ISSN: 0973-7510
E-ISSN: 2581-690X
The main dengue and Zika vector, Aedes aegypti, is a cosmotropic species. Since dengue fever cases have significantly increased in recent years, these organisms seem to be extremely detrimental. Synthetic pesticides are not biodegradable, are non-selective, and have adverse effects on beneficial organisms being handled in the vector management system. In the present study, the mosquitocidal potential of chitinase from P. putida Mb 12 was evaluated in an effort to identify risk-free options for the control of mosquitoes. Larvicidal toxicity of Pseudomonas putida Mb 12 chitinase were evaluated on IVth-instar larva of Ae. aegypti and their effect on acetylcholinesterase activity and glutathione S-transferase activity were studied. The early 4th instar larvae of Ae. aegypti were exposed to chitinase enzyme concentrations of 50U/mL, 100U/mL, 200U/mL, and 500U/mL for a period of 4 hours to assess their effectiveness. The results showed that as chitinase concentration increased, mosquito larvae mortality increased; after 4 hours, chitinase at 500 U/mL caused 100% mortality. After 4 hours of incubation, 200 U/mL was administered to achieve LC50 (which kills 50% of the exposed organisms), and after 3 hours, 500 U/mL was used to obtain LC90 values. During the study, it was discovered that different quantities of chitinase (100 U/mL, 200 U/mL, and 500 U/mL) inhibited 80% of the activity of the acetylcholinesterase enzyme. This study found that chitinase significantly increased glutathione S-transferase activity. Additionally, it was discovered that the chitinase treatment was non-hazardous to guppy fish. It was assumed that the P. putida Mb 12 chitinase tested was safe to employ in the aquatic habitat because no mortality was observed in the non-target organisms.
Chitinase, Pseudomonas putida Mb 12, Dengue, Mosquito, Aedes aegypti, Larvicidal
Four genetically distinct dengue virus (DENV) serotypes, known as DENV-1, DENV-2, DENV-3, and DENV-4, are responsible for the fastest-growing infectious disease in the world, causing 100–400 million fresh cases every year. The World Health Organization’s South East Asia region has classified India’s dengue endemicity as being in “Category A” since it is a serious public health issue, the main cause of paediatric inpatient deaths, hyperendemic with all four serotypes in circulation, and expanding to rural regions.1 The main vector underlying dengue is Ae. aegypti (Linnaeus). Several other endemic viral illnesses, including Chikungunya, West Nile fever, Mayaro virus infection, and Eastern equine encephalitis virus infection, are transmitted by Aedes mosquito species.2,3,4,5 Ae. aegypti is also a vector for zoonotic diseases, including Rift Valley fever (RVF), which is regarded as one of the more dangerous evolving zoonotic diseases.6,7 Infections transmitted by mosquitoes can be addressed by augmenting host defence mechanisms (vaccines) or eliminating the vectors. Many vector control strategies have been used to combat Aedes species, with varying degrees of success. Mosquitoes can be chemically controlled by insecticides from the classes of pyrethroids, carbamates, organophosphates, and organochlorines.8 Synthetic pesticides are not biodegradable, are non-selective, and have negative effects on beneficial organisms when applied in the vector management system. Due to their restricted movement, mosquito larvae are effectively handled, especially in areas where the primary breeding sites are man-made and readily identifiable.9 Currently, determining if a new insecticide is biodegradable and has no negative effects on creatures other than the target species is the primary priority.10 Chitin, which is found in mosquito larval, pupal, and adult integuments as well as in the peritrophic matrix of the midgut, is hydrolyzed by chitinases, making them suitable biopesticides for this use.11 Additionally, chitin has been found in the serosal cuticle of Ae. aegypti mosquitoes.12 Peritrophic membranes (PM) are film-like structures seen in insects that are embedded in a protein-carbohydrate matrix and include significant amounts of chitin. The biological management of insect pests of crop plants through the degradation of the PM by chitinases has long been extremely appealing.13,14 To our knowledge, however, very few investigations have been done on the mosquitocidal action of microorganisms’ chitinase against Ae. aegypti (Mendonsa, Ramirez).15,16 In the current work, the mosquitocidal properties of chitinase of P. putida Mb 12 were assessed in the hope of looking towards safe alternatives for mosquito management.
P. putida Mb 12, a newly identified marine bacterial strain from Cochin, Ernakulam, Kerala, India, was employed as the source of chitinase for mosquito larvicidal activity. All of the experiments used analytical grade reagents.
Mass production of chitinase enzyme
The strain P. putida Mb 12 was chosen because of its strong ability to produce chitinase. As a principal carbon source for the manufacture of chitinase, the bacterial isolate P. putida Mb 12 strain was cultivated in 100 ml of a 1% suspension of prawn shell powder and incubated for 48 hours in an incubator shaker with agitation at 100 rpm. The incubation temperature (37°C) and pH 7 were also maintained for the synthesis of the enzyme. To determine the chitinase activity, the culture was centrifuged at 8000 rpm for 20 minutes.17 The amount of enzyme that released one micromole of N-acetylglucosamine per ml per minute during the experiment was considered one unit of chitinase.
Larvicidal bioassay of P. putida Mb 12 against Ae. aegypti
Larvae maintenance
According to the Clemons et al., methodology, the test organism, Ae. aegypti, was cultivated, and eggs were harvested.18 The mosquito eggs on the filter paper were put in a plastic tray with 100 ml of distilled water and left for a few days to develop into larvae. At different phases of its life cycle, the proliferation of the mosquito is influenced by the source of food, photoperiod, temperature, and relative humidity.19 Every day, twice at a suitable interval, the diet during the developmental processes was composed of a high carbohydrate source mixed with a 10% sucrose solution, dog biscuits, and yeast extract (1:3:1). Throughout the whole experiment, a photoperiod of 14 days of sunlight and 10 hours of shade was maintained in the lab.20 The mosquito larvae were placed into a bigger glass beaker after hatching from the eggs in water and were fed fake food, a 3:1 mixture of dog biscuits and dry yeast powder. Colonies were protected from pathogens, pesticides, and repellents. Mosquito larvae were taken, examined under a microscope for their morphological characteristics, and it was determined that they belonged to Ae. aegypti spp.
Bioassay of crude chitinase enzyme against the larvae of Ae. aegypti
Different quantities of the chitinase enzyme—50 U/ mL, 100 U/mL, 200 U/mL, and 500 U/mL—were added, along with 10 Ae. aegypti larvae, to sterile cups containing each 50 ml of sterilised tap water without any chlorine. Negative controls were created using distilled water. If a larva couldn’t be roused after a certain amount of time, despite being gently nudged, it was thought to be dead. Over each interval of 30 minutes, the number of dead larvae was recorded, and the percentage of mortality was recorded for 24 hours.21,22 For each test concentration, 200 replicates and ten replicates of the untreated control were carried out.
The following formula was used Larval mortality % = Y / X × 100
where X = number of tested larvae and Y = number of dead larvae
Enzymatic studies
Studies on the impact of chitinase on the Glutathione S-transferase (GST) and Acetylcholinesterase (AChE) enzymes were carried out to examine the neurotoxicity and detoxifying effects on Ae. aegypti larvae. Early third-instar larvae were exposed to chitinase at its median lethal concentration (LC50), and the live larvae recovered after being treated with chitinase. Untreated control larvae were also used for additional biochemical analysis. The larvae were removed from the 200 U/mL chitinase solution after 4 hours and cleaned with double-distilled water before being blotted with tissue paper to remove any impurities. The recovered larvae were then put into EppendorfTM tubes and homogenised with a homogenizer in 100 mM, 0.5 ml of phosphate buffer (pH 7.2), and 1% Triton X-100.23 The supernatant from the centrifugation of the homogenate at 10,000 g for 15 min at 4°C was used to measure the enzyme activity. The Bradford method was used to estimate the amount of enzyme protein.24
Acetylcholinesterase (AChE) activity of chitinase of P. putida Mb 12
Acetylthiocholine iodide was used as a substrate for the spectrophotometric measurement of AChE activity in the whole body homogenates of larvae in accordance with a modified method of Ellman et al. 25 Malathion (250 ppm), a chemical pesticide, is maintained as a positive control (brand name pestanal Sigma-Aldrich Co, India). 450 ml of sodium phosphate buffer (100 mM, pH 7.5), 50 µl of 10 mM DTNB (Ellman’s Reagent- 5,5-dithio-bis-(2-nitrobenzoic acid), and 50 µl of 12.5 mM acetylcholine iodide were successively added to 50 µl of homogenate (200 U/mL crude enzyme). The assay was repeated three times after the sample had been incubated for 5 minutes at room temperature. The optical density of the sample was measured at 400 nm using a spectrophotometer (Shimadzu, UV-1800) and compared to a suitable reagent blank (without enzyme).
Glutathione S-transferase (GST) activity
The approach used by Vontas et al.26 to estimate GST activity was as follows: 100 μl of 30 mM CDNB 91-chloro-2,4-dinitrobenzene was mixed with 100 μl larval homogenate (200 U/mL crude enzyme), and the volume was adjusted to 500 μl using sodium phosphate buffer (100 mM, pH 7.5). After pre-incubation of the reaction mixture at 37°C for 5 min, 100 μl of 30 mM of reduced glutathione was added. At 340 nm, the spectrophotometer recorded a change in absorbance level for 5 minutes at intervals of 1 minute. The reaction mixture devoid of enzymes served as a control. The measure of GST activity was (mol CDNB hydrolyzed/min/mg protein).
Chitinase effect on Guppy Fish (Poecilia reticulata)
The guppy fish (Poecilia reticulata) is well recognised for its ability to consume insects, particularly mosquito larvae. As a result, these fish are in danger when a poisonous substance is used to kill mosquito larvae. The species is therefore a suitable non-target organism. Fishes (with a length of 3.05 ± 0.11 cm and a body weight of 1.08 ± 0.09 g) were exposed to different doses of Chitinase for 48 h while being continually monitored for harmful effects.27 Malathion was maintained as a positive control (Brand name pestanal Sigma-Aldrich Co, India).
Statistical analysis
MS Excel 2003 and SPSS were used to statistically analyse the experimental data to determine the LC50 and LC90 lethal doses, mean larval mortality, standard error, etc. Results are presented as mean±SEM. To compare the differences in enzyme inhibition between the control and treatment groups, a one-way variance analysis (ANOVA) test was run. Results with p<0.05, were deemed statistically significant.
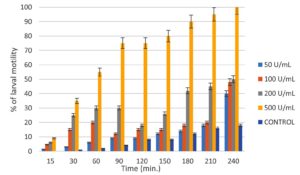
Mosquito larvicidal activity
The early 4th instar larvae of Ae. aegypti were exposed to chitinase enzyme concentrations of 50U/mL, 100U/mL, 200U/mL, and 500U/mL for a period of 4 hours to assess their effectiveness. According to the findings, mosquito larvae mortality increased as chitinase concentration increased. After 4 hours, chitinase at 500 U/mL detected 100% mortality. After 4 hours of incubation, LC50 (which kills 50% of the exposed organisms) was obtained at 200 U/mL, and after 3 hours, LC90 values were detected at 500 U/mL (Figure 1). The negative control did not exhibit any toxicity to mosquito larvae, and c2 value shows a significant difference at p < 0.05 level.
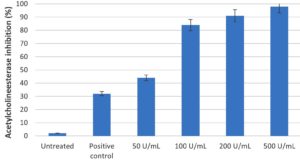
Effect of chitinase of Pseudomonas putida Mb 12 on Acetylcholinesterase (AChE) activity
Using the recognised insecticide and AChE inhibitor malathion as a positive control, the in vitro AChE inhibition by chitinase from the larval extract was investigated. It was found that different doses of chitinase (100U/mL, 200U/mL, and 500U/mL) inhibited 80% of the activity of the AChE enzyme (Figure 2). Acetylcholinesterase inhibition increased as chitinase concentration increased.
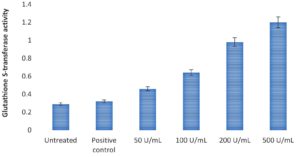
Effect of chitinase of Pseudomonas putida Mb 12 on Glutathione S-transferase (GST) activity
Figure 3 Effect of chitinase of Pseudomonas putida Mb 12 on Glutathione S-transferase (GST) activity.
In this investigation, chitinase significantly increased GST activity(Figure 3), as chitinase concentration increased, so did GST activity.
Figure 3. Effect of chitinase of Pseudomonas putida Mb 12 on Glutathione S-transferase (GST) activity.
Ecological Safety Analysis of Chitinase
Chitinase’s ecological safety examination was conducted in terms of its hazardous effects on creatures that weren’t the target. Even after 120 minutes of exposure to chitinase in guppy fish, no changes in skin color, survival rate, or swimming behaviour were noticed (Table). Results are presented as mean± SEM *p < 0.05 (ANNOVA, Tukey’s HSD test) denote significant difference compared to the control.
Table:
Effect of chitinase on the Guppy Fish (Poecilia reticulate).
| Concentration of Chitinase | Mean Mortality rate(%) | Number of fishes having difficulty in Swimming (Mean ±S.D) | Fishes with colour change | |||
|---|---|---|---|---|---|---|
| After30 min | After 60 min | After 90 min | After 120 min | |||
| 50U/mL | 0 | 0 | 0 | 0 | 0 | 0 |
| 100U/mL | 0 | 0 | 0 | 0 | 0 | 0 |
| 200U/mL | 0 | 0 | 0 | 0 | 0 | 0 |
| 500U/mL | 0 | 0 | 0 | 0 | 0 | 0 |
| Positive control | 62±2.5 | 88±1.5 | 163±1.8 | 222±0.5 | 386±6.3 | 250±9.4 |
| Negative control | 0 | 0 | 0 | 0 | 0 | 0 |
Due of the extreme toxicity of chemical pesticides to mankind, animals, and pollinating insects, they are being replaced by ecologically benign biopesticides as a substitute technique to minimise mosquito-borne diseases. It is preferable to employ specific metabolites like chitinase, which have been synthesized by bacteria, as an insecticide rather than using entire microbes.28 In Southeast Asia, the main malaria vector Anopheles dirus is combated by a strain of Serratia marcescens that secretes chitinase.29 Three species of vector mosquitoes, Anopheles stephensi, Culex quinquefasciatus, and Ae. aegypti pupae, were reported to be successfully killed by the chitinase present in the culture supernatant of P. fluorescens.30 When an emulsifiable concentrate formulation prepared with P. fluorescens was tested on C. quinquefasciatus in the field, more than 80% mortality was detected after 12 days and these exotoxins are believed to be chitinases.31 According to Mendonsa et al.15 insect cuticles can be successfully degraded by enzymes from the bacterium Myrothecium verrucaria, with 100% mortality. The extracellular mosquitocidal protein of P. fluorescens has chitinolytic activity, which is crucial for chitin catabolism and serves to weaken the insect’s peritrophic membranes and chitinous structures. It results in the mortality of the larvae or pupae treated with the proteins and ionic regulation being disrupted.32
The cholinergic neuron‘s major enzyme is acetylcholinesterase (AChE; EC 3.1.1.7), which catalyses the breakdown of acetylcholine to stop nerve impulses and release free choline, which is then absorbed back by pre-synaptic neurons for recycling. The neurotransmission at the neuromuscular junction is responsible for the absorption, reuptake, and resynthesis of ACh. However, suppression of AChE causes ACh to accumulate in the synaptic cleft, which impairs neurotransmission and kills the insect.33 Malathion is one of the most powerful AChE inhibitors and is frequently used to kill mosquitoes; therefore, because of its well-known inhibitory impact on mosquito AChE, we have chosen it as a positive control for investigations. Additionally, it has been claimed that Malathion interacts with human AChE. This is due to the fact that human AChE and the AChE enzyme found in pests are identical.34 In the current work, we are looking for a specific phytochemical that might be used to replace these commonly available carbamates and organophosphates.
The GST family of multifunctional enzymes is one of the metabolic detoxification enzymes that is widely distributed in eukaryotes and prokaryotes and is crucial for the detoxification of a wide range of both endogenous and exogenous substances. By accelerating the nucleophilic assault of thiol group in reduced glutathione (GSH) on a variety of electrophilic substrate.35,36 These substrates may include oxidative stress byproducts, insecticides, environmental contaminants, or plant allelochemicals.35,37 As a result, the increase in GST activity in Ae. aegypti larvae after chitinase treatment indicates that the larvae were attempting to detoxify chitinase molecules. Thus, compounds that block or stimulate these enzymes can cause metabolic imbalances, stunted growth, and even death in mosquito larvae.38
Guppy fish’s skin colour, survival rate, or swimming behaviour remained unchanged even after 120 minutes of exposure to chitinase. Therefore, the results confirm that chitinase is safe in terms of germination potential and toxicity to non-targeted organisms. Therefore, the results confirm that chitinase is safe to non-targeted organisms.
The study comes to the conclusion that chitinase can be employed as an effective biopesticide in the future to suppress dengue mosquito Ae. aegypti larvae. The tested chitinase was deemed safe for usage in the aquatic habitat because no death was seen in the non-target organisms. To fully understand such action against a variety of mosquito species, more research is required.
ACKNOWLEDGMENTS
None.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
None.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
This article does not contain any studies with human participants or animals performed by any of the authors.
- World Health Organization (WHO). Dengue and Severe Dengue. 2022.
- van Aalst M, Nelen CM, Goorhuis A, Stijnis C, Grobusch MP. Long-term sequelae of chikungunya virus disease: A systematic review. Travel Med Infect Dis. 2017 15:8-22.
Crossref - Long KC, Ziegler SA, Thangamani S, et al. Experimental transmission of Mayaro virus by Aedes aegypti. Am J Trop Med Hyg. 2011;85(4):750-757.
Crossref - Romero JR, Newland JG. Viral meningitis and encephalitis: traditional and emerging viral agents. Semin Pediatr Infe Dis. 2003;14(2):72-82.
Crossref - Daep CA, Munoz-Jordan JL, Eugenin EA. Flaviviruses, an expanding threat in public health: focus on dengue, West Nile, and Japanese encephalitis virus. J Neurovirol. 2014;20(6):539-560.
Crossref - Clement NM, Sharadhuli IK, John BK, Leonard EG, Mboera. Predicting distribution of Aedes aegypti and Culex pipiens complex, potential vectors of Rift Valley fever virus in relation to disease epidemics in East Africa. Infect Ecol Epidemiol. 2013;3(1):21748.
Crossref - Pepin M, Bouloy M, Bird BH, Kemp A, Paweska J. Rift Valley fever virus (Bunyaviridae: Phlebovirus): an update on pathogenesis, molecular epidemiology, vectors, diagnostics and prevention. Vet Res. 2010;41(6):61.
Crossref - Van Den Berg H, Zaim M, Yadav RS, et al. Global trends in the use of insecticides to control vector-borne diseases. Environ Health Perspect. 2012;120(4):577-582.
Crossref - Howard AFB, Zhou G, Omlin FX. Malaria mosquito control using edible fish in western Kenya: preliminary findings of a controlled study. BMC Public Health. 2007;7:199-204.
Crossref - Redwane A, Lazrek HB, Bouallam S, Markouk M, Amarouch H, Jana M. Larvicidal activity of extract from Querus lusitania var infectoria galls (oliv). J Ethnopharmacol. 2002;79(2):261-226.
Crossref - Merzendorfer H, Zimoch L. Chitin metabolism in insects: structure, function and regulation of chitin synthases and chitinases. J Exp Biol. 2003;15:206(24):4393-4412.
Crossref - Rezende GL, Martins AJ, Gentile C, et al. Embryonic desiccation resistance in Aedes aegypti: presumptive role of the chitinized Serosal Cuticle. BMC Dev Biol. 2008;8:82.
Crossref - Regev A, Keller M, Strizhov N, et al. Synergistic activity of a Bacillus thuringiensis delta-endotoxin and a bacterial endochitinase against Spodoptera littoralis larvae. Appl Environ Microb. 1996; 62(10):3581-3586.
Crossref - El-Tarabilya KA, Soliman MH, Nassar AH, et al. Biological control of Sclerotinia minor using a chitinolytic bacterium and actinomycetes. Plant Pathol. 2000;49(5):573-583.
Crossref - Mendonsa ES, Vartak PH, Rao, JU, Deshpande MV. An enzyme from Myrothecium verrucaria that degrades insect cuticle for biocontrol of Aedes aegypti mosquito. Biotechnol Lett. 1996;18:373-376.
Crossref - Ramirez-Suero M, Valerio-Alfaro, Bernal J, Ramirez-Lepe M. Synergisitic effect of chitinases and Bacillus thuringiensis israelensis spore-toxin complex against Aedes aegypti larvae. Can Entomol. 2011;143(2):157-164.
Crossref - Miller GL. Use of dinitrosalicylic acid reagent for determination of reducing sugars. Anal Chem. 1959;31:426-428.
Crossref - Clemons A, Mori A, Haugen M, Severson DW, Duman-Scheel M. Culturing and egg collection of Aedes aegypti. Cold Spring Harb Protoc. 2010(10):pdb.prot5507.
Crossref - Kauffman E, Payne A, Franke MA, Schmid MA, Harris E, Kramer LD. Rearing of Culex spp. and Aedes spp. Mosquitoes. Bio Protoc. 2017;7(17):e2542.
Crossref - Das S, Garver L, Dimopoulos G. Protocol for mosquito rearing (A. gambiae). J Vis Exp. 2007;(5):221.
Crossref - Sundarapandian S, Sundaram MD, Tholkappian P, Balasubramanian V. Mosquitocidal properties of indigenous fungi and actinomycetes against Culex quinquefasciatus Say. J Biol Control. 2002;16:89-91.
- El-Khawagh MA, Hamadah KhSh, El-Sheikh TM. The insecticidal activity of actinomycete metabolites, against the mosquitoe Culex pipiens. Egypt Acad J Biol Sci. 2011;4(1):103-113.
Crossref - Napoleao TH, Pontual EV, de Albuquerque Lima T, et al. Effect of Myracrodruon urundeuva leaf lectin on survival and digestive enzymes of Aedes aegypti larvae. Parasitol Res. 2012;110(2):609-616.
Crossref - Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976;72(1-2):248-254.
Crossref - Ellman GL, Courtney KD Jr, Andres V, Featherstone RM. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol. 1961;7(2):88-95.
Crossref - Vontas JG, Enayati AA, Small GJ, Hemingway J. A simple biochemical assay for glutathione S-transferase activity and its possible field application for screening glutathione S-transferase-based insecticide resistance. Pestic Biochem Physiol. 2000;68(3):184-192.
Crossref - Reish DL, Oshida PS. Manual of methods in aquatic environment research. FAO Fish Tech. Pap. 1987; 47-52
- Shternshis M. Biopreparations for plant protection in Siberia: application and enhancement of activity. J Agric Technol. 2005;1:1-18.
- Jupatanakul N, Pengon J, Selisana SMG. Serratia marcescens secretes proteases and chitinases with larvicidal activity against Anopheles dirus. Acta Trop. 2020;212:105686.
Crossref - Kramer KJ, Muthukrishnan S. Insect Chitinases: Molecular Biology and Potential Use as Biopesticides. Insect Biochem Mol. 1997;27(11):887-900.
Crossref - Sadanandane C, Reddy C.MR, Prabakaran G, Balaraman K. Field evaluation of Pseudomonas fluorescens against Culex quinquefasciatus larvae and pupae. Acta Trop. 2003;87:341-343.
Crossref - Usharani B, Kummankottil P. Chitinase like activity of metabolites of Pseudomonas fluorescens Migula on immature stages of the mosquito, Culex quinquefasciatus (Diptera: Culicidae). Afr J Microbiol Res. 2012;31:6(11):2718-2726.
Crossref - Yu SJ. The toxicology and biochemistry of insecticides. Toxicol Biochem Insectic. 2011.
Crossref - Yamagishi Y, Iwase H, Ogra Y. Effects of human serum albumin on post-mortem changes of malathion. Sci Rep. 2021;11:1-8.
Crossref - Hayes JD, Flanagan JU, Jowsey IR. Glutathione transferases. Annu Rev Pharmacol Toxicol. 2005;45:51-88.
Crossref - Ranson H, Hemingway J. Mosquito glutathione transferases. Methods Enzymol. 2005;401:226.
Crossref - Liu Y, Moural T, Koirala BKS, Hernandez J, Shen Z, Alyokhin A. Structural and functional characterization of one unclassified glutathione S-transferase in xenobiotic adaptation of Leptinotarsa decemlineata. Int J Mol Sci. 2021;22:11921.
Crossref - Subahar R, Aulia AP, Yulhasri Y, et al. Assessment of susceptible Culex quinquefasciatus larvae in Indonesia to different insecticides through metabolic enzymes and the histopathological midgut. Heliyon. 2022;8(12):e12234.
Crossref
© The Author(s) 2023. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.