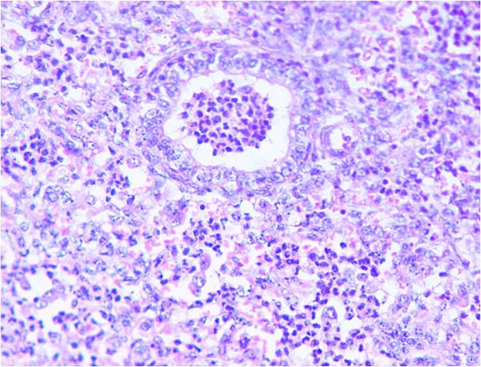 Fig. 3. Micrograph showing fibrinopurulent bronchopneumonia (H&EX400)
Fig. 3. Micrograph showing fibrinopurulent bronchopneumonia (H&EX400) 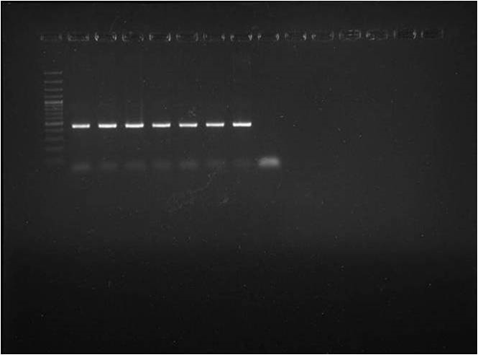 Lane M: 100 plus DNA ladder
Lane M: 100 plus DNA ladderLane 1: Positive control
Lane 2-7: Test Isolates
Fig. 4. Detection of Pasteurella multocida by species specific PCR (PM-PCR)
Pasteurella multocida is considered a commensal organism in the upper respiratory tract and tonsils and causes disease outbreaks in swine, cattle, buffalo, sheep, and goats under extreme environmental conditions, or in animals immunosuppressed by viral infections6. This organism is the most common pathogen isolated from pigs housed under poor husbandry conditions, eg, overcrowding and poor ventilation. The high mortalities observed in many outbreaks of swine pasteurellosis in India7. Similar outbreak of haemorrhagic septicaemia in pig herd has been reported earlier with variable degree of necrotic myositis. The ulcerative skin lesions were absent during recent outbreak. Oedematous swellings were noted in the pharyngeal region of some of the infected pigs which is the predominant manifestation cattle and buffaloes. P. multocida type B:2, is responsible for hemorrhagic septicemia in dairy cattle and buffalo and many outbreaks are reported every year from all over India suggesting that this serotype is transmitted between bovine species and swine8. The protein toxin from Pasteurella multocida cause necrotic changes in vascular endothelium and induce all of the major symptoms in infected animals (5). The pathological modifications viz. generalized lymphadenopathy, submandibular and brisket edema, acute fibrinous pneumonia, proctitis, acute colitis, hemorrhagic typhilitis, chronic bronchopneumonia with abscessation and pleuritis have been found to be common lesions of pneumonic pasteurellosis in pigs9. Post-mortem findings and pathological changes suggestive of swine pasteurellosis have also been reported by other workers.8, 9
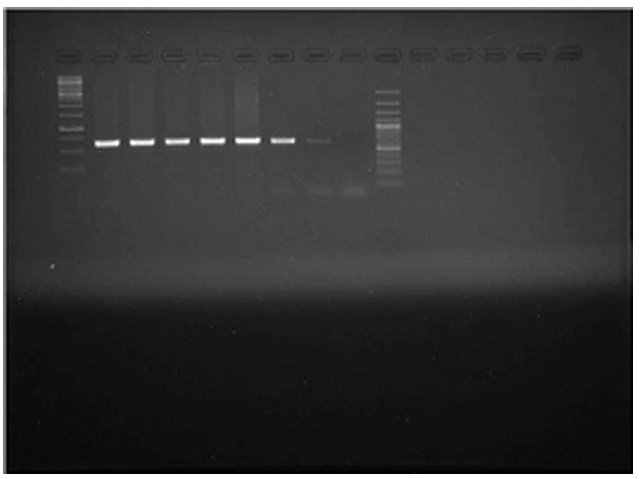 Lane M1 & M2: 100+ bp DNA ladder, Lane 1: Positive control, Lane 8: Negative control , Lane 2 to Lane 7 test isolates
Lane M1 & M2: 100+ bp DNA ladder, Lane 1: Positive control, Lane 8: Negative control , Lane 2 to Lane 7 test isolatesFig. 5. Detection of Capsular type of Pasteurella multocida by PC
The outbreak of pasteurellosis is attributed to impairment of host defences mechanism, strain and virulence of causative organism and various other physiological and environmental stress factors. Incidence of swine pasteurellosis in present case appears to have occurred due to high relative humidity during rainy season which is one of the important environmental stress factors. Thus, due to its highly contagious nature and high mortality rate, the disease has played a major role in huge economic loss in pig farmers of Chhattisgarh.
ACKNOWLEDGMENTS
Authors are thankful to the Dean, Veterinary College, Anjora, Durg, Chhattisgarh and Director, ICAR- National Research Centre on Equines, Hisar, Haryana for providing the necessary facilities to carry out the research. The authors declare that there is no interest of conflict.
- Biberstein, E.L. Handbook Series in Zonooses. In: Stoenner, H., M. Torten and W.Kaplan (Eds.), CRC Press Inc., Boca Raton, Florida,1979; pp 495-514.
- Boyce, J.D., Harper, M., Wilkie, I.W., Adler, B. Pasteurella. In: Pathogenesis of Bacterial Infections in Animals, Gyles, C.L., J.F. Prescott, G. Songer and C.O. Thoen, (Eds.).,Wiley-Blackwell, Ames, ISBN-10: 0813812372,2010; pp 664-664.
- Hawari, A.D., Obeidat, M., Awaisheh, S.S., Al-Daghistani, H.I., Al-Abbadi, A.A. Prevalence of mastitis pathogens and their resistance against antimicrobial agents in Awassi sheep in Albalqa province of Jordan. Am. J. Anim. Vet .Sci. 2014; 9:116-121. DOI:10.3844/ajavsp.2014.116.121.
- Kharb, S., Charan, S. Mouse model of haemorrhagic septicaemia: Dissemination and multiplication of Pasteurella multocida B: 2 in vital organs after intranasal and subcutaneous challenge in mice. Vet. Res. Commun. 2013; 37: 59-63. DOI: 10.1007/s11259-012-9547-5
- Faez, F.J., Affandi, S.A., Osman, A.Y., Adamu, L., Zamri-Saad, M Clinico-pathological features in mice following oral exposure to Pasteurella multocida type B: 2. J. Agric. Vet. Sci. 2013; 3: 35-39. DOI: 10.9790/23800343539.
- Aiello, S.E., ed. Merck Veterinary Manual. 6th ed Whitehouse Station, New Jersey: Merck and Co Inc.1998; pp 509-512.
- Zhao, Guosong, Thomas, Halbur, Duane, Pankratz, C. Colonization of oropharynx and nasal cavity of CDCD pigs by a nontoxigenic strain of Pasteurella multocida type D. J Swine Health Prod. 1995; 3(3):113-115.
- Kumar, H., Mahajan, V., Sharma, S., Singh, A., Arora, R., Banga, A.K., Verma, H.S., Kaur, S., Kaur, K., Meenakshi, P., Sandhu, K.S.) Concurrent pasteurellosis and classical swine fever in Indian pigs. J. Swine Health Prod. 2007; 15(5):279–283.
- Ghosh, R.C., Hirpurkar, S.D., Mondal, M. An outbreak of swine pasteurellosis in Chhattisgarh. Ind. J. Vet.. Pathol. 2011; 35(1):87-88.
© The Author(s) 2017. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


