ISSN: 0973-7510
E-ISSN: 2581-690X
The following study demonstrated the influence of 1,25D/CA and 1,25D/CUR combinations on Nrf2 expression in primary leukemic cells derived from newly diagnosed CML and AML patients of hematological department of Karaganda Regional Clinical Hospital (Karaganda, Kazakhstan). Methods. The research was conducted by using quantitative real-time PCR. Mononuclear cells were isolated by centrifugation in a Ficoll-PaqueTMPREMIUM density gradient. Cell cultures were incubated in RPMI 1640 medium with test agents. RNA extracted from mononuclear cells was used for cDNA molecules synthesis by reverse transcription. Quantitative real-time PCR was carried out in a DT-322 PCR machine. The relative expression level was quantified by using the 2-DDCt method. The statistical analysis was carried out by GraphPadPrism 6.0 program. Results. It was found 1.25D, CUR, CA alone and their combinations do not lead to an increase in the level of expression of the Nrf2 gene in peripheral blood mononuclear cells of AML and CML patients. The expression of NQO1 gene did not change much in the all cell groups of CML patients, but is pronounced in cells of AML patients grown in a medium supplemented with combinations of 1.25D/CUR, 1.25D/CA and CUR, CA alone. 1.25D/CUR and 1.25D/CA combinations do not enhance the expression level of the Nrf2 gene, but increase the amount of its protein, which in turn augments the expression level of the NQO1 gene.
Vitamin D (1.25D), curcumin, carnosic acid, Nrf2, NQO1, leukemia patients.
Leukemia is the most common malignant disease. In Kazakhstan 6741 new cases of leukemia were registered for the period from 2003-2012 (Igissinov et al. 2013). The main methods of leukemia treatment are bone marrow transplantation, radiotherapy and chemotherapy. Each of these methods has a number of limitations, including immunity suppression, radiation poisoning and development of secondary tumors which is much more resistant to all kinds of treatment. In the present day many laboratories search innovative approach of leukemia treatment. One of these ways is the differentiation therapy. It is based on the induction of leukemic blasts to mature beyond the differentiation block and, thus, to restore the normal cellular phenotype. Vitamin D, converted in the body to its hormonal form 1a,25-dihydroxyvitamin D3 (1,25D), is a potent differentiation agent which can induce maturation and/or apoptosis in different types of cancer cells (Gocek & Studzinski 2009). Although many laboratories have used 1,25D to differentiate various subtypes of AML cells in culture, the required concentrations would be lethal in vivo, principally due to hypercalcemia (Brown & Slatopolsky 2008). One approach to overcoming this problem to combine relatively low doses of 1,25D with the administration of another agent that augments the differentiation-inducing action of 1,25D but that does not enhance the levels of circulating calcium. Danilenko M. and others have previously shown that plant-derived polyphenolic antioxidants, such as carnosic acid (CA) isolated from the rosemary plant, curcumin (CUR) from turmeric and silibinin from milk thistle, markedly enhance the differentiation effects of low nanomolar concentrations of 1,25D in AML cell lines, both human (HL60) (Sokoloski, Shyam & Sartorelli 1997; Liu et al. 1997; Danilenko, Wang & Studzinski 2001; Kang et al. 2001; Danilenko et al. 2001) and murine (Sharabani et al. 2006; Shabtay et al. 2008) as well as in leukemic blasts obtained from AML patients (Zhang et al. 2010). They have shown for the first time that the transcription factor Nrf2 largely mediates the potentiating effect of plant polyphenols on 1,25D-induced differentiation in human AML cells. The enhancement of the differentiation caused by CA in U937 cells correlates with its ability to activate the Nrf2/ARE transcription system and to increase the redundant pathway for the synthesis of cellular glutathione in these cells. Activated Nrf2 binds to ARE sequences located in the promoters of many cytoprotective and antioxidant genes, the induction of which leads to increased detoxification and antioxidant potential. One of these genes is the NAD (P) quinone oxidoreductase 1 (NQO1) gene (Danilenko et al. 2001).
The aim of the research is to study the influence of 1,25D and CA, CUR alone and in the combination on the Nrf2 expression in CML and AML patients’ peripheral blood mononuclear cells.
The material of the study was 8 newly diagnosed patients peripheral venous blood from hematological department of Karaganda Regional Clinical Hospital (Karaganda, Kazakhstan) suffering from CML and AML. The diagnosis was made on the basis of blood and bone marrow examination. Written informed consent was obtained from all patients participated in this study in compliance with the guidelines of the Helsinki Committee’s approved protocol.
Mononuclear cells were isolated by centrifugation in a Ficoll-PaqueTM PREMIUM density gradient (GE HealthcareBio-SciencesAB, Uppsala, Sweden). Cell cultures were maintained in RPMI 1640 medium supplemented with 10% fetal calf serum treated with test agents. Cells were seeded at 1-8 × 106 cells/mL in plates and incubated for 120 hours with CA (10 mM), 1,25D (2.5 µM) and 1,25D (100 µM), CUR (5 mM) and combinations 1,25D (2.5 µM) with CA and CUR. The cells grown in medium supplemented with 0.1% ethanol were used as a control.
Total RNA was extracted from mononuclear cells by using a Ribo-zol-A reagents kit (AmpliSens, Moscow, Russia). The RNA concentration and purity were measured spectrophotometrically on a NanoVueplus instrument (BiochromLTD, Cambridge, England). cDNA molecules were synthesized from RNA by reverse transcription using REVERTA L reagents kit (AmpliSens, Moscow, Russia). Quantitative real-time PCR was carried out in a DT-322 PCR machine (DNA technology, Moscow, Russia). The primers for the reaction were synthesized by Sigma-Aldrich (Rehovot, Israel). The cDNA samples (5 µl) were diluted 25-fold, mixed with the specific primers (0.2mM) and then added to reaction mixture. Standard cycling conditions for this instrument were 3 min initial enzyme activation at 94°C then 42 cycles as follows: 15 sec at 94°C, 30 sec at the annealing temperature (62°C). The results were normalized by GusB mRNA content. The relative expression level was quantified by using the 2–DD Ct method.
All measurements were performed in triplicate. The statistical analysis was carried out by GraphPadPrism 6.0 program (Graph-PadSoftware, San Diego, USA).
The study was conducted from September 2016 to March 2017. Half of the patients were diagnosed with CML (4 people), which is 50%. All these patients showed expression of the chimeric bcr-abl gene, which is allowed doctors to diagnose. Also in all CML patients were observed chronic stage of the disease and leukocytosis. Four patients were diagnosed with AML (50%) in the bone marrow of which more than 45% of blast cells were detected. One of the patients with AML had a subtype M0 with co-expression of CD7. The sex ratio was as follows: 6 females and 2 males. The mean age was 61.1 ± 10.9 years.
Nrf2 gene expression
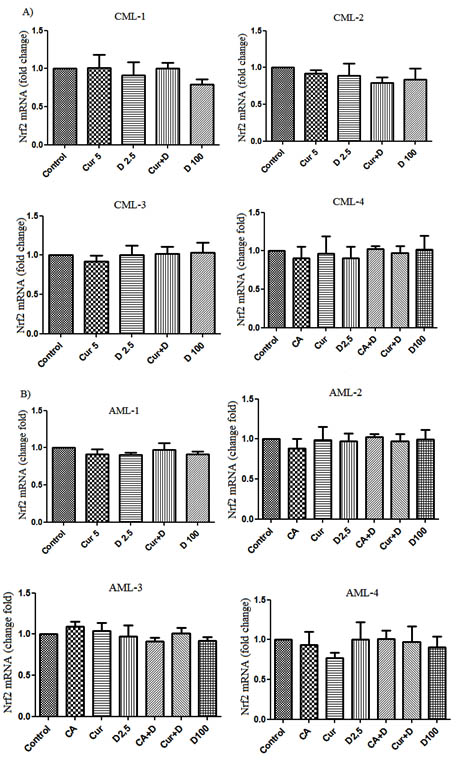
In order to study the expression of the Nrf2 gene in leukemia cells of CML patients a quantitative polymerase chain reaction in real time was conducted with cDNA obtained from cells treated with test agents. The expression level the studied gene in the control group of cells was taken as 1. Expression in the remaining groups was calculated in comparison to the control. Nrf2 gene expression in all studied cell groups of all CML patients treated with 1.25D (2.5 nM), 1.25D (100 nM), CUR, CA, and their combinations was approximately similar or lower than control group (Figure 1A).
Fig. 1. Nrf2 mRNA expression level in CML (A) and AML (B) patients leukemic cells treated with CA (10 μM), 1,25D (2.5 nM) and 1,25D (100 nM), CUR (5 μM) and combinations of 1,25D (2.5 nM) with CA and CUR. Data are the means ± SD
Further experiments were performed with treated cells of AML patients (Figure 1B). Analysis of the data obtained from the study shows that the groups of cells treated with 1.25D (2.5 nM), 1.25D (100 nM), CUR, CA, as well as and their combinations did not respond to treatment with an increase in the level of expression of the gene being studied. The values of the expression level obtained in all groups were the same or lower in comparison with the control group. All of the above suggests that 1.25D (100 nM), CUR, CA alone and their combinations do not lead to an increase in the level of expression of the Nrf2 gene in peripheral blood mononuclear cells of AML and CML patients.
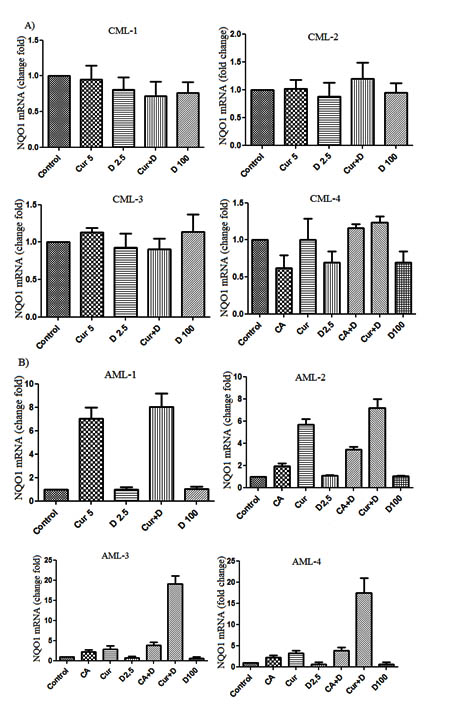
Fig. 2. NQO1 mRNA expression level in CML (A) and AML (B) patients leukemic cells treated with CA (10 μM), 1,25D (2.5 nM) and 1,25D (100 nM), CUR (5 μM) and combinations of 1,25D (2.5 nM) with CA and CUR. Data are the means ± SD
According to research of M.Danilenko, treatment of U937 cells with 1.25D does not affect the level of the Nrf2 protein, however, treatment with a combination of 1.25D and CA increased it (Huang, Nguyen & Pickett 2000; Martin et al. 2014; Xu et al. 2006). In our case, treatment with combinations of vitamin D with plant polyphenols did not show a change in the expression level of the Nrf2 gene. Our results suggest that regulation the Nrf2 gene expression occurs at the post-translational level. To confirm this hypothesis, it is necessary to study the expression level of one of the Nrf2 effectors genes. We selected the NAD(P)H gene of quinone oxidoreductase 1 (NQO1), as the induction of NQO1 gene expression occurs through the Nrf2-Keap1/ARE pathway.
NQO1 gene expression
In order to determine the NQO1 gene expression level qPCR in Real Time was performed with the same cDNA samples. The NQO1 gene expression level in CML patients’ cells is shown in Figure 2 A. The analysis of the figure suggests that the expression of the gene under study did not change much in the all cell groups. Subsequent experiments were performed with groups of cells obtained from AML patients.
The results are given in Figure 2B show that the NQO1 gene expression is most pronounced in cells grown in a medium supplemented with combinations of 1.25D/CUR and 1.25D/CA. However, the cells of these patients also reacted to a slight degree to the treatment with CUR and CA. Expression in groups of cells treated with 1.25D (2.5 nM), 1.25D (100 nM) did not change in comparison with the control group.
Thus, the obtained results make it possible to conclude that the peripheral blood mononuclear cells of AML patients were more sensitive to treatment with 1.25D/CUR and 1.25D/CA and to a lesser extent and not in all patients – with CUR and CA. This study allows suggest that 1.25 D in combinations with CUR or CA do not affect the expression level of the Nrf2 gene, but affects the amount of its protein, which in turn affects the expression level of the NQO1 gene. However, the effect of the above agents on the expression of the NQO1 gene was detected only in the cells of patients with AML. The data require further studies involving more patients to obtain statistically reliable results.
- Brown AJ, Slatopolsky E. Vitamin D analogs: therapeutic applications and mechanisms for selectivity. Mol Aspects Med 2008; 29:433-452.
- Danilenko M, Wang Q, Wang X, Levy J, Sharoni Y, Studzinski GP. Carnosic acid potentiates the antioxidant and prodifferentiation effects of 1á,25-dihydroxyvitamin D3 in leukemia cells but does not promote elevation of basal levels of intracellular calcium. Cancer Res 2003; 63:1325-32.
- Danilenko M, Wang X, Studzinski GP. Carnosic acid and promotion of monocytic differentiation of HL60-G cells initiated by other agents. J Nat Cancer Inst 2001; 93:1224-33.
- Gocek E, Studzinski GP. Vitamin D and differentiation in cancer. Critical reviews in clinical laboratory sciences 2009; 46:190-209.
- Huang HC, Nguyen T, Pickett CB. Regulation of the antioxidant response element by protein kinase C-mediated phosphorylation of NF-E2-related factor2. Proceedings of the National Academy of Sciences of the United States of America 2000; 97:75-80.
- Igissinov N, Kulmirzayeva D, Moore MA, Igissinov S, Baidosova G, Akpolatova G, Bukeyeva Z, Omralina Y. Epidemiological Assessment of Leukemia in Kazakhstan, 2003-2012. Asian Pac J Cancer Prev 2013; 15(16): 6969-6972.
- Kang SN, Lee MH, Kim KM, Cho D, Kim TS. Induction of human promyelocytic leukemia HL-60 cell differentiation into monocytes by silibinin: involvement of protein kinase C. Biochem Pharmacol 2001; 61:1487-95.
- Liu Y, Chang RL, Cui XX, Newmark HL, Conney AH. Synergistic effects of curcumin on all-trans retinoic acid- and 1á,25-dihydroxyvitamin D3 -induced differentiation in human promyelocytic leukemia HL-60 cells. 1997; 9:19-29.
- Martin D, Rojo AI, Salinas M, Diaz R, Gallardo G, Alam J, De Galarreta CM, Cuadrado A. Regulation of heme oxygenase-1 expression through the phosphatidylinositol 3-kinase/Akt pathway and the Nrf2 transcription factor in response to the antioxidant phytochemical carnosol. J Biol Chem 2004; 279: 19-29.
- Shabtay A, Sharabani H, Barvish Z, Kafka M, Amichay D, Levy J, et al. Synergistic antileukemic activity of carnosic acid-rich rosemary extract and the 19-nor Gemini vitamin D analogue in a mouse model of systemic acute myeloid leukemia. Oncology 2008; 75:203-14.
- Sharabani H, Izumchenko E, Wang Q, Kreinin R, Steiner M, Barvish Z, et al. Cooperative antitumor effects of vitamin D 3 derivatives and rosemary preparations in a mouse model of myeloid leukemia. Int J Cancer 2006; 118: 3012-21.
- Sokoloski JA, Shyam K, Sartorelli AC. Induction of the differentiation of HL-60 promyelocytic leukemia cells by curcumin in combination with low levels of vitamin D3. Oncol Res 1997; 9:31-9.
- Xu C, Yuan X, Pan Z, Shen G, Kim JH, Yu S, Khor TO, Li W, Ma J, Kong AN. Mechanism of action of isothiocyanates: the induction of ARE-regulated genes is associated with activation of ERK and JNK and the phosphorylation and nuclear translocation of Nrf2. Mol Cancer Ther 2006; 5: 18-26.
- Zhang J, Harrison JS, Uskokovic M, Danilenko M, Studzinski GP. Silibinin can induce differentiation as well as enhance vitamin D 3 -induced differentiation of human AML cells ex vivo and regulates the levels of differentiation-related transcription factors. Hematol Oncol 2010; 28: 124-32.
© The Author(s) 2018. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.