ISSN: 0973-7510
E-ISSN: 2581-690X
Plant growth promoting rhizobacteria (PGPR) are a group of useful bacteria that colonize the plant roots and significantly enhances the plant growth promotion. Keeping this in mind, an investigation was performed for the screening of potent PGPR strains for enhancing seed germination and vigour index of Hordeum vulgare (commonly called barley). Rhizobacterial strains were isolated and screened for various plant growth promoting traits, their effect on seed germination and vigour index of barley plant through pot trial, and resistant ability under various temperature and pH range. Based upon 16S rRNA sequencing data, Pseudomonas punonensis LMT03 (R1), Pseudomonas plecoglossicida R4, Pseudomonas aeruginosa DSM50071 (R2) and Alcaligenes faecalis (DBHU5) isolates were selected and showed positive result for IAA production, Phosphate solubilization, ammonia production, catalase activity, siderophore production and MR-VP test. Barley plants treated with P. punonensis and P. plecoglossicida both showed 94.44% of highest seed germination %, while P. aeruginosa and A. faecalis showed 83.11% and 77.33% respectively in comparison to the control plant shows which 49.99% seed germination only. These respective isolates also showed 2.57, 2.37, 2.0 and 1.69 fold of increase in vigour index as compare to the control plants. The above increase in fold in vigour index and seed germination is much higher as compared to earlier reports. Collectively, the data of current study underpin that addition of these PGPRs to barley rhizosphere appears a promising strategy to enhance root and shoot biomass of this important agriculture crop. To the best of our knowledge this is the first report demonstrating the effect of P. punonensis and P. plecoglossicda on barley crop.
Biopriming, PGPR characterization, Phosphate solubilization, Rhizobacteria, Seed germination, Vigour index
Due to increase in the world population leads to rise in the food demand therefore to full fill the food requirement, we are dependent on the application of hazardous chemical fertilizers and pesticides which is not appropriate for the human health as well as for our food chain. These chemical fertilizers undoubtedly increases the yield at short duration but at the same time they adversely affects the environment, causes abiotic stresses, affects the biological food chain and also are not affordable for the farmers. Inoculation of plants with beneficial microorganism such as plant growth promoting rhizobacteria (PGPR) can increase the crop productivity through increase in the seed germination, vigour index, and crop yield. Addition of PGPR has also been shown to protect the plants from abiotic and biotic stresses, and hence makes the farmers to produce more food at lower price and at minimum risk level.1
PGPR are the soil borne beneficial bacteria found in association with roots at the rhizospheric region of plants, which enhances the plant growth promotion, and also protects the plants from the soil born phytopathogens.2 The exact mechanism of action of PGPR is not well known,2 but there was two important mode of actions on the basis of which PGPR can be categorized into two groups; the first is direct which involves in the synthesis of various plant hormones like IAA, Synthesis of phosphate solubilizing compounds which exhibit solubilization of inorganic and organic insoluble phosphate into soluble form, synthesis of ACC deaminase which regulates ethylene production by hydrolysis of ACC (Precursor of ethylene) into ammonia and a-ketobutyrate, siderophore production, enhances fixation of atmospheric nitrogen, develop resistance against various abiotic stresses. The second is indirect which involves in the development of tolerance ability in plants against various phytopathogens (biotic stresses) by production of secondary metabolites, antibiotics, siderophore production, enzymatic compounds and induced systemic resistance.3,4 Treatment of these rhizobacteria enhances the seed germination percentage, vigour index, leaf surface area, root-shoot growth and development in non-stress and stress conditions through direct and indirect mechanisms.5 Phosphate is the second most fundamental nutrient for plants and PGPR treated plants can enhance their phosphate solubilizing ability and hence complete the basic nutrient requirement of plants.6 PGPR comprises species of Alcaligens, Pseudomonas, Azotobacter, Enterobacter, Azospirillum, Bacillus, Arthobacter, Serratia, and Burkholderia etc., which are known for plant growth promotion.7 Among all the bacterial strains, specific strains of Pseudomonas are known to be one of the most effective and potent PGPR, which colonizes with the roots and results in remarkable increase in the crop yield.8 Species of pseudomonas have potential to produce various phytohormones like IAA which indirectly support the water and nutrient uptake by increasing the root surface area.9 Alcaligens faecalis plays vital role as PGPR and act as bio fertilizer which enhances crop yield and hence contributed in growth promotion of plants.10
Barley is mostly cultivated in arid and semi-arid areas, and is considered as the most sacred and oldest cereal food in India, ranked fourth after wheat, rice and maize. Barley is rich in various nutritious values and for that its demand is increasing day by day. Barley has also considered as model experimental plant because of its short life cycle, morphological, and genetical traits.11,12
Therefore, the major aim of the present study was isolation, biochemical screening and molecular identification of rhizobacteria and their effect on seed germination and vigour index of barley plant for screening of potent PGPR. Further effect of Pseudomonas punonensis, Pseudomonas plecoglossicida, Pseudomonas aeruginosa and Alcaligenes faecalis on seed germination and vigour index of barley plants were also analyzed through pot trial. Growth and survivability of these rhizobacteria were also analyzed under multiple temperature and pH range. The results of the present study suggest that suitable formulation of these PGPRs could be a promising approach for the production of chemical fertilizer free and nutritionally rich barley crop which could suffice the ever increasing demand of food all over the world.
Soil sampling
Soil samples were collected from the rhizospheric region of barley crop plants for isolation of rhizobacteria. The site of soil sampling were Rajiv Gandhi South Campus, Barkachha (RGSC, Banaras Hindu University, Mirzapur, UP), Agricultural farm (AF, Banaras Hindu University, UP), and Chandauli district (UP) in India. Whole root portion were uprooted carefully from soil and kept in a sterilized autoclaved poly bag. The soils were kept at 4°C in aseptic condition and used for isolation. The soils were of good permeability and silty loam in nature.
Isolation of rhizobacteria
Bacteria were isolated through serial dilution plate technique, 1 gm of soil were dissolved in 10 ml of autoclaved distilled water through serial dilution process and appropriate dilution were spread on yeast extract mannitol agar (YEMA) media containing Yeast extract 1.00 g, mannitol 10.00 g, KH2PO4 0.50 g, MgSO4.7H2O 0.20 g, NaCl 0.10 g and agar 15.00 g, pH 6.8-7.0 in per liter of distilled water, and incubated at 30°C for 24-48 h. From the mixed cultures grown on YEMA plates, colonies were purified through streak plate method, colonies were streaked through sterilized loop and maintained as pure culture on YEMA media and stocked for further experiments. All the pure cultures were morphologically characterized through Gram staining.
Preliminary screening of bacterial isolates for plant growth promoting activities
Ten rhizobacterial isolates were isolated and morphologically characterized through Gram staining and colony morphology after then screened for multiple plant growth promoting traits.
Phosphate solubilization
All the 10 bacterial isolates were screened for phosphate solubilizing activity through pikovaskaya agar media (PVK) plate technique comprises of yeast extract 0.50 g, dextrose 10.00 g, Ca3(PO4)2 5.00 g, (NH4)2SO4 0.500 g, KCl 0.20 g, MgSO4 0.10 g, MnSO4 0.0001 g, FeSO4 0.0001 g, agar 15.00 g, Tri-calcium phosphate Ca3(PO4)2 in per liter of distilled water.13 A Loop full of selected isolates were placed on PVK media and kept for 48 h at 30°C, formation of clear transparent halo zone around the bacterial colony indicated the positive result for phosphate solubilizing activity. Phosphate solubilizing index (PSI) is calculated by following formula as described by.14
Indole acetic acid
The above isolates were further screened for auxin production ability followed by quantitative detection of IAA production.15,16 Bacteria were cultured in YEMA-broth containing 5 mM tryptophan and incubated at 30°C for 48 h for IAA production test. Cultures were centrifuged at 10,000 rpm for 10 min. 1 ml of supernatant were collected and 2 ml of salkowski reagent (Solawaski’s regent – 50 ml of 35 % Perchloric acid and 1ml of 0.5(M) FeCl3) were added in the supernatant and incubate for 1 h in dark condition at room temperature. Development of pink color confirms the IAA production and optical density was taken at 530 nm in UV-vis spectrophotometer for quantification. The concentrations produced were estimated by using standard graph obtained in µg mL-1.
Ammonia Production
Ammonia production ability of the isolates was detected by method17. 1 milliliter of each bacterial suspension (3.9×107cfu ml-1) were incubated in 10 ml of peptone water (peptone 10 g, NaCl 5 g in 1000 ml distilled water, pH 7.2±0.2) broth in sterilized test tubes and incubated for 48 h at 30°C. After incubation Nessler’s reagent (0.5 ml) in each test tube were added, a change in color from yellow to brown indicated positive result for ammonia production.
Siderophore Production
Siderophore production was detected qualitatively on solid CAS (Chromazurol S) universal blue agar plates as by18. Freshly grown cultures spots were inoculated on CAS media plate and incubated at 30°C for 3 days. Development of orange-yellow halo zone around the bacterial colony confirms the siderophore production.
Catalase activity
Catalase activities for isolates were qualitatively screened by.19 Fresh 24h old loop full of culture were placed on a clean glass slide and a drop of H2O2 (30%) was dropped over the colony and mixed very well with the help of loop. Effervescence of gas bubbles from the colony, confirms the positive catalase activity and was recorded as weak (+), moderate (++), and strong (+++).
Biochemical analysis of bacterial isolates
Methyl Red- Voges Proskaur (MR-VP) Test
MR-VP test comprises of two objectives, methyl red (MR) test is used to detect the fermentation pathway of glucose through conversion of glucose into acidic product (acetate, formate and lactate) while Voges-Proskaur is used to detect the conversion of glucose into acetoin as major product of glucose metabolism. The test was performed in two sets, bacterial culture incubated in glucose peptone broth (peptone 5.0 g; glucose 10.0 g; potassium dehydrogenate phosphate 5.0 g distilled water 1 L, pH 7.4) for 48 h at 30°C. Five drops of pH indicator methyl red (methyl red 5.0 g; dissolved in 30 ml of 95% ethanol and diluted in 50 ml of distilled water) is added in one set of bacterial culture incubated in glucose peptone broth. After addition of methyl red the culture medium turns red because of its pH at, or below 4.4 which indicates the fermentation of glucose, and positive result for MR test, while the culture which show negative result will remain yellow and have pH 6.0. 0.6 ml of α-naphthol and 0.2 ml of KOH solution is added in another 1 ml of glucose peptone culture broth set. A copper color ring like circular structure at the top of broth surface confirms the positive result for VP test.20
Temperature tolerance
Survivability of bacterial isolates at different temperature was determined in Nutrient-broth inoculated with 50 µL of bacterial cultures (109 cfu mL-1) and incubated at various temperatures 20°C, 30°C, 40°C and 45°C. Optical Density (O.D) was measured after 48 h at 420 nm in UV-vis spectrophotometer.
pH tolerance
Growth of bacteria at multiple pH was determined in nutrient broth maintained at various pH 5, 7, 9, 11 and inoculated with 50µL of isolated cultures (109 cfumL-1). O.D was measured after 48 h at 420 nm in UV-vis spectrophotometer.
Treatment of PGPR on barley seeds
Total ten bacterial isolates were screened for plant growth promoting traits out of them 4 isolates were found to show effective plant growth promoting traits. These 4 isolates were selected to evaluate its efficacy on vigour index and germination percentage of barley plant in the soil system through pot trial experiment.
These four cultures were inoculated in nutrient broth and incubated at 30°C in shaker incubator (90 rpm) for 48 h. After full growth the cultures were centrifuged at 10,000 rpm, for 20 min at 4°C. Pellets obtained were washed with 0.85% of saline solution (NaCl) so that the rest of residual culture broth from the pellets were removed out. Washed pellets were properly mixed in 1% carboxy methyl cellulose (CMC) suspension to form a uniform cell density 109 cfu mL-1.21 Seed biopriming of barley variety Manjula were procured from Institute of Agricultural sciences, Banaras Hindu University were surface sterilized with 0.2% of HgCl2 for 2 min, then rinsed thrice with autoclaved distilled water. The sterilized seeds were kept in laminar air flow on sterilized blotting paper for 2 h for air drying then suspended and mixed properly in the bacterial cell suspension as described earlier. Non-suspended CMC- treated sterile seeds were taken as control. All the four bacterial suspended and non bacterial suspended seeds were dried overnight aseptically in laminar air flow. Further for pot experiment sterilized seeds were sown in plastic pots containing autoclaved sterile soil. Six plants in each pot were maintained till experimental period, and 40% water holding capacity of soil was maintained. The effect of PGPR on seed germination percentage and vigour index of barley was evaluated. After 7 days of sowing the seeds were germinated and the seed germination percentage was calculated by following Eq. 1.
Germination %= [Total No.of germinated seeds/ Total No.of seeds sown] x 100
… Eq. 1.
All the treatments were in three replications and arranged in a completely randomized manner. The plants were uprooted after 25 days of germination and the root, shoot and leaf were collected. Root and shoot length were measured to evaluate the plant vigour index. Vigour index was calculated using formula given in Eq. 2.
Vigour index = (Mean root length+mean shoot length)×germination (%) …. Eq. 2
Molecular identification of the 4 selected potent bacterial strains
DNA extraction of the selected bacterial strains (R1, R2, R4 & DBHU5) were done as described by22 method. For amplification of 16S rRNA gene of bacterial strain R1, R2, R4 two specific primers, forward primer: 5’ GGATGAGCCCGCGGCCTA 3’ and reverse primer 5’ CGGTGTACAAGGCCCGG 3’ were used and for strain DBHU5 forward primer: 5’ ATATCGGAACGTGCCCAGTAGC 3’ and reverse primer 5’ TGACATGTCTGGGAAAG 3’ were used. PCR Amplification conditions for 25µl of total reaction volume contained 1 µl (100 ng/ µl) DNA template, 1µl each primer of both 16s forward and reverse primer, 4 µl of dNTPs (2.5 mM each), 0.33 µl (3 units/µl) Taq polymerase, 0.5 µl dNTPs, 2.5 µl 10Xx PCR buffer with 25 mM MgCl2 and 18.67 µl milli Q water was used for the PCR. PCR was performed in Prima-96 (HiMedia United States) under the given conditions: for amplification of 16s rRNA geneDNA was as follows: Initial denaturation for 3 minutes at 94°C, final denaturation for 1 minutes at 94°C, annealing at 55°C for 1 minutes followed by 35 cycles, extension at 72°C for 2 minutes and the final extension was done at 72°C for 7 minutes. Purified amplicons were sequenced by Sanger method and further analyzed by Basic Local Alignment Search Tool (BLAST) with closet culture sequence retrieved from the National Centre for Biotechnology Information (NCBI) database that finds regions of local similarity between sequences.
Statistical analysis
All the statistical analysis and calculations were performed by using IBM SPSS Statistics Ver.20 software. The statistical data were expressed as the mean of three independent replications ± standard deviation (SD) of three replicates of each experiment along with thrice repetitions data of each replicates. The data were interpreted through analysis of variance (one-way ANOVA) followed by Duncan’s multiple range test at the P≤0.001 significance level. The experiments performed in this study were done in triplicates and all the experiments repeated thrice using totally randomized design.
Isolation and morphological characterization of rhizobacteria
Total ten rhizobacterial strains were isolated from the rhizospheric region of barley plants. The rhizobacterial strains were designated as R1, R2, R3, R4, DBHU5, R6, R7, R8, R9 and R10.
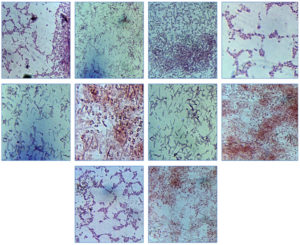
Preliminary identification of all the isolates was based on there preliminary morphological characterization were characterized as rod shaped, gram +ve and gram -ve bacteria, on the basis of Gram staining and is presented in Fig. 1; Table 1.
Table (1):
Morphological and biochemical characterization of all the 10 bacterial isolates for their specific plant growth promoting traits.
Location of soil sample collection |
Bacterial isolates |
Colony morphology |
Gram staining (G+/G-) |
Phosphate solubilization on (PSI) |
Ammonia production |
Catalase activity |
|---|---|---|---|---|---|---|
Chandauli |
R1 |
Rod shaped, non fluorescent, motile, red colony |
G- |
2 |
++ |
++ |
Chandauli |
R2 |
Rod shaped, motile |
G- |
0.9 |
++ |
++ |
RGSC Mirzapur |
R3 |
Rod shaped |
G+ |
– |
++ |
++ |
BHU |
R4 |
Rod shaped, yellow color |
G- |
1.15 |
++ |
++ |
BHU |
DBHU5 |
Rod shaped |
G- |
0.65 |
– |
++ |
BHU |
R6 |
Rod shaped |
G+ |
– |
++ |
++ |
BHU |
R7 |
Rod shaped |
G+ |
– |
++ |
++ |
Chandauli |
R8 |
Rod shaped |
G+ |
– |
++ |
++ |
RGSC Mirzapur |
R9 |
Rod shaped |
G+ |
– |
++ |
++ |
BHU |
R10 |
Rod shaped, flagella, white |
G+ |
– |
++ |
+ |
+ slightly or weak positive test, ++ moderate positive test, +++ strong positive activity, – negative (No activity), G – Gram negative
Fig. 1. Gram staining of bacterial isolates and morphological characterization of pure bacterial cultures.
In vitro plant growth promoting traits of bacterial strains
Qualitative estimation of Phosphate solubilization
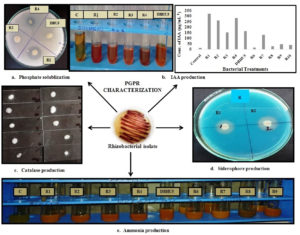
Out of ten rhizobacterial isolates, R1, R2, R4, and DBHU5 isolates showed a clear halo zone around the colony which confirmed the phosphate solubilizing ability of bacteria by solubilization of phosphorus from tri-calcium phosphate added to the Pikovskaya’s agar medium. The maximum PSI is shown by isolate P. punonensis (R1) and P. plecoglossicida (R4) i.e., 2 cm and 1.15 cm respectively. The Phosphate solubilization index of bacteria ranged from 0.65-2 cm. Further, isolate A. faecalis and P. aeruginosa showed 0.65 and 0.9 cm zone of clearance respectively. Fig. 2a; Table 1 and 2.
Table (2):
Morphological, biochemical and characterization of PGPR traits of selected most potent four PGPR isolates for screening of their plant growth promoting attributes.
Bacterial strain |
Gram staining (G+/G-) |
Phosphate solubilization (PSI) |
Ammonia production |
Catalase activity |
Methyl red |
Siderophore production |
IAA Production |
Voges- Proskaur |
|---|---|---|---|---|---|---|---|---|
P. punonensis |
G- |
2 |
++ |
++ |
++ |
+ |
++ |
++ |
P. aeruginosa |
G- |
0.9 |
++ |
++ |
++ |
+ |
++ |
++ |
P. plecoglossicida |
G- |
1.15 |
++ |
++ |
++ |
_ |
++ |
++ |
Alcaligenes faecalis |
G- |
0.65 |
++ |
++ |
_ |
_ |
++ |
++ |
+ slightly or weak positive test, ++ moderate positive test, +++ strong positive activity,
– negative (No activity), G – Gram-negative
Fig. 2. PGPR characterization of bacterial isolates. (a.) Formation of halo zone around the colony showed phosphate solubilizing activity of bacterial isolates (b.) Development of pink color indicates IAA production by bacterial isolates and bar graph represents range of IAA production in µg/ml by bacterial isolates (c.) Effervescence of gas bubbles from the bacterial colony shows catalase activity (d.) Bacterial isolate R1 & R2 shows siderophore production through formation of halo zone around the colony (e.) Development of brown color in peptone water confirms ammonia production by bacterial isolates.
Quantitative estimation of indole acetic acid production
R1, R2, R3, R4, and DBHU5 showed positive result for IAA production in tryptone yeast medium in the presence of L-tryptophan and the concentration of IAA production range varied from 323.2 to 163 µg/ml. It was found that species of pseudomonas i.e., P. punonensis, P. plecoglossicida and P. aeruginosa produce maximum of 323.2 µg/ml, 283.6 µg/ml, 260.5 µg/ml and A. faecalis showed 163.0 µg/ml of IAA production respectively, which was relatively higher production in comparison to other isolates. (Fig. 2b).
Ammonia production
Out of ten, nine isolates i.e., R1, R2, R3, R4, DBHU5, R6, R7, R8, and R9 showed change in color from yellow to brown in peptone water, indicated positive result for ammonia production. Fig. 2e.
Siderophore production
Out of ten isolates, P. punonensis and P. aeruginosa showed moderate amount of siderophore production by formation of halo zone around the colony on CAS (Chromazurol S) universal blue agar plates, irrespective of development of orange-yellow zone, which indicates very mild siderophore production. Fig. 2d.
Catalase activity
All isolates showed evolution of O2 as effervescence of gas bubbles from the colony which confirms the positive catalase activity and was recorded as weak (+), moderate (++), and strong (+++). Fig. 2c; Table 1.
Biochemical analysis of bacterial isolates
Methyl Red Test- Voges Proskaur (MR-VP)
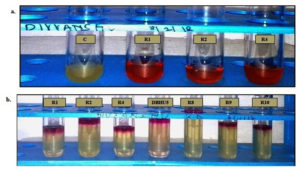
R1, R2, and R3 were screened positive for methyl red test by development of red color in the broth which indicates the fermentation of glucose while seven isolates i.e., R1, R2, R4, DBHU5, R8, R9, and R10 showed positive result for V.P test by formation of copper, red to pink color ring like circular structure at the top surface of the broth. Fig. 3 a- b.
Fig. 3. (a.) Development of red color in bacterial broth shows methyl red activity and control (without bacteria) do not show color (b.) Formation of pink ring like structure at the top surface of broth confirms Voges–Proskaur.
Effect of different pH on bacterial growth
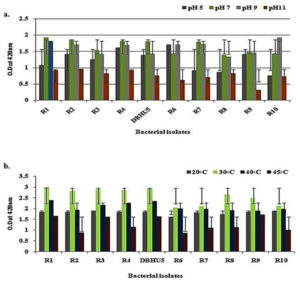
Maximum growth of all the bacterial isolates was at pH 7, however, R1 has the maximum growth at each pH as compare to other isolates which shows its tolerance ability even at unfavorable pH range. It was also concluded that ideal pH for the above bacterial growth was pH 7, while either below or above from pH 7 there is reduction in bacterial growth density. Fig. 4a.
Effect of different temperature on bacterial growth
Bacteria were incubated at various temperatures (20, 30, 40 and 45°C) and observed that, 30°C is the ideal temperature for maximum growth while temperature either rises in between 30 to 45°C or below 30°C there is uniform decrease in the growth density of bacterial cultures. Out of all, R1 and DBHU5 have maximum growth at 30°C and also showed significant growth even under unfavourable temperature i.e. higher and lower than 30°C. Fig. 4b.
Effect of PGPR strains on seed germination and vigour index
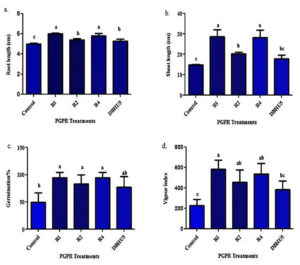
Barley plants were treated by all the ten strains through pot trial, among them the four strains R1, R2, R4, and DBHU5 showed remarkable increase in seed germination percentage and vigour index. Increase in germination percentage ranged from mean value of 77.33-94.44. It was found that barley plants treated with R1 and R4 both shows a highest 94.44%, while R2 and DBHU5 83.11% and 77.33% seed germination respectively while control plants shows only 49.99%. R1 shows the maximum vigour index of 585.73 followed by R4 (538.26), R2 (455.88) and DBHU5 (384.71), while the control plants were found to show only 227.09 vigour index. R1 treated plants showed maximum of 2.32 fold increases in shoot length, and 1.34 fold of maximum increase in root length, while R4, R2, and DBHU5 showed 1.95, 1.5 and 1.2 folds of increase on shoot length and 1.24, 1.19 and 1.10 folds of increase in root length as compared to the control plant respectively (Fig. 5 a-b; Fig. 6 a-d; Table 3). Inoculation of barley plants with P. punonensis, P. plecoglossicida both showed 1.88 fold highest increase in germination % while P. aeruginosa and A. faecalis shows 1.07 and 1.54 fold increase respectively in comparision to control plant. P. punonensis showed highest of 2.57 fold increase in vigour index of plant, while P. plecoglossicida 2.37, P. aeruginosa 2.0 and A. faecalis 1.69 fold increase in vigour index respectively.
Table (3):
Effect of selected PGPR’s on seedling growth parameters (vigour index & germination %) of barley crop with respect to control plant.
Treatments |
Mean of Seed germination% |
Mean shoot length (cm) |
Mean root length (cm) |
Mean Vigour index |
|---|---|---|---|---|
P. punonensis |
94.44±9.62a |
29.65±3.04a |
5.88±0.30a |
585.73±87.85a |
P. aeruginosa |
83.11±17.0a |
19.13±1.72d |
5.24±0.49bc |
455.88±122.08ab |
P. plecoglossicida |
94.44±9.62a |
24.9±5.24b |
5.42±0.49ab |
538.26±102.80ab |
Alcaligenes faecalis |
77.33±19.62ab |
15.8±2.33d |
4.84±0.55cd |
384.71±83.49bc |
Control |
49.99±16.66b |
12.74±1.96e |
4.37±0.60d |
227.09±59.9c |
Data represents Mean±SD, obtained from three repetitions of data from three independent replicates. SD of mean is shown as thin linear bar. (SD standard deviation). Different combinations of letters upon SD indicates data are significantly different from each other (p≤ 0.001). Results taken according to Duncan’s multiple range test at p≤0.001.
Fig. 5 a-b. Effect of bioprimed PGPR isolates [(P. punonensis (R1), P. aeruginosa (R2), P. plecoglossicida (R4) and A. faecalis (DBHU5)] on growth, vigour index and germination % of barley plants in comparison to non-inoculated control plants through pot trial.
Fig. 6. Graph representing effect of PGPRs on (a) root length , (b) shoot length (c) germination % (d) vigour index in comparison to non-inoculated control plants. Graphs were expressed as means of triplicate and vertical bars indicate standard deviation of the mean value. Bars affixed with different combinations of letters are significantly different from each other (P ≤ 0.001). Results taken according to Duncan’s multiple range test at P ≤ 0.001. where, R1 (P. punonensis), R2 (P. aeruginosa), R4 (P. plecoglossicida) and DBHU5 (A. faecalis).
Molecular Identification of the selected PGPR strains
16S rRNA gene sequencing was done for the molecular characterization and identification of bacterial isolates. Sequences obtained by gene sequencing were submitted to NCBI GenBank using BLAST (basic local alignment tool) analysis. Each isolates R1, R2, R4 and DBHU5 showed 100% similarity with Pseudomonas punonensis, Pseudomonas aeruginosa, Pseudomonas plecoglossicida, and Alcaligenes faecalis respectively. Sequences of these strains were submitted in NCBI GenBank under the accession number of bacterial isolates Pseudomonas punonensis (LMT03) (R1) MT677939, Pseudomonas aeruginosa DSM 50071 (R2) MT845116, Pseudomonas plecoglossicida (R4) MT883433, and Alcaligenes faecalis (DBHU5) MT872514. The phylogenetic trees of the above four rhizobacteria were constructed from the sequences using Mega 11 software. All the four isolates were closely related to each other. Fig.7 a-d; Table 4.
Table (4):
Selected four potent rhizobacterial isolates with their accession number.
Bacterial strain |
Accession number |
|---|---|
P. punonensis |
MT677939 |
P. aeruginosa |
MT845116 |
P. plecoglossicida |
MT883433 |
A. faecalis |
MT872514 |
In our soil system there are diversity of economically important microorganisms like plant growth promoting rhizobacteria, which is essential for overall plant growth promotion and crop productivity through various mechanisms like catalase activity, phosphate solubilization, IAA, ammonia and siderophore production.
In the present study, ten rhizobacterial isolates were isolated and screened for plant growth promoting traits. Out of them
P. punonensis, P. aeruginosa, P. plecoglossicida and A. faecalis on barley plants were found to be efficient for growth promotion. P. punonensis and P. plecoglossicida performed extremely well in seed germination and vigour index of barley plants which is an important factor for plant development and screened positive for all the biochemical analysis performed in this study.
Phosphorous is the second most essential element next to nitrogen and act as limiting factor for the plants growth and development.23 Availability of phosphorus to the plants is major concern and challenge, as in the soil phosphorus is present in insoluble form of inorganic complex and plant roots are unable to solubilize or mobilize in that form. PGPR with phosphate solubilizing ability mobilize and solubilize the phosphate and makes it available for plants by converting phosphate into organic, simple and soluble form. Phosphate solubilizing rhizobacteria Pantoea isolated from the rhizospheric region of soyabean plant induces phosphate solubilization in its culturing medium reached upto 900 mg/L.24 Pantoea cypripedii and Pseudomonas plecoglossicida significantly enhances Phosphate uptake in maize.25
Siderophore are the low molecular weight compound which have ability to chelate iron, many of the PGPRs have ability to fulfil iron requirement for plants by solubilization from organic and inorganic complexes.26 P. aeruginosa is considered as one of the most ubiquitous bacteria to acquire iron from the soil.27 Siderophore production is a mechanism of PGPR through which they protect themselves from phytopathogenic microorganism via creating a iron deficient environment for pathogens.28 PGPRs such as Aneurinibacillus aneurinilyticu inoculated with tea plant enhances siderophore production and thus plant growth promotion.29 Comparable result were found in our results by plant treated with P. punonensis and P. aeruginosa siderophore producing bacteria.
Bean plant treated with Pseudomonas fluorescence, alone or in combination with an arbuscular mycorrhizal fungus, Glomus mosseae showed enhanced catalase activity and hence performed extremely well in salt stress condition by development of antioxidant property.30 Bacteria having catalase activity shows its antioxidant property by break down of harmful free radical hydrogen peroxide into oxygen and water. Thus the plant treated by mentioned four bacteria having catalase activity as mentioned in the result which may be the reason to protect the plants from oxidative stress and thus healthy plant were obtained. Another important plant growth promoting activity was ammonia production which is source of nitrogen supply to the plants and achieves the nitrogen requirement of plants as a result increase the root-shoot length and hence favors the increase in vigour index of plants.31 The mentioned bacterial isolates were found to positive that may also be the reason to enhance the plant biomass through ammonia production.31 Bacterial isolates were also grown at various temperature and pH ranges to examine the tolerance ability and growth behavior. Further isolates R1, R2, R3, R4, and DBHU5 showed considerable and best growth density at various fluctuations of pH range and similar result was reported by.32
Production of IAA in plants also leads to increase in root length along with healthier root development.33 P. aeruginosa, showed maximum IAA production which results in suppression of charcoal Rot Disease of Chickpea.34 IAA production directly contributes in healthier and increased root length which results in increase in vigour index.32 Several reports were available on IAA production in the presence of tryptophan through PGPR treatment.35 PGPRs such as Aneurinibacillus aneurinilyticu, Sporosarcina koreensis and Bacillus megaterium associated with tea plant were known to enhance IAA production.29 P. punonensis, P. plecoglossicida, P. aeruginosa, and A. faecalis was found to show positive in indole acetic acid production hence it may be another way to increase in vigour index and healthier plant as compared to untreated one.
Inoculation of maize and rice plant with isolates belonging to genus Bacillus, Staphylococcus, Ochrobactrum, Pseudomonas, Lysinibacillus, Micrococcus, Leifsonia, Arthrobacter showed IAA, ammonia and siderophore production, and inoculation of these bacteria with rice and maize crop plants results in increase in its growth promotion.36 In support of our study similar result was found after treatment of the above isolated bacterial strains. Lentil plant inoculated with PGPR also enhanced the root-shoot elongation.37
Thus the selected four PGPR isolates were very efficient in promoting root and shoot growth during initial stage of seed germination and development and there by resulting in improved vigour index. However, after searching several literature it was found that fewer studies were done earlier on efficacy of P. punonensis, P. plecoglossicida and A. faecalis on seed germination and vigour index of barley crop plant. Increase in upto double fold of root and shoot length by the treatment of these strains were reported in the present study was much higher as compared to earlier reports on barley crop.
The applications of PGPR is an useful tool in seed biopriming technique as our result exhibits fruitful effect on enhancing vigour index, rapid seed germination, and seedling growth. Plant growth promoting rhizobacteria are the extremely valuable microorganism present in our ecosystem, which are of no cost, having no side effects on the environment as pollutant and act as natural bio fertilizers to be treated on plants so the selected bacterial strains P. punonensis, P. plecoglossicida, P. aeruginosa and A. faecalis were screened for various plant growth promoting traits, after treatment of these isolates on barley crop, performed healthy effect in plants by various plant growth promoting attributes and vigor index. As per studied literature there is very fewer studies were done on the effect of above bacterial isolates on vigour index and seed germination% of barley crop plant. Thus treatment of barley plants with PGPR strains are cost effective and eco-friendly approach to boost the agricultural productivity and co-operate with the farmers.
ACKNOWLEDGMENTS
The authors would like to thank, Department of Botany, Banaras Hindu University, Varanasi, Uttar Pradesh, India. The work was supported in part by Human Resource Development Group, Council of Scientific and Industrial Research, New Delhi, India for the award of research fellowship as CSIR JRF and further as CSIR-SRF.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
KD conceived the research, wrote the manuscript, analyzed the data, acquire the funding. KD performed the research. RSU supervised the research. All authors read and approved the final manuscript for publication.
FUNDING
This study was funded by Council of scientific and Industrial Research (award no. 09/013(0689)/2017-EMR-I), New Delhi, India.
ETHICS STATEMENT
Not applicable.
AVAILABILITY OF DATA
The DNA sequence obtained from this study have been submitted to the GenBank database with NCBI accession numbers MT677939, MT845116, MT883433, MT872514. All datasets generated or analyzed during this study are included in the manuscript.
- Keswani C, De Corato U, Sansinenea E, Adl SM, et al. Towards a new horizon of sustainable agriculture with microorganisms useful in agriculture. Rhizosphere. 2021;17:100293.
Crossref - Ahmad F, Ahmad I, Khan MS. Screening of free-living rhizospheric bacteria for their multiple plant growth promoting activities. Microbiol Res. 2008;163(2):173-181.
Crossref - Jha CK, Saraf M. Plant growth promoting rhizobacteria (PGPR): a review. J Agric Res Dev. 2015;5(2):108-119.
Crossref - Turan M, Gulluce M, Sahin F. Effects of plant-growth-promoting rhizobacteria on yield, growth, and some physiological characteristics of wheat and barley plants. Commun Soil Sci Plant Anal. 2012;43(12):1658-1673.
Crossref - Nadeem SM, Ahmad M, Zahir ZA, Javaid A, Ashraf M. The role of mycorrhizae and plant growth promoting rhizobacteria (PGPR) in improving crop productivity under stressful environments. Biotechnol Adv. 2014;32(2):429-448.
Crossref - Fonseca AA, Santos DA, Passos RR, Andrade FV, Rangel OJP. Phosphorus availability and grass growth in biochar modified acid soil: a study excluding the effects of soil pH. Soil Use Manag. 2020;36(4):714-725.
Crossref - Verma JP, Yadav J, Tiwari KN, Kumar A. Effect of indigenous Mesorhizobium spp. and plant growth promoting rhizobacteria on yields and nutrients uptake of chickpea (Cicer arietinum L.) under sustainable agriculture. Ecol Eng. 2013;51:282-286.
Crossref - Schroth MN, Hancock JG . Disease-suppressive soil and root-colonizing bacteria. Science. 1982;216(4553):1376-1381.
Crossref - Nihorimbere V, Ongena M, Smargiassi M, Thonart P. Beneficial effect of the rhizo- sphere microbial community for plant growth and health. Biotechnol Agron Soc Environ. 2011;15(2):327-337.
- Sayyed RZ, Gangurde NS, Patel PR, Josh SA, Chincholkar SB. Siderophore production by Alcaligenes faecalis and its application for growth promotion in Arachis hypogea. Indian J Biotechnol. 2010;9(3):302-307.
- Golzardi F, Vaziritabar Y, Vaziritabar Y, et al. Allelopathic Effect of Two Cynanchum acutum L. Populations on Emergence and Shoot Development of Barley. J Appl Environ Biol Sci. 2015;5(1):166-175.
- Kumar V, Khippal A, Singh J, et al. Barley research in India: Retrospect & prospects. J Wheat Res. 2014;6(1):1-20.
- Pikovskaya RI. Mobilization of phosphorus in soil in connection with vital activity of some microbial species. Mikrobiologiya. 1948;17:362-370.
- Premon ME, Moawad AM, Vlek PLG, et al. Effect of phosphate-solubilizing Pseudomonas putida on the growth of maize and its survival in the rhizosphere. 1996; (No.REP-12113. CIMMYT.).
- Bric JM, Bostock RM, Silverstone SE. Rapid in situ assay for indoleacetic acid production by bacteria immobilized on a nitrocellulose membrane. Appl Environ Microbiol. 1991;57(2):535-538.
Crossref - Loper JE, Schroth MN. Influence of bacterial sources of indole-3-acetic acid on root elongation of sugar beet. Phytopathology. 1986;76(4):386-389.
Crossref - Cappuccino JG, Sherman N. Biochemical activities of microorganisms. Microbiology, A Laboratory Manual. The Benjamin/Cummings Publishing Co. California, USA. 1992;188-247.
- Schwyn B, Neilands JB. Universal chemical assay for the detection and determination of siderophores. Anal Biochem. 1987;160(1):47-56.
Crossref - Clarke PH, Cowan ST. Biochemical methods for bacteriology. Microbiol. 1952;6(1-2):187-197.
Crossref - Sneath HAP, Halt GJ. Bergey’s manual of systematic bacteriology Vol. 2 Baltimore, M.D.: Williams and Wilkins. 1986.
- Kumari P, Meena M, Gupta P, Dubey MK, Nath G, Upadhyay RS, et al. Plant growth promoting rhizobacteria and their biopriming for growth promotion in mung bean (Vigna radiata L.) R. Wilczek). Biocatal Agric Biotechnol. 2018;16:163-171.
Crossref - Sambrook J, Fritsch EF, Maniatis T. Molecular Cloning: a Laboratory Manual, Ed 2. Cold Spring Harbor Laboratory, Cold Spring Harbor, NY. 1989; 545
- Alori ET, Glick BR, Babalola OO. Microbial phosphorus solublization and its potential for use in sustainable agriculture. Front Microbiol. 2017; 8:971.
Crossref - Son HJ, Park GT, Cha MS, Heo MS. Solubilization of insoluble inorganic phosphates by a novel salt-and pH-tolerant Pantoea agglomerans R42 isolated from soybean rhizosphere. Bioresour Technol. 2006;97(2):204-210.
Crossref - Kaur G, Reddy MS. Phosphate solubilizing rhizobacteria from an organic farm and their influence on the growth and yield of maize (Zea mays L.). J Gen Appl Microbiol. 2013;59(4):295-303.
Crossref - Arora NK, Verma M. Modified microplate method for rapid and efficient estimate on of siderophore produced by bacteria. 3 Biotech. 2017;7(6):381.
Crossref - Takase H, Nitanai H, Hoshino K, Otani T. Impact of Siderophore Production on pseudomonas aeruginosa infections in Immuno suppressed Mice. Infect Immun. 2000;68(4):1834-1839.
Crossref - Berendsen RL, Pieterse CMJ, Bakker PAHM, et al. The rhizosphere microbiome and plant health. Trends Plant Sci. 2012;17(8):478-486.
Crossref - Dutta J, Thakur D. Evaluation of multifarious plant growth promoting traits, antagonistic potential and phylogenetic affiliation of rhizobacteria associated with commercial tea plants grown in Darjeeling, India. PLoS ONE. 2017;12(8):e0182302.
Crossref - Younesi O, Moradi A. Effects of plant growth-promoting rhizobacterium (PGPR) and arbuscular mycorrhizal fungus (AMF) on antioxidant enzyme activities in salt-stressed bean (Phaseolus vulgaris L.). Agriculture. 2014;60(1):10-21.
Crossref - Marques APGC, Pires C, Moreira H, Rangel AOSS, Castro PML. Assessment of the plant growth promotion abilities of six bacterial isolates using Zea mays as indicator plant. Soil Biol Biochem. 2010;42(8):1229-1235.
Crossref - Prasad JK, Gupta SK, Raghuwanshi R. Screening multifunctional plant growth promoting rhizobacteria strains for enhancing seed germination in wheat (Triticum aestivum L.). Int J Agric Res. 2017;12(2):64-72.
Crossref - Gusain YS, Kamal R, Mehta CM, Singh US, Sharma AK. Phosphate solubilizing and indole-3-acetic acid producing bacteria from the soil of Garhwal Himalaya improve the growth of rice. J Environ Biol. 2015;36(1):301-307.
- Khare E, Arora NK. Effect of indole-3-acetic acid (IAA) produced by Pseudomonas aeruginosa in suppression of charcoal rot disease of chickpea. Curr Microbiol. 2010;61(1):64-68.
Crossref - Spaepen S, Vanderleyden J. Auxin and plant-microbe interactions. Cold Spring Harbor Perspectives in Biology. 2011:a001438.
Crossref - Bhattacharyya C, Banerjee S, Acharya U, et al. Evaluation of plant growth promotion properties and induction of antioxidative defense mechanism by tea rhizobacteria of Darjeeling, India. Sci Rep. 2020;10(1):1-19.
Crossref - Zafar M, Abbasi MK, Khan MA, Khaliq A, Sultan T, Aslam M. Effect of plant growth-promoting rhizobacteria on growth, nodulation and nutrient accumulation of lentil under controlled conditions. Pedosphere. 2012;22(6):848-859.
Crossref - Widawati S, Suliasih. The effect of plant growth promoting rhizobacteria (PGPR) on germination and seedling growth of Sorghum bicolor L. Moench. In IOP Conference Series: Earth and Environmental Science. 2018;166(1):012022.
Crossref - Majeed A, Abbasi MK, Hameed S, Imran A, Rahim N. Isolation and characterization of plant growth-promoting rhizobacteria from wheat rhizosphere and their effect on plant growth promotion. Front Microbiol. 2015;6:198.
Crossref
© The Author(s) 2022. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.