ISSN: 0973-7510
E-ISSN: 2581-690X
T-cell lymphoma is a rare disease in which T lymphocyte cells become cancerous. These lymphomas account between 10 percent and 15 percent of all cases of Non-Hodgkin lymphoma in the United States. Like the B-cell lymphomas, T-cell lymphomas are classified into two broad categories: aggressive (fast-growing) or indolent (slow-growing) (1). The G1/S checkpoint of the cell cycle is controlled by pRb protein, which functions in its hypophosphorylated form as a negative regulator of growth. p27 (Kip1), a member of CIP/KIP family of cyclin inhibitory proteins, participates in inhibition of forming complexes that allow pRb to phosphorylate and lead the cell into mitosis (2). To analyze the impact of concordant expression of p27 and HTLV-1 infection on a group of tissues with Non-Hodgkin’s lymphoma (NHL). Eighty formalin-fixed, paraffin- embedded lymph node tissues were enrolled in this study; (40) biopsies from Non-Hodgkin’s lymphoma (NHL), and (40) lymph nodes with (unremarkable pathological changes) as apparently healthy controls. Detection of HTLV-1was done by ultra-sensitive version of In situ hybridization method where as immunohisto chemistry detection system was used to demonstrate the expression of P27 gene expression. The HBZ gene of HTLV-1positive –CISH reaction was detected in (45%: 18 out of 40 cases) of Non-Hodgkin lymphoma tissues. No HTLV-1 positive – CISH reaction was detected in healthy lymph nodes tissues of the control group. The differences between the percentages of HTLV-1 detection in NHL tissues and control groups were statistically highly significant (P value = < 0.05). The positive P27-IHC reactions wase detected in 42.5% (17 out of 40 cases) of Non-Hodgkin lymphoma cases. A strong positive correlation was found between the detection, scores and intensity of p27 marker. Significant expressions of both p27 markers as well as HTLV-1 genes in Non-Hodgkin’s lymphoma could indicate for their possible roles both in lymph node pathogenesis and carcinogenesis
HTLV-1; Non-Hodgkin’s lymphoma (NHL), P27; ISH, IHC.
HTLV-1 is a complex retrovirus from the family Retroviridae3. The virus particles are 110-140 nm in size and contain the approximately 9 kb positive stranded diploid RNA genome with host cell tRNA needed for the initiation of transcription4. HTLV-1 basic leucine zipper factor (HBZ) is a unique 209 amino acid, 31 kDa protein encoded on the antisense strand that overlaps with the ORF for p12, p30 and p13. Similar to p30, it appears that HBZ acts antagonistically to Tax. It contains three main motifs, a nuclear localization motif, an N-terminus activation domain, and a C-terminus leucine zipper motif, which assist in the protein’s functions as a transcription factor5. It is expressed as three mRNA products, with a major spliced variant (sHBz) being the most dominantly expressed6. HBZ anti-Tax activity occurs mainly through indirect DNA binding. One method that it accomplishes this is through inhibition of the recruitment of CBP/p300 by Tax for viral transcription7.
Non-Hodgkin’s lymphoma (NHL) is more common in middle or elderly people. It can occur in lymph node and other organs that contain lymph tissue. This cancer may be located in one place in the body, but often located in multiple areas throughout the body8. This is because cancerous (malignant), lymphocyte often circulates through the body just like normal lymphocyte. Non-Hodgkin’s lymphoma can be either B- lymphoma or T-lymphoma depending on which type of lymphocyte become cancerous9. There are 40 different types of NHL. Some types of NHL grow very quickly; other types grow slowly10.
Most of NHLs (80-85%) are of B- cell origin, while (15-20%) being T-cell lymphomas11. NHL represent a clonal tumor of mature and immature B and T cells or natural killer (NK) cells at various stages of differentiation and can arise in nodal and extra nodal sites12.
Cyclin-dependent kinase inhibitor 1B (p27Kip1) is an enzyme inhibitor that in humans is encoded by the CDKN1B gene. It is often referred to as a cell cycle inhibitor protein because its major function is to stop or slow down the cell division cycle13.
The p27Kip1 gene has a DNA sequence similar to other members of the “Cip/Kip” family which include the p21Cip1/Waf1 and p57Kip2 genes 14. P27 cis-regulatory element is 114 nucleotides in length and is located at the very 5′ end of the 5’UTR of the p27 mRNA. It contains a small open reading frame (ORF) of 29 amino acids which is preceded by and overlaps with a G/C-rich hairpin domain. This hairpin domain is predicted to form multiple stable stem loops with similar free energy. Both the open reading frame and the stem loop elements contribute to cell cycle-regulated translation of the p27 mRNA15.
Low expression of p27kip1 protein is associated with excessive cell proliferation and has been linked to many types of human tumors, including lymphoma16, 17. Ferreri et al.,18 was reported that the activity of p27 in lymphoma by inhibiting the formation of cyclin E–CDK2 and/or cyclin D1–CDK4 complexes, thus blocking progression from G1 to S phase of the cell cycle.
Also, Møller et al.,19 was analyzed the correlation between cyclin D3 expression and high expression of p27 Kip1 in the subset of aggressive and very aggressive lymphomas with a proliferation rate exceeding 50%. However, Sáez et al.,20 and Decker et al.,21 found an abnormally high expression of p27KIP1 in NHL.
This study was done to unravel the rate as well as impact of either HTLV-1 or P27KIP1 in a group of Iraqi patients with Non-Hodgkin’s lymphoma.
The study was designed as a retrospective control cases one. It has recruited 80 selected formalin fixed, paraffin embedded lymph node tissue blocks were obtained, among them40 tissue biopsies from Non-Hodgkin’s lymphoma with different grades as well as40 lymph nodes with (unremarkable pathological changes) as apparently healthy controls. The diagnosis of these tissue blocks were based on their accompanied records. A consultant pathologist reexamined all these cases to further confirmation of their diagnosis. One section was mounted on ordinary glass slide and stained with hematoxyline and eosin, while other slides were mounted on charged slide to be used for ISH as well as IHC.
The detection of HTLV-1 by CISH kit (Zyto Vision GmbH. Fischkai, Bremerhaven. Germany) was performed on 4µm – paraffin embedded tissue sections. The sequence of oligonucleotides for HBZ- HTLV-1 used in this study was 5¯-CCA TCA ATC CCC AAC TCC TG-3¯ (nucleotide positions 645–664)22. The synthesized DNA probe was made to order by Bio-Synthesis (Lewisville, TX, USA).
For the In Situ Hybridization procedure, the slides were de-paraffinized and then treated by the standard methods of rehydration according to the details of processes for performing CISH reaction and then the probe was applied according the instructions of the manufacturing company (Zyto Vision GmbH. Fischkai, Bremerhaven. Germany). Then application of pepsin solution to the tissue section, immersion of slides in distilled water, air drying the sections, denaturation of the slides on hot plate, adding the 20 µl of cDNA probe. After that probe and target DNA were denaturized by placing in pre-warmed oven at 75°C for 8-10 minutes, slides were transferred to a pre-warmed humid hybridization chamber and incubated at 37°C for overnight. At the next day, slides were soaked in pre-warmed protein block at 37°C and remain in the buffer for 3 minutes. Then application of AP-Streptavidin to the slides and incubate for 30 min at 37°C in a humidity chamber. Then washed in wash buffer TBS and then twice times for 1 min in distilled water and application of 5-bromo3-chloro3-indoly/phosphate/nitro blue tertrazolium substrate- chromogen solution NBT/BCIP and incubated for 40 min at 37°C in humidity chamber. Slides were incubated at 37°C for 30 minutes or until color development was developed completed. Color development was monitored by viewing the slides under the microscope. A dark blue colored precipitate forms in positive cells. Then the slides were counter stained by immersion in Nuclear Fast Red stain for 30 seconds, then washing process was followed by immersion the slides for 1 minute in distilled water. After that sections were dehydrated by ethyl alcohol, cleared by xylene, then mounted with permanent mounting medium (DPX). Then final evaluation was done by light microscope.
Immunohistochemistry / Detection system (Abcam. England) was used to demonstrate the p27 gene expression (protein) in cells using a specific monoclonal antibodies, i.e. Primary antibody for that specific epitope which binds to nuclear targeted protein. The bound primary antibody is then detected by secondary antibody which contains specific label peroxidase labeled polymer conjugated to goat anti mouse immunoglobulin. The substrate is DAB in chromogen solution produced a positive reaction resulting in a brown- color precipitate at the antigen site in these tissues.
Chi –square test was used to detect the significance between variables of our study. All the statistical analysis was done by SPSS program (Version– 19) &P value was considered significant when p <0.05.
Distribution of Patients with Hodgkin and Non-Hodgkin Lymphoma According to Their Age
The archival specimens collected in this study were related to Non-Hodgkin lymphoma patients whom ages was ranged from three years to eighty years. The mean age of the patients with Non-Hodgkin lymphoma (46.20±22.067years) was highest than the mean age of the apparently healthy control (AHC) was (38.70± 22.519) years. However, there was no significant difference between NHL and AHC in age distribution Table (1).
Table (1):
Distribution of Study Groups According to the Mean and Range of Their Age (Years).
| Studied groups | No | Mean Age / Year | Std. Deviation | Std. Error |
Range | ANOVA Test (P-value) | |
|---|---|---|---|---|---|---|---|
| Min. | Max. | ||||||
| A.H. Control | 40 | 38.70 | 22.519 | 3.561 | 3 | 80 | P1=0.225 NS |
| NHL | 40 | 46.20 | 22.067 | 3.489 | 6 | 80 | |
| Total | 120 | ||||||
* Mini: Minimum, Maxi: Maximum
In Non-Hodgkin lymphoma, the most commonly affected age stratum in both 21–40 and 61-80 years were constituted (30.0%:12) for each group, followed by the age stratum of 41-60 years (27.5%:11) and lastly the lowest affected group of Non-Hodgkin lymphoma was those in the age stratum < 20 years (12.5%:5) .The statistical analysis shows non-significant differences (P>0.05) among age strata distribution of those studied groups as shown in the Table (2).
Table (2):
Statistical Analysis for the Distribution of Age Strata According to the Histopathological Diagnosis of Studied Groups.
| Age groups /Year | Groups | ||||
|---|---|---|---|---|---|
| A.H. Control |
NHL | Pearson Chi-Square (P-value) | |||
| ≤ 20 | No. | 10 | 5 | P=0.348 Non sign. (P>0.05) | |
| % | 25.0% | 12.5% | |||
| 21 – 40 (P>0.05) | No. | 12 | 12 | ||
| % | 30.0% | 30.0% | |||
| 41 – 60 | No. | 10 | 11 | ||
| % | 25.0% | 27.5% | |||
| 61 – 80 | No. | 8 | 12 | ||
| % | 20.0% | 30.0% | |||
| Total | No. | 40 | 40 | ||
| % | 100.0% | 100.0% | |||
* Non-Significant differences using Pearson Chi- square test at P>0.05 level.
Distribution of the Patients with Non-Hodgkin Lymphoma According to Their Gender
Regarding the patients whom suffering from Non-Hodgkin lymphoma, the percentage of males (57.5%: 23) was higher than the percentage of female (42.5%: 17). The male/female ratios of the patients with Non- Hodgkin lymphoma were 1.35:1. The statistical analysis showed non- significant difference (P>0.05) between lymphoma patients and control groups according to gender Table (3).
Table (3):
Distribution of Study Groups According to Their Gender.
| Gender | Studied groups | Pearson Chi-Square (P-value) | |||
|---|---|---|---|---|---|
| A.H. Control | NHL | ||||
| Male | No. | 23 | 23 | P=1.00
Non sign. (P>0.05) |
|
| % | 57.5% | 57.5% | |||
| Female | No. | 17 | 17 | ||
| % | 42.5% | 42.5% | |||
| Total | No. | 40 | 40 | ||
| % | 100.0% | 100.0% | |||
* Non-Significant differences using Pearson Chi- square test at P>0.05 level.
Grading of Lymphoma Group Cases
In this study, a the highest percentage and number of Non-Hodgkin lymphoma patients was seen in grade I and grade II (37.5%:15) and the lowest was in grade III (25%:10). The results revealed non-significant differences at (P>0.05) between Non-Hodgkin lymphomas grades shown in the Table (4).
Table (4):
Statistical Analysis for the Distribution of Lymphoma Group According to Their Grade.
| Grade | Studied groups NHL | Pearson Chi-Square (P-value) |
|
|---|---|---|---|
| I | No. | 15 | P=0.899 Non sign (P>0.05) |
| % | 37.5% | ||
| II | No. | 15 | |
| % | 37.5% | ||
| III | No. | 10 | |
| % | 25.0% | ||
| Total | No. | 40 | |
| % | 100.0% | ||
* Non-Significant differences using Pearson Chi- square test at P>0.05 level.
Human T Lymphotropic Virus Type-1 (HTLV-1)-Associated Lymphoma by In Situ Hybridization Technique (ISH)
The DNA of HTLV-1 was detected in tissue blocks of lymphoma patients. Wide spectrum HTLV-1 was detected using digoxigenin- labeled probes in a morphological preserved tissue sections of Non-Hodgkin lymphoma. The signals of CISH were detected as bright blue discoloration with blue stain and counter stained with nuclear red solution in referring to HTLV-1 at the sites of complementary sequences.
The magnitude of expression of each tested markers was measured in 2 different systems of evaluations. The first one is using a scoring system with ordered categories ranging from 1 to 4 for a positive stain [negative, low (+1), moderate (+2), high (+3)]. The second system signal intensity measure with 2 categories (no stain and ordered-grades of staining or signaling).
HTLV-1 associated with apparently healthy lymph node control tissues using wide spectrum DNA-CISH detection
In this study, the apparently healthy lymph node control tissues were tested by using wide spectrum HTLV-1-DNA-CISH detection. All cases were negative; therefore they are excluded from the statistical analysis.
Results of HTLV-1in Patients with Non-Hodgkin Lymphoma
Fig. (1) shows the positive results of HTLV-1-CISH detection, where 45% (18 of total 40) showed positive signals, while, 55% negative signals which represented 22 out of 40 cases in this group.
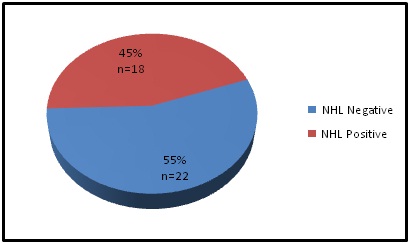 Fig. 1. Distribution of HTLV-1 Associated with Non-Hodgkin Lymphoma
Fig. 1. Distribution of HTLV-1 Associated with Non-Hodgkin LymphomaIn this study the results showed as in Table (5) that the HTLV-1associated with Non-Hodgkin lymphoma was non significant at 5 percent level (P>0.05).
Table (5):
Statistical Analysis for HTLV-1 Associated with Non-Hodgkin Lymphoma Using CISH Technique.
| HTLV-1 | NHL (n=40) |
% | P-value |
|---|---|---|---|
| Negative | 22 | 55% | Z test P=0.636
Non sign. (P>0.05) |
| Positive | 18 | 45% |
Positive HTLV-1 DNA- CISH Signal Scoring
The highest percentage of HTLV-1 score signaling (17.5%:7 out of 18) was found in the high score (score III), whereas (15%:6 out of 18) and (12.5%:5out of 18) were found within low (score I) and scores moderate ( score II), respectively. In this study the results showed that the HTLV-1associated with Non-Hodgkin lymphoma was highly significant at 1 percent level (P<0.01). Table (6) & Fig. (2).
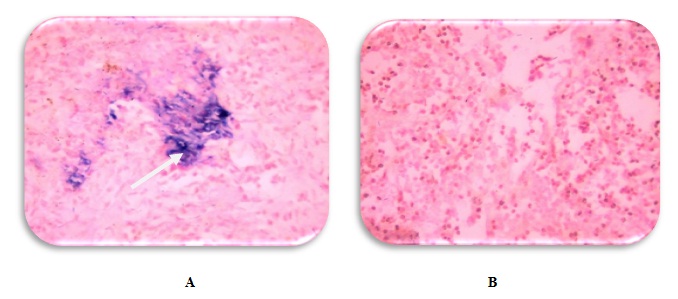 Fig. 2. In situ Hybridization (ISH) for Generic HTLV-1 Deduction Infiltrative Lymphoma Cancers Using Digoxigenin-Labeled HTLV-1 Probes; Stained with (Blue)and Counter Stained by Nuclear Fast Red (Red).A)-NHL with Positive HTLV-1 -CISH Reaction with High Score and Strong Signal Intensity (40X).B) -NHL with Negative HTLV-1–CISH Reactions. (40X)
Fig. 2. In situ Hybridization (ISH) for Generic HTLV-1 Deduction Infiltrative Lymphoma Cancers Using Digoxigenin-Labeled HTLV-1 Probes; Stained with (Blue)and Counter Stained by Nuclear Fast Red (Red).A)-NHL with Positive HTLV-1 -CISH Reaction with High Score and Strong Signal Intensity (40X).B) -NHL with Negative HTLV-1–CISH Reactions. (40X)Table (6):
Distribution of HTLV-1 Signal Scoring Associated with Non-Hodgkin lymphoma by Using ISH Technique.
| P-value | Non-Hodgkin lymphoma (no.=40) | HTLV-1signal scoring | ||
|---|---|---|---|---|
| % | No. | |||
| χ2 test P=0.00
Highly sign. (P<0.01) |
55 | 22 | Negative | |
| 45 | 18 | Positive | ||
| 15 | 6 | I | Scoring | |
| 12.5 | 5 | II | ||
| 17.5 | 7 | III | ||
Distribution of Signal Intensity of CISH for Detection of Wide Spectrum HTLV-1DNA in Non-Hodgkin Lymphoma Cases.
Table (7) show the positive results of HTLV-1 -DNA /CISH detection where 45% (18 out of 40 cases) from Non-Hodgkin lymphoma group showed positive signals included 32.5% (13 out of 18 cases) with strong signal intensity (III), followed by 10 % (4 out of 18 cases) with moderate signal intensity (II), and 2.5% (1 out of 18 cases) with weak signal intensity (I) (Fig.2). Statistically, there was highly significant difference found among them (P<0.01)
Table (7):
Distribution of HTLV-1Signal Intensity Associated with Non-Hodgkin Lymphoma by Using CISH Technique.
| P-value | Non-Hodgkin lymphoma (no.=40) | HTLV-1 signal intensity | ||
|---|---|---|---|---|
| % | No. | |||
| χ2 test P=0.00 Highly sign. (P<0.01) |
55 | 22 | Negative | |
| 45 | 18 | Positive | ||
| 2.5 | 1 | I | Intensity | |
| 10 | 4 | II | ||
| 32.5 | 13 | III | ||
The Results of Evaluation of Cyclin-Dependent Kinase Inhibitors p27KIP1in Non-Hodgkin lymphoma:
The Results of P27- Protein Expression in the Apparently Healthy Lymph Node Control Tissues.
In this study, the results of immunohistochemistry staining of P27protein using biotinylated anti- p27 protein- antibodies in the apparently healthy lymph node control tissues showed no signal was expressed to all cases; therefore they are excluded from the statistical analysis.
The Results of P27- Protein Expression in Non-Hodgkin Lymphoma.
Immunohistochemistry for P27 protein was detected as brownish discoloration at nuclear localization (Figure 3).
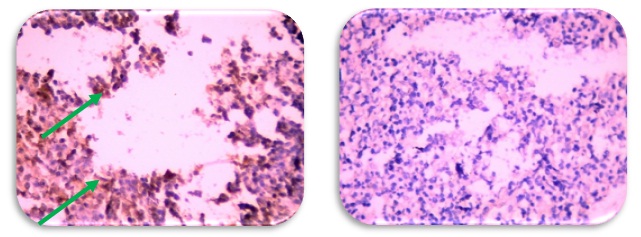
Fig. 3. Infiltrative Lymphoma Cancers Showing the Results of Immuno histochemistry Staining of p27 Protein Using Biotinylated Anti- p27 Protein- Antibodies, Stained by DAB-Chromogen (Brown) and Counter Stained by Mayer’s Hematoxylin (Blue). A) -NHL with Positive p27 -IHC Reaction (40X). B) -NHL with Negative ep27 -IHC Reactions. (40X)
Results of P27- IHC Signal Scoring
P27 protein was detected by IHC test in 42.5% (17out of 40 cases) of Non-Hodgkin lymphoma, while no signal was expressed in (57.5%:23out of 40). In the non-Hodgkin lymphoma group, 30 % (12 out of 17 cases) have low score (score I) whereas, 12.5% (5 out of 17 cases) have moderate score (score II). Statistically, highly significant differences between negative, low and moderate scoring cases at 1 percent level (P<0.01) Table (8)
Table (8):
Frequency Distribution of Immunohistochemistry Results of P27 Protein According to the Signal Scoring.
| P-value | Non-Hodgkin lymphoma (no.=40) | P27 signal scoring | ||
|---|---|---|---|---|
| % | No. | |||
| χ2test P=0.00 Highly sign. (P<0.01) |
57.5 | 23 | Negative | |
| 42.5 | 17 | Positive | ||
| 30.0 | 12 | I | Scoring | |
| 12.5 | 5 | II | ||
| 0.00 | 0/ 17 | III | ||
Results of P27- IHC Signal Intensity
Among (40) Non-Hodgkin lymphoma, only 17 cases (42.5%) showed positive P27 –IHC reactions. A percentage of 32.5% (13 cases out of 17) of P27 –IHC test in a Non-Hodgkin lymphoma group was found to have moderate signal intensity while weak signals intensity constituted 10% (4 out of 17) Fig. (3). statistically, highly significant differences between negative, weak and moderate intensity cases at 1 percent level (P<0.01) in Non-Hodgkin lymphoma group as shown in Table (9).
Table (9):
Frequency Distribution of Immuno histochemistry Results of P27 Protein According to the Signal Intensity.
| P-value | Non-Hodgkin lymphoma (no.=40) |
P27 signal intensity | ||
|---|---|---|---|---|
| % | No. | |||
| χ2test P=0.00 Highly sign. (P<0.01) |
57.5 | 23 | Negative | |
| 42.5 | 17 | Positive | ||
| 10.0 | 4 | I | Intensity | |
| 32.5 | 13 | II | ||
| 0.00 | 0/ 17 | III | ||
Correlations between Intensities of Studied Markers (Wide Spectrum HTLV-1 and P27) in Patients with Non-Hodgkin Lymphoma
HTLV-1
There are no significant correlations between the intensity of HTLV-1 and p27 Table (10).
Table (10):
Spearman’s rho Statistical Testing to Evaluate the Intensity of Studied Molecular Markers in Relation with HTLV-1 infections in Non-Hodgkin lymphoma.
| Spearman’s rho | Age groups/Year |
Grade | HTLV-1 | |
|---|---|---|---|---|
| HTLV-1 | r. | .013 | .211 | |
| P-value | .937 | .191 | ||
| P27 | r. | -.054 | .110 | -.052 |
| P-value | .742 | .501 | .748 | |
In Non-Hodgkin lymphoma, the mean age of patients was (46.20±22.067years) and the most commonly affected age stratum in both 21– 40 and 61– 80 years were constituted (30.0%:12) for each group, followed by the age stratum of 41- 60years (27.5%:11) and lastly the lowest affected group of Non-Hodgkin lymphoma was those in the age stratum < 20 years (12.5%:5) Table (1) and Table (2). Moreover, our result in Non-Hodgkin lymphoma, was closely comparable to an Iraqi study done by Ridha (23) who found that the mean age of their NHL patients was (39.70) years. Also this study was in agreement with Jordanian study done by Al-masri et al., (24) who found that the median age of their NHL patients was 43.5 years. Also, It is consistent with Pakistanian studies done by Lal et al.,(25) and Naz et al.,(26) who found the median age of their NHL patients was (48.0±13.3) years and (42.5) years, respectively.
Most people with Non-Hodgkin lymphoma are older than 60 (27, 28).These differences in age groups could be contributed to the differences in environmental and geographical risk factors affecting each study groups making NHL affecting relatively middle age group in Iraq and neighboring countries.
The ratio of male to female patients with Non- Hodgkin lymphoma was 1.35:1 Table (3).The results of the current study are in concurrence with the results of most other studies. Hussein et al., (29) was found that NHL is more common in male (70%) than female (30%). Moreover, Smith et al., (30) who found in NHL that the affected men were more than their counter part women. Also, Boffetta, (31), was found that the NHL is the 8th most commonly diagnosed cancer in men and the 11th in women.
Lymphoma and infections in males higher than females suggests that the well known differences in immunity may be responsible for this dichotomy. Besides immune surveillance, genome surveillance mechanisms also differ in efficiency between males and females. Other obvious differences include hormonal ones and the number of X chromosomes .Also the difference might be due to the peculiar characteristics of the referral centers, smaller case numbers, or geographic distribution.
The highest percentage and number of Non-Hodgkin lymphoma patients was seen in grade I and grade II (37.5%:15) for each and the lowest was found in grade III (25%:10).The present study is mostly concurring with study made by Laurini et al., (32).The distribution of low-grade of NHL was (47.1%) in Central and South America and high-grade of NHL North America was (37.5%). However ,in the same study were little differences in high-grade of NHL (52.9%) in Central and South America and in low grade of NHL (62.5%) in North America. In addition ,the results of our study is generally consent with Villuend as et al., (33) who revealed that the ratio of grade I, grade II and grade III in NHL were 26.7%, 26.9% and 46.4% ,respectively .In addition, the patients often present themselves to the medical care system at much later stages of the diseases where the low grade lymphomas have evolved into secondary type of high grade once. For a more precise understanding of this phenomenon, it is necessary to examine the etiology and the epidemiology of the lymphomas in Iraq as well as Iran because there is the possibility that higher grade lymphomas are of the primary type and some important factors may be involved in high occurrences that is a subject that need further investigation, since there has been little research done in this area (34). In my opinion this could be contributed to the heterogeneity ,risk factors (exposure to certain chemicals and drugs, radiation exposure, a weakened immune system, Race, ethnicity, and geography) and small samples size.
The positive results of HTLV-1-CISH detection, where 45% showed positive signals, while, 55% negative signals which represented 31 out of 40 cases in this group. Also, as in negative results of HTLV-1-DNA in NHL are probably related to the absence of HTLV-1-DNA in these biopsies or could be related its presence in the cells at different regions of that tissue. The presence of different type of HTLV-1 other than these type used in this study is another possibility. The HTLV-1-seropositive cases of Non-Hodgkin lymphoma in this study may predominantly constitute with the results obtained by Adedayo and Shehu, (35) who revealed that was a significant association between HTLV-1 seropositivity and Non-Hodgkin lymphoma, as 44.4% of positive cases. The present study also in a broad harmony with study done by Manns et al., (36), they revealed that overall patients with NHL were 10 times more likely than were controls to be seropositive for HTLV-I in Jamaica, Trinidad and Tobago. They found that the association between NHL and HTLV-I was greatest for T-cell lymphomas. Panelatti (37), was found that the serological test was positive for HTLV-1 antibodies in NHL patient. In addition, Birckhead et al., (38) who found that a close relation between HTLV-1 and lymphoma. While, Miyagi et al., (39) who found that the ratio of positive cases in NHL with HTLV-1 was 26.1%. While, Hatano et al., (40) found19.35% of NHL had HTLV-1 proviral DNA.
Whereas, the difference between these studies and recent study regarding the ratio of HTLV-1 with NHL may be related to the peculiar characteristics of the referral centers, smaller case numbers, or geographic distribution enrolled in these studies. In the other hand, The molecular conducted by Gualco et al.,(41) for pro viral HTLV-1DNA using Tax gene amplification was negative in all cases of NHL patients. While, in the present study for pro viral HTLV-1DNA using HBZ gene. Viruses are seldom complete carcinogenesis and are essential but not sufficient factors even in those with viral carcinogenesis such as HPV, EBV, HTLV-1 related carcinogenesis (42). In this respect, we believe the detected HTLV-1is thought to have a synergistic effect in the pathogenesis of NHL along with many other etiological factors such as chemical, radiations, and genetic factors.
Previous studies have shown that a subset of aggressive and very aggressive lymphomas with high p27Kip1 expression exceeding (50%) (43).
In the current study, the P27 protein was detected by IHC test in 42.5% of Non-Hodgkin lymphoma, while no signal was expressed in 57.5% Table (8).
Wang et al., (44) was revealed that the low expression of p27kip1 protein is associated with excessive cell proliferation and has been linked to many types of human tumors; including lymphoma.p27KIP1 staining was found most frequently in a low to intermediate proportion of the tumoural cells.
The P27protien positive cases of Non-Hodgkin lymphoma in this study may predominantly constitute with the results obtained by Møller et al., (45) and Li et al., (46) were found 43% and 51.9% of p27 positive cases of NHL, respectively.
However, there are no significant correlations among HTLV-1 and P27 in NHL (P-value > 0.05). In my opinion this could be contributed to the heterogeneity, geographic distribution and small samples size.
- Parkin, D.M., The global health burden of infection associated cancers in the year 2002. International journal of cancer, 2006; 118(12), pp.3030-3044.
- Cinti C, Leoncini L, Nyongo A, Ferrari F, Lazzi S, Bellan C, Vatti R, Zamparelli A, Cevenini G, Tosi GM, Claudio PP, Maraldi NM, Tosi P and Giordano A. Genetic alterations of the retinoblastoma-related gene RB2/p130 identify different pathogenetic mechanisms in and among Burkitt’s lymphoma subtypes. Am J Pathol, 2000; 156: 751–760
- Lairmore, M. and Franchini, G., Human T-cell leukemia virus types 1 and 2. Fields virology, 2007; 5: pp.2071-2106.
- Lawson, V.A., Lee, J.Y., Doultree, J.C., Marshall, J.A. and McPhee, D.A., Visualisation of phenotypically mixed HIV-1 and HTLV-I virus particles by electron microscopy. Journal of biomedical science, 2000; 7(1): pp.71-74.
- Hivin, P., Frédéric, M., Arpin-André, C., Basbous, J., Gay, B., Thébault, S. and Mesnard, J.M., Nuclear localization of HTLV-I bZIP factor (HBZ) is mediated by three distinct motifs. Journal of cell science, 2005; 118(7), pp.1355-1362.
- Murata, K., Hayashibara, T., Sugahara, K., Uemura, A., Yamaguchi, T., Harasawa, H., Hasegawa, H., Tsuruda, K., Okazaki, T., Koji, T. and Miyanishi, T., A novel alternative splicing isoform of human T-cell leukemia virus type 1 bZIP factor (HBZ-SI) targets distinct subnuclear localization. Journal of virology, 2006; 80(5), pp.2495-2505.
- Clerc, I., Polakowski, N., André-Arpin, C., Cook, P., Barbeau, B., Mesnard, J.M. and Lemasson, I., An interaction between the human T cell leukemia virus type 1 basic leucine zipper factor (HBZ) and the KIX domain of p300/CBP contributes to the down-regulation of tax-dependent viral transcription by HBZ. Journal of Biological Chemistry, 2008; 283(35), pp.23903-23913.
- López-Guillermo, A., Colomo, L., Jiménez, M., Bosch, F., Villamor, N., Arenillas, L., Muntañola, A., Montoto, S., Giné, E., Colomer, D. and Beà, S., Diffuse large B-cell lymphoma: clinical and biological characterization and outcome according to the nodal or extranodal primary origin. Journal of clinical oncology, 2005; 23(12), pp.2797-2804.
- NCI,National Cancer Institute. Adul non-Hodgkin lymphoma treatment: Cellular classification of a dult non-Hodgkin lymphoma pagez. 2010; Accessed September 2.
- ACS,American Cancer Society (2005). Cancer Facts & Figures 2005.Atlanta : American Cancer Society 2003. Non-Hodgkin’s lymphoma.Avaiable at: http://www3.cancer.org/cancer info/.
- NCCN ,National Comprehensive Cancer Network.( 2012). Clinical Practice Guidelines in Oncology: Non-Hodgkin’s Lymphomas.1.
- Illes, A., Varoczy, L., Papp, G., Wilson, P.C., Alex, P., Jonsson, R., Kovacs, T., Konttinen, Y.T., Zeher, M., Nakken, B. and Szodoray, P.. Aspects of B Cell Non Hodgkin’s Lymphoma Development: A Transition from Immune Reactivity to Malignancy. Scandinavian journal of immunology, 2009; 69(5): pp.387-400.
- Nadeem, L., Brkic, J., Chen, Y.F., Bui, T., Munir, S. and Peng, C., Cytoplasmic mislocalization of p27 and CDK2 mediates the anti-migratory and anti-proliferative effects of Nodal in human trophoblast cells. J Cell Sci, 2013; 126(2): pp.445-453.
- Sgambato, A., Cittadini, A., Faraglia, B. and Weinstein, I.B., Multiple functions of p27Kip1 and its alterations in tumor cells: a review. Journal of cellular physiology, 2000; 183(1): pp.18-27.
- Göpfert, U., Kullmann, M. and Hengst, L., Cell cycle-dependent translation of p27 involves a responsive element in its 52 -UTR that overlaps with a uORF. Human molecular genetics, 2003; 12(14): pp.1767-1779.
- Hatta, Y. and Koeffler, H.P., Role of tumor suppressor genes in the development of adult T cell leukemia/lymphoma (ATLL). Leukemia, 2002; 16(6), pp.1069-1085.
- Wang, Y., Fei, M., Cheng, C., Zhang, D., Lu, J., He, S., Zhao, Y., Wang, Y. and Shen, A., Jun activation domain-binding protein 1 negatively regulate p27kip1 in non-Hodgkin’s lymphomas. Cancer biology & therapy, 2008 7(3): pp.460-467.
- Ferreri, A.J., Ponzoni, M., Pruneri, G., Freschi, M., Rossi, R., Dell’Oro, S., Baldini, L., Buffa, R., Carboni, N., Villa, E. and Viale, G., Immunoreactivity for p27KIP1 and cyclin E is an independent predictor of survival in primary gastric non Hodgkin’s lymphoma. International journal of cancer, 2001; 94(4), pp.599-604.
- Møller, M.B., Nielsen, O. and Pedersen, N.T., Cyclin D3 expression in non-Hodgkin lymphoma: correlation with other cell cycle regulators and clinical features. American journal of clinical pathology, 2001; 115(3), pp.404-412.
- Sáez, A.L., Sanchez, E., Sanchez-Beato, M., Cruz, M.A., Chacon, I., Munoz, E., Camacho, F.I., Martinez-Montero, J.C., Mollejo, M., Garcia, J.F. and Piris, M.A., p27KIP1 is abnormally expressed in diffuse large B-cell lymphomas and is associated with an adverse clinical outcome. British journal of cancer, 1999; 80(9), p.1427–1434.
- Decker, T., Schneller, F., Hipp, S., Miething, C., Jahn, T., Duyster, J. and Peschel, C., Cell cycle progression of chronic lymphocytic leukemia cells is controlled by cyclin D2, cyclin D3, cyclin-dependent kinase (cdk) 4 and the cdk inhibitor p27. Leukemia, 2002; 16(3), pp.327-334.
- Shimizu-Kohno, K., Satou, Y., Arakawa, F., Kiyasu, J., Kimura, Y., Niino, D., Sugita, Y., Ishikawa, F., Matsuoka, M. and Ohshima, K., Detection of HTLV 1 by means of HBZ gene in situ hybridization in formalin fixed and paraffin embedded tissues. Cancer science, 2011; 102(7), pp.1432-1436.
- Ridha, W.K., Epstein Barr virus-mediated deregulation of cell cycle pathways in malignant lymphomas: Implication of NFKB, p53, p27 and the mutant p21 Ras using in situ technique (Doctoral dissertation, Ph. D. Thesis, 2004; University of AL-Nahrain, Iraq).
- Al-masri, N.M., Habashneh, M.A. and Khalidi, H.S., Non-Hodgkin lymphoma in Jordan. Types and patterns of 111 cases classified according to the WHO classification of hematological malignancies. Saudi medical journal, 2004; 25(5), pp.609-614.
- Lal, A., Bhurgri, Y., Vaziri, I., Rizvi, N.B., Sadaf, A., Sartajuddin, S., Islam, M., Kumar, P., Adil, S., Kakepoto, G.N. and Masood, N., Extranodal non-Hodgkin’s lymphomas—a retrospective review of clinico-pathologic features and outcomes in comparison with nodal non-Hodgkin’s lymphomas. Asian Pac J Cancer Prev, 2008; 9(3), pp.453-458.
- Naz E, Mirza T, Aziz S, Danish F, Tariq S, Ali A: Frequency and Clinics pathologic Correlation of Different types of NHL According to WHO classification . J Pak Med Assoc;2011; 61(3): 260-357
- Patte, C., Auperin, A., Gerrard, M., Michon, J., Pinkerton, R., Sposto, R., Weston, C., Raphael, M., Perkins, S.L., McCarthy, K. and Cairo, M.S., Results of the randomized international FAB/LMB96 trial for intermediate risk B-cell non-Hodgkin lymphoma in children and adolescents: it is possible to reduce treatment for the early responding patients. Blood, 2007; 109(7), pp.2773-2780.
- Ansell, P., Simpson, J., Lightfoot, T., Smith, A., Kane, E., Howell, D., Newton, R., McGonagle, D., Jack, A. and Roman, E., Non Hodgkin lymphoma and autoimmunity: Does gender matter?. International journal of cancer, 2011; 2: pp.460-466.
- Hussein, M.R., Al-Sabae, T.M. and Georgis, M.N., Analysis of the Bcl-2 and p53 protein expression in the lymphoproliferative lesions in the Upper Egypt. Cancer biology & therapy, 2005; 4(3), pp.324-328.
- Smith, A., Crouch, S., Lax, S., Li, J., Painter, D., Howell, D., Patmore, R., Jack, A. and Roman, E., Lymphoma incidence, survival and prevalence 2004–2014: sub-type analyses from the UK’s Haematological Malignancy Research Network. British journal of cancer, 2015; 112(9), pp.1575-1584.
- Boffetta, P., I. Epidemiology of adult non-Hodgkin lymphoma. Annals of oncology, 22(suppl_4), 2011; pp. 427-431.
- Laurini, J.A., Perry, A.M., Boilesen, E., Diebold, J., MacLennan, K.A., Müller-Hermelink, H.K., Nathwani, B.N., Armitage, J.O. and Weisenburger, D.D., Classification of non-Hodgkin lymphoma in Central and South America: a review of 1028 cases. Blood, 2012; 120(24), pp.4795-4801.
- Villuendas, R., Sánchez-Beato, M., Martínez, J.C., Saez, A.I., Martinez-Delgado, B., García, J.F., Mateo, M.S., Sanchez-Verde, L., Benítez, J., Martínez, P. and Piris, M.A., Loss of p16/INK4A protein expression in non-Hodgkin’s lymphomas is a frequent finding associated with tumor progression. The American journal of pathology, 1998; 153(3), pp.887-897.
- Hashemi, M. and Parwaresh, M.R., A Study of lymphomas in selected centers in Theran, Based on the updated Kiel classification. Arch Iran Med, 2001; 4: pp.59-66.
- Adedayo, O.A. and Shehu, S.M., Human T cell lymphotropic virus type 1 (HTLV 1) and lymphoid malignancies in Dominica: A seroprevalence study. American journal of hematology, 2004; 77(4), pp.336-339.
- Manns, A., Cleghorn, F.R., Falk, R.T., Hartge, P., Benichou, J., Blattner, W.A., Jaffe, E.S., Hanchard, B. and Bartholomew, C., Role of HTLV-I in development of non-Hodgkin lymphoma in Jamaica and Trinidad and Tobago. The Lancet, 1993; 342(8885), pp.1447-1450.
- Panelatti, G., Plumelle, Y., Arfi, S., Pascaline, N., Caplanne, D. and Jean-Baptiste, G., Sarcoidosis and leukemia/T-cell lymphoma associated with HTLV-1 virus infection in adults (apropos of a case). La Revue de medecine interne, 1992; 13(4), pp.299-301.
- Birckhead, B.J., Peterson, J.L., Patel, A.B. and Miller, R.C., Palliative radiation for leukemic arthropathy from human T-cell lymphoma virus-associated adult T-cell leukemia lymphoma. Rare tumors, 2015; 7(3), pp.102-104
- Miyagi, J.I., Toda, T., Uezato, H., Ohshima, K., Miyakuni, T., Takasu, N. and Masuda, M., Detection of Epstein-Barr virus and human T-cell lymphotropic virus type 1 in malignant nodal lymphoma, studied in Okinawa, a subtropical area in Japan. International journal of hematology, 2002; 75(1), pp.78-84.
- Hatano, B., Ohshima, K., Katoh, A., Kanda, M., Kawasaki, C., Tsuchiya, T., Shimazaki, K., Haraoka, S., Sugihara, M., Suzumiya, J. and Kikuchi, M., Non HTLV 1 associated primary gastric T cell lymphomas show cytotoxic activity: clinicopathological, immunohistochemical characteristics and TIA 1 expression in 31 cases. Histopathology, 2002; 41(5), pp.421-436.
- Gualco, G., Chioato, L., Weiss, L.M., Harrington Jr, W.J. and Bacchi, C.E., Analysis of human T-cell lymphotropic virus in CD25+ anaplastic large cell lymphoma in children. American journal of clinical pathology, 2009; 132(1), pp.28-33.
- Brooks, G. F; Carroli, K. C. ; Butel, J. S. and Morse, S. A. Jawetz, Melnick and Adelberg,S.Medical Microbiology., 2010; 24 ed.McGraw Hill Companies.USA.
- Sánchez-Beato, M., Camacho, F.I., Martýìnez-Montero, J.C., Sáez, A.I., Villuendas, R., Sánchez-Verde, L., Garcýìa, J.F. and Piris, M.A., Anomalous high p27/KIP1 expression in a subset of aggressive B-cell lymphomas is associated with cyclin D3 overexpression. p27/KIP1—cyclin D3 colocalization in tumor cells. Blood, 1999; 94(2), pp.765-772.
- Wang, Y., Fei, M., Cheng, C., Zhang, D., Lu, J., He, S., Zhao, Y., Wang, Y. and Shen, A., Jun activation domain-binding protein 1 negatively regulate p27kip1 in non-Hodgkin’s lymphomas. Cancer biology & therapy, 2008; 7(3), pp.460-467.
- Møller, M.B., Skjødt, K., Mortensen, L.S. and Pedersen, N.T., Clinical significance of cyclin dependent kinase inhibitor p27Kip1 expression and proliferation in non Hodgkin’s lymphoma: independent prognostic value of p27Kip1. British journal of haematology, 1999; 105(3), pp.730-736.
- Li, Y.N., Feng, D.Y. and Zheng, H., Expression of PTEN and p27 proteins in non-Hodgkin’s lymphoma. Hunan yi ke da xue xue bao= Hunan yike daxue xuebao= Bulletin of Hunan Medical University, 2003; 28(3), pp.233-236.
© The Author(s) 2017. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


