ISSN: 0973-7510
E-ISSN: 2581-690X
Marine endophytic fungi are rich sources of bioactive metabolites due to their peculiar life style and extreme environmental conditions of the marine ecosystem. However, many of the marine endophytic fungi are either unexplored or obscure due to lack of proper staining and culture techniques. In the present work an endophytic fungal species Cladosporium uredinicola was isolated from the marine brown alga Dictyota dichotoma and identified through molecular characterization. Further, an effort was also made to understand the life cycle of the endophytic fungus and its growth pattern. Different stages in the life cycle of the fungus was observed with the improvised staining techniques and phase contrast microscopic observations.
Marine endophytic fungi, Cladosporium uredinicola, Dictyota dichotoma, differential staining
Endophytes are the microorganisms that reside in the living tissues of the host without causing any apparent infection(Suryanarayanan 2013).Due to this unique adaptation the endophytes thrive in the metabolically aggressive hostile environment by constantly encountering the host defence metabolites (Cabral et al.1993, Schulz et al.1999, Selim et al.2012).This hostile environment may contribute to the evolution of the potential synthetic ability of the endophytes(Owen and Hundley 2004).Further the survival of the marine endophytes in the extreme physical and chemical conditions of the marine environment imparts special characters which make them prospective candidates for screening of natural products with various pharmaceutical and other applications.
Even though the endophytes are worth exploring, many of them remain unexplored due to lack of proper techniques in their culture and morphological observation (Ghimire and Hyde 2008). Majority of the endophytic fungi belongs to the division Ascomycota (Geisen et al. 2017).
In the present work an endophytic ascomycetes fungus Cladosporium uredinicola was isolated from the marine brown alga Dictyota dichotoma and identified through molecular characterization .An attempt was also made to observe different stages in the life cycle of this endophytic fungus through simple and improvised isolation and staining techniques. Different stages in the life cycle of the fungus viz. Conidiospores, coronate ascocarp, ascospores, 4-celled ascospores, 8-celled ascospores, transmission of endophyte from host to its offspring were observed under phase contrast and light microscopes.
Samples
The algal samples were collected from Bay of Bengal at Mypadu beach (coordinates 14.5068º N 80.1790 ºE), SPSR Nellore district, Andhra pradesh state of India. The fungus was isolated from the brown alga Dictyota dichotoma.
Isolation and identification of the endophytic fungus
Isolation
The algal sample was washed with sterile distilled water and surface sterilized by immersing in 70 per cent ethanol for 5 seconds. The algal sample was dried on sterile paper towels, cut into bits and inoculated on 2 per cent malt extract agar aseptically (Andrew J Flerwelling et al.2015). The plates were incubated at 250C and monitored regularly for the growth of the endophytic fungi.
Sub culturing of the fungus
The isolated endophytic fungus was inoculated into the conventional media used for fungal growth viz .potato dextrose agar, GPY(glucose peptone yeast extract) agar and malt extract agar. But the fungus was unable to grow hence a new medium named MGM medium was formulated and the fungus was successfully grown on it.
Identification of the fungus
The isolated endophytic fungus which was subcultured and obtained as a pure culture was used for identification by molecular characterization through sequence analysis of D2 region of LSU (Large Sub Unit: 28S r DNA).
Molecular characterization
The DNA was isolated from the culture filtrate of endophytic fungus and it was amplified by AB Thermoanalyzer using BDV31 cycle sequencing kit by utilizing forward and reverse primers of 28s rDNA. The PCR amplicon was purified and processed for further sequencing. Consensus sequence of D2 region of LSU gene was generated from forward and reverse sequence data using aligner software.
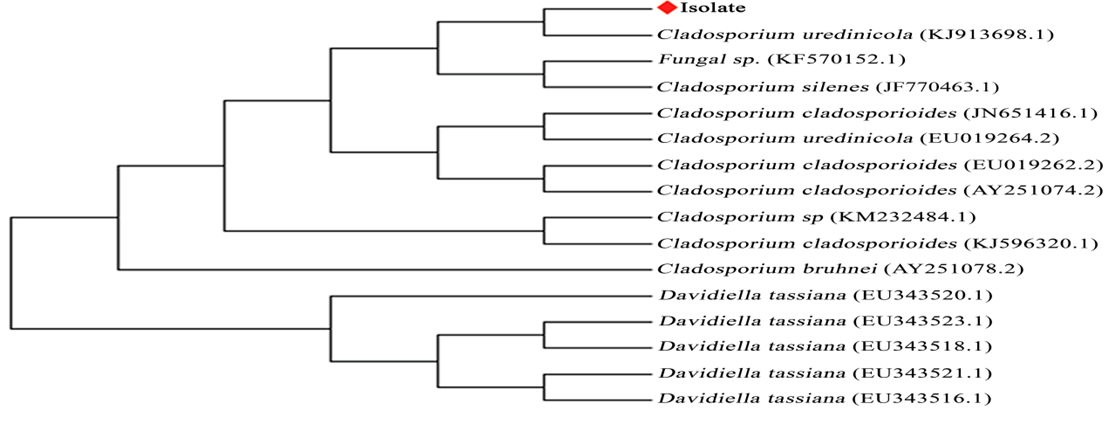
The D2 region of 28S r DNA sequence was subjected to homology search analysis. Based on maximum identity score, first fifteen sequences were selected and aligned using multiple alignment software program clustal W. Distance matrix was generated using RDP data base and the phylogenetic tree was constructed using MEGA 5 (Tamura et al.2011).
Monitoring different stages in the life cycle of the endophytic fungus
Transverse section of the brown alga
The algal sample was surface sterilized with 70 per cent ethanol and a thin transverse section was made with the help of a stainless steel blade. The section cutting was transferred to a clean grease-free glass slide. Coverslip was placed and observed under 10X of phase contrast microscope.
Lactophenol cotton blue staining of the transverse section
In order to ascertain the presence of fungus in the algal sample, Lactophenol cotton blue staining was carried out. The algal sample was placed in sterile marine water for 30 days and observed for the growth of fungal hyphae. Thin transverse section cuttings were made from the sample and stained with Lactophenol cotton blue. Cover slip was placed and observed under 10X objective of phase contrast microscope.
Differential staining of the transverse section
In order to differentiate the fungal hyphae from host tissue a differential staining method was developed using Lactophenol cotton blue and saffranin stain. The algal section cutting was stained with Lactophenol cotton blue and after one minute the stain was washed with ethanol. Saffranin was used as a counter stain. Cover slip was placed and observed under phase contrast microscope.
Staining of the isolated fungus from algal bit and subcultured fungus
In order to confirm that the fungus isolated from the algal bit and that was obtained by subculture are same or not ,Lactophenol cottonblue staining was performed for both the samples.
Observation of different forms of the endophytic fungus
The isolated fungus Cladosporium uredinicola belongs to Ascomycota division which are known for their ability to exist in different forms. The isolated fungus forms a very thick mat on the medium which makes the staining and microscopic examination of the fungus very difficult. In order to overcome this problem a new method was developed to facilitate the clear observation of different forms of the fungus. The fungus was grown in the newly formulated MGM broth medium (Umamaheswari devi and Vasavi.2016, Indian patent published-application number 201641020940) and grown for 28 days .The thick mat was removed from the medium with the help of a forceps and the broth was filtered using the Whatman no.1 filter paper. The debris left on the filter paper was scrapped carefully using a sterile lancet and suspended in Lactophenol cotton blue solution on a glass slide. Cover slip was placed and observed under microscope.
Isolation
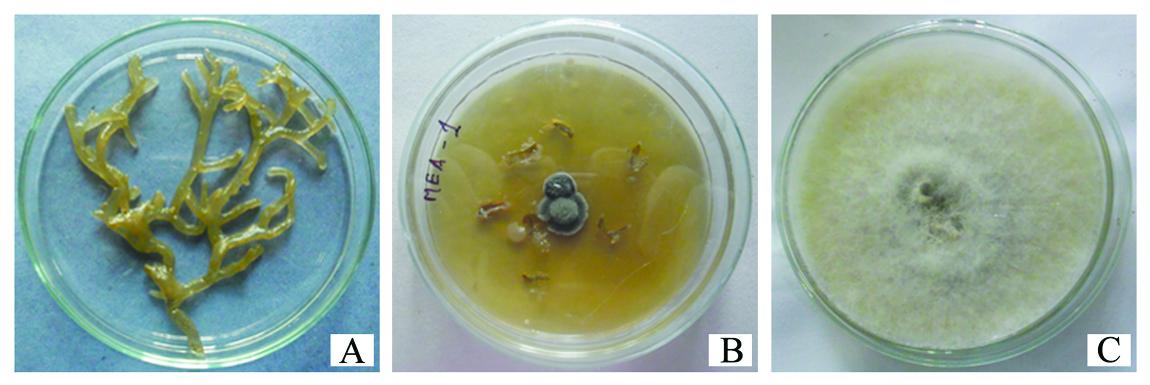
An endophytic fungus was observed from the cut edges of the Dictyota dichotoma bit inoculated in the 2% malt extract agar plates on the 12th day of incubation.Along with the endophytic fungus a bacterial species was also isolated (Figure 1).
Fig.1. (A) Marine brown alga Dictyota dichotoma (B) isolated endophytic fungus and bacte-ria (C)Subcultured fungus
Subculturing of the isolated endophytic fungus
Upon subculturing the isolated fungus for three consecutive times it was observed that the isolate showed same morphological and growth characteristic pattern measured through dry weights .
Molecular characterization
The isolated endophytic fungus was identified as cladosporium uredinicola by 28 s rDNA sequence analysis. The sequence was deposited at the Genbank under the accession number MG719993. Cladosporium uredinicola belongs to Davidiellaceae family and Ascomycota division of Kingdom Fungi (Figure 2).
Observation of the transeverse section of the alga
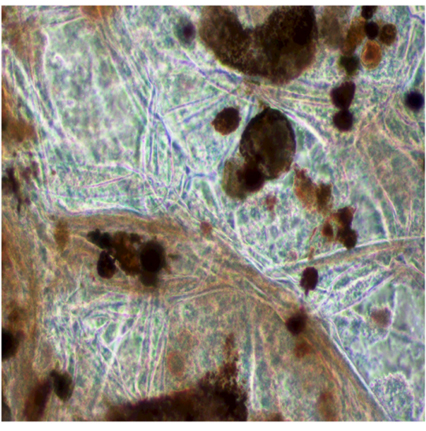
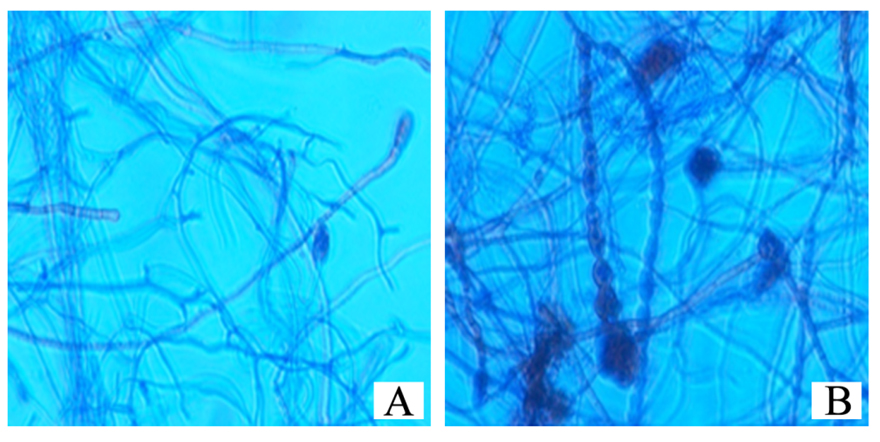
When the transverse section of the brown algal sample was observed without staining under the phase contrast microscope, fungal hyphae were observed around the fucosan granules of the alga (Figure 3). This observation indicates the dependence of fungal endophyte for nutrition on its host.
Lactophenol cotton blue staining of the section cutting of the alga
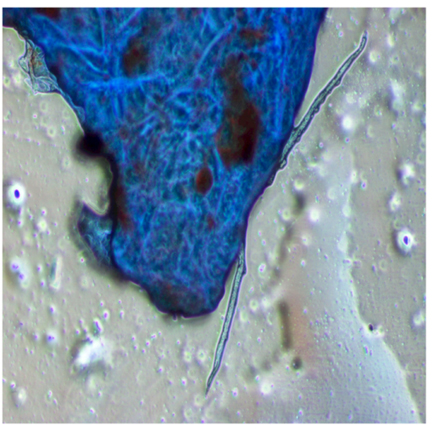
In order to observe the growth of the endophytic fungus ,the cuttings of the algal sample were stained with Lactophenol cotton blue and observed under phase contrast microscope.The fungal hyphae were observed to be protruded from the cut edges of the brown alga (Figure 4).
Differential staining
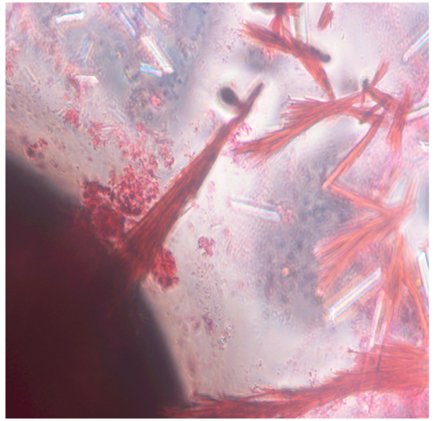
Differential staining technique was employed, to distinguish the fungal endophyte from algal tissue.. The fungal hyphae were found to be in dark blue colour on the back ground of the saffranin stained algal tissue. The spore structures of the fungus appeared darker in colour when compared to the fucosan granules of the host (Figure 5). The micrographs also revealed the transfer of fungal spores from one generation of the host alga to the next through the zygote (Figure 6).
Staining of the isolated fungus from algal bit and subcultured fungus
From the results of the Lactophenol cotton blue stainig of the fungus and its subculture it was observed that both of them have the similar morphological characteristics
Observation of different forms of Cladosporium uredinicola
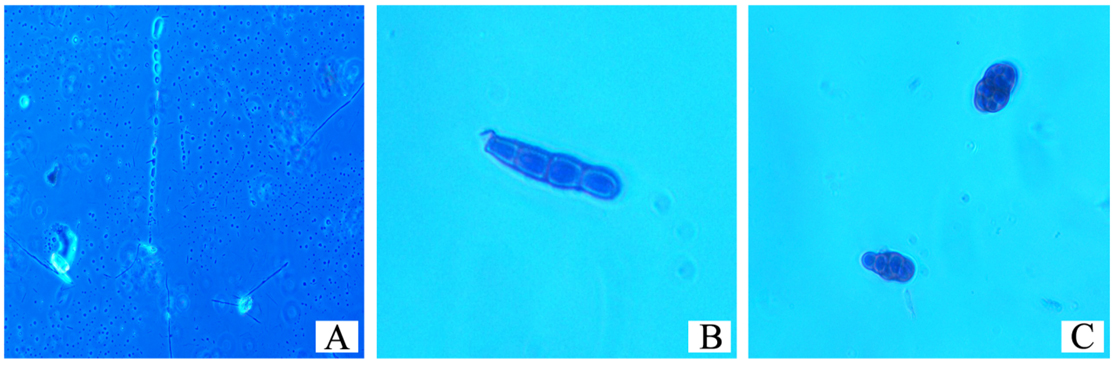
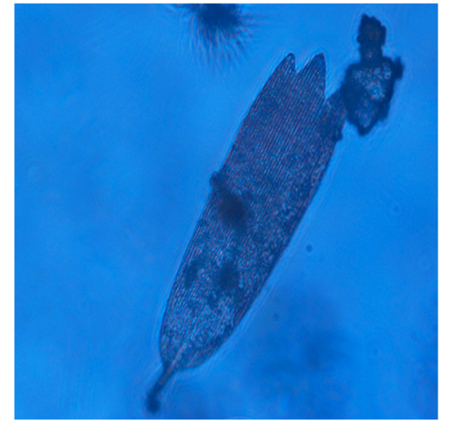
From the microscopic observation of the Lactophenol cotton blue stained scrapings from the Whatman filter paper, different forms of the isolated cladosporium uredinicola were observed. A highly woven thick mycelium without spores was observed initially,followed by the appearance of Asci(Figure 7).Different types of spores namely conidiospores, and spore masses with Four celled and 8celled ascospores were also observed (Figure 8).Coronate ascocarp was also observed clearly under 40 X of the phase contrast microscope(Figure 9).
Marine environment is a rich source of biodiversity due to its extreme conditions of temperature, pressure and salinity (Debbab et al.2011). In such a harsh environment organisms evolve to combat the different unfavourable conditions (Johnson et al.1997).Endophytism is such an adaptation which is beneficial for both host and endophyte(Doliska et al.2011).In the present work an endophytic relationship between the fungus Cladosporium uredinicola and marine brown alga Dictyota dichotoma was studied. By using improvised staining techniques different stages of the endophytic life cycle were observed.
Cladosporium spp. of fungi are worldwide in distribution and they live as decomposers (Debbab et al.2011).Cladosporium uredinicola lives as a hyper parasite on rust fungi Dugan and Glawe 2006 and ,Barrows et al.1999) .Reports are available to support the occurrence of Cladosporium uredinicola as an endophyte from the terrestrial plants(Thakur et al.2013).In the present work the endophytic fungus isolated from the marine brown alga Dictyota dichotoma was identified as Cladosporium uredinicola by molecular characterization .Microscopic examination using simple and improvised techniques revealed different forms of the fungus and stages of its association with the host.
As far as our knowledge is concerned the present work is the first report of the occurrence of Cladosporium uredinicola as endophyte in Dictyota dichotoma. The results of the present work conclude that the modified growth medium and the simple staining techniques shall be useful to study the different forms of Ascomycetes fungi and their endophytic life style pattern.
ACKNOWLEDGMENTS
The authors are grateful to DST-CURIE (Sri Padmavathi Mahila Visvavidyalayam,Tirupati) for their support .
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
- Andrew J Flerwelling, Jessica Currie, Christopher A Gray , John A Johnson. Endophytes from marine macroalgae: promising sources of novel natural products. Current Science. 2015; 109(1):7-18
- Barros S T, Oliveira N, SidneyT, Bastos T G,Mala L C. Hyperparasitism of cladosporium uredinicola over Puccinia puta on the host Ipomea fistulosa. Plant Dis. 1999; 13(1):23-24.
- Cabral D, Stone JK, Carroll GC. The internal mycobiota of Juncus pp.microscopic and cultural observations of infection patterns. Mycological Research. 1993; 97: 367-376.
- Debbab, A, Aly A H, Proksch, P. Bioactive secondary metabolites from endophytes and associated marine derived fungi . Fungal Divers. 2011; 49: 1–12.
- Doliska T M, Bartkowska A, Schollenberger M. Light and scanning microscope observations of cladosporium uredinicola growth on rust fungi. Phytopathologia. 2011; 61:37-44
- Dugan F M, Glawe D A. Phyllactinia gutatta is host for cladosporium uredinicola in Washington state. Pac.Norwest Fungi, 2006; 1(1):1-5.
- Geisen S, Kostenko O, Cnossen M C, ten Hooven F C, Vreš B and van der Putten WH. Seed and root endophytic fungi in a range expanding and a related plant species. Front. Microbiol. 2017; 8: 1645.
- Ghimire S R, Hyde K D. Fungal endophytes. In:Varma A.,Abbott L.,Werner D.,Hampp R.(eds) Plant Surface Microbiology. Springer, Berlin, Heidelberg 2008.
- Johnson NC, Graham JH, Smith FA. Functioning of mycorrhizal associations along the mutualism-parasitism continuum. New phytologist. 1997; 135: 575-585
- Owen N L, Hundley N. Endophytes –the chemical synthesizers inside plants. Science Progress. 2004; 87:79-99.
- Schulz B, Rommert AK, Dammann U,Aust H J,Strack D. The endophyte –host interaction: a balanced antagonism ? Mycological Research. 1999; 103: 1275-1283.
- Selim KA, El-Beih AA, AbdEl-Rahman TM, El-Diwany AI. Biology of endophytic fungi. Current Research in Environmental & Applied Mycology. 2012; 2(1):31-82.
- Suryanarayanan TS. Endophyte research: going beyond isolation and metabolite documentation. Fungal ecology. 2013; 6: 561-568.
- Tamura K., Peterson D., Peterson N., Stecher G., Nei M., and Kumar S. MEGA5: Molecular Evolutionary Genetics Analysis using Maximum Likelihood, Evolutionary Distance, and maximum parsimony methods. Molecular biology and evolution. 2011; 28:2731-2739
- Thakur A, Singh V, Kaur A, Kaur S. Insecticidal potential of an endophytic fungus cladosporium uredinicola against spodoptera litura Phytoparasitica 2013; 41(4): 373-382.
- Umamaheswari devi P and Vasavi T. ‘MGM medium’. Indian patent application number 201641020940( http://ipindiaservices.gov.in)
© The Author(s) 2018. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.