ISSN: 0973-7510
E-ISSN: 2581-690X
Studies have suggested that modulation of gut microbiota is a viable therapeutic possibility for diabetes. This study evaluated the ability of an edible plant, Gongronema (G.) latifolium Benth (Asclepiadaceae), to modulate the gut microbiome and reduce blood glucose of alloxan-induced diabetic rats. Thirty (30) young, male, albino rats were divided into 6 groups of 5 rats each: Group 1 comprised normal rats; Groups 2 to 4, diabetic rats treated with 200, 400 and 800 mg/Kg body weight of hydro-alcoholic leaf extract, respectively; Group 5, diabetic rats treated with 0.2 mg/Kg glibenclamide (an anti-diabetic drug); and Group 6 comprised untreated diabetic rats. Following induction of diabetes with alloxan injections, the treatments were administered twice daily on a 12-hourly basis by orogastric intubation for 21 days. Thereafter, faecal samples were collected from the animals and subjected to metagenomic analysis, to ascertain the composition and relative abundance of the gut microbiota. There were five dominant bacterial phyla in the rat gut: Firmicutes, Bacteroidetes, Actinobacteria, Spirochaetea and Proteobacteria. Induction of diabetes resulted in observable dysbiosis in the rats. However, treatment of the diabetic rats with G. latifolium extract, ameliorated the state of dysbiosis and resulted in significant increase in species like Lactobacillus (L.) johnsonii, L. reuteri and Prevotella corpri, which are associated with improved glucose metabolism. The plant extract produced the best result at the dose of 400 mg/Kg. The results from this study show that G. latifolium may be used as a therapeutic option for restoration of the microbiome in diabetic patients.
Diabetes, Gongronema latifolium, dysbiosis, microbiome, hyperglycaemia, gut flora
The role of the microbiome, particularly the gut microbiome, in health cannot be overemphasized. Studies have shown that the microbes of the gut microbiome impact the host physiology, through the gut-brain axis1,2. The normal microbial community in the gut contributes to host nutrient metabolism, xenobiotic and drug metabolism, maintenance of structural integrity of the gut mucosal barrier, immunomodulation, protection against pathogens and production of various bioactive compounds, thereby leading to overall improvement of host health3,4. On the other hand, an imbalance of this microbial community may result in any of various diseases, including metabolic diseases such as obesity and diabetes. Recent studies have also shown that fecal therapy, probiotics, prebiotics and symbiotics may be new approaches to the management of diabetes, through modulation of the gut flora1,4.
The term diabetes describes a group of metabolic disorders that are characterized by hyperglycaemia in the absence of treatment. Two major types of diabetes are known, type 1 diabetes (T1DM) and type 2 diabetes (T2DM); generally distinguished by the underlying mechanism, which are inadequate production of insulin and inadequate sensitivity of cells to insulin action, respectively. New classifications have been developed by the World Health Organization, recently, but while the therapeutic approach may differ for these types of diabetes, dietary management remains essential for all types5.
Increasingly, diabetes management is involving non-conventional drugs. It is estimated that 25 to 57% of people with diabetes have at one time or another resorted to complementary and alternative medicine, including medicinal plants6. The use of plants in the treatment of diseases dates back to the early history of man and recent times are witnessing resurgence in the use of plant products as an alternative to modern medicine; due to increasing health care costs, inadequate health care facilities, religious beliefs, adverse drug reactions and problems of drug resistance, among others7,8. Some clinical and laboratory studies, carried out in different parts of the world, on a variety of herbs, spices and vitamins, evaluated their effectiveness in diabetes therapy and some of these products were found promising6,9,10.
In Nigeria, various plants and plant products are claimed to be useful in managing diabetes. One such plant is G. latifolium, known as amaranth in English and utazi among the Ibos of eastern Nigeria. The plant belongs to the family Asclepiadaceae. It is a climbing shrub that grows up to 5 m long and the leaves are used either as vegetable or spice. In ethnomedicine and folkloric use, individuals having diabetes or elevated blood glucose chew the bitter leaves of the plant or use them as a salad in meals, often in an unregulated manner, or as much as the individual can stand the bitter taste. Sometimes, the leaves are used in conjunction with conventional drugs, raising questions about the true anti-diabetic effect of the plant. However, in a study by Akah et al.11 leaf extracts of the plant were found to reduce the blood glucose of alloxan-induced diabetic rats in a dose dependent manner, validating the traditional medicine claims.
Although this plant and some other medicinal plants are claimed to have antidiabetic action, their exact mechanisms of action and effects on the microbiome have not been studied, particularly as it has been suggested that the body’s microbiota plays a strong role in glucose metabolism. This study therefore aimed at evaluating the modulatory effect of G. latifolium extract on the gut microflora and blood glucose of induced diabetic rats.
Collection and processing of plant materials
Fresh leaves of G. latifolium were purchased from traditional dealers at a local market in Nsukka, southeastern Nigeria. Nsukka is located at longitudes 7° 13 00 – 7° 35 30 and latitudes 6° 43 30 – 7° 00 to the equator. Leaf samples were immediately taken to the Herbarium Unit of the Department of Pharmacognosy and Environmental Medicine, University of Nigeria, Nsukka, and authenticated by a Plant Taxonomist/Curator. A voucher specimen was deposited at the Herbarium for reference purposes (Voucher No. PCG/UNN/0343). Extraction was carried out by a modification of the method described by Ezeonu et al.12 Within 3 hours after purchase, fresh leaves were washed with clean tap water and air dried under a shade for 7 days. Thereafter, the dried leaves were pulverized into coarse powder with an electric blender. A hydro-alcoholic extract was prepared by soaking 250 g portion of powder in 1 L of 30% (v/v) ethanol (Sigma-Aldrich, St. Louis USA) for 48 h, with intermittent stirring and then filtered through Whatman No. 1 filter paper. The resulting filtrate was evaporated to a sticky paste under a constant stream of cool air for 48 h and stored at 4°C until required for use. The hydro-alcoholic preparation was chosen over 100% aqueous extraction, because it was less prone to contamination in preliminary studies. The extraction yield was calculated using the formula:
Percentage yield (%) = [Weight of Extract / Weight of pulverized
plant material] (g) X100
The percentage yield was approximately 4.2%.
Experimental animals
A total of 30 young, male albino rats, weighing between 160-190 g (average weight 172 + 14g), were obtained from the animal house of the Faculty of Veterinary Medicine, University of Nigeria, Nsukka. Male rats were chosen because they are believed to be more physiologically stable and less subject to hormonal fluctuations that may affect results. The animals were housed in well-ventilated cages in the animal house and allowed free access to feed (Vital Feed, Nig. Ltd) and clean water. The animals were handled with humane care according to NIH guidelines for use and care of laboratory animals12. Ethical approval for this study was also obtained from the University of Nigeria Faculty of Veterinary Medicine Animal Care and Use Committee (approval number: FVM-UNN-IACUC-2020-1059). The guidelines set out by the University of Nigeria Faculty of Veterinary Medicine Ethics Committee for Medical and Scientific Research (MSR) which among others include good, clean and hygienic housing, provision of clean water and humane handling of animals during sample collections were strictly followed in the experiment.
Induction of diabetes
This was done as described by11,14. A stock solution of alloxan was prepared by dissolving 4.5 g alloxan monohydrate (Sigma-Aldrich, USA) in 30 ml of sterile distilled water to give a stock concentration of 150 mg/ml. Diabetes was induced in 24 rats by intraperitoneal injection of the alloxan solution, at a dose of 150 mg/Kg body weight, following a 12 h fast. The alloxanized animals were allowed free access to food and water and monitored daily. Animals with fasting blood glucose >140 mg/dl for 5 consecutive days were considered as having developed experimental diabetes and selected for the study.
Experimental groups and treatments
On the sixth day, following induction, the rats were divided into 6 different experimental groups of 5 rats per group by simple randomization. Group 1 served as normal control and comprised 5 uninduced rats. Groups 2, 3 and 4, received treatments of 200, 400 and 800 mg/Kg body weight of the hydro-alcoholic G. latifolium extract, respectively. The doses were fixed as previously described by Akah et al.11 Group 5 was treated with standard antidiabetic drug (glibenclamide) given at 0.2 mg/Kg body weight, while Group 6 comprised untreated diabetic rats. The treatments were administered twice daily on a 12-hourly basis by orogastric intubation for 21 days.
Measurement of blood glucose
The animals were allowed to fast for about 12 h. Blood samples were obtained from the tail veins of the rats and tested for fasting blood glucose level, using an Oncall Glucometer (ACON Laboratories Inc, USA). Measurements were taken at 3-day intervals until the end of treatment and every week, for three weeks after termination of treatment.
Microbiome analysis
Faecal samples were collected at the end of the treatment period. All samples were collected directly from the animals’ rectum into sterile vials, containing DNA/RNA shield transport and storage medium (Zymo Research Corp, CA, USA). Samples were stored at 40C until courier-dispatch to CosmosID®, Rockville, MD. DNA extraction, shotgun DNA sequencing and metagenomic analysis were carried out on each sample to ascertain the composition and relative abundance of bacteria, viruses and fungal species in individual faecal samples. DNA from the faecal samples was isolated using the Zymo Miniprep Kit (Zymo Research Corp.), according to the manufacturer’s protocol. DNA libraries were prepared using the Illumina Nextera XT library preparation kit, according to the manufacturer’s protocol, while Library quantity and quality was assessed with Qubit (ThermoFisher Scientific, USA) and Tapestation (Agilent Technologies, CA, USA). Libraries were then sequenced on Illumina HiSeq platform 2×150 bp. Unassembled sequencing reads were directly analyzed by CosmosID bioinformatics platform (CosmosID Inc., Rockville, MD).
Euthanasia of experimental animals
All 5 untreated diabetic rats in group 6, died shortly after the 21-day test period. The rats in the other groups were monitored for an additional 21 days for stability of the blood glucose levels. Thereafter, the rats were sacrificed by cervical dislocation. Liver tissues from each experimental group were harvested and frozen for further studies. The euthanasia and harvest of organs were carried out by qualified veterinarians from the Faculty of Veterinary Medicine, University of Nigeria, Nsukka.
Statistical analysis
Data obtained were subjected to one-way Analysis of Variance (ANOVA), using IBM SPSS Statistics software version 23. Values were expressed as mean + SEM. Significance was accepted at p ≤ 0.05.
Effect of treatments on gut flora
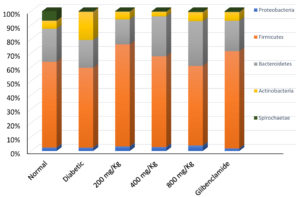
Although metagenomic analysis of the animals’ faecal samples provided information about the relative abundance of bacteria, fungi, protists and viruses in the samples, this study focused only on the gut bacteria. Metagenomic analyses of faecal samples of the experimental animals revealed the presence of bacteria from 5 major phyla, namely: Firmicutes, Bacteroidetes, Actinobacteria, Spirochaetae and Proteobacteria. In normal, non-diabetic rats, the relative abundance of the different phyla was: 61%, 24%, 6%, 7% and 2%, respectively. In diabetic rats, however, there was reduction in abundance of all phyla, except Actinobacteria, which increased more than three-fold. The relative abundance of the phyla in diabetic rats was 57%, 19%, 20%, 0.4% and 2%, respectively. All treatment groups showed a reduction in relative abundance of phylum Spirochaetae. There was no significant difference between normal rats and the treatment groups in the other phyla (Fig. 1).
Fig. 1. Relative abundance of bacterial phyla in faecal samples from normal, diabetic and treated rats. Diabetic rats showed significant increase in Phylum Actinobacteria with decrease in all other phyla, while all treatment groups showed reduction in Phylum Spirochaetae.
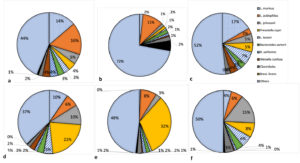
At the species level, significant (P < 0.05) differences were observed in the relative abundance of individual bacterial species within the phyla. Major bacterial species identified in the normal rat samples included: Lactobacillus murinus, L. acidophilus, L. johnsonii, Prevotella copri, L. leuteri, Bacteroides (B.) sartorii, B. uniformis, Weisella confusa, Clostridiales, and Brevibacterium linens among others. These ten bacterial species accounted for more than 50% of the total bacteria, with L. murinus and L. acidophilus being the most dominant (Fig. 2a and 3a). In diabetic rats, however, there was a noticeable disruption in both the diversity and abundance of the flora. Most of the core bacteria reduced in relative abundance, while other species such as Brevibacterium spp, Brachybacterium spp, Corynebacterium variable, Desulfovibrio, Lactobacillus amylovorous etc., previously present in relatively lower abundance, increased in abundance (Fig. 2b and 3b). In rats treated with glibenclamide and G. latifolium hydro-alcoholic extract, the diversity of the bacterial flora was restored (Fig. 2c – 2f). However, differences were observed in the relative abundance of the bacteria in different treatment groups. Rats treated with moderate (400 mg/Kg body weight) and high dose (800 mg/Kg body weight) of G. latifolium extract showed enrichment of L. johnsonii and Prevotella copri, with a reduction in the other species (Fig. 2d and 2e). A more balanced mix was observed at 400 mg/Kg bw (Fig. 2d and 3c).
Fig. 2. Relative abundance of major bacterial species in the colonic flora of rat groups: (a) normal rats (b) untreated diabetic rats (c) rats treated with 200 mg/Kg bw G. latifolium (d) rats treated with 400 mg/Kg bw G. latifolium (e) rats treated with 800 mg/Kg bw G. latifolium (f) rats treated with glibenclamide. Note significant disruption of composition of flora of diabetic rats compared with groups treated with G. latifolium, in which there was increase in species associated with improved glucose metabolism. Dosage of 400 mg/Kg bw produced the best results.
Effect of treatments on blood glucose levels
Intraperitoneal injection of alloxan monohydrate to rats caused elevated blood glucose levels (diabetes) in the rats, with blood glucose levels rising from baseline levels of about 85 mg/dl in normal rats to about 350 mg/dl in alloxan-treated rats. Administration of G. latifolium extract to the diabetic rats over a period of 21 days effectively reduced the blood glucose levels in a dose dependent manner (Table 1). Moreover, the effect was lasting, as the treated rats maintained normal blood glucose levels up to three weeks after treatment was terminated. The best anti-diabetic effect was observed with the dose of 400 mg/Kg body weight. All the 5 untreated diabetic rats in group 6, died shortly after the 21 days experimental period.
Table (1):
Fasting blood sugar levels in rats before and after treatment with different concentrations Gongronema latifolium leaf extract.
Stages |
Normal rats |
Diabetic Rats |
Rats treated with 200 mg/kg bw G.l |
Rats treated with 400 mg/kg bw G.l |
Rats treated with 800 mg/kg bw G.l |
Rats treated with (Glibenclamide) |
|---|---|---|---|---|---|---|
Baseline |
76.33± 10.02ab |
88.00 ± 2.00b |
68.33 ± 7.64a |
87.67 ± 1.53b |
91.67± 10.69b |
84.00 ± 14.00ab |
After induction |
94.00± 8.72a |
310.33± 43.89b |
217.67± 140.01ab |
320.00± 121.51b |
254.00± 72.13ab |
330.33 ± 69.97b |
Day 3 |
88.67± 7.57a |
446.67± 46.15d |
352.00± 112.80cd |
272.00± 26.15bc |
324.00± 57.24bc |
231.00 ± 35.37b |
Day 6 |
89.67± 7.51a |
483.00± 73.82c |
110.67± 29.28ab |
121.67± 15.50ab |
175.67± 33.29b |
90.00 ± 6.00a |
Day 9 |
93.33± 6.43a |
412.67± 44.12b |
150.67± 61.49a |
137.33± 41.55a |
132.00± 57.82a |
82.33 ± 10.02a |
Day 12 |
89.67 ± 4.51a |
514.00± 41.76b |
130.67± 42.58a |
107.33± 37.22a |
99.00 ± 16.46a |
86.00 ± 2.00a |
Day 15 |
63.67± 7.37a |
442.33± 69.50b |
107.00± 42.04a |
92.67 ± 28.02a |
94.67 ± 6.43a |
90.33 ± 1.53a |
Day 18 |
65.00± 7.81a |
378.33± 96.72b |
105.33± 37.75a |
93.00 ± 7.94a |
82.67 ± 7.57a |
72.67 ± 7.02a |
Day 21 |
80.67± 7.02a |
552.00± 56.32b |
94.33 ± 21.78a |
88.00 ± 5.29a |
64.00 ± 5.29a |
66.67 ± 13.32a |
1 week after treatment |
85.33± 5.69ab |
– (animals dead) |
89.00 ± 6.56b |
85.00 ± 9.54ab |
72.67 ± 7.02a |
84.00 ± 7.21ab |
2 weeks after treatment |
82.33 ± 7.37a |
– (animals dead) |
87.33 ± 8.51a |
82.67 ± 5.51a |
82.33 ± 9.51a |
84.00 ± 6.00a |
3 weeks after treatment |
81.33± 4.16a |
– (animals dead) |
97.67 ± 3.51c |
80.67 ± 5.86a |
90.00 ± 2.00b |
84.00 ± 2.00ab |
Mean values with different alphabets as superscripts in a row differ significantly (p ≤ 0.05)
Mean values with different numbers as superscripts for a parameter in a column differ significantly (p ≤ 0.05)
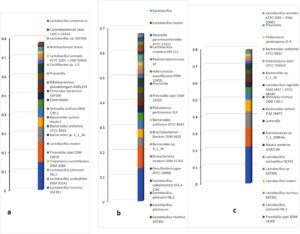
Fig. 3. Dominant bacterial species in the colonic flora of (a) normal rat, (b) untreated diabetic rat and (c) rat treated with 400 mg/Kg bw G. latifolium. Note depletion of the normally dominant species in diabetic rat and proliferation of other species (dysbiosis). Note also, restoration and improvement with G. latifolium treatment.
The gastrointestinal tract of vertebrates is a delicately balanced ecosystem, containing a variety of microbial species that interact with the host and with each other. These microbial species co-exist in a delicate balance, which when disrupted – a condition known as dysbiosis – often has negative consequences for the host’s health. Studies have established a strong link between the gut microbiome and metabolic diseases such as diabetes and suggested that these diseases may be managed through modulation of the gut flora4,15-18. This study was conducted to evaluate the modulatory effect of an edible plant, G. latifolium, claimed to have anti-diabetic activity in traditional medicine, on the gut flora of drug-induced diabetic rats.
A previous study had evaluated and reported the antidiabetic activities of aqueous and methanolic extracts of G. latifolium11. This study focused mainly on the effects of a hydro-alcoholic (30% ethanol) preparation of the leaves of the plant on the gut microbiome of diabetic rats.
In this study, metagenomic analyses of faecal samples of normal rats showed the presence of bacteria, fungi, viruses, protists as well as various resistance genes and virulence factors within the rat gut. The study however focused on only the bacteria. Five major phyla of bacteria were identified in normal non-diabetic rats, in the order – Firmicutes > Bacteroidetes > Spirochaetae > Actinobacteria > Proteobacteria. The Firmicutes and Bacteroidetes accounted for more than 80% of all the bacteria; having 61% and 24% relative abundance, respectively. This is consistent with the reports of Clarke et al.19 and Li et al.20 that the Firmicutes and Bacteroidetes are the most predominant phyla in the rat gut. However, this order was altered in diabetic rats, with significant decreases in relative abundance of bacteria in three of the phyla (Firmicutes, Bacteroidetes and Spirochaetae), in favour of Actinobacteria, which became the second most abundant phylum (Fig. 1).
In healthy animals, gut bacteria comprise a great diversity of bacterial species, including beneficial or friendly bacteria and potentially harmful bacteria. The friendly bacteria keep a check on the abundance of harmful organisms, through a variety of mechanisms, including lowering of pH, production of inhibitory substances, competitive exclusion of receptors and nutrients among others. A depletion of the protective friendly bacteria, by any mechanism, including antibiotic use or disease, affects this protective shield and creates opportunity for overgrowth of the harmful bacteria, a condition known as dysbiosis. Dysbiosis, in turn, has various negative consequences for the host, depending on the organisms that eventually dominate. In this study, some bacterial species were identified as major bacteria in the normal rat gut, based on recurrence in different rats and their relative abundance. These include: L. murinus, L. acidophilus, L. johnsonii, Prevotella copri, L. leuteri, B. sartorii, B. uniformis, Weisella confusa, Clostridiales, and Brevibacterium linens; mostly of the Firmicutes and Bacteroidetes phyla. The relative abundance of these bacteria was therefore compared in untreated diabetic rats versus rats treated with G. latifolium leaf extract and standard anti-diabetic drug (glibenclamide). The results showed that induction of diabetes in the rats caused a significant disruption of the gut bacterial flora as L. acidophilus became the most abundant organism (11.01% + 2.00%) in the diabetic animals, followed by Brevibacterium linens (5.28% + 2.96%), with appearance of species such as Brachybacterium paralongomeratum, Corynebacterium variable, Brevibacterium sp VCM10, L. amylovorus, L. plantarum and L. coleohominis; while L. murinus, L. johnsonii, L. reuteri and Prevotella copri decreased in abundance (Fig. 1b and 2b). Induction of diabetes in the rats, thus resulted in dysbiosis, with members of the Actinobacteria replacing some of the core bacteria and becoming the predominant flora. This observation is in agreement with one study suggesting that there is disturbance of microflora with onset of diabetes21. Many studies in both animal models and humans have shown that there are indeed differences in the microflora of diabetic and non-diabetic individuals4,15,18,22, but while most of these studies suggest that the composition of the flora may predispose the host towards development of diabetes (that is, composition as the cause and diabetes as effect), the results in this study show clearly that diabetes can be the cause of dysbiosis. The association between dysbiosis and diabetes can therefore be likened to a vicious cycle; dysbiosis predisposes towards development of diabetes and diabetes in turn, causes dysbiosis, in which there is increased abundance of opportunistic pathogens.
The condition was however ameliorated by treatment of the rats with G. latifolium leaf extract and glibenclamide, in this study. At all three test doses of G. latifolium, as with glibenclamide, there was noticeable restoration of the microflora as compared with that of untreated diabetic rats (Fig. 2 and 3). Moreover, the treatments produced increases in relative abundance of species like Prevotalla copri and L. johnsonii, which are associated with improved glucose metabolism3,23,24. The best results were observed with G. latifolium extract at the dosage of 400 mg/Kg body weight. An interesting observation in this study was the fact that although there were no overt changes in ratios of the different phyla between the animal groups, the changes were observed in relative abundance of species and strains within the phyla. As seen in Fig. 1, besides the dramatic increase in abundance of the phylum Actinobacteria in diabetic rats, the Firmicutes and Bacteroidetes remained the predominant phyla in most other groups. However, a look at the strain level showed that there were significant changes in abundance of individual species (Fig. 3). These findings suggest that studies to evaluate changes in the microbiome should ideally be at the strain level and not at the phylum level.
Tracking of the fasting blood glucose of the rats over a six-week period showed that G. latifolium leaf extract also effectively normalized the blood glucose levels of the diabetic rats and the effect was sustained for up to three weeks following cessation of treatment. This finding is consistent with previous reports that G. latifolium has anti-diabetic activity11. The best result was again observed at the dose of 400 mg/Kg body weight. Although the dose of 800 mg/Kg produced equally good blood glucose results, in terms of effect on microbiome, this high dose appeared to cause an over-enrichment of only one or two bacterial species, while causing an overt reduction in some core bacterial flora, including L. murinus, L. johnsonii, L. leuteri, B. sartorii and Weisella confusa. This finding suggests that treatments aimed at modulating the microbiome may be better at moderate rather than high doses, even if the treatment agent is non-toxic.
The results from this study show that hydro-alcoholic leaf extracts of G. latifolium, administered at moderate doses, can effectively control diabetes through modulation of the gut microbiome and provides support for management of metabolic diseases through modulation of the microbiome.
ACKNOWLEDGMENTS
The authors wish to thank Dr. Remigius Onoja and Mr. Anthony Agbo of the Faculty of Veterinary Medicine, University of Nigeria, Nsukka, for their assistance in the handling and care of the experimental animals.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
EIM conceptualized, organized and supervised the research work. She also prepared the final versions of the manuscript.
CIK carried out the experiments and prepared the first draft of the manuscript.
AMC assisted with the genomic DNA extractions and some other molecular aspects of the research. She also assisted in sample collection.
UKO assisted with measurements of blood glucose and other hematological parameters during the project. He also reviewed the initial draft of the manuscript.
FUNDING
None.
ETHICS STATEMENT
The animals were handled with humane care according to NIH guidelines for use and care of laboratory animals. The protocol was also reviewed and approved by the University of Nigeria Faculty of Veterinary Medicine Animal Care and Use Committee (approval number: FVM-UNN-IACUC-2020-1059).
AVAILABILITY OF DATA
The metagenomic data are available on the CosmosID® portal, http://app.cosmosid.com
Other data generated are included in this manuscript.
- Shreiner AB, Kao JY, Young VB. The gut microbiome in health and disease. Curr Opin Gastroenterol. 2015;31(1):69-75.
Crossref - Mohajeri MH, Brummer RJM, Rastall RA, et al. The role of the microbiome for human health: from basic science to clinical applications. Eur J Nutr. 2018;57(Suppl 1):S1-S14.
Crossref - Shah NJ, Swami OC. Role of probiotics in diabetes: a review of their rationale and efficacy. EMJ Diabet. 2017;5:104-110.
- Belizario JE, Faintuch J, Garay-Malpartida M. Gut microbiome dysbiosis and immunometabolism: New frontiers for treatment of metabolic diseases. Mediat Inflamm. 2018;2018:2037838.
Crossref - World Health Organization. Diabetes Facts Sheet. 2020; Available: https://www.who.int/news-room/fact-sheets/detail/diabetes
- Grossman LD, Roscoe R, Shack AR. Complementary and Alternative Medicine for Diabetes. Can J Diabetes. 2018;42(Suppl 1):S154-S161.
Crossref - Pandey A, Tripathi P, Pandey R, Srivatava R, Goswami S. Alternative therapies useful in the management of diabetes: a systematic review. J Pharm Bioallied Sci. 2011;3(4):504-512.
Crossref - Ekor M. The growing use of herbal medicines: issues relating to adverse reactions and challenges in monitoring safety. Front Pharmacol. 2014;4:177.
Crossref - Yakubu MT, Sunmonu TO, Lewu FB, Ashafa AOT, Olorunniji FJ, Eddouks M. Medicinal plants used in the management of diabetes mellitus 2015. ECAM Comp & Alternative Med. 2015;2015:467196.
Crossref - Kooti W, Farokhipour M, Asadzadeh Z, Ashtary-Larky D, Asadi-Samani M. The role of medicinal plants in the treatment of diabetes: a systematic review. Electron Physician. 2016;8(1):1832-1842.
Crossref - Akah PA, Uzodimma SU, Okolo CE. Antidiabetic activity of aqueous and methanol extract and fractions of Gongronema latifolium (Asclepidaceae) leaves in alloxan diabetic rats. J Appl Pharm Sci. 2011;1:99-102.
- Ezeonu IM, Akobueze EU, Chah KF. Prebiotic effects of Vernonia amygdalina and Ocimum gratissimum aqueous leaf extracts in rabbit (Oryctolagus cuniculus). Afr J Biotech. 2012;11(10):2537-2547.
Crossref - National Research Council (US) Committee for the Update of the Guide for the Care and Use of Laboratory Animals. Guide for the Care and Use of Laboratory Animals. 8th edition. Washington (DC): National Academies Press (US); 2011. Available: https://www.ncbi.nlm.nih.gov/books/NBK54045/
- Iweala EJ, Okeke CU. Comparative study of the hypoglycaemic and biochemical effects of Caranthus roseus and chlorpropamide on alloxan-induced diabetic rats. Biokemistri. 2005;17(2):149-159.
Crossref - Brunkwall L, Orho-Melander M. The gut microbiome as a target for prevention and treatment of hyperglycaemia in type 2 diabetes: from current human evidence to future possibilities. Diabetologia. 2017;60:943-951.
Crossref - Sandberg J, Kovatcheva-Datchary P, Bjorck I, Backhed F, Nilsson A. Abundance of gut Prevotella at baseline and metabolic response to barley prebiotics. Eur J Nutr. 2019;58:2365-2376.
Crossref - Guerreiro CS, Calado A, Sousa J, Fonseca JE. Diet, microbiota, and gut permeability-the unknown triad in rheumatoid arthritis. Front Med. 2018;5:349.
Crossref - Liu Y, Lou X. Type 2 diabetes mellitus-related environmental factors and the gut microbiota: emerging evidence and challenges. Clinics. 2020;75:e1277.
Crossref - Clarke SF, Murphy EF, Nilaweera K, et al. The gut microbiota and its relationship to diet and obesity: new insights. Gut Microbes. 2012;3(3):186-202.
Crossref - Li D, Chen H, Mao B, et al. Microbial biogeography and core microbiota of the rat digestive tract. Sci Rep. 2017;7:45840.
Crossref - Yan X, Feng B, Li P, Tang Z, Wang L. Microflora disturbance during progression of glucose intolerance and effect of sitagliptin: an animal study. J Diabetes Res. 2016;2016:2093171.
Crossref - Egshatyan L, Kashtanova D, Popenko A, et al. Gut microbiota and diet in patients with different glucose tolerance. Endocr Connect. 2016;5(1):1-9.
Crossref - Zhang Y, Zhang H. Microbiota associated with type 2 diabetes and its related complications. Food Sci Hum Well. 2013;2(3-4):167-172.
Crossref - Kovatcheva-Datchary P, Nilsson A, Akrami R, et al. Dietary fibre-induced improvement in glucose metabolism is associated with increased abundance of Prevotella. Cell Metab. 2015;22(6):971-982.
Crossref
© The Author(s) 2021. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.