ISSN: 0973-7510
E-ISSN: 2581-690X
Radiological investigations are essential for the diagnosis and classification of fungal rhinosinusitis; however, radiological findings might occasionally be misleading. Computed tomography (CT) scan and magnetic resonance imaging (MRI) complement each other, facilitating clinicians to arrive at a diagnosis. Hence, even with the best radiological modalities, correlation between clinical and microbiological findings is crucial for the accurate diagnosis of fungal rhinosinusitis. In addition, the role of traditional methods such as KOH microscopy and culture should be accurately evaluated. To this end, we aimed to diagnose rhinosinusitis with a fungal etiology based on radiological findings and subsequently correlate these findings with those of microbiological techniques, namely culture and KOH microscopy. A total of 57 clinically suspected fungal rhinosinusitis cases were included in the study. Radiological investigations were performed using either CT or MRI. Tissues samples were processed and analyzed using KOH microscopy and culture. The results of the study suggest that using a single method for the diagnosis of fungal rhinosinusitis is inadequate. Rather, the diagnosis should be based on radiological as well as microbiological findings, especially for cases that are clinically ambiguous.
Rhinosinusitis, Mucormycosis, Computed tomography, Magnetic resonance imaging, , Microscopy, Culture
Radiological investigations have become a vital tool in the diagnosis and classification of fungal rhinosinusitis. However, at times, their findings can be misleading; the presentation in allergic fungal rhinosinusitis is very specific, while in invasive fungal rhinosinusitis, it may range from vague sinusitis to an apparently normal scan. Computed tomography (CT) scanning and magnetic resonance imaging (MRI) are adjunct to each other. Thus, even with the best radiological modalities, clinical and microbiological parallelism is necessary to accurately diagnose this ambiguous condition known as fungal rhinosinusitis. In addition, the role of commonly used methods such as culture and KOH microscopy needs to be examined. Rhinosinusitis is an inflammatory disorder of the nasal cavity and paranasal sinuses that affects 20% of the human population. Fungal settlement in the nose and paranasal sinuses occurs in diseased and healthy individuals. In recent years, fungal rhinosinusitis has increased in prevalence, especially in tropical countries like India; this increase may be attributed to advances in diagnostic imaging and other adjunct laboratory investigations.1 CT helps to differentiate fungal rhinosinusitis from nonfungal rhinosinusitis based on specific features. In 1885, Schubert discovered a non-invasive Aspergillus species and diagnosed the first particular variant of fungal rhinosinusitis. In 1897, Oppe first described invasive aspergillosis, in a case where the fungus had extended into the cranium through the sphenoid sinus. Fungal rhinosinusitis can be classified as either non-invasive (allergic fungal rhinosinusitis and fungal ball) or invasive (acute invasive, chronic invasive, and granulomatous invasive fungal sinusitis).2 In this study, we analyzed fungal rhinosinusitis on the basis of radiological diagnosis and then correlated the results with those of microbiological techniques, including culture and KOH microscopy.
Type of Study
An observational, cross-sectional study was conducted at the Microbiology Department, in collaboration with the departments of Radiodiagnosis and ENT, at Sri Guru Ram Das University of Medical Sciences and Research, Sri Amritsar, India, over a period of 6 months (February 2021–July 2021). A total of 57 clinically doubtful fungal rhinosinusitis cases were included in the study on the basis of the following inclusion and exclusion criteria.
Inclusion Criteria
Patients fulfilling at least two major criteria or one major criterion and two minor criteria were included in the study, as described previously by Lanza and Kennedy.3 Major criteria included obstruction of the nasal pathway, facial pain/fullness, hyposmia/anosmia and fever, and postnasal discharge. Minor criteria included headache, fatigue, halitosis, dental pain, ear pain/fullness, and cough.
Exclusion Criteria
Patients with conditions such as congenital mucociliary disorder, chronic sinusitis, chronic granulomatous disease, or atrophic rhinitis were excluded from the study. Patients with any malignancy of the paranasal sinuses, who were unwilling to participate in the study, or who were lost to follow-up were also excluded from the study.
After obtaining written informed consent from the patients, patient information, including age, sex, clinical presentation, and immune status was recorded. Of the 57 clinically suspected cases, 39 underwent CT scans of the paranasal sinuses and orbit, while 18 underwent MRI.
CT Protocol and Findings
The following features observed on CT scan help in the diagnosis of fungal rhinosinusitis: hyper-attenuation, air-fluid levels, bony erosions, neo-osteogenesis, and extra-sinus association. CT is superior to MRI for assessing bone changes. However, MRI is better than CT for the evaluation of intracranial and intraorbital extensions in fungal rhinosinusitis. The invasion of the periantral soft tissues into the maxillary sinus is indicative of an invasive disease.
Microbiological Findings
Following radiological assessment, sinonasal tissue biopsy samples were collected for microbiological correlation. During the study period of 6 months, samples were cumulated from February to July 2021. The samples were collected in vials containing normal saline and were processed in a microbiology lab, specifically a mycology lab for KOH microscopy and culture.
For microscopic examination, the tissue samples were examined in 20% KOH. Sabouraud dextrose agar (SDA) with chloramphenicol was used for the culture, and the cultures were incubated at 25°C and 37°C, respectively, for 28 days. The results of the fungal microbiological examination were correlated with the radiological findings. Hence, a radiological–mycological approach was used to arrive at the final diagnosis and categorization of fungal rhinosinusitis.
Ethical Approval
This study was approved by the Institutional Ethical Committee of Sri Guru Ram Das University of Health and Sciences, Sri Amritsar, Punjab, India.
Statistical Analysis
Statistical analysis of the data was conducted using the SPSS software. Percentages and proportions were used to express the qualitative data sets. Quantitative data were expressed as mean and standard deviation. Associations were measured using the chi-square test.
Analysis of the collected data was as follows:
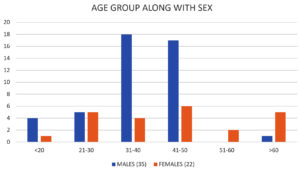
In this study, the age of the patients with fungal rhinosinusitis ranged from 15 to 68 years, while the majority of patients (39%) belonged to the 41–50 year age group, and the male to female ratio was 1.69:1 (Fig. 1). Majority of the patients presented with symptoms of obstruction of the nasal airway and headache, followed by nasal discharge, facial pain, and fullness. Of the 57 patients, 45 (79%) were immunocompromised due to diabetes. The COVID-19 RT-PCR test was positive in 25 out of 57 cases (44%).
Fig. 1. Figure showing variations in the age group and sex in the patients of fungal rhinosinusitis.
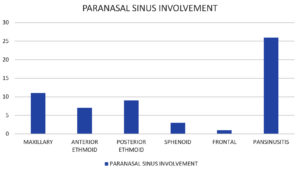
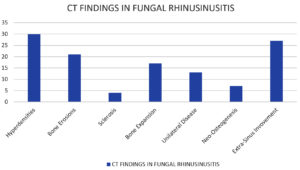
Pansinusitis was the most common CT finding, followed by involvement of the maxillary sinus and the posterior ethmoid sinus (Fig. 2). The CT findings evaluated are presented in Fig. 3. In addition, various MRI findings were evaluated.
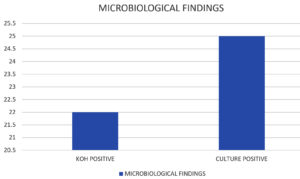
The KOH mount positivity rate in this study was 39% (22 out of 57). Of the 57 sinonasal specimens, 25 (44%) were culture-positive for fungi. The distribution of fungi isolated from clinically and radiologically diagnosed fungal rhinosinusitis patients is shown in Fig. 4.
Fig. 4. Figure showing results of microbiological evaluated data in the form of KOH positivity and Culture positivity.
In total, 22 of the 25 fungal pathogens were hyaline hyphomycetes belonging to Aspergillus species (88%), of which 15 (68%) and 7 (32%) isolates were identified as A. flavus and A. fumigatus, respectively. The remaining fungi were phaeoid and yeast-like fungi, accounting for 9% and 3%, respectively.
Rhinosinusitis is an inflammation of the paranasal sinus and nasal mucosa. It is a common disease affecting approximately 20% of the population and can at times interfere with day-to-day life.4 It was earlier considered rare but is now reported with increasing frequency not only in the Indian subcontinent but also worldwide. This disorder is being increasingly described in northern regions. Emerging evidence demonstrates that fungi play an important role in the exacerbation and spread of mucosal inflammation. In a large number of patients diagnosed with rhinosinusitis, fungi were the main etiological agents. Studies have reported 9%–38% prevalence of sinonasal mycotic infections.5
Accurate diagnosis of fungal rhinosinusitis is necessary to administer the appropriate treatment and to prevent careless use of antibiotics and unnecessary repeated surgical interventions. Clinical scrutiny should not be the final unquestionable modality in diagnosing fungal rhinosinusitis. Rather, based on clinical suspicion, the diagnosis should be confirmed by radiological and microbiological examination. Few studies have been conducted in this regard. Thus, the present study was conducted to correlate clinical presentation with radiological and microbiological findings in fungal rhinosinusitis.
This study included 57 patients with clinically suspected rhinosinusitis who satisfied both the inclusion and exclusion criteria and presented to the Radiodiagnosis department of Sri Guru Ram Das University of Medical Sciences and Research for cross-sectional imaging between February and July 2021. Clinical and radiological characteristics along with microbiological results were analyzed. The study aimed to discover both radiological and microbiological perspectives of fungal rhinosinusitis.
In this study, 41–50 years was the most common age group affected by fungal rhinosinusitis; coincidently, Michael et al. reported 45 years as the mean age in their study.6
Among the clinical symptoms, obstruction of the nasal pathway was the most common presentation seen in all the cases (100%), followed by headache (82%), discharge from the nasal cavity (58%), facial pain (52%), disturbances of olfaction (48%), halitosis (31%), bleeding from the nose (30%), and fever (15%). Similar results were also observed by Chakrabarti et al.7 Majority of the patients (79%) had diabetes and were, hence, immunocompromised.
From the beginning of the COVID-19 pandemic, opportunistic infections have emerged as another disease spectrum. Among these opportunistic infections, fungal rhinosinusitis has become a matter of concern owing to a rapid increase in the number of cases, similar to the pre-COVID-19 era. Cases reported in post-COVID-19 patients can be attributed to their immunocompromised state. Additionally, pre-existing comorbidities, namely diabetes, contribute to worse prognosis.8
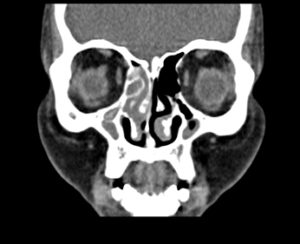
Fig. 5. CT Coronal image showing polypoidal mucosal thickening in bilateral maxillary sinus, right frontal, ethmoid and sphenoid sinuses, extending into the nasal cavity on right side. A high attenuation hyperdense area is also noted within the mucosal thickening which is associated with absorption of the ethmoid septum.
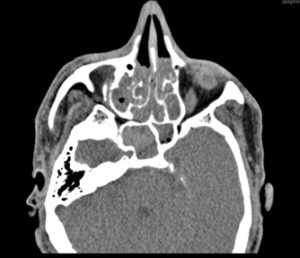
In previous studies, CT findings of patients revealed intra-sinus high attenuation density areas, sinus expansion (complete opacification), bone erosion of the elaborated sinus, transformation with thinned out walls of the sinuses, bony erosions of the walls of the paranasal sinuses, and extension of the disease into the adjacent sinuses. A 28% incidence of bone erosion with extension into the brain and orbit has been reported.9 In the present study, sinus expansion and bone erosion were observed in 17 (30%) and 21 (37%) cases, respectively (Fig. 5). Heterogeneous “high-attenuation” areas within sinuses, reflecting allergic mucin, on CT scans are relatively characteristic for fungal rhinosinusitis.10 This observation is consistent with our findings: 30 cases (54%) had hyperattenuating areas in the involved sinuses. These findings should make the clinician vigilant to the opportunity of fungal rhinosinusitis and timely make other diagnostic studies to clinch the diagnosis.11 The incidence of bone sclerosis was 7% (4 patients) in our study (Fig. 6). Taking into account the laterality of the disease, 13 (22%) patients had unilateral fungal rhinosinusitis, while 27 (48%) patients had extra sinus involvement. Additionally, neo-osteogenesis was observed in 27 (48%) patients. The results of our study correlate well with the findings of Das et al., who reported 42.7% fungal rhinosinusitis in 665 cases of chronic rhinosinusitis (CRS) over a period of five years. In another five-year study from South India, out of 138 CRS biopsies, 63 (45.7%) were confirmed to be positive for fungal rhinosinusitis.
Fig. 6. CT Coronal image showing marked mucosal hypertrophy in bilateral maxillary, ethmoid, frontal and sphenoid sinuses with hyperdense contents seen within them. There is widening of accessory ostia of maxillary sinuses with associated rarefaction of the ethmoid trabeculae. Mild thinning of walls of bilateral maxillary sinuses is also noted.
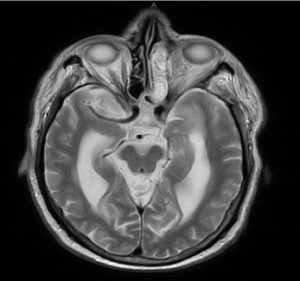
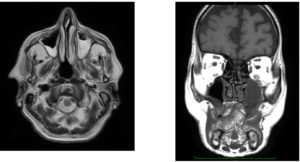
The MRI features of fungal rhinosinusitis involve an extensive spectrum of imaging findings, including a range from hypo-intensities of the sinus fillings on T1-weighted images to high signal intensity and mixed to low, intermediate, and high signal intensity on T2-weighted images in these patients. In addition, there was a low signal intensity or signal void on T2-weighted images (Fig. 7).12 The presence of this T2 signal void can be attributed to the high concentration of metal ions, such as iron, magnesium, and manganese, used by the fungi, as well as high protein content. Furthermore, T2 signal voids may be the result of an extremely low amount of free water present in allergic mucin. The paranasal sinuses have an inflamed mucosal lining, which is usually hypoattenuating on non-contrast CT and hyperintense on T2-weighted images with contrast enhancement. There was no enhancement in the central sinus contents. Sclerosis and thickening of the bony walls of the sinuses were observed. In addition, expansion and thinning of the walls with focal areas of erosion due to pressure necrosis were observed.13 The fungus ball, owing to its low free water content, displays a hypointense signal on T1- and T2-weighted images (Fig. 8A, 8B). Although this fungal ball is a rare finding seen in the study. Signal voids on T2-weighted images may also result because of the presence of paramagnetic metals, such as iron, magnesium, and manganese, and calcifications.
Fig. 7. T2W Image showing Sinonasal thickening with soft tissue proliferation of the left air cells of ethmoid sinus with irregularity along lamina papyracea with significant inflammatory changes.
Fig. 8A, 8B. Axial T1W and Coronal T1W images showing abnormal thickening involving the mucosa of the sinuses with the left maxillary sinus showing collection together with frontal sinuses, which is predominantly low signal intensity on T1 images and heterogeneously high signal intensity on T2W images.
The prognosis and treatment of fungal rhinosinusitis differs depending on the causative fungal species. Therefore, accurate diagnosis is necessary, and microbiological investigation plays a vital role in the process. While clinical presentation of the disease provides a basis for diagnosis, accurate diagnosis cannot be made without radiological and microbiological findings.14 Supplementary modalities can be used either for verification or when the fungus has been identified, but the patient is not responding to the antimycotic agents.
Of these methods, culture was found to be quite sensitive, followed by KOH microscopy. In our study, strict measures were taken to prevent laboratory contamination. Culture positivity was higher than KOH positivity (Fig. 9, 10); this result may be attributed to sample collection errors or inter-observer variability; for instance, the sample may not have been taken from the inflamed site but from neighboring areas showing inflammation.
Many studies have also shown that the use of mucolytic agents before inoculation increases the culture yield by 96%.15 Therefore, proper processing of tissue samples is very important to reduce the aforementioned issues.
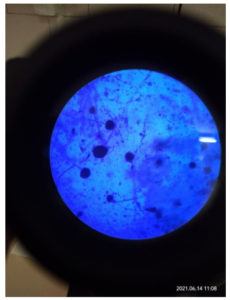
In contrast to the immunity of the host, infections may exist as colonization without causing any harm or may entail noninvasive or invasive forms. Thus, there is an enhancement of the diagnosis made by the radiologists due to the acquaintance and affinity of the characteristic imaging patterns seen in fungal rhinosinusitis (Fig. 11). Microbiological tests facilitate the identification of etiological agents, thereby helping physicians with regards to further evaluation and providing guidance for initiating the appropriate treatment at the right time to prevent long-term effects.
The present observational study was conducted during the COVID-19 pandemic to study the microbiological and radiological perspective of fungal rhinosinusitis in a tertiary care centre in the northern region of India. In this study, culture was found to be more sensitive in detecting fungal rhinosinusitis than KOH microscopy, with 44% and 39% positive cases, respectively. The most common fungi identified through culture were hyaline hyphomycetes belonging to the Aspergillus species. However, some cases were positive for phaeoid and yeast-like fungi. Radiological findings also contributed to the diagnosis of the disease. Of the fungal rhinosinusitis cases, 44% tested positive for coronavirus, and a large proportion of the patients had diabetes. These results suggest that the body’s immunity plays a vital role in the pathogenesis of the fungal entity affecting the nose and paranasal sinuses.
ACKNOWLEDGMENTS
We would like to thank the Department of Microbiology, Radiodiagnosis and ENT of Sri Guru Ram Das University of Medical Sciences for facilitating laboratory and radiological investigations throughout the study.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a direct, substantial and intellectual contribution to the work, and approved it for publication.
FUNDING
None.
ETHICS STATEMENT
The study was approved by the institutional Ethics committee, Sri Guru Ram Das Institute of Medical Sciences and Research, Sri Amritsar, Punjab, India.
AVAILABILITY OF DATA
All datasets generated or analyzed during this study are included in the manuscript.
INFORMED CONSENT
Written informed consent was taken from all individual participants of the study.
- Krishnan KU, Agatha D, Selvi R. Fungal rhinosinusitis: A clinicomycological Perspective. Indian J Med Microbiol. 2015;33(1):120-124.
Crossref - Navya BN, Vivek TG, Sudhir, Kariappa TM, Shwetha VP, Ahalya R. Role of Histopathology in the Diagnosis of Paranasal Fungal Sinusitis. Journal of Dental and Medical Sciences. 2015;14(1):97-101.
- Lanza D, Kennedy DW. Adult rhinosinusitis defined. Otolaryngol Head Neck Surg. 1997;117(3 Pt 2):S1-7.
Crossref - Chatterjee SS, Chakrabarti A. Epidemiology and medical mycology of fungal rhinosinusitis. Otorhinolaryngol Clin Int J. 2009;1(1):1-13.
Crossref - Karthikeyan PV. Nirmal coumare. Incidence and presentation of Fungal Sinusitis in Patient Diagnosed with chronic Rhinosinusitis. Indian J Otolaryngol Head Neck Surg. 2010;62(4):381-385.
Crossref - Michael RC, Michael JS, Ashbee RH, Mathews MS. Mycological profile of fungal sinusitis: An audit of specimens over a 7-year period in a tertiary care hospital in Tamil Nadu. Indian J Pathol Microbiol. 2008;51(4):493-496.
Crossref - Chaterjee SS, Chakrabarti A. Epidemiology and mycology of fungal rhinosinusitis. Otorhinolaryngology Clinics. 2009;1(1):1-13.
Crossref - Bhattacharyya A, Sarma P, Sharma DJ, et al. Rhino-orbital-cerebral-mucormycosis in COVID-19: A systematic review. Indian J Pharmacol. 2021;53(4):317-327.
Crossref - Mukherji SK, Figueroa RE, Ginsberg LE, et al. Allergic fungal sinusitis: CT findings. Radiology. 1998;207(2):417-422.
Crossref - Dhiwakar M, Thakar A, Bahadur S et al. Preopeerative diagnosis of allergic fungal sinusitis. Larynggoscope. 2003;113(4):688-694.
Crossref - Middlebrooks EH, Frost CJ, De Jesus RO, Massini TC, Schmalfuss IM, Mancuso AA. Acute invasive fungal rhinosinusitis: A comprehensive update of CT findings and design of an effective diagnostic imaging model. AJNR Am J Neuroradiol. 2015;36:1529-1535.
Crossref - Manning SC, Merkel M, Kriesel K, Vuitch F, Marple B. Computed tomography and magnetic resonance diagnosis of allergic fungal sinusitis. Laryngoscope. 1997;107(2):170-176.
Crossref - Mossa-Basha M, Ilica AT, Maluf F, Karakoc O, Izbudak I, Aygun N. The many faces of fungal disease of the paranasal sinuses: CT and MRI findings. Diagn Interv Radiol. 2013;19(3):195-200.
Crossref - Kimura M. [Histopathological Diagnosis of Fungal Sinusitis and Variety of its Etiologic Fungus]. Med Mycol J. 2017;58(4):127-132.
Crossref - Ickrath P, Sprugel L, Beyersdorf N, Scherzad A, Hagen R, Hackenberg S. Detection of Candida albicans-Specific CD4+ and CD8+ T Cells in the Blood and Nasal Mucosa of Patients with Chronic Rhinosinusitis. J Fungi (Basel). 2021;7(6):403.
Crossref
© The Author(s) 2021. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.