ISSN: 0973-7510
E-ISSN: 2581-690X
The present study envisaged to optimise the culture parameters for improved bioactive secondary metabolite production by Geosmithia pallida (KU693285), a potent endophytic fungi, isolated from endangered medicinal plant Brucea mollis Wall ex. Kurz. G. pallida was isolated from healthy, surface sterilized leaf, bark and root tissues of B. mollis. Sporulation was induced in sterile G. pallida by growing it on modified PDA media. Identification of G. Pallida was confirmed by ITS r-DNA sequence analysis. The culture parameters were optimized for enhanced production of metabolites. Maximum metabolite production was observed at 25°C incubation temperature and 6.5 pH on 28th day of inoculation, when lactose and yeast extract were added in the basal medium respectively as carbon and nitrogen source along with 3.5 g/l NaCl. The inhibition zone showed by G. pallida was significantly higher during the optimum culture conditions than that of the control (basal media). From the present study, it is concluded that for the maximum production of bioactive metabolite the culture parameters need to be optimized. These results would steer the authors to carry out further studies on purification, characterization, and identification of bioactive metabolites produced by G. pallida (KU693285) for pharmaceutical as well as agricultural applications.
Endophytic fungi, Geosmithia pallida (KU693285), Bioactive secondary metabolite, optimization, Cultural parameters
Endophytes are the microbes that colonize living internal tissues of plants without causing any immediate, overt negative effects1. Endophytic fungi are proven source of bioactive metabolites in today’s world2. They are the nature’s hidden resources of antimicrobials, antioxidants, insecticides, herbicides etc.3,4. The endophytic fungal diversity is more or less unexplored. Proper exploration of medicinal, endemic, rare, endangered etc. plants is required for the enumeration of novel endophytic fungi residing inside them for further study.
Appearance of multi-drug resistant strains of pathogens has guided researchers to search for new noble antimicrobial compounds. The endophytic microorganisms are one of the potential biological resources of potent active molecules because of the pristine specialized niche they have occupied5. It is observed that these organisms may produce metabolites identical to the one produced by their hosts6. The production of bioactive metabolites by fungi is affected by the culture parameters like, pH, temperature, incubation period, different nitrogen and carbon sources, NaCl concentration etc. of the medium and the availability of the metal ions in the media used7,8. Optimization and maintenance of suitable culture parameters are necessary criteria to achieve maximum production of bioactive metabolites by microbial strains.
B. mollis of family Simaroubaceae, is a small tree and is recorded as endangered species in North East India by CAMP survey (Annonymous 2003). It is traditionally used as antimalarial and antimicrobial agent but has antiplasmodial, cytotoxic, anticancer, diuretic, cardiovascular properties as well9. Being endangered and growing in biodiversity hotspot region of the world, B. mollis may have noble endophytic fungi having antimicrobial, antimalarial, anticancer etc. properties like that of the host. Use of endophytic fungi in place of the plants for the production of the compounds having antimicrobial, antimalarial, anticancer etc. properties, may help in conserving the host plants as their use will be lessen. Keeping this hypothesis in mind, an attempt was made to conserve endangered medicinal B. mollis through exploration of endophytic fungi associated with it, for antimicrobial compound production for pharmaceutical purposes in place of the plant.
The present investigation envisaged to optimise the medium for optimum production of metabolite by endophytic G. pallida (KU693284) isolated from B. mollis.
Isolation and identification of endophytic fungi
Leaf, bark and root samples were washed and cut into about 2 cm long and 0.5 cm broad segments with sterile knife. Segments were sequentially immersed in 70% ethanol for 3 minutes, 4% aqueous solution of sodium hypochlorite (NaClO) for 5 minutes and 70% ethanol for 1 minute and finally in 0.1% Mercuric chloride (HgCl2) for 3 minutes10. The segments thereafter were rinsed with sterile de-ionized water to remove the traces of HgCl2 and then surface-dried. The effectiveness of surface sterilization was tested for every segment using imprint method11. The segments were then inoculated in Petri plates (90×15 mm) containing different media viz., Czapeck-Dox-Agar (CDA), Potato-Dextrose-Agar (PDA), Malt Extract Agar (MEA), Sabouraud Dextrose Agar (SDA), V8 juice agar (V8JA), nutrient agar (NA) and water agar media. All the media were procured from Himedia, India. Streptomycin (50 µg/ml) was used to prevent bacterial contamination.
As the endophyte Geosmithia pallida didn’t sporulate, sporulation was induced using different media viz., potato dextrose agar, czapeck dox agar and malt extract agar amended with different concentrations of dextrose, peptone, yeast extract and sucrose. Modified media were ME-YEA (Malt extract-yeast extract agar), PDA-PS-YEA (Potato dextrose-peptone sucrose-yeast extract agar) and YE-RB-SA (Yeast extract-rose bengal-sucrose agar). In ME-YEA yeast-extract (0.4 g/l) was amended in malt extract agar media; in PDA-PS-YEA peptone (2 g/l), yeast-extract (2 g/l) and sucrose (2 g/l) were amended in potato dextrose media; in YE-RB-SA sucrose (15 g/l) and rose-bengal (2 g/l) were amended in yeast extract agar. After sporulation it was identified based on morphological characters using manuals of Nagamani et al. (2006)12. Taxonomical identity of the G. pallida was confirmed using molecular tools. The sequence was submitted to the GeneBank.
Optimization of in vitro physico-chemical parameters for optimum bioactive metabolite production
Standardization of the basal media
To standardise the basal media for optimum metabolite production by G. pallida, different media namely Czapeck-Dox-broth (CDB), Potato-Dextrose-Broth (PDB), Malt Extract Broth (MEB), Sabouraud Dextrose Broth (SDB), V8 juice Broth and Nutrient broth media were used7. All the media were procured from HiMedia Laboratories, Mumbai, India. Conical flasks containing 50 ml broth media having initial pH 6.6±0.2 was inoculated with 7 days old inoculum of the isolate and flasks were then incubated at stationary condition at 27 ± 2 °C for 20 days. The media in which the fungus produced highest metabolite was used as basal media for further work.
Effect of carbon and nitrogen sources
Different Carbon and Nitrogen sources at 10 g/l (w/v) were added in the 50 ml basal media13. Glucose, Glycerol, D-mannitol, Lactose, Sucrose, Galactose, Xylose, Starch, Maltose, Sorbitol, Na-citrate as carbon sources and Beef extract, Yeast extract, Peptone, Ammonium sulphate, Ammonium chloride, Sodium nitrate as nitrogen sources were used. After inoculation the conical flasks were incubated at stationary condition for 20 days, at 27±2 °C temperature and pH 6.6±0.2.
Effect of NaCl concentration
The effect of NaCl concentration on the bioactive metabolite production by G. pallida was carried out by incubating the fungus in 50 ml basal media containing different concentrations of NaCl ranging from 1 g/l to 10 g/l, for 20 days, at 27±2°C temperature and pH 6.6±0.2 at stationary condition14.
Effect of temperature
The fungal strain G. pallida was inoculated into the 50-ml basal medium and incubated at different temperatures ranging from 10 °C to 55 °C, at stationary condition for 20 days, at pH 6.6±0.2. Fungal metabolite production at each temperature was determined15.
Effect of pH
To study the effect of pH on the bioactive metabolite production by G. pallida was tested using 50 ml basal media containing different pH levels ranging from 3.5 to 8.5. The pH of the basal medium was adjusted by adding 0.1N NaOH or 0.1N HCl and incubated at 27±2 °C temperatures for 20 days at stationary condition16.
Effect of incubation period and shaking condition
Incubation periods ranging from 2 to 40 days were used to determine the effect of incubation period on the active metabolite production by G. pallida and other conditions were maintained as like that of the basal media7.
To determine the effect of shaking condition on active metabolite production, culture flasks with basal media were incubated at 27±2 °C in an orbital shaker at 150 rpm at for 20 days, at pH 6.6±0.2.
Determination of production of bioactive metabolite
Production of bioactive metabolite by G. pallida was determined by antimicrobial activity assay of the metabolite against some microbes.
Extraction of the metabolite
Fungal mycelial mat was separated by filtration. After that bioactive secondary metabolite was extracted three times from the filtrate using ethyl acetate17. The ethyl acetate extract was concentrated to dryness by using rotary vacuum evaporator (Model: EYELA/NVC-2100) at 40 °C according to the protocols of Phongpaichit et al. (2007)17. The resulting extracts of the isolate was diluted with dimethyl sulfoxide (DMSO) at a concentration of 10 mg/ml. The solution was sterilized by filtration through 0.4 µm Cellulose Acetate (hydrophilic) filter and kept for antimicrobial activity assay.
Antagonistic activity assay
Antimicrobial activity of the ethyl acetate extract of the metabolite produced by G. pallida was tested against two gram-negative, viz., Escherichia coli (MTCC 443) and Klebsiella pneumoniae (MTCC 619), two gram-positive, viz., Bacillus subtilis (MTCC 441) and Staphylococcus epidermidis (MTCC 435) bacteria and two fungi i.e., Candida albicans (MTCC 183) and C. Tropicalis (MTCC 1000). The test organisms except S. epidermidis and Candida sp. were collected from the Institute of Microbial Technology (IMTECH), Chandigarh, India. S. epidermidis was collected from Regional Institute of Medical Sciences (RIMS), Imphal, India and Candida albicans (MTCC 183) was collected from Defense Research Laboratory (DRL), Tezpur, India. Kirby-Bauer disc diffusion method was followed for determination of antimicrobial activity of the metabolite18. For this, plates with PDA and NA medium were inoculated with 0.2 ml of cultured test microorganisms7. The inoculum in the plates was evenly spread out with the help of a sterile cotton swab. A fixed volume of 20 µl of the extracted metabolite solution of the isolate was then applied to the sterile discs (6.0 mm diameter, Whatman antibiotic assay discs). The diameters of the inhibition zones formed surrounding the discs after 48-72 h at 25±2 ºC were recorded. Three replicates were maintained in each case. Larger was the zone diameter, more was the metabolite production7,13,16.
Statistical analysis
All experiments were carried out in triplicates and results are expressed as mean ± SD (n=3). To analyse the differences among mean values of different assays one-way analysis of variance (ANOVA) was conducted followed by least significant difference (LSD) test in Microsoft office excel 2016. P values of less than 0.05 were considered to indicate statistical significance.
Identification of Geosmithia pallida
G. pallida was identified based on morphological and reproductive characteristics. It was sterile in nature and creamy white in appearance when isolated and did not sporulate in unamended media. It sporulated when cultured on MEYEA and PD-PS-YEA media. Sporulation was the best in PD-PS-YEA media. The isolate, however, did not grow in YERBSA media. The ITS region of rDNA of G. pallida was sequenced to confirm its identification. The sequence was submitted to GeneBank-NCBI (Accession number KU693285).
Optimization of in vitro physico-chemical parameters for bioactive metabolite production
Standardization of Basal media
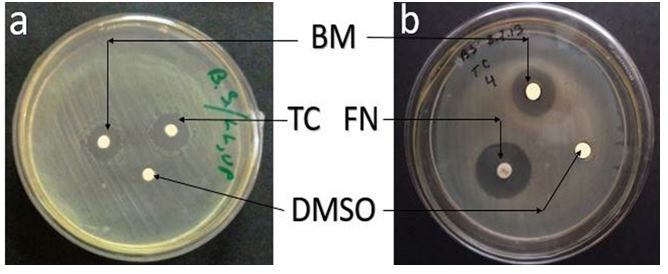
Maximum zone of inhibition was recorded in Potato dextrose broth (PDB) medium (Table 1). The zone of inhibition was significantly higher when PDB was used for metabolite production by the fungal strain than that of the other media used (p<0.05) (Table 1). This indicated that the ingredients of potato dextrose media induced bioactive metabolite production by the fungal strain. Therefore, PDB was selected as the basal medium for further experiments. Variations in the nutritional constituents of the culture media used affect the bioactive metabolite production by the strain. Photo-plates of antimicrobial activity shown by bioactive metabolite produced by G. pallida (KU693285) against Bacillus subtilis (Fig.1a) and Candida albicans (Fig.1b), co-assayed with antibiotic discs- Tetracycline (standard antibacterial) and Fluconazole (standard antifungal) as well as disc dissolved in 100% DMSO, have been shown in Fig.1.
Table (1):
Effect of different culture media on the bioactive metabolite production by G. pallida (KU693285) against test microbes.
| Zone of inhibition (mm) | ||||||
|---|---|---|---|---|---|---|
| Media | Gram- positive bacteria | Gram- negative bacteria | Fungi | |||
| Se | Bs | Kp | Ec | Ca | Ct | |
| PDB | 13±0.43a | 14.07±0.12a | 18.17±0.76a | 11±0.09a | 23.93±0.12a | 17.72±0.21a |
| CDB | 13.07±0.51a | 12.14±0.39b | 10.31±0.29b | 9.19±0.23b | 21.18 ± 0.5b | 18.23±0.45b |
| MEB | 11.91±0.23b | 10.19±0.3c | 11.29±0.41c | 9±0.86b | 16.45 ± 02c | 17.28±0.34a |
| SDB | 12.63±0.19b | 12.38±0.11b | 10.09±0.52b | 11.33±0.17a | 20.28 ± 0.48b | 13.83±0.12c |
| V8JB | 9.17±0.27c | 7.18±0.28d | 7.21±0.37d | 6.81±0.18c | 18.24 ± 0.25d | 11.21±0.63d |
| NB | 3.61±0.33d | 4.58±0.42e | 3.31±0.47e | 0 | 7.89 ± 0.19e | 6.43±0.35e |
| Antibiotic disc | 16.67±0.58e | 25.27±0.25f | 10.91±0.36b | 20.0±0.29d | 15±0.64c | 19.0±0.39b |
| Negative control | 0 | 0 | 0 | 0 | 0 | 0 |
PDB=Potato dextrose broth, CDB=Czapek dox broth, MEB=Malt extract broth, SDB=Sabouraud dextrose broth, V8JB=V8 juice broth, NB=Nutrient broth. Se=S. epidermidis, Bs=B. subtilis, Kp= K. pneumoniae, Ec=E. coli, Ca=C. albicans, Ct= C. tropicalis. Positive control: Co-assayed antibiotics (Tetracycline-30 µg/disc, Fluconazole-10 µg/disc). Negative control: Sterile disc (5mm diameter) immersed in Dimethyl sulphoxide (DMSO). Data mean of three replicates ±SE (n=3). Means with different letters within a column were significantly different (p < 0.05).
Fig. 1. Antimicrobial activity of bioactive metabolite (BM) produced by G. pallida (KU693285) against (a) Bacillus subtilis and (b) Candida albicans (MTCC183), Positive Control-(a) Tetracycline (TC-30 µg/dsc) and (b), Fluconazole (FN-10 µg/disc), Negative Control-Dimethyl sulfoxide (DMSO)
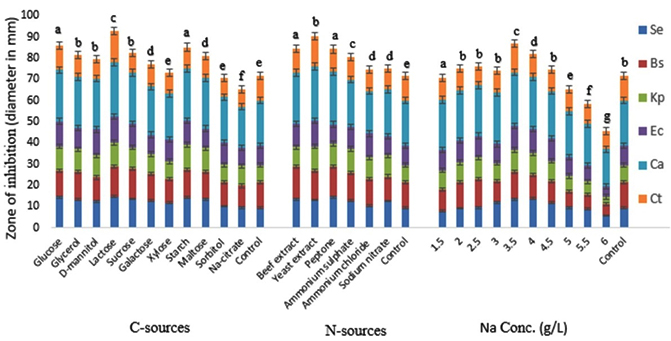
Fig. 2. Effect of different C-source, N-sources and NaCl concentration on the bioactive metabolite production by G. pallida (KU693285) against test microbes. Se=S. epidermidis, Bs=B. subtilis, Kp= K. pneumoniae, Ec=E. coli, Ca=C. albicans, Ct= C. tropicalis. Vertical bars represent standard deviation (±SD, n=3). Means with different letters within an assay were significantly different (p < 0.05)
Effect of Carbon and Nitrogen Sources
During the present investigation G. pallida was grown in the basal media by replacing the carbon sources as mentioned in material and methods. Lactose had maximum influence on antimicrobial metabolite production followed by starch (Fig.2). The zone of inhibition against C. albicans using lactose and starch respectively were 25.47±0.63 mm and 24.69± 0.25 mm. Among the N-sources used, Yeast extract was most effective followed by peptone for maximum antimicrobial metabolite production in terms of zone of inhibition against C. albicans, which were 25.38±0.51 mm and 24.91±0.44 mm respectively in yeast extract and peptone amended media (Fig.2). The inhibition was significantly higher in the basal media treated with lactose and yeast extract as C- and N-sources respectively than that of the control (basal media) (p<0.05).
Effect of NaCl concentrations
At 3.5 g/l concentration of NaCl, G. pallida produced maximum antimicrobial metabolite (zone of inhibition 25.34±0.19 mm) (Fig.2). After that, the metabolite production gradually decreased with the increase in salt concentration in the basal media. The inhibition was significantly higher in the media treated with 3.5 g/L NaCl than that of the control (p<0.05).
Effect of temperature
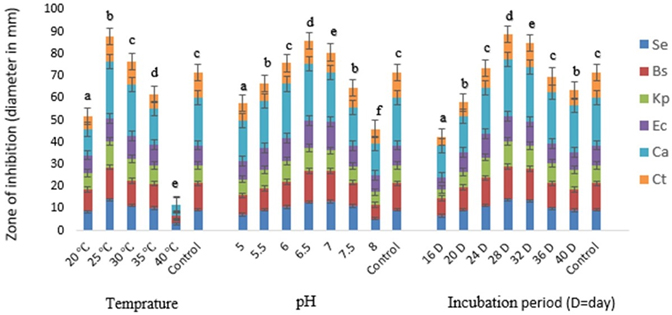
A varied range of incubation temperatures 10 to 55 °C was used to assess its impact on the bioactive metabolite production by G. pallida. The increase of the incubation temperatures from 25 to 30 ºC enhanced the production of bioactive metabolite (Fig.3). At 25 °C G. pallida produced maximum metabolite (inhibition zone 25.33 ± 0.61 mm). However, no antagonistic activity was observed at ≤ 15 °C and ≥ 50 °C temperatures. The inhibition was significantly higher when incubation temperature was 25 °C than the incubation temperature of the control (p<0.05).
Fig. 3. Effect of different temperature, pH and incubation period on the bioactive metabolite production by G. pallida (KU693285) against test microbes. Se=S. epidermidis, Bs=B. subtilis, Kp= K. pneumoniae, Ec=E. coli, Ca=C. albicans, Ct= C. tropicalis. Vertical bars represent standard deviation (±SD, n=3). Means with different letters within an assay were significantly different (p < 0.05)
Effect of pH
The antagonistic activities of G. pallida were also influenced by pH of the medium. Maximum antimicrobial metabolite production (zone of inhibition 25.79±0.44 mm against C. albicans) was recorded at pH 6.5 (Fig.3). The inhibition was significantly higher when pH of the media was 6.5 than the pH of the control (p<0.05).
Effect of incubation period and shaking conditions
The isolate required incubation period of 28 days for maximum production of antimicrobial metabolite (zone of inhibition 25.71 ± 0.13 mm against C. albicans) under stationary conditions (Fig.3). The antimicrobial activity of bioactive secondary metabolite reduced when the conditions changed from stationary to shaking. In terms of zone of inhibition, the zone of inhibition decreased to 16.89±0.32 mm from 23.93±0.12 mm, while the culture broth (basal medium) was incubated at shaking conditions from stationary condition. The inhibition was significantly higher on 28th of incubation than the incubation period of the control (p<0.05).
Advances in technology have sparked a renaissance in the discovery of natural antimicrobial product from microbial sources, like actinomycetes, cyanobacteria, bacteria and fungi19. More recently, researches on endophytic fungi, has resulted in several newly discovered antibiotics as they are the source of a wide range of bioactive compounds fighting against various pathogens20. However, different researchers have proved that the culture parameters, i.e., the nutrient supplements, nutrient concentration and the culture conditions of the media effects the metabolite production by the fungi.
In the present study, maximum antimicrobial metabolite production by G. pallida (KU693285) was observed on Potato dextrose broth (PDB) medium which was used as basal media for further experiment (Table 1). This result supports the findings of Rabbani et al. (2011) and Bhattacharya and Jha (2011)7,21. While Kalyani et al. (2016) found maximum growth and metabolite production by Aspergillus niger (MTTC 961) using sabouraud dextrose broth19.
Miao and Qian (2005) studied the effect of culture medium on mycelial growth, metabolite profile and antimicrobial compound yield by a marine derived fungus Arthrinium c.f. saccharicola suggesting the need of optimal culture parameters to achieve maximal bioactivity by the fungus22. Merlin et al. (2013) observed that the endophytic fungi Fusarium solani isolated from Tylophoraindica, had optimum growth and metabolite production when cultured for 9 days in the media containing 4% dextrose, 0.05% yeast extract and aspartic acid (0.01%) as carbon, nitrogen and amino acid sources respectively23. Bhattachryya and Jha (2011) observed that the most effective C- and N- sources for maximum bioactive metabolite production by Aspergillus strain TSF 146 respectively was sucrose and asparagine7. Merlin et al. (2013) optimized the growth and metabolite production by Fusarium solani using dextrose as C-source and yeast extract as N-source in the basal medium23. But in the present study lactose was the most effective followed by sucrose as C-source, while yeast-extract was the most effective followed by peptone as N-source for bioactive metabolite production by G. pallida (KU693285) (Fig.2). This result supports the findings of Gogoi et al. (2008) and Mathan et al. (2013)16,24.
Gogoi et al. (2008) found that the NaCl concentration 2.5–3.0 % was the optimum for maximum production of bioactive metabolite by an antagonist fungus, Fusarium sp.16. Bhattachryya and Jha (2011) observed that the NaCl concentration of 5 g/l was optimal for maximum production of bioactive metabolite7. In the present study, optimal NaCl concentration was 3.5 g/l for G. pallida (KU693285) and the values gradually decreased with the increase in NaCl concentration in the basal media (Fig.2). This result supports the findings of Bhattachryya and Jha (2011) and Gogoi et al. (2008)7,16.
Temperature is one of the major parameters influencing the growth rate of antagonist25. Jain and Pundir (2011) also reported 25 ºC temperature as optimal for maximum production of antimicrobial metabolite by Aspergillus terreus26. Maximum growth and metabolite production by Aspergillus niger (MTTC-961) was observed at 30 oC19. During the present study, G. pallida (KU693285) produced maximum bioactive metabolite at incubation temperature of 25 ºC supporting the findings of Ritchie et al. (2009), Jain and Pundir (2011) and Mathan et al. (2013)24, 26,27.
According to Thongwai and Kunopakarn (2007) microorganisms can synthesize antimicrobial compounds at pH ranging from 5.5 to 8.528. Mathan et al. (2013) and Jain and Pundir (2011) observed the maximum production of antibacterial metabolite by Aspergillus terreus respectively at pH 6.0. and 5.524,26. During the present study, the antagonistic activity of ethyl acetate extract of metabolite produced by G. pallida was also influenced by pH of the medium and 6.5 was the optimum pH for maximum antimicrobial metabolite production (Fig.3).
Alberts et al. (1990) observed that the production of metabolite occurred after 12 days, reached maximum at 91st day and gradually decreased after 91 days of incubation when effect of incubation period was investigated on its production by Fusarium moniliforme29. Aspergillus TSF 146 required 16 days for optimum production of antimicrobial metabolite7. Optimum incubation period for maximum metabolite production by Aspergillus niger (MTTC-961) was 144 hr19. However, G. pallida showed maximum antimicrobial metabolite production on 28th day of incubation followed by 30 days of incubation period and gradually it decreased.
During the present study, shaking condition of incubation did not enhance the antimicrobial metabolite production by G. pallida supporting the findings of Kalyani et al. (2016), Alberts et al. (1990) and Bundale et al. (2015), as compared to the shaking condition19,29,30. From the results obtained during the present study, it can be drawn that to obtain maximum production of bioactive metabolite, the physico-chemical properties have to be optimized.
The results of this study represent that endophytic fungi may serve as a potential natural source of antimicrobial agents. Endophytic fungi are recommended as an organism of pharmaceutical importance with high medicinal value. Therefore, optimization of culture conditions for maximum production of bioactive metabolite by endophytic fungi is very important. Geosmithia pallida (KU693285) produced maximum bioactive metabolite in 25°C incubation temperature and 6.5 pH, on 28th day of inoculation, when lactose and yeast extract were used respectively as carbon and nitrogen source along with 3.5 g/l NaCl. However, the toxicity profiling, structural elucidation of the bioactive compounds produced by G. pallida are yet to be done for pharmaceutical as well as agricultural uses.
ACKNOWLEDGMENTS
The first author is thankful to the University Grant Commission (UGC), New Delhi for providing Basic Scientific Research (BSR) fellowship under Special Assistance Program-Departmental Research Support-I (SAP, DRS-I), Department of Botany, Gauhati University, Guwahati, Assam.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
- Stone, J.K., Bacon, C.W., White, J.F.: An overview of endophytic microbes endophytism defined. In: Microbial Endophytes (Bacon CW, White JF, ed). New York: Taylor & Francis, 2000; pp 3-29.
- Strobel, G.A., Daisy, B., Castillo, U., Harper, J. Natural product from endophytic microorganisms. J. Nat. Prod., 2004; 67: 257-268.
- Malinowski, D.P., Belesky, D.P. Ecological importance of Neotyphodium sp. Grass endophytes in agroecosystems. Grassland Sci., 2006; 52(1): 23-28.
- Knop, M., Pacyna, S., Voloshchuk, N., Kjant, S., Mullenborn, C., Steiner, U., Kirchmair, M., Scherer, H.W., Schulz, M. Zea mays: Benzolinone Detoxification under sulfer deficiency conditions- A complex allelopathic alliance including endophytic Fusarium verticillioides. J. Chem. Ecol., 2007; 33(2): 225-237.
- Krabel, D., Morgenstern, K., Herzog S. Endophytes in changing environments – do we need new concepts in forest management?, iForest– Biogeosci. Forestry., 2004; 6: 109-112.
- Stierle, A., Strobel, G.A. Stierle, D. Taxol and taxane production by Taxomyces andreanae. Science, 1993; 260: 214-216.
- Bhattacharyya, P.N., Jha, D.K. Optimization of cultural conditions affecting growth and improved bioactive metabolite production by a subsurface Aspergillus strain tsf 146. Int. J. App. Biol. Pharm. Technol., 2011; 2(4): 133-143.
- Thakur, D., Bora, T.C., Bordoloi, G.N., Mazumdar, S. Influence of nutrition and culturing conditions for optimum growth and antimicrobial metabolite production by Streptomyces sp. 201. J. Med. Mycol., 2009; 19: 161-167.
- Kakati, D., Barthakur, S.K. Morphological and anatomical studies of vegetative parts of Brucea mollis Wall. ex Kurz, – an endemic medicinal plant of Assam, NeBIO – An Int. J. Environ. Biodivers., 2016; 7: 57-65.
- Strobel, G., Ford, E., Worapong, J., Harper, J.K., Arif, A.M., Grant, D.M. Isopestacin, a unique isobenzofuranone from Pestalotiopsis microspora possessing antifungal and antioxidant properties. Phytochem., 2002; 60: 179-183.
- Schulz, B., Wanke, U., Draeger, S., Aust, H.J. Endophytes from herbaceous plants and shrubs: effectiveness of surface sterilization methods. Mycol. Res., 1993; 97: 1447–1450.
- Nagamani, A., Kunwar, S.K., Monoharachary, C. (ed): Handbook of Soil Fungi. India: New Delhi, 2006; pp 101-427.
- Ripa, F.A., Nikkon, F., Zaman, S., Khondkar, P. Optimal Conditions for Antimicrobial Metabolites Production from a New Streptomyces sp. RUPA-08PR Isolated from Bangladeshi Soil. Mycobiol., 2009; 37(3): 211-214.
- Naik, M.K., Hiremath, P.C., Hegde, R.K. Physiological and nutritional studies on Colletotrichum gloeosporioides. A causal agent of anthracnose of beetle vine. Mysore J Agri Sci., 1988; 22: 471-474.
- Singh, L.S., Mazumder, S., Bora, T.C. Optimization of process parameters for growth and bioactive metabolite produced by a salt-tolerant and alkaliphilic actinomycete, Streptomyces tanashiensis strain A2D. J Med. Mycol., 2009; 19: 225-233.
- Gogoi, D.K., Deka-Boruah, H.P., Saikia, R., Bora, T.C. Optimization of process parameters for improved production of bioactive metabolite by a novel endophytic fungus Fusarium sp. DF2 isolated from Taxus wallichiana of North East India. World J. Microbiol. Biotechnol., 2008; 24: 79-87.
- Phongpaichit, S., Nikom, J., Rungjindamai, N., Sakayaroj, J., Hutadilok-Towatana, N., Rukachaisirikul, V., Kirtikara, K. Biological activities of extracts from endophytic fungi isolated from Garcinia plants. FEMS Immunol. Med. Microbiol., 2007; 51: 517-525.
- Bauer, A.W., Kirby, W.M.M., Sherris, J.C., Turck, M. Antibiotic susceptibility testing by a standardized single disk method. Am. J. Clin. Pathol., 1996; 45: 493-496.
- Kalyani, P., Geetha, S., Hemalatha, K.P.J. Optimization of cultural conditions for improved production and bioactive metabolites by Aspergillus niger (MTCC-961). Eur. J. Pharm. Med. Res., 2016; 3(10): 255-260.
- Bhardwaj, A., Agrawal, P.K. A review fungal endophytes: As a store house of bioactive compound. World J. Pharm. Sci., 2014; 3(9): 228-37.
- Rabbani, N., Bajwa, R., Javaid, A. Influence of culturing conditions on growth and sporulation of Drechslera hawaiiensis, the foliar blight pathogen of Marsilea minuta L. Afr J Biotechnol., 2011; 10: 1863-1872.
- Miao, L., Qian, P.Y. Antagonistic antimicrobial activity of marine fungi and bacteria isolated from marine biofilm and seawaters of Hong Kong. Aquat. Microb. Ecol., 2005; 38: 231-238.
- Merlin, J.N., Christhudas, I.V.S.N., Kumar, P.P., Agastian, P. Optimization of growth and bioactive metabolite production: Fusarium solani. Asian J. Pharm. Clin. Res., 2013; 6(3): 98-103.
- Mathan, S., Subramanian, V., Nagamony, S. Optimization and antimicrobial metabolite production from endophytic fungi Aspergillus terreus KC 582297. Eur. J. Exp. Biol., 2013; 3(4): 138-144.
- Kok, C.J., Papert, A. Effect of temperature on in vitro interactions between Verticillium chlamydosporium and other Meloidogyne-associated microorganisms. BioControl., 2002; 47: 603-606.
- Jain, P., Pundir, R.K. Effect of fermentation medium, pH and temperature variations on antibacterial soil fungal metabolite production. J. Agri. Sci. Technol., 2011; 7: 247-269.
- Ritchie, F., Bain, R.A., McQuilken, M.P. Effects of nutrient status, temperature and pH on mycelial growth, sclerotial production and germination of Rhizoctonia solani from potato. J. Plant Pathol., 2009; 91: 589-596.
- Thongwai N, Kunopakarn J. Growth inhibition of Ralstonia solanacearum PT1J by antagonistic bacteria isolated from soils in the Northern part of Thailand. Chiang Mai. J. Sci., 2007; 34: 345-354.
- Alberts, J.F., Gelderblom, W.C., Gthiel, P.G., Marasas, W.F., Schalkwyk, D.J.V.Z., Behrend, Y. Effects of temperature and incubation period on production of Fumonisin B1 by Fusarium moniliforme. J. Appl. Environ. Microbiol., 1990; 56: 1729-1733.
- Bundale, S., Begde, D., Nashikkar, N., Kadam, T., Upadhyay, A. Optimization of Culture Conditions for Production of Bioactive Metabolites by Streptomyces spp. Isolated from Soil. Adv. Microbiol., 2015; 5: 441-451.
© The Author(s) 2018. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.