ISSN: 0973-7510
E-ISSN: 2581-690X
Candida is one of the most common microorganisms forming biofilms. The present study was aimed to evaluate biofilm formation in different clinical Candida isolates and their adhesion to urinary catheter materials. The study comprised 150 Candida isolates from clinical samples. Colonies of Candida were identified to species level using standard tests. Biofilm formation was studied on microtitre plates. Adhesion assay for the biofilm producers was performed on urinary catheter. Statistical Analysis was done by Chi square test. From 150 isolates collected; highest number of candida isolates were recovered from blood culture (44%) followed by urine (22.7%). Though C. albicans was the commonest isolate (44%), C. krusei was the most common species isolated from blood cultures (42 of 66; 63.6%). A total of 41 Candida isolates were found to produce Biofilm (27.3%; 41/150). The proportion of the biofilm producers in blood (27.3%), urine (32.4%) and exudates (29.6%) was almost identical. Lowest proportion of biofilm producers was found on dentures (17.4%) (Non-significant; p value 0.53). Higher biofilm producing tendency in urinary isolates may be contributory to their potential to cause UTI in catheterized patients. Higher isolation of C. krusei from blood samples was a noteworthy finding.
Candida, Biofilm, adhesion, urinary catheter.
Biofilms are structured microbial communities attached to surfaces. They secrete exopolymeric material often referred to as matrix1. Candida species are one of the most common fungi producing biofilms. Ability of candida to produce hyphal structures provides architectural integrity to the biofilm. The structure of biofilm helps candida to survive harsh environmental conditions. Once lodged, Candida cells are capable of forming matured biofilms in 20hrs 2. Surfaces of implants, temporary intravascular devices or removable aids like denture frequently provide suitable surfaces for biofilm formation. Biofilms are difficult to dislodge. They are the continuous source of infection leading to disseminated infections or septicemia 3. The fungal biofilms are becoming irksome clinical and economical problem with higher mortality rates. Infections due to biofilm forming Candida are difficult to manage and the hospital stay in these cases is usually prolonged 4.
Of late, the non-albicans Candida (NAC) are frequently reported causing clinical infections and a number of these isolates are shown to produce biofilms. In fact, some workers have reported higher biofilm production in NAC isolates from blood stream infections.
Aim and Objectives
The present study was aimed at testing for two important factors influencing pathogenicity of Candida isolates, i.e., identification of biofilm producers and adhesion to urinary catheter materials, with the following objectives.
- Isolation and speciation of Candida isolates from clinical specimens.
- Detection of biofilm producers among the clinical Candida isolates.
- Evaluate adhesion of Candida isolates to urinary catheter material.
- Comparison of the biofilm formation in both C. albicans and NAC spp. isolated from clinical specimens.
The study was begun after obtaining permission from the institutional ethical committee. We included 150 Candida species isolated from clinical samples received at Clinical Microbiology laboratory of our hospital, from November 2014 to November 2015. The Candida isolates were identified by standard methods including Gram’s stain, germ tube test, chlamydospore formation; carbohydrate assimilation and temperature sensitivity test 5. The speciated candida isolates were stored on Sabouraud’s Dextrose Agar (SDA) slants at 4oC for further study. The identified Candida isolates were subjected to biofilm formation and adhesion tests. The pure isolates were subcultured on SDA (Hi Media, Mumbai) and a single colony was transferred to tubes having Sabouraud’s Dextrose broth. The tubes were incubated in a shaking incubator at 37 ºC at 120 rpm for 18 hrs. Cells were harvested by centrifugation and washed thrice with sterile phosphate-buffered saline (PBS, pH: 7.2). The yeast cells were re-suspended in fresh buffer and standardized to a concentration of 1×107 to 1×106 cells/mL. Estimation of Biofilm formation in different Candida species.
Biofilm formation was studied on sterile, polystyrene; flat-bottom 96-well microtitre plates (Laxbro, India) using the method explained by Taff et al., (2011) 6.
Incubation of Candida for biofilm formation
Each well of the microtitre plate was filled with 100 µl of a standardized cell suspension (107 cells/ml). The plate was incubated for 1.5 hours at 37 oC at 75 rpm to allow the yeast cells adhere to microtitre wells. A known biofilm producing Candida albicans isolate and a known biofilm negative Candida albicans isolate from our laboratory were used as positive and negative controls. With each test run, 100 µl of PBS was added to a well and 100 µl of SD broth were used as blank and media control respectively. After the incubation, the supernatant was carefully removed and the wells were gently washed two times with 200 µl of PBS to remove unbound cells. The washed wells were filled with 100 µL SD broth, and the plates were incubated at 37°C in a Shaking incubator at 75 rpm. The SD broth was replaced with fresh medium two times a day and incubation was continued up to 48 hours. The isolates were tested in triplicate. All the inoculations and media replacements were done in a bio-safety cabinet Class 2 A 2.
Estimation of biofilm development by Crystal violet assay
After the completion of incubation the wells were washed twice with 200 µL of PBS and allowed to dry at room temperature for 45 minutes. The washed wells were stained with 100 µL of 0.4% crystal violet solution for 45 minutes. Each well was washed manually four times with 350 µL of sterile distilled water to remove excess crystal violet. For de-staining 200 µL of 95% ethanol was added to the wells and plates were kept at room temperature for 45 minutes. 100 µL of solution from the wells was transferred to wells in a new microtitre plate. The optical density was measured at 600 nm (Tecan, Sunrise, Austria).
Adhesion assay on the urinary catheter
Adhesion assays were performed using a modification of the technique described by Uzunoglu et al., (2014) 7. Briefly, urinary catheter materials (PVC, RUCH, Germany) were cut to get several 1 cm pieces using sterile surgical blades inside the biosafety cabinet. The pieces were put in a Petri plate. The standardized C. albicans cell suspension in PBS (1 × 106 cells/mL) was added to each Petri plate to cover the pieces and incubated for 2 hours at 37 ºC to facilitate adhesion. After incubation, the pieces were gently washed twice with 5 mL PBS to remove the unadhered cells.
Counting of adhered Candida cells
Washed catheter pieces were put in a sterile test tube consisting 5ml normal saline and then were pulse agitated using a vortex mixer at the maximum speed for 30 seconds and left at room temperature for 5 minutes and 100µl of the suspension was spread uniformly on SDA plates. The plates were incubated at 37oC for 24 hours and colonies were counted. The colony forming units (CFU) per millilitre for Candida were calculated.
All the 41 biofilm producing Candida isolates and 40 non-biofilm producers Candida isolates were studied for adhesion to the urinary catheter material.
Statistical Analysis
Chi square test was used for statistical analysis to know the significance of test results.
Isolation of Candida from clinical specimens with their speciation
A total of 150 clinical Candida isolates were recovered during in the study period. Table 1., shows sample wise distribution of the isolates. Of the 150 isolates 66 (44%) were C. albicans and 84 (56%) were NAC. Highest number of isolates were obtained from blood samples (44%) followed by urine samples (22.7%). Though overall isolation of NAC was higher than C. albicans; in all the samples except from blood, C. albicans isolation was higher compared to NAC. C. krusei (37.3%) was the commonest isolate among the NAC group and also from each sample type. The isolation of C. krusei from blood (36.7%), however, outnumbered the other sample groups. Other NAC species obtained were C. tropicalis, C. guilliermondii, C. glabrata, C. parapsilosis, C. lusitaniae, C. kefyr and C. dubliniensis.
Table (1):
Isolation of Candida species from various clinical isolates biofilm producing and non biofilm producing.
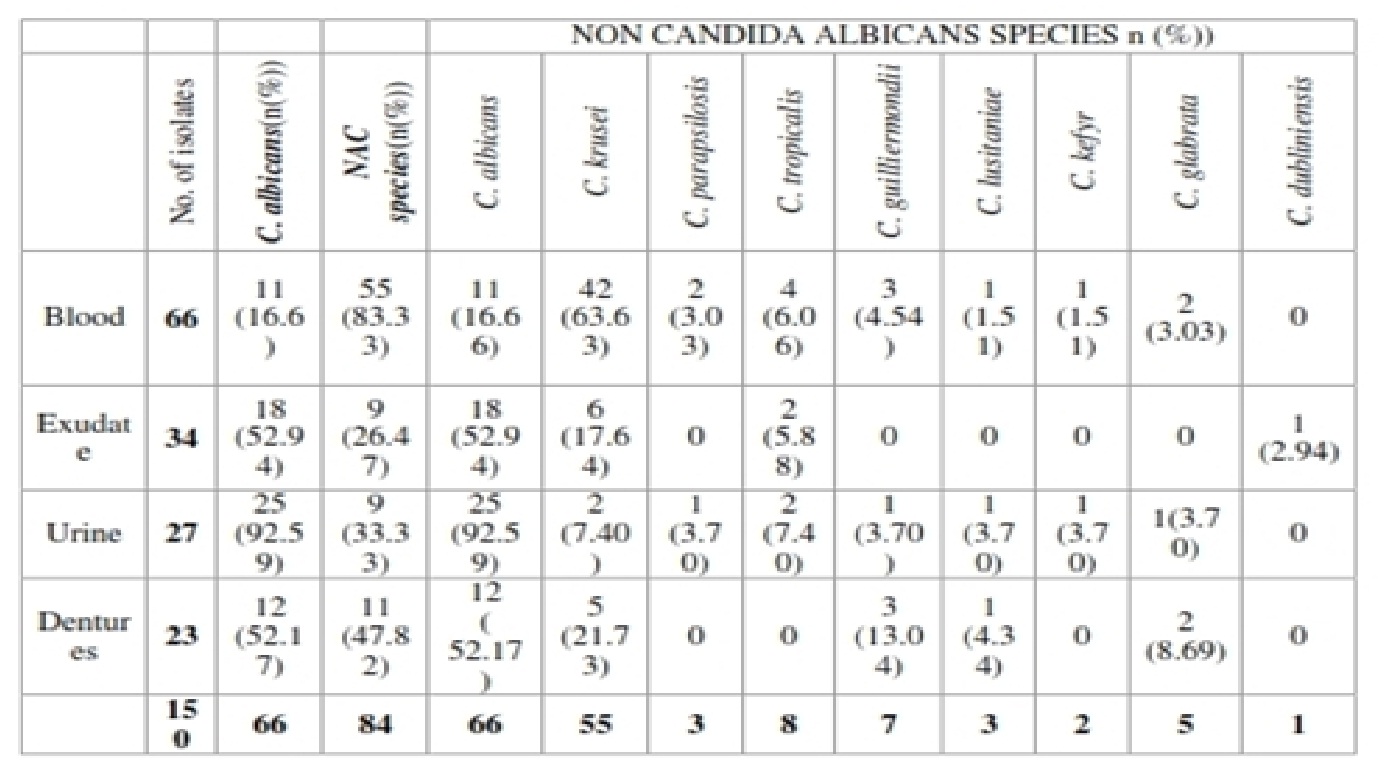 Majority of the isolates in the present study were from blood (66/150) followed by 34 from urine, 27 from exudates and 23 from dentures. C. krusei was the commonest isolate from blood. Out of the 66 blood isolates 42 were C. krusei while C. albicans were 11. C. albicans was the commonest isolate from urine, exudates and dentures. C. krusei was the second common isolate in each sample type. A comparison of proportion of C. albicans and NAC isolation was done by Chi2 test. For blood the difference in the proportion was highly significant (Chi square is 24.79, p<0.001).
Majority of the isolates in the present study were from blood (66/150) followed by 34 from urine, 27 from exudates and 23 from dentures. C. krusei was the commonest isolate from blood. Out of the 66 blood isolates 42 were C. krusei while C. albicans were 11. C. albicans was the commonest isolate from urine, exudates and dentures. C. krusei was the second common isolate in each sample type. A comparison of proportion of C. albicans and NAC isolation was done by Chi2 test. For blood the difference in the proportion was highly significant (Chi square is 24.79, p<0.001).
Biofilm production
The biofilm production by all the 150 isolates was assessed by the method described by Taff et al. (2011) 6. As shown in Table 2. among total Candida isolates, 41 (26.7%) showed biofilm production. Of 66 C. albicans isolates 20 produced biofilm (30.3%) as against 21 of the 84 (25%) NAC showing biofilm production. C. krusei (12/55; 21.8%) was the highest biofilm producer among the NAC isolates. C. dublinensis, C. glabrata and C. kefyr did not show biofilm production. C. albicans was the commonest biofilm producer from all the samples except blood. In urinary biofilm producers 7 of 11, from exudates 7 of 8 and from dentures, 3 of 4 were C. albicans. Thus, except for blood C. albicans outnumbered NAC in the biofilm producers. In blood, of 18 biofilm producers 10 were C. krusei.
Table (2):
Biofilm producers among the clinical isolates.
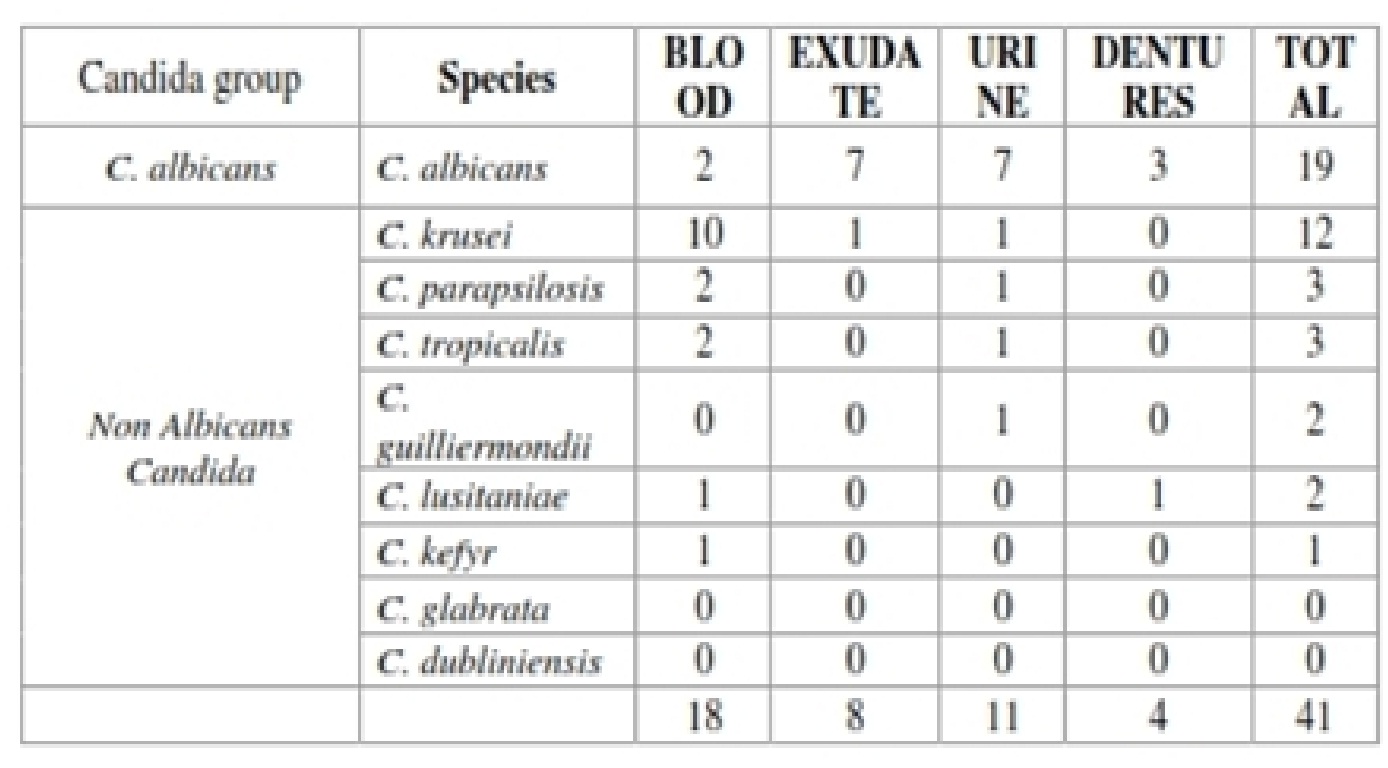 The proportion of the biofilm producers in urine (11/34; 32.4%) and exudates (8/27; 29.6%) was almost identical followed by 27.3% blood isolates showing biofilm production. Lowest proportion of biofilm producers was found on dentures (17.4%). C. albicans isolates from exudates were more frequent biofilm producers (7/18; 38.9%) while for blood, C. krusei were more common in biofilm production.
The proportion of the biofilm producers in urine (11/34; 32.4%) and exudates (8/27; 29.6%) was almost identical followed by 27.3% blood isolates showing biofilm production. Lowest proportion of biofilm producers was found on dentures (17.4%). C. albicans isolates from exudates were more frequent biofilm producers (7/18; 38.9%) while for blood, C. krusei were more common in biofilm production.
On statistical analysis (Chi2 test), there was no significant difference in percentage of biofilm producers among different clinical specimens (p=0.28).
Adhesion pattern in biofilm producing Candida isolates
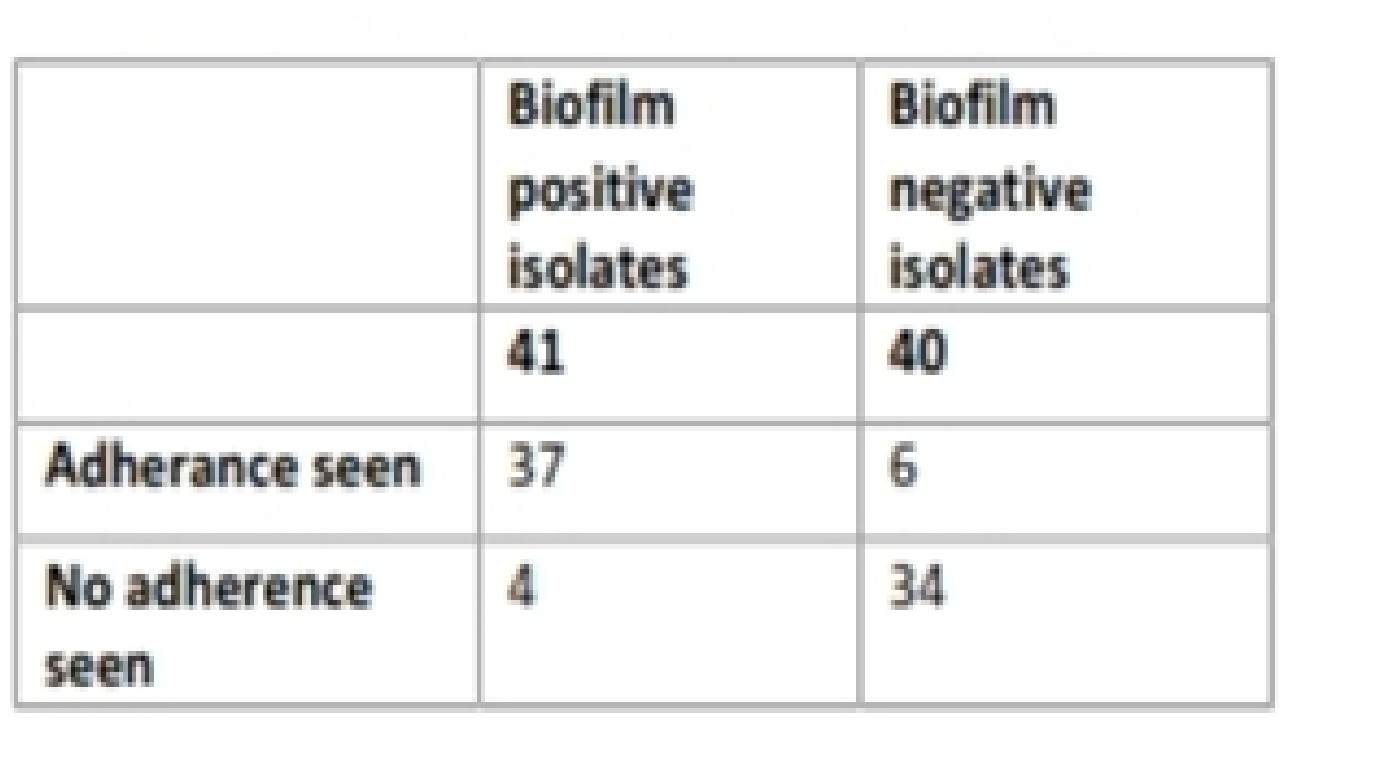
All the 41 biofilm producing isolates were subjected to adhesion study 7. The details are shown in Table 3, Graph 1. Both C. albicans and NAC showed adherence to urinary catheter material, the C. albicans isolates gave higher CFUs compared to NAC isolates. Species and sample wise prevalence of adhesion is shown in Table 3. There were no quantitative differences among different species on comparison of their CFUs except two C. albicans isolates from dentures showing strong adherence. Mean adhesion in all different clinical isolates was shown to be significant (p = 0.039) by Kruskal Wali’s test. However mean adhesion between the clinical groups compared by Mann-Whitney test was non significant.
Table (3):
Adhesion pattern in biofilm producing Candida isolates.
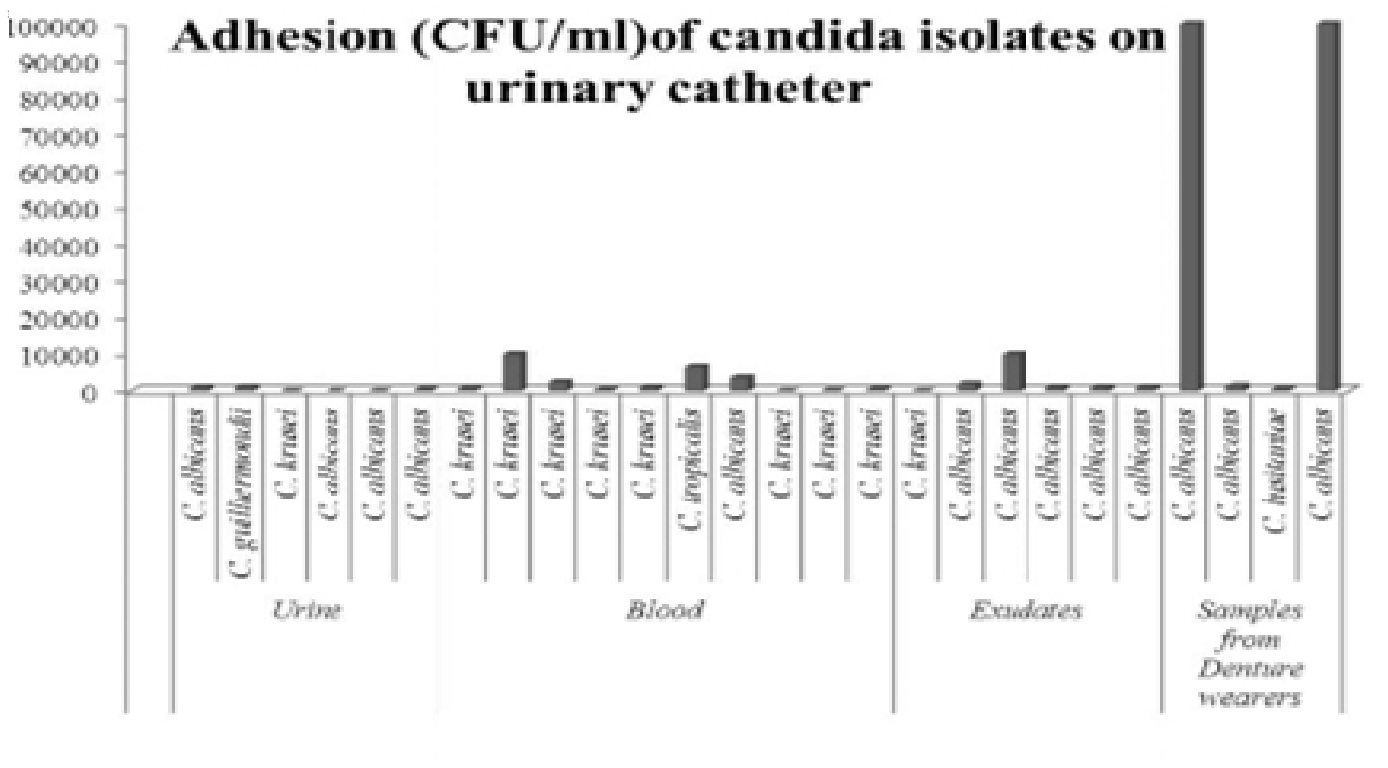
Graph 1: Adhesion pattern in biofilm producing Candida isolates
Adhesion pattern in non-biofilm producing Candida isolates
The results were further compared with the adhesion pattern of non-biofilm producing Candida isolates. A total of 40 non-biofilm isolates were subjected to adhesion test (table 3). Only 6 of the 40 isolates showed adherence. Association of adherence to biofilm production in biofilm positive and biofilm negative isolates showed very high significance p value < 0.00000.
Candida infections are gaining a lot of importance of late. Candida albicans was, so far, considered to be the pathogenic species and there was a tendency to neglect the non albicans isolates (NAC). The scenario is changing fast. Influenced by several predisposing factors like use of immunosuppressive agents, antibiotics and long term hospitalization etc. Candida infections have become frequent 8,9,10. Among the virulence factors, biofilm production is considered as an important virulence attribute 11. The present study was conducted to speciate Candida isolates from clinical specimens and to study their adherence to urinary catheter material and also to test the isolates for biofilm production. The study was conducted at Department of Microbiology, SDM College of Medical Sciences and Hospital, Dharwad from November 2014 to November 2015. A total of 150 Candida isolates from blood, urine, exudates and denture surfaces were speciated (Table 1). NAC (56%) outnumbered C. albicans (44%) and a total of 8 different NAC species were encountered in this work. C. krusei was the commonest NAC isolate and was the predominant isolate from blood. Except C. krusei, all the other seven isolates tallied in single figure.
Candida isolates from blood should not be neglected and dismissed as contaminants 12. Frequent isolation of C. krusei, especially from neonatal samples, was alarming. We, therefore, did environmental sampling of neonatology unit. Samples from hands of neonatology staff were also collected (Data not shown). However, none of these samples yielded Candida species. Vinitha and Mamata (2011), showed C. krusei as the most prevalent NAC isolate (60. 36%) 10. Similarly studies from Mujika et al., 2004 showed prevalence of C. krusei from respiratory tract samples 13. C. krusei, though less virulent, is recognized for its fluconazole resistance 14. In all the samples other than blood C. albicans were proportionally higher than NAC. Except for blood the comparison of proportion of C. albicans and NAC was non significant for all the samples. For blood the difference was highly significant (p < 0.006).
We observed high Candida isolation from newborns rather than from other age groups. Wadile and Bhate (2015) showed isolation of Candida in 32.26% of neonatal septicemia cases among which NAC accounted for 35% 15. Juyal et al. (2013) found 80.30% NAC species in neonatal candidemia, where C. parapsilosis (25.0%) and C. tropicalis (21.97%) were the most predominant species 16. The prevalence of NAC species could be dependent upon geographical area and type of clinical specimen. During our previous work we had speciated 534 Candida isolates from oral cavity and found C. guilliermondii as the second common isolate 17. Geographical variations in the distribution of Candida species have been described. It is also reported that a species preponderant in a geographical area may subsequently be replaced by other species 2.The increased number of NAC isolation and high prevalence of C. krusei in the present study suggests the need for speciation of clinical Candida isolates.
All the isolates were tested for biofilm production by tissue culture plate method (TCP). Biofilm producing isolates were marginally higher in C. albicans group. Out of 42 C. krusei from blood 10 showed biofilm production. Biofilm producing ability is an important virulence attributes. Biofilm formation is known to be the important virulence factor in the infection process 18. Among the 150 Candida isolates of our study 41 were positive for biofilm production (26.7%). In the studies done by Mohandas and Ballal ., 2007 and 2011 they showed 50 and 60% of the isolates being biofilm producers respectively 10, 19. Udayalaxmi et al., (2014), showed that 70% of the isolates produced biofilm 20. In our study, the proportion of the biofilm producers in urine and exudates was almost identical (urine – 29.4% and exudates – 29.6%) followed by blood giving 27.3% biofilm producers. Lowest proportion of biofilm producers was found on dentures (17.4%). The exudates samples obtained were commonly from in association with various intravenous tubes like endotracheal tubes etc. Similarly a few studies claim that some isolates are more prone to adhere and form biofilm depending on their isolation site (Shin et al., 2002; Hasan et al., 2009; Silva et al., 2010a, 2011; Mohandas and Ballal, 2011). In a study done at Manipal, India by Mohandas and Ballal (2011).,C. albicans isolates recovered from blood exhibited lower percentage of biofilm positivity. In the study of done by Udayalaxmi et al, (2014), biofilm production was more in Candida isolated from urine samples in comparison to vaginal isolates20.
Except for 5 samples from dentures none of the other samples gave mixed Candida growth.
The samples, isolates and their biofilm forming ability were statistically analyzed. However, there was no significant relationship between these factors.
Adhesion is an important attributes of pathogenicity of Candida . Adhesion facilitates colonization which is the initial step for most pathogens proceeding to clinical infections. The materials of artificial devices could be critical factor in deciding the success of colonization. Adhesion on the surface is the initial event in Biofilm formation 22 .However, the materials of artificial devices could be critical factor in deciding the colonization 23. We, therefore, studied adhesion in all the 41 biofilm producers recovered in the study. We also subjected 40 biofilm negative isolates with specimen-wise matching numbers for adhesion.
Urinary catheters are one of the most common devices related to Candida infections 24, 25. As this is a routine device used in the management of various conditions, we chose urinary catheter material to study adherence. The catheters used were made of PVC, RUCH, Germany. Of the 41 biofilm producers 37 isolates showed adherence where CFUs ranged from 20 to 105 per ml. The highest number of CFU of 105 , was seen in two C. albicans isolates from dentures. The maximum CFUs yielded by biofilm producers were 105/ml whereas for non biofilm producers the maximum CFU number was only 102/ml. Only six of the 40 non biofilm producers tested showed adherence.
Though adhesion is facilitated by biofilm production, several other factors including zeta potential, contact angle, mucin, other bacteria in the niche and hydrophobicity play important role in adhesion 26, 18. Studies by Tamura and Gasparetto (2003) showed adherence of C. albicans was significantly greater than C. parapsilosis on latex, but it was comparable on silicon 23. Catheter related candidemia, age related Candida infections, etc. are increasing in recent years 1, 8, 9, 10, 13. Literature suggests that, almost invariably, an implanted device such as an intravascular or urinary catheter or endotracheal tube, is associated with Candida infections and a biofilm can be detected on the surface of these device 20, 24, 25. Present study didn’t reveal any significant association between in biofilm production of the isolates and the clinical samples from which they were coming. Though, adherence to the urinary catheter and biofilm forming ability significantly co-existed, there was no significant difference in adherence between the different Candida species observed.
- D’Antonio D, Romano F, Pontieri E, Fioritoni G, Caracciolo C, Bianchini S, Olioso P, Staniscia T, Sferra R, Boccia S, Vetuschi A, Federico G, Gaudio E, Carruba G. Catheter-related candidemia caused by Candida lipolytica in a patient receiving allogeneic bone marrow transplantation. J Clin Microbiol. 2002; 40(4):1381-6.
- Guinea J. Global trends in the distribution of Candida species causing candidemia. Clin Microbiol Infect. 2014; 20(6):5-10.
- Shin JH, Kee SJ, Shin MG, Kim SH, Shin DH, Lee SK, Suh SP, Ryang DW. Biofilm production by isolates of Candida species recovered from nonneutropenic patients: comparison of bloodstream isolates with isolates from other sources. J Clin Microbiol. 2002; 40(4):1244-8.
- Ramage G, Mowat E, Jones B, Williams C, Lopez-Ribot J. Our current understanding of fungal biofilms. Crit Rev Microbiol. 2009; 35:340 – 55.
- Larone DH., Medically Important Fungi: A Guide to Identification, 4th Edition ASM. Washington D.C. 2002.
- Taff HT, Nett JE, Andes DR. Comparative analysis of Candida biofilm quantization assays. Med Mycol. 2012; 50(2):214-8.
- Uzunoglu E, Yildirim Bicer AZ, Dolapci I, Dogan A. Biofilm-forming ability and adherence to poly-(methyl-ethacrylate) acrylic resin materials of oral Candida albicans strains isolated from HIV positive subjects. J Adv Prosthodont. 2014; 6(1):30-4.
- Ramage G, VandeWalle K, Wickes BL, López-Ribot JL. Biofilm formation by Candida dubiliensis. J Clin Microbiol. 2001; 39:3234–40.
- Mohan das V, Ballal M. Proteinase and phospholipase activity as virulence factors in Candida species isolated from blood. Rev Iberoam Micol. 2008; 25(4):208-10.
- Mohandas V, Ballal M. Distributionof Candida species in different clinical samples and their virulence: biofilm formation, proteinase and phospholipase production: a study on hospitalized patients in southern India. J Glob Infect Dis. 2011; 3(1):4-8.
- Hasan F, Xess I, Wang X, Jain N, Fries BC. Biofilm formation in clinical Candida isolates and its association with virulence. Microbes Infect. 2009; 11:753–61.
- Bhatt M, Sarangi G, Paty BP, Mohapatra D, Chayani N, Mahapatra A, Das P, Sahoo D. Biofilm as virulence marker in Candida species in Nosocomial blood streaminfection and its correlation with antifungal resistance. Indian J Med Microbiol. 2015; 33: 112-4.
- Mujika MT, Finquelievich JL, Jewtuchowicz V, Iovannitti CA. Prevalence of Candida albicans and Candida non-albicans in clinical samples during 1999-2001. Rev Argent Microbiol. 2004; 36:107–12.
- Krcmery V, Barnes AJ. Non-albicans Candida spp. causing fungaemia: pathogenicity and antifungal resistance. J Hosp Infect. 2002; 50(4):243-60.
- Wadile RG, Bhate VM. Studyof clinical spectrum and risk factors of neonatal candidemia. Indian J Pathol Microbiol. 2015; 58(4):472-4.
- Juyal D, Sharma M, Pal S, Rathaur VK, Sharma N. Emergenceof non-albicans Candida species in neonatal candidemia. N Am J Med Sci. 2013; 5(9):541-5.
- Jain PA, Kulkarni RD, Ajantha GS, Shubhada C. A comparative evaluation of oral candida carriage in HIV-infected individuals and HIV seronegative healthy individuals in North Karnataka. J Biosci Tech. 2011; 2: 232–7.
- Silva-Dias A, Miranda IM, Branco J, Monteiro-Soares M, Pina-Vaz C, Rodrigues AG. Adhesion, biofilm formation, cell surface hydrophobicity, and antifungal planktonic susceptibility: relationship among Candida spp. Front Microbiol. 2015: 12;6:205. doi: 10.3389/fmicb.2015.00205.
- Vinitha M, Mamata B. Biofilm as Virulence Marker in Candida Isolated from Blood. World J Med Sci. 2007; 2: 46-48.
- Udayalaxmi, Jacob S, D’Souza D. Comparison Between Virulence Factors of Candida albicans and Non-Albicans Species of Candida Isolated from Genitourinary Tract. Clin. Diagn. Res. 2014; 8(11):DC15-7.
- Silva S, Henriques M, Oliveira R, Williams D, Azeredo J. In vitro biofilm activity of non-Candida albicans Candida species. Curr Microbiol 2010a; 61: 534– 40.
- Klotz SA, Drutz DJ, Zajic JE. Factors governing adherence of candida species to plastic surfaces. Infect Immun.1985; 50(1):97-101.
- Tamura NK, Gasparetto A, Svidzinski TI. Evaluation of the adherence of Candida species to urinary catheter. Mycopathologia. 2003; 156(4):269-72.
- Julia D. Candida biofilms and their role in infection.Trends Microbiol. 2003; 11:30- 36.
- Sardi JCO, Scorzoni L, Bernardi T, Fusco-Almeida AM, Mendes Giannini MJ. Candida species; Current epidemiology, pathogenicity, biofilm formation, natural antifungal products and new therapeutic options. J Med Microbiol. 2013; 62:10-24.
- Karaagaclioglu L, Can G, Yilmaz B, Ayhan N, Semiz O,Levent H. The adherence of Candida albicans to acrylic resin reinforced with different fibers. J Mater Sci Mater Med. 2008; 19: 959-63.
© The Author(s) 2017. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


