ISSN: 0973-7510
E-ISSN: 2581-690X
Palmitic acid is used in cosmetics as well as skin care products, and its extraction from plants remains major source of production. The conventional process of extraction is quite laborious and may lead to loss of plant diversity. Therefore, the current investigation aims at producing palmitic acid from coconut oilcake using probiotics isolated from marine fish. A total of 18 bacterial strains were isolated from Indian oil Sardine fish (Sardinella longiceps) and Obtuse barracuda fish (Sphyraena obtusata). Among various strains isolated, Mm12 strain was able to synthesize 60.57 mg/100mL fatty acids. The production of palmitic acid was optimized using RSM with the most appropriate values of the tested parameters by using Design-Expert 13 to resolve the correlation coefficients binomial equation’s reciprocal matrix as temperature 10°C, pH 5.5, and 40 g oil cake substrate. The highest quantity of palmitic acid that could be produced under ideal circumstances has been estimated to be 19.97 mg/100 mL which was confirmed by GC–MS fatty acid methyl ester (FAME) analysis. Production of palmitic acids from the oil cake was not only a novel method of valorizing industrial waste using probiotic bacteria and but also a promising method of producing palmitate using cheaper substrates.
Probiotics, Fatty Acids, Coconut Oilcake, Palmitic Acid, GC–MS FAME Analysis
About 6–10% of fatty acids found in oils, extracted from plants such as soy, safflower, peanut, or sunflower, are made up of palmitic acid.1 Over the past 20 years, there has been a scarcity of palmitic acid produced through the microbial conversion of agro-industrial wastes. The fatty acids synthesized from microbes are equally good as the many natural fatty acids identified in oil cakes. Bacteria are capable of producing palmitic acid and they grow easily than any other organisms. Even though the microbial world has a great diversity of fatty acids,2 there may be benefits for many of the minor fatty acids that are not widely accessible.3
Palmitic acid, myristic acid, lauric acid, and stearic acid are prevalent among animal (dairy products, fish) and vegetable fats (palm kernel, coconut, and nutmeg). These contain more unsaturated fatty acids, making them the preferred oils for cooking at high temperatures.4 The dietary saturated fatty acid produced by endogenous synthesis is palmitic acid (PA) (16:0). Palmitate is the component of cell membranes, secretory, transport lipids and plays an important role in protein palmitoylation and palmitoylation signaling molecules.5
About 20–30% membrane phospholipids (PL) and triglyceride are made up of PA.6 A significant portion of PA is present in dairy, cocoa butter, palm oil, and olives oil. Similarly, 20–30% of total lipids in breast milk are composed of PA,7 and daily consumption of PA is required between 20–30 g.8 The main constituents of fats and oils, triglycerides, and wax esters contain fatty acids as essential constituents. However, phospholipids and glycolipids are important; especially the latter compounds are well known for their interesting biological activities. Therefore, marine organisms also contain eicosapentaenoic acid and docosahexaenoic acid, as well as the essential polyunsaturated fatty acids (PUFAs). Recently, many unique chemical structures of fatty acids have been discovered, especially in sponges and prickly pears.9
Dehydrated coconut meat can be used to produce coconut cake that are produced 1.931 million tons worldwide in 2013–2014, with India second only to the Philippines (230,000 tons).10 The common use of copra cake as an animal feed formulation has been compromised by tightening regulations on aflatoxin B1 and by having lower protein content (20–25%) than other oil cakes, which results in a low nutritional value. The raw material (copra cake) fatty acid analysis demonstrated that saturated fat accounted for the majority (86.2%), monounsaturated fat accounted for 10.3%, and polyunsaturated fat accounted for 3.5%. The saturated fraction is primarily made up of myristic acid (C14:0, 21.58%) and lauric acid (C12:0, 44.66%). These are followed by palmitic acid (C16:0, 11.53%), caprylic acid (C8:0, 3.62%), capric acid (C10:0, 2.19%), and stearic acid (C18:0, 2.63%). To produce their corresponding alkaline salts, which are then used as emulsifiers, emollients, and lubricants in numerous cosmetic cakes, pastes, creams, and soaps, oleic, lauric, palmitic, myristic, and stearic acids are frequently employed as intermediates. Skincare, shaving, non-shaving preparations and personal hygiene products include lauric, palmitic and myristic acids. In addition to being used as reagents in the creation of other food-grade additives, oleic, lauric, palmitic, myristic, and stearic acids are used in food as plasticizers, lubricants, binders, and defoamers. Recent clinical applications of fatty acids include conjugation with antibodies to enhance protein incorporation into membranes and conjugation with antigens to enhance immunity. Derivatives of stearic acid are frequently employed as paramagnetic probes in radioactivity and electron spin resonance spectroscopy studies of film fluidity. In positron emission tomography, palmitic acid is used as a diagnostic radioactive tracer.11
Fish oil, sea buckthorn (Hippophae rhamnoides L.) berries, and macadamia nuts are the main sources of this fatty acid. These sources, however, fall short of the rising demand for palmitic acid. Synthesizing palmitic acid (217 mg/L) from 1% glucose is possible in the presence of the least amount of sodium oleate. Plasmid-mediated amplification of fasB led to a 1.47-fold increase in palmitic acid production (320 mg/L), whereas the disruption of fasB tends to result in minimal fatty acid production and also the efflux of malonic acid (30 mg/L). The Pseudomonas nitroreducens 9-desaturase genes DesBC were then inserted into the producer of palmitic acid to turn into a palmitoleic acid producer (C16:1-9). Production tests showed that one of these mutant M-1s generated 17 mg/L of palmitoleic acid and 173 mg/L of palmitic acid simultaneously.12 Current research predominantly focuses on the synthesis and optimization of palmitic acid using coconut oilcake as substrate by using fatty acids-producing organism obtained from the guts of marine fish. Furthermore, there have been no previous reports of microbial studies on the synthesis of palmitic acid from marine fish gut using coconut oil cake which is considered as a less expensive substrate.
Isolation of fatty acid producing bacteria
Owing to isolate probiotic bacteria capable of producing fatty acids, Sardinella longiceps and Obtuse barracuda were collected from fishing area of Rameswaram, Tamil Nadu, India (9.2876° N, 79.3129° E) and brought to the laboratory in an ice box. Intestine parts of fishes were excluded from the fish in pieces no wider than 5 cm. In addition, each distinct section was put into a 250 mL Erlenmeyer flask that held 100 mL of PYM medium (5 g/L peptone, 3 g/L yeast, 3 g/L malt extract, 55mM glucose), and it was incubated at 15°C for one day. The sample was diluted three times, and 10 µL was streaked onto ZoBell’s marine agar. It was then incubated for two to four days at 15°C. To confirm the strains’ purity, bacterial colonies were separated from these plates and re-cultured on ZoBell’s marine agar plates.13
Primary screening of fatty acids producer
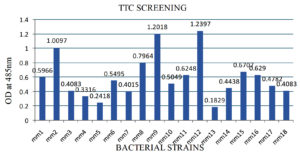
Tetrazolium Red reduction assay was used to screen isolates for the production of fatty acids. Tetrazolium Red will be reduced in to TF (Triphenyl red formazan) in liquid culture using ZoBell’s marine medium used as primary screening methods for fatty acid production. The medium was introduced into two sterile 50 mL polystyrene tubes (Corning) and a single colony of bacteria that were isolated from the gut of marine fish was seeded inside each tube. Following three days of incubation at 15°C, triphenyl tetrazolium chloride (TTC) was added to the cultures till a final concentration of 0.1% w/v. The cultures were, therefore, cultured for an extra hour at the same temperature, and an optical density (OD) of 485 nm was measured using a Shimadzu UV 160A spectrophotometer to assess the results.13
Production of palmitic acid using marine bacteria
Palmitic acid production was carried out in a 250 mL sterile Erlenmeyer flask with 100 mL of ZoBell’s marine broth. In an orbital shaker incubator, the seeded broth was incubated for 96 hours at a temperature of 15°C. Each flask’s final biomass was collected into a 50 mL falcon centrifuge tube, and it was centrifuged for 10 minutes at 1,957 × g. After being freeze-dried, the cell pellets proceed with FAME-GC–MS analysis.
Extraction fatty acids
The Soxhlet apparatus’s thimble held a 50 g sample of the dried oil cake. Thick filter paper is used to make the thimble, and it is put into the Soxhlet extractor’s main chamber. A flask filled with 500 mL of extraction solvent (Hexane) is placed on top of the Soxhlet extractor. After that, a condenser is equipped to the Soxhlet and reflux is reached by heating the solvent. Hexane, the solvent, creates vapors that rise up a distillation arm and pour into the chamber containing the solid thimble. Solvents vapor that starts to cool would be directed into the chamber containing the solid material by the condenser. The warm solvents gradually fill the chamber with the solid material. The heated hexane will then cause a few of the desired compounds to dissolve. The siphon side arm automatically empties the Soxhlet chamber when it is nearly full, sending hexane back to the distillation flask. At different times, this cycle was repeated for 3 days. Hexane dissolves a portion of the oil during each cycle. The desired oil was concentrated in the distillation flask after numerous cycles of this kind. Hexane was eliminated from the extracted compound by evaporation and GC–MS FAME analysis was used to examine it.
Optimization of palmitic acid production using response surface methodology
Box-Behnken design (BBD) for the production of palmitic acid had been carried out.14 Palmitic acid production (mg/100 mL) was optimized with temperature, pH, and the concentration of oil cake substrate. Three levels (-1, 0, +1) were used to analyze the values of pH, temperature, and oil cake substrate concentration from the lowest to the highest levels. Design-Expert 13.0.0.0 (Stat-Ease, trial version) was used to acquire experimental data. The quadratic model was used to analyze the data. When interpreting experimental data for RSM, Design-Expert software provided a glossary of statistical terms and their meaning.15 Second-order polynomials that are linearly fitted to the experimental data yield the regression equation.16 ANOVA and fit statistics were used to interpret the data in order to choose the optimal model.17 Using ANOVA, the models’ significance was ascertained. The fact that the models are valid indicates that the triplicate values were calculated for the expected values and validated under ideal circumstances. The production of palmitic acid is dependent upon temperature, pH, and the concentration of the oil cake substrate. (X1, X2, …, Xn). The relationship between environmental parameters and the production of palmitic acid was represented by
Y = f(X1, X2, X3) + ו………………… (1)
Where, f- The real response function (unknown), ו- The residual error
The correlation between the production of palmitic acid and various factors such as temperature, pH, and substrate concentration are represented by the response surface. These factors, collectively known as RSM, are correlated with X1, X2, …, Xn. Equation (2) describes the quadratic response model, including the squared terms, linear terms, and interaction effects. Where, b0 is referred as Constant, bi is referred as Slopes or linear effect, and bij is referred as Interaction of the quadratic effects (2)
Y = bo + ΣbiXi + ΣbiiXi2 + ΣbijXiXj…….. (2)
Characterization of fatty acids using GC–MS-FAME
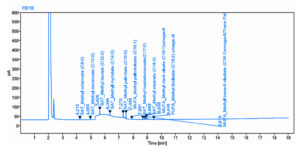
The stored bacterial pellets were directly transmethylated to produce fatty acid methyl esters.13 Biomass was mixed with 10 µl of the internal standard solution (23:0 FAME in toluene) with 0.5 mL of 1% (v/v) sodium methoxide in methanol, and the mixture was incubated for 30 minutes at 80°C. The incubation cycle was repeated after 0.5 mL of 5% methanolic HCl had been added to the vial, and it had cooled to room temperature. Hexane (1 mL) was used to extract the FAMEs. With the help of argon gas, hexane was evaporated, and the FAME residue was then re-dissolved in 100 mL of chloroform. Gas chromatograph (GCMS) with a flame ionization detector and mass spectrometry detector was used to analyze the samples. The use of an RTX5ms capillary column and detector splitting system (30 m x 0.25 mm i.d., 0.25 m) enabled the division of a column flow direction to capture data signal with both a mass spectrometry detector and a flame ionization detector. A carrier gas, 1/20 split helium, was used. The injector and detector had respective temperatures of 280°C and 320°C. The oven’s temperature program was set to begin at 70°C (hold on for 2 minutes), then climb to 320°C at a rate of 4°C per minute, and stay there for 15 minutes. MS spectra were used to locate FA peaks. The chromatographic peak’s area is regressed linearly in relation to the peak of an internal standard at a known concentration, and thus, FAME quantification was achieved (256.43 g/mol).
A total of 36 strains were also recovered similarly from the intestine of an Obtuse barracuda fish, and 8 of these isolates produced a red color during TTC screening. The Mm12 strain was able to synthesize fatty acids with a maximum OD value at 485 nm than other bacteria, demonstrating the effectiveness of fatty acid-producing bacterial strains using the colorimetric method (Figures S1 and Figure 1). DNA was isolated from the bacterial strain and PCR was performed using with universal primers (27f/1492r) of 16S rRNA. Further, the PCR product was sequenced by ABI dye terminator method. The 16S rRNA sequencing was used to identify the bacterial strain Mm12, which is responsible for palmitic acid production, and it was identified as Aeromonas veronii. The gut contents of the deep sea fish, Centroscyllium fabricii, which was caught at a depth of 1000 m, were similarly isolated aseptically and diluted tenfold before being spread onto nutrient agar (supplemented with 2% NaCl) and incubated at 28°C for a week. Several of the 137 isolates, isolated from fish gut using culture-dependent techniques, were found to have antibacterial activity.18 Shellfish, shrimps, and crabs were purchased from the Songkhla wet market and sourced directly from fishing boats moored on the Songkhla coast. Fish, crabs, shrimp, and crustaceans comprised 24 species of marine fish, from which lactic acid bacteria were isolated. Colonies that demonstrate antibacterial activity against pathogenic E. coli was cultured concurrently on MRS agar plates covered in nutrient agar that contained pathogenic E. coli.19
Previous reports have shown that the fish Leucostictus, Mossambicus, and Suratenis were collected from the Velar estuary on the south-eastern coast of India (11°29’N, 79°46’E). The total heterotrophic bacterial load ranged from 1.8 x106 to 4.8 x 106 CFU/g in gut samples, with a maximum of 4.5 x 106 CFU/g in the Mossambicus fish intestine, 3.1 x 106 CFU/g in the Leucostictus fish intestine, and a minimum of 0.8 x 106 CFU/g in the Suratenis fish intestine. The three estuarine fish had a total of 188 bacterial strains, which were divided into nine taxonomic groupings, including Acinetobacter Spp. (2), Bacillus Spp. (22), Enterobacteriaceae (43), Vibrio Spp. (21), Alcaligenes Spp. (12), Photobacterium Spp. (9), Pseudomonas Spp. (33), Aeromonas Spp. (32), and Flavobacterium Spp.20 Previous report has obtained lactogenic bacteria were isolated from the gastrointestinal tract of fish: Tilapia Oreochromis mossambicus fish was collected from the river Kaveri, a freshwater river in Erode District, Tamil Nadu, India.21 TTC was able to be converted into TPF by 95 isolates. 31 of them were able to develop quickly, and as a result, those were taken into consideration for additional research to confirm the isolates’ capacity to make LC-PUFAs because TTC colorimetric screening has been known to create false-positive results.22 The TTC screening procedure, Jack mackerel, tarakihi, bluenose, cardinal, and scorpion fish were all used in a single specimen. Each fish was cleaned and then both TTC-containing and TTC-free marine agar plates were seeded and incubated according to the instructions in the procedures section. Only 36 bacteria out of a pool in mixed flora of 2.0 x 108 CFU/mL were successfully converted into TTC to TC, and among them only 16 bacterial strains were confirmed being capable of EPA production by using gas chromatography (GC). The EPA-producing strains were all gram-positive rod bacteria, according to a subsequent gram staining, but all non-producing strains were gram-negative cocci.13
Table (1):
Optimization of palmitic acid production using Mm12 bacterial strain
Fatty acids |
Mm12-Strain mg/100 mL |
Oil Cake mg/100 mL |
Optimized conditions |
|---|---|---|---|
Cis-9 Oleate (C18:1) – Omega-9 |
16.624 |
5.41 |
15.97 |
Cis-10 heptadecenoate (C17:1) |
3.122 |
– |
– |
Palmitate (C16:0) |
16.132 |
10.16 |
19.97 |
Stearate (C18:0) |
1.004 |
3.58 |
4.80 |
Decanoate (C10:0) |
0.939 |
– |
– |
Laurate (C12:0) |
4.107 |
– |
– |
Myristate (C14:0) |
1.000 |
– |
– |
Tridecanoate (C13:0) |
0.311 |
– |
– |
Myristoleate (C14:1) |
0.196 |
– |
– |
Pentadecenoate (C15:0) |
2.793 |
– |
– |
Cis-10-pentadecenoate (C15:1)10 |
2.540 |
– |
– |
Linoleic acid (C18:2) |
11.87 |
1.38 |
3.23 |
Capric acid (C10:0) |
– |
4.64 |
– |
Lauric acid (C12:0) |
– |
46.27 |
– |
Myristic acid (C14:0) |
– |
22.38 |
– |
Elaidic acid (C18:1n9t) |
– |
0.32 |
– |
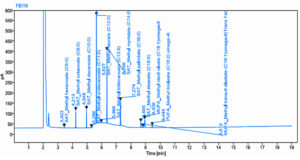
Palmitic acid production was validated by using GC–MS FAME analysis. The Mm12 strain, which was isolated from the fish gut, produced 16.132 mg of palmitic acid per 100 mL
(Table 1). The coconut oil cake’s fatty acid content was 5.41% Cis-9 Oleate – Omega-9, 10.16% Palmitate, 3.58% Stearate, 1.38% Linoleic acid, 4.64% Capric acid, 46.27% Lauric acid, 22.38% Myristic acid, and 0.32% Elaidic acid (Figure S2 and Figure 2). Similarly, a Soxhlet extraction was performed using algae powder, time, and temperature to obtain algal oil. Five oilseeds and soluble dry distillate grains were selected and ground. Hexane extraction was used to remove excess oil. Crude protein, starch, and total water-soluble carbohydrates increased after extraction. DDGs had the highest glucan content (20.8%), followed by canola, peanuts, and soybeans. Canola, sunflower, and sesame cakes had 16, 13, and 20% lignin content, while soybean meal, peanut meal, and DDGs had less than 10%. The biodiesel sample contained a higher amount of methyl ester group.23
The optimal level of palmitic acid production was achieved by analyzing the interaction among different parameters such as pH, temperature, and oil cake substrate concentration. Optimization of palmitic acid production was done using three parameters including substrate concentration, temperature and pH. These parameters were examined from the lowest to the highest range at three levels (-1, 0, and +1) in the model. A model with 17 experiments was created and run in triplicate. From the data that were analyzed, a quadratic model for optimization was suggested. As per the BBD experimental design, the expected and observed response 1 (palmitic acid production mg/100 mL) for each experiment was presented (Table 2). The outcome of the research is a representation of the BBD design matrix and its associated findings, along with response 1 statistical model (palmitic acid production mg/100 mL). For the BBD standardization of independent variables, the three zenith points were utilized. The response 1 polynomial equation in second order of palmitic acid synthesis (mg/100 mL) is
(Y) = + 7.75724 * A + -1.1567 * B + 0.476464 * C + -0.0333333 * AB + -0.0190476 * AC + -0.0109143 * BC + -0.658667 * A2 + 0.04192 * B2 + 0.00423837 * C2.
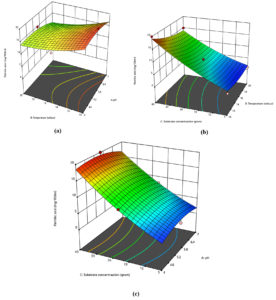
The total effects of all input factors equal the generation of palmitic acid (mg/100 mL). The ideal concentrations of the selected variables were found by looking at the response surface contour, solving the regression equation, and making surface plots. Table S1 lists the results of the analysis of variance (ANOVA) for the chosen quadratic polynomial model.1 The lack of fit statistic was used in this study to assess how well the model represented the experimental domain data at certain times that were excluded from the regression. The model was valid because the lack-of-fit had a significant F-value (p = 0.0115). Regression equations are graphically represented as three-dimensional (3D) response surfaces. They offer a technique for visualizing the interactions between any two tested factors as well as the relationship between the answer and each variable. Analyzing the 3D response surfaces allowed us to quickly determine the ideal ranges for each variable to maximize response, making it simple to study the interactions between any two variables. Three-dimensional response surface plots were created using the Design-Expert software 13.0.0.3 (Figure 3).
Figure 3. Optimization of palmitic acid production using BBD; a) pH (A) vs temperature (B); b) temperature (B) vs substrate concentration (C); c) pH (A) vs substrate concentration (C)
Table (2):
Optimization of palmitic acid production using BBD
| Run | Factor 1 | Factor 2 | Factor 3 | Response 1 Palmitic acid production mg/100 mL | |
|---|---|---|---|---|---|
| A:Temperature | B: pH | C:Substrate Concentration | Actual value | Predicted value | |
| 1 | 10 | 7 | 22.5 | 8.00 | 8.01 |
| 2 | 10 | 5.5 | 5 | 2.15 | 3.24 |
| 3 | 15 | 4 | 40 | 14.00 | 15.10 |
| 4 | 15 | 4 | 5 | 1.0000 | 0.1475 |
| 5 | 15 | 5.5 | 22.5 | 7.00 | 7.18 |
| 6 | 10 | 4 | 22.5 | 9.00 | 8.76 |
| 7 | 20 | 7 | 22.5 | 4.00 | 4.23 |
| 8 | 15 | 5.5 | 22.5 | 7.00 | 7.18 |
| 9 | 15 | 7 | 40 | 12.00 | 12.85 |
| 10 | 15 | 5.5 | 22.5 | 7.00 | 7.18 |
| 11 | 20 | 5.5 | 5 | 1.0000 | 1.87 |
| 12 | 15 | 7 | 5 | 1.0000 | -0.1025 |
| 13 | 15 | 5.5 | 22.5 | 7.00 | 7.18 |
| 14 | 20 | 4 | 22.5 | 6.00 | 5.98 |
| 15 | 20 | 5.5 | 40 | 15.00 | 13.91 |
| 16 | 10 | 5.5 | 40 | 19.97 | 19.10 |
| 17 | 15 | 5.5 | 22.5 | 7.92 | 7.18 |
The production of palmitic acid was influenced by pH, temperature, substrate concentration, and their interactions, as shown by the response surface in Figure 3. Thus, the three investigated factors’ ideal ranges to attain the highest palmitic acid production were additionally determined to be: pH = 4.0–7.0, temperature = 10–20°C, and oil cake substrate concentration = 5–40 g. Response surface method in Figure 3a showed the impact of temperature and pH on palmitic acid synthesis. When the temperature dropped to 10°C and the pH 5.5, there was an increase in the production of palmitic acid. The response surface method in Figure 3b showed that the temperature and oil cake substrate concentration affect palmitic acid synthesis. With a drop in temperature to 10°C and an increase in substrate concentration to 40 g, there was increase in palmitic acid production. Response surface in Figure 3c illustrated that the impact of pH and oil cake substrate concentration on palmitic acid synthesis. Design-Expert 13 solved the inverse matrix of a regression polynomial to obtain the optimal values for the tested parameters in the uncoded units: temperature, pH, and quantity of oil cake substrate were 10°C, 5.5 molar, and 40 grammes, respectively. The greatest quantity of palmitic acid that might be produced under ideal circumstances was estimated to be 19.97 mg/100 mL (Table 3 and Figure 4). Experimental testing was carried out using the derived ideal conditions to validate the model equation for predicting the optimal response value. The actual studies produced a mean value of palmitic acid production of 19.97 mg/100 mL under the pre-determined conditions, which was somewhat higher than the maximum value (19.10 mg/100 mL) predicted. When the Student’s t-test was performed, no notable difference between the experimental and anticipates yields was found, showing that the model was appropriate and suitable for simulating the anticipated optimization. The weight ratios of the fatty acid compositions of the biodiesel were investigated using a gas chromatography (GC) analyzer. The results of the waste fish oil GC–MS analysis showed the major fatty acid composition to be as follows: 1.07% Muriatic, 20.91% Palmitic, 6.81% Palmitoleic, 0.69% Heptadecanoic, 0.36% Heptadecenoic, 6.14% Stearic, 40.34% Oleic, 13.86% Linoleic, 2.64% Linolenic, 0.44% Eicosapentaenoic, and 4.75% Docosahexaenoic.24 The amount of cetane number in diesel fuels is influenced by these fatty acids. The specifications of fish oil and biodiesel made from waste oil and rapeseed were compared. The findings demonstrated that fish oil had a lower flash point than other oils and a higher kinematic viscosity than rapeseed oil and waste oil. Rapeseed oil has the highest density when compared to other oils, while waste oil has the lowest density.25
Table (3):
Mass Fragmentation Chromatogram
No. |
Fatty acid |
M12 with Oilcake fermentation – 10g |
|---|---|---|
1. |
Cis-9 Oleate (C18:1) – Omega-9 |
15.97 |
2. |
Tricosanoate (C23:0) |
– |
3. |
Palmitoleate (C16:1) |
27.70 |
4. |
Nervonate (C24:1) |
– |
5. |
Lignocerate (24:0) |
– |
6. |
Cis -10 heptadecenoate (C17:1) |
– |
7. |
Palmitate (C16:0) |
19.97 |
8. |
Stearate (C18:0) |
4.80 |
9. |
Decanoate (C10:0) |
– |
10. |
Laurate (C12:0) |
– |
11. |
Myristate (C14:0) |
– |
12. |
Tridecanoate (C13:0) |
– |
13. |
Myristoleate (C14:1) |
– |
14. |
Pentadecenoate (C15:0) |
– |
15. |
Cis-10-pentadecenoate (C15:1)10 |
– |
16. |
Linoleic acid (C18:2) |
3.23 |
17. |
Heptadecenoate (C17:0) |
0.41 |
18. |
Trans-9-Eladiate (C18:1) |
– |
19. |
Linolenic acid (C18:3) |
– |
20. |
Arachidic acid |
– |
21. |
Cis-11-Eicosanoate (C20:1) |
– |
22. |
Behenate (C22:0) |
– |
23. |
Capric acid (C 10:0) |
1.45 |
24. |
Lauric acid (C 12:0) |
15.57 |
25. |
Myristic acid (C 14:0) |
7.44 |
26. |
Elaidic acid (C 18:1n9t) |
1.71 |
27. |
Alpha-Linolenic Acid (C 18:3 n3) |
– |
It has been accounted for that trans-esterification can deliver unsaturated fat methyl esters from utilized cotton seed oil. 27.76% of Palmitic acid and 42.84% of linoleic acids were determined to be the two primary fatty acids in the GC–MS analysis of WCCO (Waste Coconut Cooking Oil) oil. The oil’s molecular mass was 881.039 g/mol. The reaction temperature, time, methanol/oil ratio, and catalyst loading rate that produced the highest yield of 92% biodiesel were 60°C, 50 min, 12:1, and 3% (wt.%), respectively. The catalyst made from calcined egg shell was developed and characterized. It was suggested to partially purify fatty acid methyl esters to improve engine performance and increase biodiesel quality. The biodiesel was found to have a glimmer point and fire point of 128°C and 136°C separately. At maximum load, the WCCO b10 biodiesel’s brake thermal efficiency was 26.04%. Conversely, the specific fuel consumption of diesel was 32 kg/kWh. By lowering carbon monoxide and hydrocarbon emissions while marginally raising nitrogen oxide (NOx) emissions, the use of biodiesel blends improved emission characteristics.26
Palmitic acid produced by the gut bacteria was optimized using RSM with the most appropriate values of the tested parameters in un-coded units were achieved by using Design-Expert 13 to solve the inverse matrix of the regression polynomial equation: temperature, pH, and quantity of oil cake substrate were 10 °C, 5.5, and 40 grammes, respectively. The greatest quantity of palmitic acid that might be produced under ideal circumstances was estimated to be 19.97 mg/100 mL which was confirmed by GC-MS FAME analysis. Fatty acids production in the marine atmosphere becomes challenging, because it is well known that some synthesized product must have disadvantages such as marine pollution, algal odor, and an extravagant production process. So there is a need for extensive research to find alternate production process and its use would meet high environment and safety requirements. In accordance with the results in this research, it can be concluded that the study could demonstrate the optimization of palmitic acids production from Mm12 strain using RSM-BBD. It is recommended that the optimization of the palmitic acid should be done by using oilcake as substrate to obtain higher production and reduce the cost of production.
Additional file: Additional Table S1 and Figure S1 and S2.
ACKNOWLEDGMENTS
None.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
None.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
Not applicable.
- Gavrilova V, Shelenga T, Porokhovinova E, et al. The diversity of fatty acid composition in traditional and rare oil crops cultivated in Russia. Bio Commun. 2020;65(1):68-81.
Crossref - Shah AM, Yang W, Mohamed H, Zhang Y, Song Y. Microbes: A Hidden Treasure of Polyunsaturated Fatty Acids. Front Nutr. 2022;9:827837.
Crossref - Jovanovic S, Dietrich D, Becker J, Kohlstedt M, Wittmann C. Microbial production of polyunsaturated fatty acids – high-value ingredients for aquafeed, superfoods, and pharmaceuticals. Curr Opin Biotechnol. 2021;69:199-211.
Crossref - Powell L, Wallace EC. Fatty Acid Metabolism, 5th Ed. Textbook of Natural Medicine. 2020;79:584-592.e4.
Crossref - Gianfranca C, Elisabetta M, Sebastiano B, Claudia M. Palmitic Acid: Physiological Role, Metabolism and Nutritional Implications. Front Physiol. 2017;8:8:902.
Crossref - Carta G, Murru E, Lisai S, et al. Dietary Triacylglycerols with Palmitic Acid in the sn-2 Position Modulate Levels of N-Acylethanolamides in Rat Tissues. PLoS One. 2015;10(3):e0120424.
Crossref - Osorio D, Pinzon A, Martin-Jimenez C, Barreto GE, Gonzalez J. Multiple Pathways Involved in Palmitic Acid-Induced Toxicity: A System Biology Approach. Front Neurosci. 2020;13:1410.
Crossref - Sette S, Le Donne C, Piccinelli R, Arcella D, Turrini A, Leclercq C. The third Italian National Food Consumption Survey. INRAN-SCAI2005-06 – part 1: Nutrient intakes in Italy. Nutr Metab Cardiovasc Dis. 2011;21:922-932.
Crossref - Berge JP, Barnathan G. Fatty Acids from Lipids of Marine Organisms: Molecular biodiversity; Role as Biomarkers, Biologically Active Compounds and Economical Aspects. Adv Biochem Eng Biotechnol. 2005;96:49-125.
Crossref - Fu Zh, Liu J, Zhong LB, et al. Screening of cellulose-degrading yeast and evaluation of its potential for degradation of coconut oil cake. Front Microbiol. 2022; 13.
Crossref - Seiki T, Yosuke H, Kako K, et al. Metabolic engineering to produce palmitic acid or palmitoleic acid in an oleic acid-producing Corynebacterium glutamicum strain. Metabol Eng. 2023;78:148-158.
Crossref - Jason R, Hannah F, Sandra V, Mikhail V, Gabriel V. A rapid method for the isolation of eicosapentaenoic acid-producing marine bacteria. J Microbiol Methods. 2010;82(1):49-53.
Crossref - Venil CK, Zakaria ZA, Ahmad WA. Optimization of culture conditions for flexirubin production by Chryseobacterium artocarpi CECT 8497 using response surface methodology. Acta Biochim Polonica. 2015;62(2):185-90.
Crossref - Montgomery DC, Verbeeck DV.Control estadístico de la calidad.Limusa Wiley. 2004;658:562 M66.
- Zhang YJ, Li Q, Zhang YX, Wang D, Xing JM. Optimization of succinic acid fermentation with Actinobacillus succinogenes by response surface methodology (RSM). J Zhejiang Univ Sci B. 2012;13(2):103-110.
Crossref - Muthukumar M, Mohan D, Rajendran M. Optimization of mix proportions of mineral aggregates using Box-Behnken design of experiments. Cem Concr Compos. 2003;25(7):751-758.
Crossref - Bindiya ES, Tina KJ, Raghul SS, Bahar SG. Characterization of Deep Sea Fish Gut Bacteria with Antagonistic Potential, from Centroscyllium fabricii (Deep Sea Shark). Probiotics Antimicrob. Proteins. 2015;7(2):157-163.
Crossref - Nirunya B, Suphitchaya C, Tipparat H. Screening of lactic acid bacteria from gastrointestinal tracts of marine fish for their potential use as probiotics, Songklanakarin. J Sci Technol. 2008;30:141-148.
- Sivasubramanian K, Ravichandran S, Kavitha R. Isolation and Characterization of Gut Micro Biota from Some Estuarine Fishes. Marine Sci. 2012;2(2):1-6.
Crossref - Pandian V, Thirugmanasambandam S. Glycosaminoglycans (GAG) from Backwater Clam Marcia opima (Gmelin). Iranian J Pharmacol Therapeut. 2008;1735-2657.
- Roberta R, Noura R, Fabio F. Mediterranean Sea bacteria as a potential source of long-chain polyunsaturated fatty acids. FEMS Microbiol Lett. 2020;367(16):fnaa132.
Crossref - Toparea NS, Jadhav SR, Vilas CR, Khedkar VS, Chavan YP, Bhagat SL. Extraction of Oil From Algae By Solvent Extraction and Oil Expeller Method. Int J Chem Sci. 2011;9(4):1746-1750.
- Yahyaee R, Ghobadian B, Najafi G. Waste fish oil biodiesel as a source of renewable fuel in Iran. Renew Sustain Energy Rev. 2013;17:312-319.
Crossref - Kara K, Ouanji F, El Lotfi M, El Mahi M, Kacimi M, Ziyad M. Biodiesel production from waste fish oil with high free fatty acid content from Moroccan fish-processing industries. Egy J Petrol. 2018;27(2):249-255.
Crossref - Duple S, Murugavelh S. Biodiesel production from waste cotton seed oil using low cost catalyst: Engine performance and emission characteristics. Perspect Sci. 2016;8:237-240.
Crossref - Chu M, Noh E, Lee KG. Analysis of oxidation products and toxic compounds in edible and blended oil during the deep-frying of French fries. Food Sci Biotechnol. 2024.
Crossref
© The Author(s) 2024. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.