ISSN: 0973-7510
E-ISSN: 2581-690X
The major problem encountered in recent days due to the advent of emerging antimicrobial resistance strains is Hospital Acquired Infections (HAIs). The spread of HAI is mainly through the hospital staff and the hospital settings’ inanimate surfaces. The study includes the surveillance of hospital surfaces, including wards and ICUs, by taking swabs dipped in normal saline. Pseudomonas aeruginosa (53.8%) was found to be the most common pathogen isolated, followed by Klebsiella species (18%), Methicillin-Sensitive Staphylococcus aureus (16%), Coagulase-Negative Staphylococci (6%), Methicillin-Resistant Staphylococcus aureus (3%). The widespread presence of bacterial sensitivity to antimicrobials and the modifications insensitivity forms the basis for designing antibacterial therapy’s practical recommendations and rational use of antimicrobials. Proper display of all Information, Education, and Communication (IEC) materials at appropriate places play a significant role in preventing nosocomial infections. Audiovisual aids and training to the staff play utmost importance in preventing the spread of HAIs. All these can reduce the occurrence and outbreak of nosocomial conditions. Overall, these minimize health care costs.
Hospital Acquired Infections, Pseudomonas aeruginosa, Information, Education and Communication, Klebsiella species, antimicrobial resistance
Hospital Acquired Infections (HAIs) serves as one of the major threat in the medical field even after the beginning of much antimicrobial therapy and progress in supportive care management.1 The hospital environment, objects/instruments, and health-care workers are likely to get populated by various microbial agents. Disease transmission leads to morbidity and mortality due to contact with things handled by infected and colonized patients and direct communication with such patients1,2 and also at least 90,000 of them die due to HAI.3,4 Healthcare-associated infections are the fifth major cause of death in critical-care hospitals. The proportion of HAI patients in developing countries is neither recorded nor appropriately reported due to various reasons like difficulty in collecting reliable data and improper follow-up of HAI surveillance system.5 A summary of evidence has estimated the frequency of HAIs to be 7.6% in high-income countries and 10.1% in middle-income countries.6
These HAIs produce mortality at high rates in neonates, especially in premature and low birth weight babies. Worldwide, out of the five million neonatal deaths, 4% to 56% of neonatal deaths are due to this hospital-acquired infections.7 Moreover, these HAI occur mainly in developing countries; there is the fact that about 98% of these neonatal deaths occur in developing nations. In high-income countries, the prevalence of HAI is about 7.6% which is less than 10.1% in middle-income countries.6,10 Multidrug-resistant pathogens are now emerging as a global threat, and neonates are most commonly affected by their low immune status. Most frequently, the contaminated hands of the health care workers and the hospital environmental surfaces serve as the primary source of infection for all these HAIs. Methicillin-Resistant Staphylococcus aureus (MRSA) strains continue to be the main threat to community and hospital-acquired diseases. These staphylococcus shows high resistance to the drug Vancomycin, one of the most common susceptible drugs for Staphylococcus aureus.8 In this study, about 17.5% were MRSA strains, and 5% of vancomycin-resistant strains were identified.8 Certain studies tell us the persistence of bacterial pathogens on the hospital floors and wards inanimate surfaces. These serve as the primary source of transmission of nosocomial pathogens and remain a danger to susceptible patients or health care workers.9
The present study aims to detect the potential source of infection responsible for HAI in various wards, labor rooms, ICUs and identification of bacterial isolates along with their antibiotic resistance pattern.
A prospective surveillance study of pathogens causing a potential infection source at various tertiary care hospital wards was conducted. The study period was about four months, from May 2019 to August 2019. A total of 150 samples were obtained from sterile swabs dipped in normal saline. The samples were taken from the floors, walls, air conditioning vent, door handles, medicine racks, oxygen cylinders, bed, and cots. After collection, the swabs were immediately transferred to the laboratory and inoculated into the brain heart infusion broth for 24 hours. After incubation, the samples were sub-cultured in Blood agar and MacConkey agar. Thereafter,incubated at 37°C for the next 24-hour period. All positive cultures were identified using biochemical tests such as-indole, sugars, citrate, urease, and triple sugar iron medium. The disc diffusion method in Muller Hinton Agar (MHA) determined the antibiotic sensitivity profile of each identified organism, as per the Clinical and Laboratory Standards Institute (CLSI) 2019 guidelines. Resistance to Methicillin was detected on all Staphylococcus aureus isolates using Cefoxitin (30mcg).
Around 150 samples were collected from the medical, surgical, paediatric ICUs and wards, labor room, and post-operative ward. Among the samples, 43.3% (65) were positive for bacterial isolates. Table 1 identifies possible sources of infection within various wards. In this, 93.3% of bacteria isolated from surgical wards, 73.3% from paediatric wards, 66.7% from medicine wards, 30% from labor room, and 20% from medical, surgical, and paediatric ICUs. Pseudomonas aeruginosa was the predominant organism isolated in major wards and ICUs, followed by Klebsiella species, Methicillin-Sensitive Staphylococcus aureus (MSSA), Coagulase-Negative Staphylococci (CONS), Methicillin-Resistant Staphylococcus aureus (MRSA). Apart from these, Enterococcus spp was also isolated rarely. Overall, the study revealed a predominant presence of strains of Pseudomonas aeruginosa from the medical, surgical, paediatric, work room and post-operative intensive care rooms from which the swabs were taken.
Table (1):
Positive Bacterial growth in different areas of the hospital.
S.No |
Places where swabs taken |
Number of swabs taken |
Positive culture |
Percentage (%) |
|---|---|---|---|---|
1 |
Medicine ICU |
20 |
4 |
20 |
2 |
Surgical ICU |
25 |
5 |
20 |
3 |
Paediatric ICU(NICU&PICU) |
20 |
4 |
20 |
4 |
Medicine Ward |
15 |
10 |
66.7 |
5 |
Surgical ward |
15 |
14 |
93.3 |
6 |
Paediatric ward |
15 |
11 |
73.3 |
7 |
Labour room |
20 |
6 |
30 |
8 |
Postop |
20 |
11 |
55 |
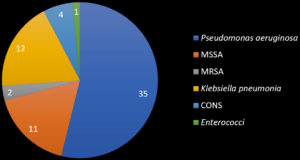
Among the 150 cultures done, 65 plates showed growth in the blood agar and MacConkey agar which was followed by biochemical tests as a further step to identify the pathogenic species. Among those 65 positive cultures, 53.8% (35) were isolated as Pseudomonas aeruginosa, 18% (12) isolates were identified as Klebsiella pneumonia, 16% (11) isolates were Methicillin-Sensitive Staphylococcus aureus (MSSA), 6% (4) were Coagulase-Negative Staphylococcus (CONS), 3% (2) were Methicillin-Resistant Staphylococcus aureus (MRSA) and 1.6% (1) were identified as Enterococcus species.
The antibiotic sensitivity testing for all the isolates was done using the Kirby-Bauer disc diffusion method as per standards to identify the multidrug-resistant strains (MDR) and MRSA. The various drugs used for Gram negative bacilli were ampicillin, piperacillin-tazobactam, ceftazidime, cefepime, cefaperazone-sulbactam, amikacin, gentamicin, ciprofloxacin, imipenem, meropenem and cotrimoxazole. For Gram-positive isolates, amoxicillin-clavulanic acid, vancomycin, linezolid, cefoxitin, erythromycin, clindamycin and tetracyclines were used. The predominant isolate of this study was Pseudomonas aeruginosa. Among 65 positive cultures identified, 53.8% (35) were positive for Pseudomonas. It was identified by using various biochemical tests such as oxidase positive, citrate positive, TSI(K/K-), indole negative and also with the help of pigment production.
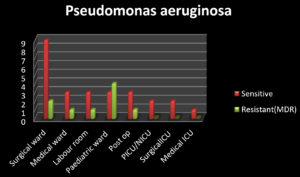
Among 35 isolates of Pseudomonas aeruginosa identified, 74% (26) were sensitive to piperacillin-tazobactam, ceftazidime, cefepime, cefaperazone-sulbactam, amikacin, gentamicin, ciprofloxacin, imipenem, meropenem and 26% (9) were identified as resistant to all the above drugs and sensitive only to higher group of drugs like colistin. All isolates of Pseudomonas aeruginosa identified were intrinsically resistant to Cotrimoxazole by disc diffusion method. Fig. 2 illustrates the sensitive strains and Multi drug-resistant Pseudomonas aeruginosa strains.
Klebsiella pneumonia was the next predominate organism that was isolated. Among 65 (53.8%) positive cultures, 12 (18%) were identified as Klebsiella pneumonia by doing biochemical tests such as indole negative, citrate positive, Mannitol motility medium showing its fermentation of mannitol and also its non-motile behaviour. Klebsiella pneumonia was isolated from all wards except surgical ward.
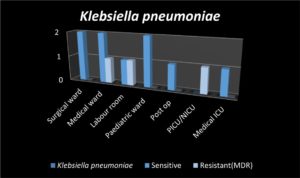
Among twelve isolates, nine of them were sensitive to drugs like amoxicillin-clavulanic acid, cefotaxime, ceftrioxzone, ceftazidime, imipenem, amikacin, gentamicin and cotrimoxazole and three isolates were resistant to all the above drugs. Klebsiella pneumonia is intrinsically resistant to ampicillin. Fig. 3 illustrates the sensitive strains and resistant strains of Klebsiella pneumonia.
From sixty five positive cultures, seventeen (26%) were identified as Gram-positive cocci in clusters and only one (1.5%) isolate was seen as Gram-positive cocci in pairs. Staphylococcus aureus identified by doing positive catalase test, coagulase test and cefoxitin disc for identification of MSSA and MRSA. Only two isolates were found to be MRSA in postoperative wards. Enterococcus species was identified by positive bile esculin test and is sensitive to all drugs including high level gentamicin. Table 2 shows the distribution of Multi-drug resistant strains and Methicillin-Resistant Staphylococcus aureus (MRSA).
Table (2):
Distribution of Resistant strains and Methicillin-Resistant Staphylococcus aureus (MRSA).
Name of the isolate |
Ward /ICUS |
Number of isolate |
Sensitive strains |
Resistant strains |
|---|---|---|---|---|
Pseudomonas aeruginosa |
Surgical ward Medical ward Labour room Paediatric ward Post op PICU/NICU SurgicalICU Medical ICU |
11 4 4 7 4 2 2 1 |
9 3 3 3 3 2 2 1 |
2 1 1 4 1 0 0 0 |
Klebsiella pneumonia |
Surgical ward Medical ward Labour room Paediatric ward Post op PICU/NICU Medical ICU |
2 3 2 2 1 1 1 |
2 2 1 2 1 0 1 |
0 1 1 0 0 1 0 |
MRSAs |
Postop |
2 |
1 |
1 |
Infectious bacteria found in different parts of hospitals are often known to be the common cause of HAI. Infected patients, colonized with bacteria and healthcare providers are the main source of contamination. The main source of infection is coughing, sneezing, talking and uncontrolled movement within and outside the hospital. A hospital monitoring study in Ghana found that 91% of swabs were positive for various micro-organisms such as coagulase-negative staphylococcus, Bacillus sp, E.coli, Klebsiella sp., and Pseudomonas aeruginosa.11,12 In another study by poline et al, Staphylococcus aureus, Pseudomonas aeruginosa, Klebsiella pneumonia, Enterococcus species are responsible for nosocomial infections.
These organisms are constantly present in the hospital, whereas, in our study, 53.8% (35) were isolated as Pseudomonas aeruginosa, 18% (12) isolates were identified as Klebsiella pneumonia, 16% (11) isolates were Methicillin-Sensitive Staphylococcus aureus (MSSA), 6% (4) were Coagulase-Negative Staphylococcus (CONS), 3% (2) were Methicillin-Resistant Staphylococcus aureus (MRSA) and 1.6% (1) were identified as Enterococcus species.
Pseudomonas aeruginosa was the predominant microorganism causing HAIs. Certain studies show that 34% of Pseudomonas aeruginosa were multidrug-resistant, and 22% were sensitive strains compared to our research, which shows 26% were MDR pseudomonas and 74% were sensitive strains.12 Staphylococcus aureus was identified as the predominant cause of HAIs. It spreads through the hands of health care personnel. Different studies from South Africa and Switzerland have reported the appearance of resistance to antibacterial agents by Klebsiella and Enterobacter strains has caused Healthcare-associated infections related to the gram-negative bacteria.13,14 In our study, 12 (18%) were identified as Klebsiella pneumonia causing the nosocomial attacks. Among twelve isolates, nine were sensitive to drugs like amoxicillin-clavulanic acid, cefotaxime, ceftrioxzone, ceftazidime, imipenem, amikacin, gentamicin, and cotrimoxazole and three isolates were resistant to all the drugs.
In the study done by Marisa et al., 25% of isolates in the labor room were Staphylococcus aureus. In our study, 16% (11) isolates were Methicillin-Sensitive Staphylococcus aureus (MSSA), 6% (4) were Coagulase-Negative Staphylococcus (CONS), 3% (2) were Methicillin-Resistant Staphylococcus aureus (MRSA), and 1.6% (1) was identified as Enterococcus species15. A study done in 1992 by Omori et al. showed the presence of MRSA in environmental samples collected by wiping of floors.16 A further study in China found that 60% of MRSA isolates came from environmental samples, while in our study only 3% were identified as MRSA.
The prevention and control measures of all nosocomial infections are based on minimizing microorganisms’ transmission among the health care personnel and the patients. Also, the prevalence of bacterial sensitivity to antimicrobials forms the basis for designing antibacterial therapy’s practical recommendations and for more rational use of antimicrobials. Many measures are taken to control and prevent HAIs, such as hand hygiene technique, wearing personal protective equipment, proper donning and doffing of PPE kits used, aseptic non-touch techniques especially in neonatal and paediatric ICUs, and maintaining good respiratory hygiene. Any act of carelessness in the hospital premises like improper maintenance of the wards, poor hand hygiene can lead to nosocomial outbreaks. Proper display of all Information, Education, and Communication (IEC) materials at appropriate places play a significant role in preventing nosocomial infections. Audiovisual aids and training to the staff play utmost importance in preventing the spread of Hospital Acquired Infections. These all can reduce the occurrence and outbreak of nosocomial infection and reduce the health care cost.
ACKNOWLEDGMENTS
None.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
None.
ETHICS STATEMENT
Not applicable.
AVAILABILITY OF DATA
All datasets generated or analyzed during this study are included in the manuscript.
- Gravel D, Taylor G, Ofner M, et al. Canadian Nosocomial Infection Surveillance Program. Point prevalence survey for healthcare-associated infections within Canadian adult acute-care hospitals. Journal of Hospital Infection. 2007;66(3):243-248.
Crossref - Dancer SJ. Importance of the environment in Methicillin-resistant Staphylococcus aureus acquisition: the case for hospital cleaning. Lancet Infect Dis. 2008;8(2):101-113.
Crossref - Stone PW, Braccia D, Larson E. Systematic review of economic analyses of health care-associated infections. Am J Infect Control. 2005;33(9):501-509.
Crossref - Sydnor ER, Perl TM. Hospital epidemiology and infection control in acute-care settings. Clin Microbiol Rev. 2011;24(1):141-173.
Crossref - Allegranzi B, Pittet D. Preventing infections acquired during health-care delivery. Lancet. 2008;372(9651):1719-1720.
Crossref - Magill SS, Edwards JR, Bamberg W, Beldavs ZG, Dumyati G, Kainer MA, Lynfield R, Maloney M, McAllister-Hollod L, Nadle J, Ray SM. Multistate point-prevalence survey of health care-associated infections. New England Journal of Medicine. 2014 Mar 27;370(13):1198-208.
- Wagh A, Sinha A. Prevention of healthcare-associated infections in paediatric intensive care unit. Child’s Nerv Syst. 2018;34(10):1865-1870.
Crossref - Dilnessa T, Bitew A. Prevalence and antimicrobial susceptibility pattern of methicillin resistant Staphylococcus aureus isolated from clinical samples at Yekatit 12 Hospital Medical College, Addis Ababa, Ethiopia. BMC Infect Dis. 2016;16(1):398.
Crossref - Kramer A, Schwebke I, Kampf G. How long do nosocomial pathogens persist on inanimate surfaces? A systematic review. BMC Infect Dis. 2006;6(1):130.
Crossref - Ogwang M, Pramatti D, Molteni T, et al. Prevalence of hospital-associated infections can be decreased effectively In developing countries. J Hosp Infect. 2013;84(2):138-142.
Crossref - Newman MJ. Neonatal intensive care unit: reservoirs of Nosocomial pathogens. West Afr J Med. 2002;21(4):310- 312.
Crossref - Polin RA, Saiman L. Nosocomial infections in the Neonatal Intensive Care. NeoReviews. 2003;4(3):e81-e89.
Crossref - Cotton MF, Wasserman E, Pieper CH, et al.. Invasive disease due to extended spectrum beta-lactamase-producing Klebsiella pneumoniae in a neonatal unit: the possible role of cockroaches. J Hosp Infect. 2000;44(1):13-17.
Crossref - Harbarth S, Sudre P, Dharan S, Cadenas M, Pittet D. Outbreak of Enterobacter cloacae related to understaffing, overcrowding, and poor hygiene practices. Infect Control Hosp Epidemiology. 1999;20(9):598-603.
Crossref - Mussi-Pinhata MM, Nascimento SD. Neonatal nosocomial infections. J Pediatric (Rio J). 2001;77(Supl.1):s81-s96.
Crossref - Omori A, Takahashi A, Watanabe K, et al. Isolation of MRSA from inpatients, staff and environment in the hospital. Kansenshogaku Zasshi. 1992;66(10):1396-1403.
Crossref
© The Author(s) 2021. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.