Recently, the attention to exploring and applying probiotics has been increased. Probiotics are living microscopic organisms that have valuable impacts on health when consumed in adequate amounts. One of the most popular widespread utilized probiotics for many years until now is the Lactobacillus species which exemplifies the hugest heterogeneous group among the lactic acid bacteria (LAB) that normally occupy many environments that contain high nutrient levels such as soil, humans, waters, plants, animals and even air without causing any illness. The purpose of this work is to review and demonstrate the wide diversity of lactobacilli isolation sources and related health benefits applications of the strains discovered in order to break the familiar sources of lactobacillus spp., thereby encouraging more research into unconventional sources and opening up opportunities for clinical applications of Lactobacillus spp. As a result, the isolation sources of lactobacillus spp. in this review have been split into two groups based on the environmental condition, including fermented and non-fermented habitats described as lactobacilli probiotic sources. The unique Lactobacilli isolated from various sources are regarded as potential therapies for a variety of illnesses and physiological abnormalities, including immunomodulation, suppression of carcinogenesis, and cholesterol reduction, in addition to their capacity to guard against infections. This will be highly significant for revealing updated information about Lactobacillus spp. that has attracted considerable attention due to its potential application in clinical practices.
Lactobacillus spp., LAB, Probiotics, Health Benefits, Isolation Sources
Probiotics
The probiotic term refers to nonpathogenic living microorganisms that are intended to have beneficial health to humans and animals when consumed in adequate volumes.1,2 The term was created from the Greek words Pro (favor) and bios (life).3
Probiotics have earned enormous attention due to their valuable impacts on health. Furthermore, probiotics can be considered a healthy alternative therapeutic method to minimize drug resistance due to the excessive use of antibiotics to fight infections in people and animals. They can be helpful for the host by enhancing the intestinal microbial balance in the gastrointestinal tract, strengthening the epithelial barrier, and improving mucosal immunity.4,5 In addition, boost-up the immune system and its own antagonistic activity against many pathogens by producing antimicrobial compounds such as bacteriocins.6,7 Probiotics can prevent and heal many health issues and diseases including decreasing liver lipids concentrations,8 common cold, Fever blisters, Lyme disease, improving the general digestion problems,9 constipation,10 cancers, cardiovascular diseases, diabetes,11 allergic diseases,12 Helicobacter pylori infection,13 and even relief of anxiety and depression.14 According to the statement of a conducted study related to antibiotics, probiotics particularly the ones that belong to the genus lactobacillus found powerful enough to kill the HIV-1 virus when given at a high dosage. Moreover, Ligilactobacillus salivarius had the ability to inhibit the formation of carcinogenic biofilms of Streptococcus mutans and Candida albicans reducing its pathogenicity.15
Probiotics contain several genera and species of bacteria, yeast, and molds such as Lactobacillus, Bifidobacterium, Candida pintolopesii, Streptococcus, Aspergillus niger, Leuconostoc, Aspergillus oryzae, Propionibacterium, Bacillus, Pediococcus, Saccharomyces cerevisiae, and Enterococcus.16 Among the above-mentioned, the widely used genera belong to bacteria and particularly to the lactic acid bacteria (LAB) mainly represented by Bifidobacterium and Lactobacillus.17 The LAB that belongs to the genus of Lactobacillus has been one of the most popular and widely used probiotics for many years until now which exemplifies the hugest heterogeneous group among LAB.18
Lactobacilli are members of LAB, a diversified group of catalase-negative, Gram-positive, non-spore-forming and anaerobic or aero tolerant, coccobacilli, or rod-shaped that is mainly characterized by the lactic acid production as the main end product of their carbohydrate fermentation.19
Lactobacilli occupy many nutrient-rich habitats and environments such as spoiled and fermented food and are found in animal feed, soil, and body of invertebrate and vertebrate animals.20 They live naturally in our body by inhabiting the digestive tract, oral cavity, genital and urinary system.21 Members of the Lactobacilli group have been generally renowned as safe (GRAS) by Food and Drug Administration (FDA) due to their frequent abundance in our food like cheese and yogurt without causing illnesses.22
Lactobacillus genus characterized by a massive genome possesses a high grade of genetic diversity and physiology which make them a targeted candidate in many important applications, such as the production of fermented food, food bio preservation strategies to prolong the shelf life while guaranteeing their safety, and in health improvement applications.23,24
The reason behind the focus on Lactobacillus is due to their vast impact on the host immune system, considered safe in use, show a competitive exclusion, and own a high adhering ability.25 This work is aimed to review the wide variety of lactobacilli isolation sources and associated health benefits applications of the strains discovered to break the acquainted sources of lactobacillus spp., thus also inspiring more research to isolate new strains from unconventional sources and providing possibilities for more clinical applications.
Taxonomy of Lactobacilli
Lactobacilli were one of the earliest classified bacteria due to their ubiquitous existence in many fermented products. Numerous classifications have been improved since the first description of the Lactobacillus genus by Beijerink in 1901.26
The base of the initial taxonomy of lactobacilli was depend on the observable and measurable traits including cell morphology, gram stain, colony appearance, optimum growth temperature, pH tolerance, oxygen requirements, and pathways for carbohydrate fermentation which classified lactobacilli according to the type of fermented sugars and fermentation products into three general groups; obligate homofermentative, facultative heterofermentative and obligate heterofermentative.27,28
In the 1980s, with the entrance of bacterial taxonomy the genetic age, genotypic and chemotaxonomic characteristics were used in classification. The availability of 16S rRNA sequencing allowed the first phylogenetic analysis of the Lactobacillus which uncovered an extensive diversity by the generation of a true phylogenetic tree that contributed to the development of the taxonomy of lactobacilli.27,28 After the evolution of molecular techniques, most species have been named again and some reclassified to other genera.
Based on NCBI taxonomy, the current taxonomic hierarchy for Lactobacillus is Domain: Bacteria, Phylum: Firmicutes, Class: Bacilli, Order Lactobacillales, Family Lactobacillaceae, Genus: Lactobacillus.
The Lactobacillus genus implicates more than 200 species distinguished by phylogenetic and metabolic diversity. About 540 species and 54 subspecies of this genus have been identified according to the online database of List of Prokaryotic names with standing in Nomenclature (LPSN) (http://www.bacterio.net/lactobacillus.html) which is classified by the taxonomy requirements of the International Code of Nomenclature of Prokaryotes such as L. achengensis.19,20,29
Isolation sources of Lactobacillus spp.
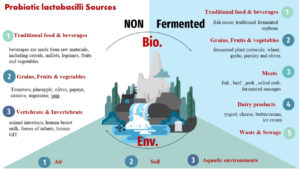
Biological sources
Fermented biological sources
Lactobacillus spp. are food spoilage organisms that control the vast majority of the microbiota of fermented foods.30,31 Fermented vegetables, silage, also various grains rely on the basic material type as an inoculum source. Other fermented sources such as the majority of dairy fermentations, fermented meats, and sourdough, are produced by the manufactured environments of diverse lactic acid bacteria isolated from natural sources.32,33 Microorganisms in fermented products are subjected to centuries of growing continuously, becoming accustomed to the fermented product. 34, 35 As a consequence of lactobacillus‘ capacity to adapt to multiple conditions in different environments, lactobacilli isolation may be inoculated into many different types of goods or natural environments in order to produce novel strains and, as a result, prospective therapeutic uses.
Traditional fermented foods have recently been investigated as potential natural probiotic bacteria sources, according to recent studies. The Lactobacillus genus is home to the bulk of microorganisms isolated from fermented environments.36 The Genus of Weissella was obtained from traditional food under fermentation conditions in Nigeria followed by an evaluation of potential probiotics abilities in a recent investigation.37
Lactobacillus delbrueckii subsp. bulgaricus appears to be adapting to conditions in fermented products, as evidenced by a rapid and continuing decrease in the size of the genome.37 As a result, despite the fact that the vast majority of the strain types were obtained from fermented foods, they are improbable to serve as Lactobacillus‘ main habitat. Lactobacillus species can be isolated directly from a variety of natural and man-made fermented environments and products:
Dairy products
Probiotics are used in a variety of dairy products, including sour/fermented milk, cheese, yogurt, butter/cream, new-born formula, and ice cream, according to the dairy industry. Probiotics are also utilized as starting cultures in conventional starters, either alone or in combination, or integrated into dairy products after fermentation, where their presence adds many functional characteristics to the fermented product (for example, improved taste, aroma, and textural characteristics), as well as conferring many health-promoting effects.38
The fermented milk includes multiplex species of LAB and hence serve as an excellent probiotic supply. It should come as no surprise that Streptococcus thermophilus and L. delbrueckii subsp bulgaricus are the most prevalent microbiological communities were found recently in the fermented yak milk (Kurut).39 Furthermore, probiotic lactobacillus strains and yeasts were identified from kefir grains, Koumissm, a fermented milk drink; and Masai milk; these bacteria have the capacity to favorably alter immunological responses.40,41
Some products, such as yogurt, cheese, and butter, can benefit from the addition of starting cultures made from milk or milk alone. In comparison to the traditional Luria-Bertani medium, milk whey, a dairy by-product from the cheese manufacturing industry, is utilized to develop LAB to create more lactic acid. As a result, it might be one of several sources from which advantageous LAB strains can be obtained42 (Figure 1).
Meats
LAB strains used to be detected in a variety of traditional fermented foods, such as beef,43 fish,44 pork,45 and seafood.46 Enterococcus is the most common LAB strain recovered from fermented crustaceans and fishes, whereas Lactobacillus is the most common LAB obtained from fermented meats and plants.47,48 Due to the complex components of meats, LAB extracted from such environments is likely to be more diversified functionally and physiologically and it is likely to provide further therapeutic applications than LAB isolated from other sources (Figure 1).
Grains, Fruits & vegetables
Lactobacilli strains from Plant-derived LAB are the most halophilic and flourished at NaCl concentrations greater than 22%. Significance species of lactobacillus were collected from fermentation condition of the plant parts are L. plantarum and L. fermentum. In addition, the various basic substance of dry wheats, garlic, olives, as well as parsley used in fermented food are high in LAB and may be tested for probiotic strains.45
Lactobacilli isolated from the natural brine of Algren, green olive fermented, was discovered in recent research.48 Several health benefits can be obtained from L. buchneri P2 as a potential probiotic collected from pickle juice was proved to have properties of antimicrobial activity, cholesterol reduction, and tolerance to acid and bile juice.49
Lactobacillus spp. or Leuconostoc spp. Were represented the majority of LAB isolated in a study from sauerkraut fermentations and investigated to discover their probiotic properties and determined genotypic and phenotypic characterization of the isolates were found in four different sauerkraut fermentations50 (Figure 1).
LAB isolated from manufactured fermented products that basically contain plant parts are generated due to the contamination conditions during production processes or from endophytes bacteria. Endophytes lactobacillus spp. recovered from plant parts have the capacity to survive in a wide variety of PH gradients, allowing LAB to be separated from mild PH conditions of plant tissues or surfaces. lactobacilli did, however, exhibit a definite preference for improved development in a low pH environment.
Traditional food & beverages
Many of the isolated species of Traditional fermented foods and drinks flourished in NaCl concentrations of less than 6%.51 Traditional Thai salted crab also contains NaCl-like LAB (Poo-Khem). The fish dressing is identified as meal prepared under fermentation condition created from various basic ingredients including fish and shellfish. Probiotic isolates of L. plantarum was isolated from fermented soybean, popular traditional food in India, in addition, other genera of LAB including Lactobacillus, Vagococcus, Weissella, and Enterococcus were discovered.52 After conducting several tests to determine LAB genera isolated from natural fermented dry sausage, L. plantarum and, L. brevis have been identified as the crucial lactobacillus spp.53 The majority of L. plantarum isolates from LAB came from traditional fermented Turkish beverages (Boza). To overcome Bacteria that cause disease including B. subtilis, B. cereus, E. coli, P. aeruginosa, L. monocytogenes, and K. pneumoniae, the antagonistic activity of isolates was exanimated by produced substances (primarily hydrogen peroxide and organic acids).54 In addition, various studies reported that probiotic LAB was collected from ungerminated cereal crops mixed with millet cereals known as (Kunu-zaki) as a traditional fermented beverage in Nigeria55 (Figure 1).
Lactobacilli utilized in traditional meals are either mesophilic or thermophilic, depending on the kind of ingredients and preparation circumstances, and may live at low temperatures as Psychrophilic bacteria. Lactobacillus spp. thrive in a wide range of temperatures, putting them at the forefront of bacterial species found in traditional meals.
Waste & Sewage
L. delbrueckii is one of the other species of lactobacillus that can be obtained from Fermented veggies that were discarded and inhibited the development of various harmful bacteria such as E. coli, P. aeruginosa and P. vulgaris.56
Lactobacilli can be discovered in sewage due to fecal contamination.57 potential probiotics had been located in diffusion of areas of the human body, consisting of human feces remoted from each healthful adult and youngsters58 and breast-fed newborns59 (Figure 1).
Lactobacilli found in waste or sewage come from a variety of original environments, including humans, animals, and plants. Because of the abundance of complex components and the availability of growth nutrients, waste circumstances enable optimal development for Lactobacillus spp.
From non-fermented biological sources
Traditional food & beverages
In numerous traditional non-fermented drinks have an excessive number of probiotics. Non-fermented drinks are created from a range of ingredients, including grains, millets, fruits, legumes, and vegetables.60
These meals and beverages are mostly composed of fresh items infected with lactobacillus spp., such as “waldorf” salad61 (Figure 1).
Grains, Fruits & vegetables
Microorganisms containing Probiotic properties may be isolated from raw materials of plant origin including fruits and their liquids,62 honeycomb,63 cereals,64 and dirt.65,66 L. plantarum specifically and many other LAB strains were isolated in a study from different kind of fruit juices, whereases L. mesenteroides is seldom isolated from fruits, however, it has been found in tomato.62 Another growing medium may be utilized to produce LAB and isolate lactobacilli from origin including pineapple waste and tomato juice.67,68 Lactobacillus kunkeei is one of the most common strains from the majority of Lactobacillus spp. among LAB. These species have been shown to have antibacterial properties against yeast development as well as spoilage-related effects in honey by yeast.69 Several studies have indicated that fruit juice, vegetables, and long grass may be used to isolate LAB. Green olives were used to isolate L. plantarum, L. pentosus, and L. paracasei, whereas L. mesenteroides and L. pentosus were isolated from dark olives.70,71
Several characteristics were examined to identify the probiotic potential of specific strains after isolation in a propitiate nutrient media of LAB. Plants Sources like papaya, yam, cassava, sugarcane, and taro leaves were confirmed to contain isolates of lactobacillus spp. by reading the sequence of 16S rDNA of the samples, species were recognized as L. paraplantarum and L. plantarum isolated from crops leaves.72
According to one study, these LAB isolated from plant sources may not be present in the regular microflora of plants grown recently, Lactobacillus spp. can be isolated from dead plants or raw materials such as olives, cabbage, carrots, beets, and fruits (for example pears and grapes).73 However, identification of lactobacillus spp. from surfaces of flowers or in the tissues of the other plant parts is restricted.74,75
The leaves of P. aquilinum and, Aloe Barbadensis, also seeds of L. usitatissimum, roots of D. carota, and flowers of P. cerasifera were investigated, and 14 distinct LAB were identified as Lactococcus and Lactobacillus, as well as 8 various enterococci. species were initially identified by API kit, and then again analyzed given information by PCR technology, which corroborated the biochemical outcomes. The strains of LAB were isolated from plants’ parts comprised of Lactobacillus delbrueckii subsp. Bulgaricus, Lactobacillus brevis, Lactococcus lactis, Lactobacillus acidophilus, and some species of Leuconostoc76 (Figure 1).
The epiphyte Lactobacillus spp. outnumbered the endophyte bacteria, the reason may back to the species coming from surrounding sources such as animals, humans, waste, and soil. Climate, geographical factors, plant type, resident microbial communities, and Physical factors of the environment can impact lactobacilli dispersion in plants.
Vertebrate & Invertebrate
Animal intestines, according to Siddiqee et al., are the most likely source of LAB.77 Several probiotic strains have been identified from human sources, including human breast milk, from various regions of the human body.78
the non-sterile breast milk, even if amassed aseptically, still makes the chance of containing isolates of lactobacillus spp. in high odds.79 Breast milk bacteria were long thought to be the result of cutaneous or fecal infections. Although genotypes of lactobacillus spp. in breast milk differ from the genes of the isolates lactobacilli collected from the skin,80 also the strains of LAB detected in breast milk also were discovered inside newborns’ excrement.78 Lactobacillus spp. can be found in numerous amounts in breast milk as a natural source of probiotics to be for incorporation into food products.74
Lactobacillus species are among the most common bacteria isolated from breast milk,81 also its consumption promotes the predominance of one more source of probiotics in the human intestinal tract. over 500 various species of bacteria can be obtained from the stomach of adult humans Indeed, many of today’s probiotic strains, including L. gasseri and L. reuteri, were isolated from this source.82 It’s a common misconception that the digestive tract should be the sole residence for the probiotics strains to be more beneficial.83
Probiotics are present in the stomachs of many animals, including rats, pigs, and even chickens.84 Lately, strains of Lactobacillus johnsonii, collected from Apis mellifera stomach, were proven to boost honeybee colonies.85 Probiotics’ isolates were also identified from the digestive tract of freshwater and marine fish, including Carassius auratus gibelio,86 rainbow trout,87 and shrimp.88
In vitro research has shown that some strains of bacteria obtained from meats (lactobacillus sakei, Staphylococcus carnosus, and lactobacillus curvatus) and fruits (L. plantarum and L. paracasei) exhibit functional and metabolic features comparable to bacteria of adult’s intestine.89
Lactobacilli may be isolated consistently from a broad variety of insects, including bees and flies, as well as vertebrates such as rodents, birds, people, and agricultural animals. Because scientific research has mostly concentrated on domesticated animals and people, the host range is likely to be substantially greater.90-92 Lactobacilli tend to favor food storage organs in animal hosts, such as the crop and forestomach. Insects including bees, flies, bumblebees, and vertebrates’ organs were used to isolate lactobacillus species. digestive tract, mouth cavity, vagina, and saliva and feces of healthy adults can be considered as non-fermented sources of lactobacillus isolates.93 Tajabadi et al. extracted LAB from honey collected from massive honeybees.94
Lactic Acid Bacteria (LAB) from ten healthy wild giant freshwater prawns’ stomachs and hepatopancreas, Macrobrachium rosenbergii, were identified in a recent study to investigate native probiotic strains for additional uses. According to the findings, two LAB isolates, L. plantarum and E. faecalis, have been recommended as prospective Probiotics derived from huge freshwater prawns to enhance the aquaculture sector95 (Figure 1).
The presence of Lactobacillus spp. species in many terrestrial and marine organisms gives a clear indication of the concept of one health approach. Therefore, the balance of these species in other organisms and environments can provide solutions for environmental balance, food security, and public health alike.
Environmental sources
Environments around food products, for example, have also been isolated as unconventional sources of high potential probiotics.
Air
The air in the surrounding environs, as well as the air in a bakery’s storage and working rooms, can be a source of lactobacilli during the sourdough preparation process. L. plantarum was discovered in the samples, which is comparable to the one collected from the dough. in addition, Lactobacilli were isolated from the air around palm plants56,96 (Figure 1).
Soils
Probiotic bacteria isolated from soil in Japan and Taiwan have been discovered and categorized. In order to isolate LAB in Japan and Taiwan, 68 soil samples were collected from various animal areas including the area surrounding the horse farm and floors of the henhouses. Several tests were conducted Physiologically and genetically to determine strains of LAB. 54 out of 68 isolates identified as LAB. This clearly indicates the existence of LAB in significant numbers. According to the findings, dirt may be a frequent source of lactic acid bacteria isolation.
The initial habitat of many isolates came from soil sources. These lactobacillus isolates can be transferred to any product or habitat derived from the rhizosphere or phyllosphere of plants57,74,97 (Figure 1).
Aquatic environments
In aquatic environments, scientists are interested in isolates from the gastrointestinal tract of marine organisms. Lactobacillus spp. Were collected from the GIT of several fish and shrimp. However, the environment surrounding and hydro planktons that form as nutrients for the marine organisms are rich in probiotic strains (Figure 1).
The significant roles of lactobacillus species in our life
Human has very tight contact with an enormous number of microorganisms that exist in the mouth, skin, or in gut. Some of them possess remarkable health functions such as inducing the immune system, owning an agnostic activity against invading pathogens, enhancing the digestion process, and amending many diseases.98
There are many identified Lactobacillus species that have been reported as probiotics and have beneficial effects on health (Figure 2).
Protection against pathogens
Inhibiting the growth of pathogens is one of the ways probiotics benefit human health. These probiotics compete for resources that pathogens would use for growth and proliferation. As a result, they provide protection against pathogens colonization by causing direct death and competing for resources.99
In the gastrointestinal tract
Probiotics are one of the most effective ways to influence human health and disease by balancing the gut flora.100
Several studies demonstrated the role of Lactobacillus as a probiotic against many pathogens such as Lactobacillus acidophilus is safe for human epithelial colorectal cells and efficacious enough to be utilized as a supplementary treatment for reducing toxin generation in Vibrio cholerae and Shigella dysenteriae caused acute infectious diarrhea.101
Furthermore, it was reported that taking Lactobacillus rhamnosus HN001 on a regular basis lowered the risk of Staphylococcus aureus carriage in the human gastrointestinal tract (GI). Competitive inhibition is the key possible mechanism of L. rhamnosus HN001, in which pathogen colonization is limited by beneficial commensal microbes out-competing them for vital resources in the GI tract.102
In another study, Jun Meng and others found that the surface layer protein (SLP) of Lactobacillus casei fb05 might reduce Escherichia coli and Salmonella’s harmful effects on the digestive tract in two ways: by lowering pathogen adherence and avoiding apoptosis caused by pathogens.103
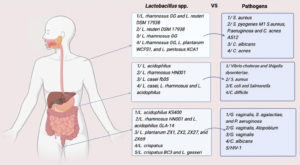
The leading cause of disruption of the normally protective gastrointestinal microbiota is Clostridium difficile infection (CDI) and antibiotic usage is a common cause of this condition. However, the supplementation of a certain probiotic combining Lactobacillus casei, L. rhamnosus, and L. acidophilus strains has been shown to be effective in inhibiting a number of undesirable consequences., including CDI and antibiotic-associated diarrhoea.104 The information about the gut microbiome is constantly accumulating which has heightened the interest in gut microbiome-based healthcare (Figure 3).
Figure 3. Different Lactobacillus species identified as human body protectors against various pathogens in the gastrointestinal tract, the vaginal tract, and the skin
Because of the strong connections between gut microbiota, health, and disease, there is a lot of interest in utilizing probiotics to alter the gut microbiota in order to treat or prevent certain diseases. However, more research is required to ascertain which probiotic species and strains, dosage, and duration of treatment are efficient. It is also necessary to look into any potential probiotic-diet interactions and interindividual variability that may result in disparate responses to probiotics.
In the vaginal tract
There are a variety of normal mycobiota and microbiota that colonize the human vagina, but Lactobacillus is the most prevalent bacteria found in the normal vaginal tract. These vaginal lactobacilli have been claimed as having the ability to prevent disease invasion by controlling their population.105
Lactobacillus has been shown to have fundamental probiotic effects against infections in the vaginal tract in several investigations. Fermentation of Lactobacillus acidophilus KS400 has been shown to generate bacteriocin that prevents urogenital pathogens growth like G. vaginalis, S. agalactiae, and P. aeruginosa.106
In addition, Lactobacillus acidophilus GLA-14 and Lactobacillus rhamnosus HN001exhibit antibacterial activities against several pathogens that were responsible for aerobic vaginosis (Staphylococcus aureus and Escherichia coli) and bacterial vaginosis (Atopobium vaginae and Gardnerella vaginalis) by means of a co-culturing assay.107
The L. Plantarum new isolates called ZX1, ZX2, ZX27, and ZX69 were found to limit Gardnerella vaginalis growth, the Bacterial vaginosis causative agent.108
In another study, they demonstrated that the major Lactobacillus species linked to a healthy vagina is L. crispatus may effectively block the growth of C. Albicans and the formation of hyphae. In an NRG1-dependent way, L. crispatus may also suppress the hyphae-specific gene expression (ALS3, HWP1, and ECE1). Furthermore, L. crispatus B145 is an excellent candidate for probiotic research.109
Also, a Lactobacilli dominated vaginal ecology has the ability to protect the host from human immunodeficiency virus (HIV) and sexually transmitted infections (STIs).110,111
L. crispatus BC3 and L. gasseri BC12 protect isolated cells and ex vivo tissues from infection with the Human Immunodeficiency Virus 1 (HIV-1) by releasing their extracellular vehicles (EVs) this finding could lead to new HIV-1 prevention measures for male-to-female sexual transmission112 (Figure 3).
Moreover, Lactobacillus shows to be a promising influence to use in the prevention of vaginal infections such as vulvovaginal candidiasis (VVC) and bacterial vaginosis (BV).105
To reduce the need for antimicrobial medications, a complementary method involving Lactobacillus to correct the imbalance of vaginal microbiota is urgently needed. To treat the heterogeneity of probiotic effectiveness, there should be more scientific investigations on the effectiveness of probiotic lactobacilli against vaginal infection.
In Skin health
The skin is one of the largest organs in the human body, and it serves as a vital barrier and protective barrier against pathogen invasion.113
For many years, various studies on Lactobacillus and skin health have been published, including prevention of skin illness and improvement of skin diseases.
Several studies have shown that exogenously applied lactobacilli, for example Lactobacillus rhamnosus GG and Lactobacillus reuteri DSM 17938 have been found to have anti-pathogenic properties both directly and indirectly. Lactobacilli or their lysates, through direct binding interactions, can prevent infections from adhering to keratinocytes or actively displace adherent pathogens, as proven in vitro against S. aureus. For L. rhamnosus GG at least in part, these effects appear to be caused by SpaCBA pili, it has a high level of adhesion to human keratinocytes.114-116
Furthermore, live L. reuteri DSM 17938 demonstrated antibacterial activity against pathogens on the skin including (Streptococcus pyogenes M1 Staphylococcus aureus, Pseudomonas aeruginosa, and Cutibacterium acnes AS12).116
Exopolysaccharides and the primary peptidoglycan hydrolase Msp1 were identified as important components of L. rhamnosus GG that reduced C. albicans hyphae production and attachment to host epithelial cells.117
Under the influence of Lactobacillus pentosus KCA1, L. rhamnosus GG, and L. plantarum WCFS1 has greatly reduced the lipase activity of Cutibacterium acnes that helps in the conversion of sebum into free fatty acids, contributing to acne (Figure 3).
These mechanistic insights have already been converted into positive therapeutic outcomes in a few clinical trials, demonstrating that topically applied lactobacilli can colonize the skin for a short period of time and improve skin health, However, in order to generate in vivo mechanistic insights and in-depth skin microbiome analyses, more and larger clinical studies are required.118,119
Numerous novel probiotic products are being developed for the treatment of various skin problems as a result of these studies on the gut and skin microbiomes, which have produced some extremely insightful findings. Additionally, Eczema, atopic dermatitis, acne, and allergic inflammation are just a few of the skin conditions that probiotic bacteriotherapy may help prevent and treat. It may also help reduce skin hypersensitivity and help protect against UV-induced skin damage and wounds. It can also be applied topically.
Combating physiological disorders
Lactobacillus probiotics have demonstrated effectiveness in a variety of clinical disorders and conditions ranging from diarrhea, female urogenital infection, Helicobacter pylori infections and inflammatory bowel disease to cancer98,119 (Table).
Table:
Uses of Lactobacillus spp. as probiotic against various human diseases. the clinical outcomes had been conducted on humans as a subject.
| Probiotic | Effective against | Clinical outcomes | Ref. |
|---|---|---|---|
| L. reuteri | Bowel diseases | Improved inflammation along with increased cytokine expression levels | 120 |
| Restoration of gut flora and the prevention of diarrhea | 121 | ||
| Coxsackieviruses | Reduce the antiviral activity | 28 | |
| Periodontitis | reduced abundance of the pathogenic bacterium | 122 | |
| Gingivitis | Decreased bleeding during chewing gums containing probiotics | 123 | |
| Type 2 diabetes mellitus | Beneficial effects on patients | 124 | |
| Diarrhea | Minimize the frequency and period of diarrhea | 125 | |
| Respiratory tract infection | Decrease the rate of the infection | 125 | |
| H. pylori infection | Lower the pathogen load in the stomach. | 126 | |
| Cholesterol | Reduce cholesterol levels. | 127 | |
| L. salivarius | Halitosis | enhanced physiologic halitosis and appeared beneficial impacts on bleeding from the periodontal pocket | 128 |
| Atopic dermatitis | Enhance scoring atopic dermatitis and itch values from baseline | 129 | |
| L. gasseri | Obesity | Reduce the risk of obesity by Lowering NEFA levels | 130 |
| Chronic Stress | Enhance sleep quality and mental state | 131 | |
| L. casei | Influenza virus (H3N2) | Increasing survival rate and preventing weight loss. | 132 |
| respiratory tract infection | Decreased prevalence of symptoms | 133 | |
| lowering the infection occurrence and duration of the upper respiratory tract. | 134 | ||
| L. fermentum | respiratory tract infection | Decreased prevalence of symptoms | 133 |
| Conjunctivitis | Lowered the incidence of conjunctivitis in infants. | 135 | |
| L. johnsonii | Type 1 diabetes | provide prevention against T1D | 136 |
| L. acidophilus | Autism disorder | Improved concentration and carrying out orders | 137 |
| Perennial allergic rhinitis | Reduced the symptoms | 138 | |
| L. plantarum | Irritable bowel syndrome | Relief the abdominal pain and reduce the bloating | 139 |
| Autism Disorder | improve some autism symptoms | 140 | |
| L. helveticus | Neurological and psychiatric diseases | Beneficial psychological effects in healthy humans | 141 |
| Cognitive fatigue | Enhancement of cognitive function. | 142 | |
| L. paracasei | respiratory tract infection | Decreased prevalence of symptoms | 133 |
| Allergic rhinitis | Reduction of nasal symptoms | 143 | |
| L. delbrueckii | influenza virus | Help by preventing the infection | 144 |
| L. crispatus | Urinary tract infections | Decreased recurrence | 145 |
| Vaginal wounds | expedite vaginal epithelial cell wound healing | 146 | |
| L. rhamnosus | severe watery diarrhea | Reduce the duration and frequency of diarrhea | 147 |
| Dental caries | lowered Streptococcus mutans counts | 148 | |
| Bacterial vaginosis | Decreased pH and the rate of recurrence | 149 | |
| allergic disease | Protect and reduce the eczema symptoms. | 150 | |
| Particularly to prevent or treat food allergies Cow’s milk intolerance | 151 | ||
| Respiratory and gastrointestinal tract infections | Decreased the danger of infection, and the duration of symptoms | 152 |
The impact of gut microbiota on host physiological regulation has recently gotten a lot of attention, especially in crucial areas including the immune system and metabolism.153
Immunomodulation
The immunomodulatory activities of probiotics can act both directly and indirectly. Directly such as by increasing the activity of natural killer cells or macrophages modulating the secretion of cytokines or immunoglobulins. Indirectly by competitive exclusion of other (pathogenic) bacteria, enhancing the gut epithelial barrier and altering the mucus secretion.154
Anti-commensal antibodies, innate cytokines, and Th17 responses were all reduced after oral gavage with L. reuteri, reducing immunological hyper-reactivity (Table). Also, autochthonous Lactobacillus may also aid in the restoration of immunological homeostasis and the resolution of infectious illness.155
Lactobacillus-free (LF) mice were used to establish the physiological relevance of L. reuteri‘s immunological effects in vivo.156,157 Proinflammatory cytokines and chemokines generated by intestinal epithelial cells in the ileum and jejunum were activated as a result of the administration of L. reuteri in this animal.158
Furthermore, probiotics have been shown in multiple studies to strengthen the integrity of the intestinal barrier by increasing gene expression in tight junction signaling. By tight junctional protein phosphorylation, enhancing (actinin, occludin) cytoskeletal or preserving (actin, ZO-1). L. acidophilus inhibited enter invasive E. coli invasion and adhesion in the human colon cancer cell line (HT29) and Caco-2 cells.159
Inhibition of carcinogenesis
It is well proven that Lactobacillus can modulate-ameliorate specific mechanisms against cancers via stimulating anti-cancer effects like enhancing apoptosis of cancer cells and protecting them from oxidative stress.
Probiotic bacteria, particularly Lactobacillus microorganisms, have been reported to cause anti-cancer action through increasing apoptosis of cancer cells and the protection against oxidative stress.160,161
In similar research involving colon cancer, it was demonstrated that the administration of L. fermentum NCIMB 5221 and L. acidophilus ATCC 314 in a murine colon cancer model had been discovered to stop or slow the growth of the tumor by triggering an anti-cancer immune response.162
Moreover, in mice models of colorectal cancer and melanoma, it has been shown that the administration of live L. rhamnosus GG orally enhanced the antitumor effectiveness of anti-programmed cell death 1 (PD-1) immunotherapy by boosting tumor-infiltrating dendritic cells (DCs) and T cells.163
In addition, L. paracasei, L. casei, L. fermentum, and L. rhamnosus were the most frequently isolated species from head and neck cancer patients. moreover, L. paracasei had the highest proportion of antimicrobial activity and the best acid-producing capacity against S. mutans.164
Probiotics might alter the micro-environment of the intestine, causing pro-inflammatory cytokines to decrease. As in the study of Zaharuddin and his group they have shown that pro-inflammatory cytokines were reduced in colorectal cancer patients by using probiotics consisting of six live Lactobacillus and Bifidobacteria strains.165
In addition, in breast cancer metastases in a mouse model, it has been shown that the fermentation of Milk by Lactobacillus casei CRL431 (probiotic fermented milk (PFM) help in breast cancer metastases by altering the immune response.166
Furthermore, probiotic L. rhamnosus MD 1 produced metabiotics that exhibited both cytotoxic and antigenotoxic potential against colon cancer.167
Another study has shown that lactobacillus E6-1 can inhibit the proliferation of Cal-27 and effectively induce apoptosis of tumor cells in vitro.168
The exopolysaccharide (EPS) generated by L. plantarum-12 was found to suppress the colon cancer cell line’s proliferation HT-29 in humans via the mitochondrial route.169
The findings of a study about colorectal cancer (CRC) suggested that consuming Lactobacillus acidophilus in persons with polyps and people that have a family history of CRC may be a means to prevent, treat, or lower the severity of CRC.170
Because of their activity against cervix cancer cells, other research suggests that Lactobacillus casei SR1, L. casei SR2 and L. paracasei SR4 strains derived from the breast milk of humans can be employed as topical therapy with a potential therapeutic index.171
The results of recent investigations support the hypothesis that probiotics may be used in cutting-edge cancer treatments. Therefore, to validate these findings and develop treatment plans, additional research on the subject is required. This could theoretically result in a breakthrough in many areas of medicine, including the support of immunotherapy in the treatment of cancer and the development and production of novel vaccines, as well as the improvement of drug delivery in the treatment of other bowel diseases while simultaneously preventing and reducing inflammation.
Reduction of cholesterol level
Numerous studies have demonstrated Lactobacillus‘ potential to lower cholesterol levels. Such as In C57BL/6 mice fed a hypercholesterolemia diet, L. plantarum H6 lowered serum cholesterol levels. It improved the microbial community of intestinal structure in mice by preventing the pathway of farnesoid X receptor to increase the synthesis of bile acids in vivo via promoting the expression of the CYP7A1 gene.172 (Table).
It was shown that L. plantarum YS5 can reduce the level of cholesterol by 84% due to its ability to remove cholesterol in vitro. furthermore, in male Wistar rats, probiotic supplementation was observed to lower serum total cholesterol, low-density lipoprotein cholesterol, and triglyceride levels.173
There is evidence that consuming probiotic-rich dairy products lowers blood cholesterol, which could help prevent diabetes, obesity, cerebral stroke, and cardiovascular disease.174
Lactobacillus, such as L. fermentum, L. plantarum, and L. acidophilus decrease cholesterol and ameliorate the advancement of nonalcoholic steatosis. Thus, Lactobacillus can be used as a beneficial strategy for non-alcoholic fatty liver disease (NAFLD) treatment.175
Furthermore, the administration of L. rhamnosus BFE5264 resulted in a major reduction in serum cholesterol in a mouse model fed a high-cholesterol diet and changes in the gut flora and the production of short-chain fatty acids were also observed (SCFA). Also, this strain has a positive impact on the metabolism of cholesterol in the liver in a manner like that resulting from statin treatment, which is a drug that can inhibit the biosynthesis of cholesterol in the liver.176
L. kefiri DH5 has anti-obesity benefits via lowering the lumen cholesterol and upregulating the PPAR gene in adipose tissues.177
Along the AMP-activated protein kinase (AMPK) pathway, lactobacilli could exert cholesterol-lowering properties by AMPK phosphorylation that lowered 3-hydroxy-3-methylglutaryl-coenzyme A reductase (HMGCR) expression.178
L. plantarum (LP06CC2) was tested for its effect on cholesterol metabolism in mice fed a cholesterol-rich diet. The consumption of LP06CC2 inhibited the hepatic damage indices as well as the increase of liver cholesterol. The deconjugation of bile acids was clearly boosted by LP06CC2, indicating that bile acid absorption was reduced. LP06CC2 appears to be a promising Lactobacillus for lowering cholesterol levels by modulating deconjugation of bile acid.179
Probiotics’ effects on total cholesterol levels have been thoroughly studied. Researchers have made an effort to elucidate the mechanisms of probiotics on the hypocholesterolemic effect through in vitro and in vivo experiments in order to explain the variable cholesterol-lowering effect displayed by distinct probiotic strains. Numerous theoretical explanations and experimental data focusing on cholesterol-lowering effects are still debatable. Therefore, better-designed in vivo studies may reveal more information that can help resolve disagreements, better understand underlying mechanisms, and determine whether a substance is safe to consume before it is used.
Probiotics as “One Health” Perspective
According to the World Health Organization, One Health is an integrated, unified strategy aimed at continuously balancing and optimizing the health of humans, animals, and ecosystems; in order to intimately connect the interdependence of all these domains.
Increasing the risks of antibiotic resistance and pathogen transmissibility between humans and animals, changing climatic conditions, people’s migrations, and increasing the rate of communication are all factors that have contributed to disease spread and will require the application of the concept of “one health.” Some of the most often used antimicrobial medications in animals, for example, are likewise reserved for the most difficult cases in humans.
Beneficial domestic multi-dominant agents such as probiotics, which are used to dwell in ecosystems and organisms, can apply the “one health” principle. Ecosystems contain and maintain the most biodiversity on the planet, which may be transmitted to humans via animal and plant products. Probiotics found in soil and plants are primarily favorable to humans and animals. Probiotics, which demonstrate dynamic relationships with different bacteria across the animal and plant kingdoms, developed alongside plants, invertebrates, and vertebrates, forming mutualism, symbiosis, commensalism, or even parasitic behavior with their hosts. Lactobacillus spp., with their ability to colonize the phyllosphere, endosphere, and rhizosphere, as well as the fruits and flowers of many plants, make these probiotics species suitable for achieving the “one health” notion.
The diverse lactobacilli isolation sources, as well as the species’ capacity to transmit and reside in multiple locations and habitats, enhance the implementation of the “one health” concept. Many studies have shown that Lactobacillus spp. can restore environmental balance and enhance human and animal health. For example, in human health, the remarkable flexibility and adhesion of lactobacilli in the host’s gastrointestinal system may help enhancement of the host’s gut health. Furthermore, lactobacilli species may be useful as plant growth promoters. Probiotics with the ability to ferment a wide range of carbohydrates, probiotic benefits on human and animal health, and are found in vegetables, dairy products, pickles, sauerkraut, cheeses, fish products, and sausages are likely the greatest candidates for “One Health.”
Improving the quality of animal products, increasing production and food security, and lowering the risk of pathogenic agents can all be accomplished by using probiotics as nutritional supplements in scientifically studied ways, in addition to providing an alternative solution to the use of antibiotics. Despite its critical and helpful properties, a high concentration of lactobacillus spp. in food can induce food spoilage.
Almost a century after Lactobacillus species were initially characterized, researchers are constantly working to identify new Lactobacillus species from previously unknown sources. Probiotics may be found in a number of locations; however, as development techniques have been employed, scientists have begun to seek probiotics in unorthodox sources that can aid in the discovery of novel probiotic strains such as non-dairy fermented goods and inanimate surroundings. Lactose-intolerant persons are given probiotics obtained from plant sources, which also demonstrate potential new antibiotics activities of isolates that can be utilized as a replacement or supplement to current chemical antibiotics. Lactobacillus species may be separated from fermented and non-fermented biological sources, as well as from environmental sources. The lactobacilli‘s unique habitat features and wide range of environments indicate the possibility of searching for and isolating new lactobacillus probiotics strains from unexpected sources.
It’s far viable that they may grow to be greater effective strategies inside the remedy of diverse issues which includes immune gadget stimulation, high cholesterol level, oral infections, urinary tract infections, vagina inflammation, diarrhea symptoms, and so forth as an alternative or supplement to other well-known treatments in the future. It is a natural remedy for newborn animals and humans, as well as for nutrition and sickness treatment. Aside from therapy, they are now widely used in newborn diets and milk. In terms of liver disease, lowering liver enzymes and bilirubin levels can be achieved by Lactobacilli isolated from sugar cane following the fermentation process.
Probiotics are vital for restoring normal gut flora, increasing the development of good bacteria, and decreasing the chance of acquiring chronic conditions including cardiovascular disease. As a result, there is widespread interest in these molecules as components for the development of innovative meals with functional qualities. seeking to the roles of native microbiota affecting health and balance restoration, the most current scientists in this regard are still in the early stages of discovering connections and relationships between native probiotics and intestinal microbiota, which might enable for successful customization of probiotic treatment, the commitment of supplementation time, and description of optimal doses for patients in order to improve cardiovascular efficiency or treat certain disorders.
Gut microbiome research is a young and difficult topic with methodologies that are a way to be uniform and coordinated. Massive datasets created from the microbiome, metabolomics, genomics, and transcriptomics are analyzed as consolidated investigation data could be a challenge to interpret, and a variety of complicated bioinformatics approaches have been conducted. the requirement for improving this field of inquiry is crucial through empirical and disarranged controlled studies. To increase existing understanding, well-designed clinical studies including all components of life routine, intestinal microbiome, metabolite, and genetics data are better to be improved.
Although additional clinical evidence is needed to further assess probiotics’ therapeutic value, procaryotes such as Lactobacilli strains are now among the most extensively utilized agents in GIT diseases. According to in vivo investigations, Lactobacillus survives well in the human GI tract. Lactobacilli are one of the most common LAB species that have played important therapeutic functions since their discovery. The increasing amount of research shows that it may be beneficial for diarrhea prevention and alleviation, as well as the treatment of recurring Clostridium difficile colitis.
By upgrading methodologies, tools, and techniques for isolation and identification, new pathways for probiotics will open, resulting in additional discoveries of local probiotic strains. New approaches, such as those that enable real-time human study and monitoring of a microbe’s integration into an established microbiome, are being developed, along with technologies capable of assessing hit points, which can move this area ahead. after moving on to physical tests henceforth, information on the presence of bacteria, their interaction with the host, and the effect of environmental stimuli (including medications, and nutrition) going to be routine. revolutionary pattern technology will disclose how a probiotic interacts with the host in several stages such as immune machine, all microbiome components, and metabolism. The integrated approach will aid in the establishment of dose-response connections for diagnosis, as well as in coordinating what gets into our systems, which probiotic bacteria have the most predicted impact, and how they will be absorbed.
Probiotic strains isolated from marine organisms have several applications and can be used for Aquaculture species and to prevent microbial pathogens that could threaten marine life. Studies that aim to isolate novel strains of probiotics from aquatic organisms present alternative treatments for the human and marine organisms’ pathogens and shed the light on the significant importance to intensify research for probiotics isolated from marine organisms in the future.
ACKNOWLEDGMENTS
The authors would like to thank Deanship of Scientific Research, Vice Presidency for Graduate Studies and Scientific Research & Department of Biological Sciences, King Faisal University, for their endless encouragement and support to accomplish this work.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
This work was supported by the Deanship of Scientific Research, Vice Presidency for Graduate Studies and Scientific Research, King Faisal University, Saudi Arabia Project No.
Grant727.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
This article does not contain any studies with human participants or animals performed by any of the authors.
- Sornplang P, Piyadeatsoontorn S. Probiotic isolates from unconventional sources: a review. J Anim Sci Technol. 2016;58(1):26.
Crossref - Montoro B, Benomar N, Lerma LL, Gutierrez SC, Galvez A, Abriouel H. Fermented Alorena Table Olives as a Source of Potential Probiotic Lactobacillus pentosus Strains. Front Microbiol. 2016;7:1583.
Crossref - Chauhan A, Ranjan A, Kumar Basniwal R, Jindal T. Probiotic, Prebiotic and Synbiotics in the Prevention of Lifestyle Disorders. Int J Curr Microbiol Appl Sci. 2016;5(12):933-947.
Crossref - Kaur M, Kaur G, Sharma A. Isolation of newer probiotic microorganisms from unconventional sources. J Appl Nat Sci. 2018;10(3):847-852.
Crossref - Wu H, Xie S, Miao J, et al. Lactobacillus reuteri maintains intestinal epithelial regeneration and repairs damaged intestinal mucosa. Gut Microbes. 2020;11(4):997-1014.
Crossref - Darbandi A, Ghanavati R, Asadi A, et al. Prevalence of bacteriocin genes in Lactobacillus strains isolated from fecal samples of healthy individuals and their inhibitory effect against foodborne pathogens. Iran J Basic Med Sci. 2021;24(8):1117-1125.
Crossref - Fuochi V, Coniglio MA, Laghi L, et al. Metabolic characterization of supernatants produced by lactobacillus spp. with in vitro anti-legionella activity. Front Microbiol. 2019;10:1403.
Crossref - Plaza-Diaz J, Ruiz-Ojeda F, Gil-Campos M, Gil A. Mechanisms of Action of Probiotics. Advances in Nutrition. 2019;10(suppl_1):S49-S66.
Crossref - Arshad F, Mehmood R, Hussain S, Khan A, Khan MS. Lactobacilli as Probiotics and their Isolation from Different Sources. Br J Res. 2018;5(3):43.
Crossref - Vandenplas Y, Huys G, Daube G. Probiotics: an update. J Pediatr (Rio J). 2015;91(1):6-21.
Crossref - Bazireh H, Shariati P, Azimzadeh Jamalkandi S, Ahmadi A, Boroumand MA. Isolation of Novel Probiotic Lactobacillus and Enterococcus Strains From Human Salivary and Fecal Sources. Front Microbiol. 2020;11:597946.
Crossref - Chamberlain C, Hatch M, Garrett T. Metabolomic profiling of oxalate-degrading probiotic Lactobacillus acidophilus and Lactobacillus gasseri. PLoS One. 2019;14(9):e0222393.
Crossref - Terai T, Kato K, Ishikawa E, et al. Safety assessment of the candidate oral probiotic Lactobacillus crispatus YIT 12319: Analysis of antibiotic resistance and virulence-associated genes. Food Chem Toxicol. 2020;140:111278.
Crossref - Pirbaglou M, Katz J, de Souza RJ, Stearns JC, Motamed M, Ritvo P. Probiotic supplementation can positively affect anxiety and depressive symptoms: A systematic review of randomized controlled trials. Nutr Res. 2016;36(9):889-898.
Crossref - Krzysciak W, Koscielniak D, Papiez M et al. Effect of a Lactobacillus Salivarius Probiotic on a Double-Species Streptococcus Mutans and Candida Albicans Caries Biofilm. Nutrients. 2017;9(11):1242.
Crossref - Luchansky JB, Muriana PM, Klaenhammer TR. Application of electroporation for transfer of plasmid DNA to Lactobacillus, Lactococcus, Leuconostoc, Listeria, Pediococcus, Bacillus, Staphylococcus, Enterococcus and Propionibacterium. Mol Microbiol. 1988;2(5):637-646.
Crossref - Darukaradhya J, Phillips M, Kailasapathy K. Selective enumeration of Lactobacillus acidophilus, Bifidobacterium spp., starter lactic acid bacteria and non-starter lactic acid bacteria from Cheddar cheese. Int Dairy J. 2006;16(5):439-445.
Crossref - Bull M, Plummer S, Marchesi J, Mahenthiralingam E. The life history of Lactobacillus acidophilus as a probiotic: a tale of revisionary taxonomy, misidentification and commercial success. FEMS Microbiol Lett. 2013;349(2):77-87.
Crossref - Ojha A, Shah N, Mishra V. Conjugal Transfer of Antibiotic Resistances in Lactobacillus spp. Curr Microbiol. 2021;78(8):2839-2849.
Crossref - Duar RM, Lin XB, Zheng J, et al. Lifestyles in transition: Evolution and natural history of the genus Lactobacillus. FEMS Microbiol Rev. 2017;41(Supp_1):S27-S48.
Crossref - Fijan S. Microorganisms with Claimed Probiotic Properties: An Overview of Recent Literature. Int J Environ Res Public Health. 2014;11(5):4745-4767.
Crossref - Widyastuti Y, Febrisiantosa A, Tidona F. Health-Promoting Properties of Lactobacilli in Fermented Dairy Products. Front Microbiol. 2021;12:673890.
Crossref - Raveschot C, Cudennec B, Coutte F, et al. Production of Bioactive Peptides by Lactobacillus Species: From Gene to Application. Front Microbiol. 2018;9:2354.
Crossref - Barcenilla C, Ducic M, Lopez M, Prieto M, Alvarez-Ordonez A. Application of lactic acid bacteria for the biopreservation of Meat Products: A systematic review. Meat Sci. 2022;183:108661.
Crossref - Lievin-Le Moal V, Servin AL. Anti-Infective Activities of Lactobacillus Strains in the Human Intestinal Microbiota: from Probiotics to Gastrointestinal Anti-Infectious Biotherapeutic Agents. Clin Microbiol Rev. 2014;27(2):167-199.
Crossref - Zheng J, Wittouck S, Salvetti E, et al. A taxonomic note on the genus lactobacillus: Description of 23 novel genera, emended description of the genus lactobacillus beijerinck 1901, and union of Lactobacillaceae and Leuconostocaceae. Int J Syst Evol Microbiol. 2020;70(4):2782-2858.
Crossref - Felis GE, Dellaglio F. Taxonomy of Lactobacilli and Bifidobacteria. Curr Issues Intest Microbiol. 2007;8(2):44-61. PMID: 17542335
- Ang LY, Too HK, Tan EL, et al. Antiviral activity of lactobacillus reuteri Protectis against Coxsackievirus A and enterovirus 71 infection in human skeletal muscle and colon cell lines. Virol J. 2016;13:111.
Crossref - Salvetti E, Torriani S, Felis GE. The genus lactobacillus: A taxonomic update. Probiotics Antimicrob Proteins. 2012;4(4):217-226.
Crossref - Ganzle MG. Lactic metabolism revisited: metabolism of lactic acid bacteria in food fermentations and food spoilage. Current Opinion in Food Science. 2015;2:106-117.
Crossref - Scheirlinck I, Van der Meulen R, De Vuyst L, huys VG. Molecular source tracking of predominant lactic acid bacteria in traditional Belgian sourdoughs and their production environments. J Appl Microbiol. 2009;106(4):1081-1092.
Crossref - Ripari V, Ganzle MG, Berardi E. Evolution of sourdough microbiota in spontaneous sourdoughs started with different plant materials. Int J Food Microbiol. 2016;232:35-42.
Crossref - Van de Guchte M, Penaud S, Grimaldi C, et al. The complete genome sequence of Lactobacillus bulgaricus reveals extensive and ongoing reductive evolution. Proc Natl Acad Sci U S A. 2006;103(24):9274-9279.
Crossref - Vogel C, Silva GM, Marcotte EM. Protein Expression Regulation under Oxidative Stress. Mol Cel Proteomics. 2011;10(12):M111.009217.
Crossref - Won TJ, Kim B, Lim YT, et al. Oral administration of Lactobacillus strains from Kimchi inhibits atopic dermatitis in NC / Nga mice. J Appl Microbiol. 2011;110(5):1195-1202.
Crossref - Ayeni FA, Sanchez B, Adeniyi BA, de Los Reyes-Gavilan CG, Margolles A, Ruas-Madiedo P. Evaluation of the functional potential of Weissella and Lactobacillus isolates obtained from Nigerian traditional fermented foods and cow’s intestine. Int J Food Microbiol. 2011;147(2):97-104.
Crossref - Gao J, Li X, Zhang G, et al. Probiotics in the dairy industry-Advances and opportunities. Compr Rev Food Sci Food Saf. 2021;20(4):3937-3982.
Crossref - Sun Z, Liu W, Gao W, et al. Identification and characterization of the dominant lactic acid bacteria from kurut: The naturally fermented yak milk in Qinghai, China. J Gen Appl Microbiol. 2010;56(1):1-10.
Crossref - Lopitz-Otsoa F, Rementeria A, Elguezabal N, Garaizar J. Kefir: a symbiotic yeasts-bacteria community with alleged healthy capabilities. Rev Iberoam Micol. 2006;23(2):67-74.
Crossref - Ya T, Zhang Q, Chu F, et al. Immunological evaluation of Lactobacillus casei Zhang: a newly isolated strain from koumiss in Inner Mongolia, China. BMC Immunol. 2008;9(1):68.
Crossref - Shiphrah VH, Sahu S, Thakur AR, Chaudhrui SR. Screening of Bacteria for Lactic Acid Production from Whey Water. Am J Biochem Biotechnol. 2013;9(2):118-123.
Crossref - Sonplang P, Uriyapongson S, Poonsuk K, Mahakhan P, Angkititrakul S. Antimicrobial Susceptibility of Lactic Acid Bacteria Isolated From Native Chicken Feces. KKU Res J (GS). 2007;7(3):1-8.
Crossref - Paludan-Muller C, Huss HH, GramL. Characterization of lactic acid bacteria isolated from a Thai low-salt fermented fish product and the role of garlic as substrate for fermentation. Int J Food Microbiol. 1999;46(3):219-229.
Crossref - Siripornadulsil W, Tasaku S, Buahorm J, et al. Probiotic Properties of Lactic Acid Bacteria Isolated from Fermented Food. Zenodo. 2014.
Crossref - Golneshin A, Adetutu E, Ball AS, May BK, Van TTH, Smith AT. Complete Genome Sequence of L. plantarum Strain B21, a Bacteriocin-Producing Strain Isolated from Vietnamese Fermented Sausage Nem Chua. Genome Announc. 2015;3(2):e00055-15.
Crossref - Sonsa-Ard N, Rodtong S, Chikindas ML, Yongsawatdigul J. Characterization of bacteriocin produced by Enterococcus faecium CN-25 isolated from traditionally Thai fermented fish roe. Food Control. 2015;54:308-316.
Crossref - Parlindungan E, Lugli GA, Ventura M, van Sinderen D, Mahony J. Lactic Acid Bacteria Diversity and Characterization of Probiotic Candidates in Fermented Meats. Foods. 2021;10(7):1519.
Crossref - Abriouel H, Benomar N, Perez Pulido R, Canamero MM, Galvez A. Annotated Genome Sequence of Lactobacillus pentosus MP-10, Which Has Probiotic Potential, from Naturally Fermented Alorena Green Table Olives. J Bacteriol. 2011;193(17):4559-4560.
Crossref - Zeng XQ, Pan DD, Guo YX. The probiotic properties of Lactobacillus buchneri P2. J Appl Microbiol. 2010;108(6):2059-2066.
Crossref - Touret T, Oliveira M, Semedo-Lemsaddek T. Putative probiotic lactic acid bacteria isolated from sauerkraut fermentations. PLOS ONE. 2018;13(9):e0203501.
Crossref - Miyashita M, Yukphan P, Chaipitakchonlatarn, W, et al. 16S rRNA gene sequence analysis of lactic acid bacteria isolated from fermented foods in Thailand. Microbiol Cult Coll. 2012;28(1):1-9. http://jsmrs.jp/journal/No28_1/No28_1_1.pdf
- Thokchom S, Joshi SR. Antibiotic resistance and probiotic properties of dominant lactic microflora from Tungrymbai, an ethnic fermented soybean food of India. J Microbiol. 2012;50(3):535-539.
Crossref - Cvrtila Fleck Z, Savic V, Kozacinski L, Nijari B, Zdolec N, Filipovic I. Identification of lactic acid bacteria isolated from dry fermented sausages. Veterinary Archives. 2012;82(3):265-272. https://hrcak.srce.hr/clanak/119820
- Kivanc M, Yilmaz M, Cakir E. Isolation and identification of lactic acid bacteria from boza, and their microbial activity against several reporter strains. Turk J Biol. 2011;35(3):313-324.
Crossref - Oluwajoba SO, Akinyosoye FA, Oyetayo OV. In Vitro Screening and Selection of Probiotic Lactic Acid Bacteria Isolated from Spontaneously Fermenting Kunu-Zaki. Adv Microbiol. 2013;03(04):309-316.
Crossref - Jini R, Swapna H, Rai A, et al. Isolation and characterization of potential lactic acid bacteria (LAB) from freshwater fish processing wastes for application in fermentative utilisation of fish processing waste. Braz J Microbiol. 2011;42(4):1516-1525.
Crossref - Kvasnikov E, Kovalenko N, Nesterenko O. Lactic acid bacteria in nature and the national economy [USSR, excludes the dairy industry] [1983]. Appl Biochem Microbiol. 2012;8:665-676 https://agris.fao.org/agris-search/search.do?recordID=US19840057754
- Sook CS, Yong KB, Jun CM, et al. Safety Assessment of Potential Lactic Acid Bacteria Bifidobacterium longum SPM1205 Isolated from Healthy Koreans. J Microbiol. 2005;43(6):493-498. PMID: 16410764
- Reis NA, Saraiva MAF, Duarte EAA, de Carvalho EA, Vieira BB, Evangelista-Barreto NS. Probiotic properties of lactic acid bacteria isolated from human milk. J Appl Microbiol. 2016;121(3):811-820.
Crossref - Krishnamoorthy M, Arjun P. Probiotic and antimicrobial activity of bacteria from fermented toddy of Cocus nucifera. J Acad Indus Res. 2012;1(3):127-131.
- Franz CM, Toit MD, Olasupo NA, Schillinger U, Holzapfel WH. Plantaricin D, a bacteriocin produced by Lactobacillus plantarum BFE 905 from ready-to-eat salad. Lett Appl Microbiol. 1998;26(3):231-235.
Crossref - Naeem M, Ilyas M, Haider S, Baig S, Saleem M. Isolation characterization and identification of lactic acid bacteria from fruit juices and their efficacy against antibiotics. Pak J Bot. 2012;44:323-328. https://www.pakbs.org/pjbot/PDFs/44(SI1)/48.pdf
- Tajabadi N, Mardan M, Manap MYA, Mustafa S. Molecular identification of Lactobacillus spp. isolated from the honey comb of the honey bee (Apis dorsata) by 16S rRNA gene sequencing. J Apic Res. 2013;52(5):235-241.
Crossref - Hamet MF, Londero A, Medrano M, et al. Application of culture-dependent and culture-independent methods for the identification of Lactobacillus kefiranofaciens in microbial consortia present in kefir grains. Food Microbiol. 2013;36(2):327-334.
Crossref - Chen YS, Yanagida F, Shinohara T. Isolation and identification of lactic acid bacteria from soil using an enrichment procedure. Lett Appl Microbiol. 2005;40(3):195-200.
Crossref - Yanagida F, Chen Y, Shinohara T. Searching for bacteriocin-producing lactic acid bacteria in soil. J Gen Appl Microbiol. 2006;52(1):21-28.
Crossref - Patel A, Prajapati JB, Holst O, Ljungh A. Determining probiotic potential of exopolysaccharide producing lactic acid bacteria isolated from vegetables and traditional Indian fermented food products. Food Bioscience. 2014;5:27-33.
Crossref - Yoon KY, Woodams EE, Hang YD. Probiotication of Tomato Juice by Lactic Acid Bacteria. J Microbiol. 2004;42(4):315-318. https://www.koreascience.or.kr/article/JAKO200430710475434.pdf
- Olofsson TC, Vasquez A. Detection and Identification of a Novel Lactic Acid Bacterial Flora Within the Honey Stomach of the Honeybee Apis mellifera. Curr Microbiol. 2008;57(4):356-363.
Crossref - Argyri AA, Zoumpopoulou G, Karatzas KAG, et al. Selection of potential probiotic lactic acid bacteria from fermented olives by in vitro tests. Food Microbiol. 2013;33(2):282-291.
Crossref - Doulgeraki AI, Pramateftaki P, Argyri AA, Nychas G-JE, Tassou CC, Panagou EZ. Molecular characterization of lactic acid bacteria isolated from industrially fermented Greek table olives. LWT – Food Sci Technol. 2013;50(1):353-356.
Crossref - Samedi L, Charles AL. Viability of 4 Probiotic Bacteria Microencapsulated with Arrowroot Starch in the Simulated Gastrointestinal Tract (GIT) and Yoghurt. Foods. 2019;8(5):175.
Crossref - Jay JM, Loessner MJ, Golden DA. Modern Food Microbiology, 7th Ed. Springer Science & Business Media; 2008. https://books.google.com.sa/books?hl=en&lr=&id=KbTDBAAAQBAJ&oi= fnd&pg=PA2&dq=LAB+isolated+from+ plants+sources+may+not+ be+present+in+the+ regular+microflora+of+ plant+grown+recently
- Arboleya S, Binetti A, Salazar N, et al. Establishment and development of intestinal microbiota in preterm neonates. FEMS Microbiol Ecol. 2011;79(3):763-772.
Crossref - Martins G, Lauga B, Miot-Sertier C, et al. Characterization of Epiphytic Bacterial Communities from Grapes, Leaves, Bark and Soil of Grapevine Plants Grown, and Their Relations. PLoS One. 2013;8(8):e73013.
Crossref - Anacarso I, Bassoli L, Sabia C, Iseppi R, Condo C. Isolation and identification of lactic acid bacteria from plants and other vegetable matrices and microbial recombination with Enterococcus spp. Am Res Thoughts. 2015;1: 1503-1515.
Crossref - Siddiqee MH, Sarker H, Shurovi KM. Assessment of probiotic application of lactic acid bacteria (LAB) isolated from different food items. Stamford Journal of Microbiology. 2013;2(1):10-14.
Crossref - Martin R, Olivares M, Perez M, et al. Identification and evaluation of the probiotic potential of lactobacilli isolated from canine milk. Vet J. 2010;185(2):193-198
Crossref - West PA, Hewitt JH, Murphy OM. The Influence of Methods of Collection and Storage on the Bacteriology of Human Milk. J Appl Bacteriol. 1979;46(2):269-277.
Crossref - O’Hara AM, Shanahan F. The gut flora as a forgotten organ. EMBO Reports. 2006;7(7):688-693.
Crossref - Perez-Cano FJ, Dong H, Yaqoob P. In vitro immunomodulatory activity of Lactobacillus fermentum CECT5716 and Lactobacillus salivarius CECT5713: two probiotic strains isolated from human breast milk. Immunobiology. 2010;215(12):996-1004.
Crossref - Ryan KA, Jayaraman T, Daly P, et al. Isolation of lactobacilli with probiotic properties from the human stomach. Lett Appl Microbiol. 2008;47(4):269-274.
Crossref - Madsen K, Cornish A, Soper P, et al. Probiotic bacteria enhance murine and human intestinal epithelial barrier function. Gastroenterology. 2001;121(3):580-591.
Crossref - Petrof EO. Probiotics and Gastrointestinal Disease: Clinical Evidence and Basic Science. Antiinflamm Antiallergy Agents Med Chem. 2009;8(3):260-269.
Crossref - Audisio MC, Benitez-Ahrendts MR. Lactobacillus johnsonii CRL1647, isolated from Apis mellifera L. bee-gut, exhibited a beneficial effect on honeybee colonies. Benef Microbes. 2011;2(1):29-34.
Crossref - Chu W, Lu F, Zhu W, Kang C. Isolation and characterization of new potential probiotic bacteria based on quorum-sensing system. J Appl Microbiol. 2010;110(1):202-208.
Crossref - Perez-Sanchez T, Balcazar JL, Garcia Y, et al. Identification and characterization of lactic acid bacteria isolated from rainbow trout, Oncorhynchus mykiss (Walbaum), with inhibitory activity against Lactococcus garvieae. J Fish Dis. 2011;34(7):499-507.
Crossref - Hill JE, Baiano JCF, Barnes AC. Isolation of a novel strain of Bacillus pumilus from penaeid shrimp that is inhibitory against marine pathogens. J Fish Dis. 2009;32(12):1007-1016.
Crossref - Haller D, Colbus H, Ganzle MG, Scherenbacher P, Bode C, Hammesb WP. Metabolic and Functional Properties of Lactic Acid Bacteria in the Gastro-intestinal Ecosystem: A comparative in vitro Study between Bacteria of Intestinal and Fermented Food Origin. Syst Appl Microbiol. 2001;24(2):218-226.
Crossref - Endo A, Futagawa-Endo Y, Sakamoto M, Kitahara M, Dicks LM. Lactobacillus florum sp. nov., a fructophilic species isolated from flowers. Int J Syst Evol Microbiol. 2010;60(10):2478-2482.
Crossref - Engel P, James RR, Koga R, Kwong WK, Mcfrederick Q, Moran NA. Standard methods for research on Apis mellifera gut symbionts. J Apic Res. 2013;52(4):1-24.
Crossref - Sartore-Bianchi A, Trusolino L, Martino C, et al. Dual-targeted therapy with trastuzumab and lapatinib in treatment-refractory, KRAS codon 12/13 wild-type, HER2-positive metastatic colorectal cancer (HERACLES): a proof-of-concept, multicentre, open-label, phase 2 trial. Lancet Oncol. 2016;17(6):738-746.
Crossref - Walter J. Ecological Role of Lactobacilli in the Gastrointestinal Tract: Implications for Fundamental and Biomedical Research. Appl Environ Microbiol. 2008;74(16):4985-4996.
Crossref - Ugras S. Isolation, identification and characterization of probiotic properties of bacterium from the honey stomachs of Yigilca honeybees in Turkey. Turk J Entomol. 2017;41(3).
Crossref - Mohamad N, Manan H, Sallehhuddin M, Musa N, Ikhwanuddin M. Screening of Lactic Acid Bacteria isolated from giant freshwater prawn (Macrobrachium rosenbergii) as potential probiotics. Aquaculture Reports. 2020;18:100523.
Crossref - Faparusi SI. Origin of Initial Microflora of Palm Wine from Oil Palm Trees (Elaeis guineensis). J Appl Bacteriol. 1973;36(4):559-565.
Crossref - Yanagida F, ChenY, Sato H, Suzui K-I. An attempt at isolation of lactic acid bacteria from soil samples collected in Aokigahara Jukai Forest, Japan. Japanese Journal of Lactic Acid Bacteria. 2006;17(2):138-141.
Crossref - Gupta V, Garg R. Probiotics. Indian J Med Microbiol. 2009; 27(3): 202-209.
Crossref - Galdeano CM, Cazorla SI, Dumit JML, Velez E, Perdigon G. Beneficial effects of probiotic consumption on the immune system. Ann Nutr Metab. 2019;74(2):115-124.
Crossref - Lee E-S, Song E-J, Nam Y-D, Lee S-Y. Probiotics in human health and disease: from nutribiotics to pharmabiotics. J Microbiol. 2018;56(11):773-782.
Crossref - Alamdary SZ, Bakhshi B. Lactobacillus acidophilus attenuates toxin production by Vibrio cholerae and shigella dysenteriae following intestinal epithelial cells infection. Microbial Pathogenesis. 2020;149:104543.
Crossref - Eggers S, Barker A, Valentine S, Hess T, Duster M, Safdar N. Effect of Lactobacillus rhamnosus HN001 on carriage of Staphylococcus aureus: results of the impact of probiotics for reducing infections in veterans (IMPROVE) study. BMC Infect Dis. 2018;18(1):129.
Crossref - Meng J, Wang Y-Y, Hao Y-P. Protective function of surface layer protein from Lactobacillus casei fb05 against intestinal pathogens in vitro. Biochem Biophys Res Commun. 2021;546:15-20.
Crossref - McFarland L, Ship N, Auclair J, Millette M. Primary prevention of Clostridium difficile infections with a specific probiotic combining Lactobacillus acidophilus, L. casei, and L. rhamnosus strains: assessing the evidence. J Hosp Infect. 2018;99(4):443-452.
Crossref - Chee WJY, Chew SY, Than LTL. Vaginal microbiota and the potential of Lactobacillus derivatives in maintaining vaginal health. Microb Cell Fact. 2020;19(1):203.
Crossref - Gaspar C, Donders G, Palmeira-de-Oliveira R, et al. Bacteriocin production of the probiotic Lactobacillus acidophilus KS400. AMB Express. 2018;8(1):1-8.
Crossref - Bertuccini L, Russo R, Iosi F, Superti F. Effects of Lactobacillus rhamnosus and Lactobacillus acidophilus on bacterial vaginal pathogens. Int J Immunopathol Pharmacol. 2017;30(2):163-167.
Crossref - Qian Z, Zhao D, Yin Y, Zhu H, Chen D. Antibacterial activity of Lactobacillus strains isolated from Mongolian yogurt against Gardnerella vaginalis. BioMed Res Int. 2020;1-9.
Crossref - Wang S, Wang Q, Yang E, Yan L, Li T, Zhuang H. Antimicrobial compounds produced by vaginal Lactobacillus crispatus are able to strongly inhibit Candida albicans growth, hyphal formation and regulate virulence-related gene expressions. Front Microbiol. 2017;8:564.
Crossref - van de Wijgert JHHM. The vaginal microbiome and sexually transmitted infections are interlinked: Consequences for treatment and prevention. PLoS Med. 2017;14(12):1-4.
Crossref - Chehoud C, Stieh D, Bailey A, et al. Associations of the vaginal microbiota with HIV infection, bacterial vaginosis, and demographic factors. AIDS. 2017;31(7):895-904.
Crossref - Nahui Palomino R, Vanpouille C, Laghi L, et al. Extracellular vesicles from symbiotic vaginal lactobacilli inhibit HIV-1 infection of human tissues. Nat Commun. 2019;10(1):5656.
Crossref - Gallo RL. Human skin is the largest epithelial surface for interaction with microbes. J Invest Dermatol. 2017;137(6):1213-1214.
Crossref - Spacova I, O’Neill C and Lebeer S. Lacticaseibacillus rhamnosus GG inhibits infection of human keratinocytes by Staphylococcus aureus through mechanisms involving cell surface molecules and pH reduction. Benef Microbe. 2020;11(7):703-715
Crossref - Mohammedsaeed W, McBain A, Cruickshank S, O’Neill CA. Lactobacillus rhamnosus GG inhibits the toxic effects of Staphylococcus aureus on epidermal keratinocytes. Appl Environ Microbiol. 2014;80(18):5773-5781.
Crossref - Khmaladze I, Butler E, Fabre S, Gillbro JM. Lactobacillus reuteri DSM 17938-a comparative study on the effect of probiotics and lysates on human skin. Exp Dermatol. 2019;28(7):822-828.
Crossref - Delanghe L, Spacova I, Van Malderen J, Oerlemans E, Claes I, Lebeer S. The role of lactobacilli in inhibiting skin pathogens. Bioch Soc Trans. 2021;49(2):617-627.
Crossref - Lebeer S, Oerlemans E, Claes I, et al. Topical cream with live lactobacilli modulates the skin microbiome and reduce acne symptoms. bioRxiv. 2018.
Crossref - Higaki S. Lipase inhibitors for the treatment of acne. J Mol Catal B Enzym. 2003;22(5-6):377-384.
Crossref - Oliva S, Di Nardo G, Ferrari F, et al. Randomised clinical trial: The effectiveness of Lactobacillus reuteri ATCC 55730 rectal enema in children with active distal ulcerative colitis. Aliment Pharmacol Ther. 2011;35(3):327-334.
Crossref - Wang H, Zhou C, Huang J, Kuai X, Shao X. The potential therapeutic role of Lactobacillus reuteri for treatment of inflammatory bowel disease. Am J Transl Res. 2020;12(5):1569-1583. PMCID: PMC7270012. Accessed 05 September 2022.
- Teughels W, Durukan A, Ozcelik O, Pauwels M, Quirynen M, Haytac MC. Clinical and microbiological effects of lactobacillus reuteri probiotics in the treatment of chronic periodontitis: A randomized placebo-controlled study. J Clin Periodontol. 2013;40(11):1025-1035.
Crossref - Twetman S, Derawi B, Keller M, Ekstrand K, Yucel-Lindberg T, Stecksen-Blicks C. Short-term effect of chewing gums containing probiotic lactobacillus reuteri on the levels of inflammatory mediators in gingival crevicular fluid. Acta Odontol Scand. 2009;67(1):19-24.
Crossref - Hsieh M-C, Tsai W-H, Jheng Y-P, et al. The beneficial effects of lactobacillus reuteri ADR-1 or ADR-3 consumption on type 2 diabetes mellitus: A randomized, double-blinded, placebo-controlled trial. Sci Rep. 2018;8(1):16791.
Crossref - Gutierrez-Castrellon P, Lopez-Velazquez G, Diaz-Garcia L, et al. Diarrhea in preschool children and lactobacillus reuteri: A randomized controlled trial. Pediatrics. 2014;133(4):e904-9.
Crossref - Mu Q, Tavella VJ, Luo XM. Role of lactobacillus reuteri in human health and diseases. Front Microbiol. 2018;9:757.
Crossref - Jones M, Martoni C, Prakash, S. Cholesterol lowering and inhibition of sterol absorption by Lactobacillus reuteri NCIMB 30242: a randomized controlled trial. Eur J Clin Nutr. 2012;66(11):1234-1241.
Crossref - Iwamoto T, Suzuki N, Tanabe K, Takeshita T, Hirofuji T. Effects of probiotic lactobacillus salivarius WB21 on halitosis and Oral Health: An open-label pilot trial. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2010;110(2):201-208.
Crossref - Niccoli AA, Artesi AL, Candio F, et al. Preliminary results on clinical effects of probiotic lactobacillus salivarius LS01 in children affected by atopic dermatitis. J Clin Gastroenterol. 2014;48(Suppl 1):S34-S36.
Crossref - Ogawa A, Kadooka Y, Kato K, et al. Lactobacillus gasseri SBT2055 reduces postprandial and fasting serum non-esterified fatty acid levels in Japanese hypertriacylglycerolemic subjects. Lipids Health Dis. 2014;13:36.
Crossref - Nishida K, Sawada D, Kuwano Y, Tanaka H, Rokutan K. Health benefits of Lactobacillus gasseri CP2305 tablets in young adults exposed to chronic stress: a randomized, double-blind, placebo-controlled study. Nutrients. 2019;11(8):1859.
Crossref - Jung Y-J, Lee Y-T, Ngo VL, et al. Heat-killed lactobacillus casei confers broad protection against influenza A virus primary infection and develops heterosubtypic immunity against future secondary infection. Sci Rep. 2017;7(1):17360.
Crossref - Zhang H, Yeh C, Jin Z, et al. Prospective study of probiotic supplementation results in immune stimulation and improvement of upper respiratory infection rate. Synth Syst Biotechnol. 2018;3(2):113-120.
Crossref - Shida K, Sato T, Iizuka R, et al. Daily intake of fermented milk with Lactobacillus casei strain Shirota reduces the incidence and duration of upper respiratory tract infections in healthy middle-aged office workers. Eur J Nutr. 2015;56(1):45-53.
Crossref - Pastor-Villaescusa B, Hurtado JA, Gil-Campos M, et al. Effects of Lactobacillus fermentum CECT5716 Lc40 on infant growth and health: a randomised clinical trial in nursing women. Benef Microbes. 2020;11(3):235-244.
Crossref - Marcial G, Ford AL, Haller MJ, et al. Lactobacillus johnsonii N6. 2 modulates the host immune responses: a double-blind, randomized trial in healthy adults. Front Immunol. 2017;8:655.
Crossref - Kalużna-Czaplinska J, Blaszczyk S. The level of Arabinitol in autistic children after probiotic therapy. Nutrition. 2012;28(2):124-126.
Crossref - Ishida Y, Nakamura F, Kanzato H, et al. Clinical effects of lactobacillus acidophilus strain L-92 on perennial allergic rhinitis: A double-blind, placebo-controlled study. J Dairy Sci. 2005;88(2):527-533.
Crossref - Ducrotte P, Sawant P, Jayanthi V. Clinical trial: Lactobacillus plantarum 299v (DSM 9843) improves symptoms of irritable bowel syndrome. World J Gastroenterol. 2012;18(30):4012.
Crossref - Liu Y, Liong M, Chung Y, et al. Effects of Lactobacillus plantarum PS128 on children with autism spectrum disorder in Taiwan: a randomized, double-blind, placebo-controlled trial. Nutrients. 2019;11(4):820.
Crossref - Messaoudi M, Lalonde R, Violle N, et al. Assessment of psychotropic-like properties of a probiotic formulation (lactobacillus helveticusR0052 and bifidobacterium longumR0175) in rats and human subjects. Br J Nutr. 2010;105(5):755-764.
Crossref - Chung Y, Jin, H, Cui Y, at el. Fermented milk of Lactobacillus helveticus IDCC3801 improves cognitive functioning during cognitive fatigue tests in healthy older adults. J Funct Foods. 2014;10:465-474.
Crossref - Yonekura S, Okamoto Y, Okawa T, et al. Effects of daily intake of lactobacillus paracasei strain KW3110 on Japanese cedar pollinosis. Allergy Asthma Proc. 2009;30(4):397-405.
Crossref - Yamamoto Y, Saruta J, Takahashi T, et al. Effect of ingesting yogurt fermented with Lactobacillus delbrueckii ssp. bulgaricus OLL1073R-1 on influenza virus-bound salivary IgA in elderly residents of nursing homes: a randomized controlled trial. Acta Odontol Scand. 2019;77(7):517-524.
Crossref - Stapleton AE, Au-Yeung M, Hooton TM, et al. Randomized, placebo-controlled phase 2 trial of a lactobacillus crispatus probiotic given intravaginally for prevention of recurrent urinary tract infection. Clin Infect Dis. 2011;52(10):1212-1217.
Crossref - Pan M, Hidalgo-Cantabrana C, Goh YJ, Sanozky-Dawes R, Barrangou R. Comparative Analysis of Lactobacillus gasseri and Lactobacillus crispatus Isolated From Human Urogenital and Gastrointestinal Tracts. Front Microbiol. 2020;10:3146.
Crossref - Basu S, Paul DK, Ganguly S, Chatterjee M, Chandra PK. Efficacy of high-dose lactobacillus rhamnosus GG in controlling acute watery diarrhea in Indian children. J Clin Gastroenterol. 2009;43(3):208-213.
Crossref - Nase L, Hatakka K, Savilahti E, et al. Effect of long-term consumption of a probiotic bacterium, lactobacillus rhamnosus gg, in milk on dental caries and caries risk in children. Caries Res. 2001;35(6):412-420.
Crossref - Recine N, Palma E, Domenici L, et al. Restoring vaginal microbiota: Biological control of bacterial vaginosis. A prospective case-control study using Lactobacillus rhamnosus BMX 54 as adjuvant treatment against bacterial vaginosis. Arch Gynecol Obstet. 2015;293(1):101-107.
Crossref - Wickens K, Barthow C, Mitchell E, et al. Effects of Lactobacillus rhamnosus HN001 in early life on the cumulative prevalence of allergic disease to 11 years. Pediatr Allergy Immunol. 2018;29(8):808-814.
Crossref - Canani RB, Di Costanzo M, Pezzella V, et al. The Potential Therapeutic Efficacy of Lactobacillus GG in Children with Food Allergies. Pharmaceuticals. 2012;5(6):655-664.
Crossref - Hojsak I, Snovak N, Abdovic S, Szajewska H, Misak Z, Kolacek S. Lactobacillus GG in the prevention of gastrointestinal and respiratory tract infections in children who attend day care centers: A randomized, double-blind, placebo-controlled trial. Clin Nutr. 2010;29(3):312-316.
Crossref - Tang TWH, Chen HC, Chen CY, et al. Loss of gut microbiota alters immune system composition and cripples postinfarction cardiac repair. Circulation. 2019;139(5):647-659.
Crossref - La Fata G, Weber P, Mohajeri MH. Probiotics and the gut immune system: indirect regulation. Probiotics Antimicrob Proteins. 2018;10(1):11-21.
Crossref - Heeney DD, Gareau MG, Marco ML. Intestinal Lactobacillus in health and disease, a driver or just along for the ride? Curr Opin Biotechnol. 2018;49:140-147.
Crossref - Livingston M, Loach D, Wilson M, Tannock GW, Baird M. Gut commensal Lactobacillus reuteri 100-23 stimulates an immunoregulatory response. Immunol Cell Biol. 2010;88:99-102.
Crossref - Hoffmann M, Rath E, Hoelzwimmer G, et al. Lactobacillus reuteri 100-23 transiently activates intestinal epithelial cells of mice that have a complex microbiota during early stages of colonization. J Nutr. 2008;138(9):1684-1691.
Crossref - Walter J, Britton R A, Roos S. Host-microbial symbiosis in the vertebrate gastrointestinal tract and the Lactobacillus reuteri paradigm. Proc Natl Acad Sci U S A. 2011;108(Suppl 1):4645-4652.
Crossref - Resta-Lenert S, Barrett KE. Live probiotics protect intestinal epithelial cells from the effects of infection with enteroinvasive Escherichia coli (EIEC). Gut. 2003;52(7):988-997.
Crossref - Badgeley A, Anwar H, Modi K, Murphy P, Lakshmikuttyamma A. Effect of probiotics and gut microbiota on anti-cancer drugs: mechanistic perspectives. Biochim Biophys Acta Rev Cancer 2021;1875(1):188494.
Crossref - Dasari S, Kathera C, Janardhan A, Kumar AP, Viswanath B. Surfacing role of probiotics in cancer prophylaxis and therapy: a systematic review. Clin Nutr. 2017;36(6):1465-1472.
Crossref - Kahouli I, Malhotra M, Westfall S, Alaoui-Jamali MA, Prakash S. Design and validation of an orally administrated active L. fermentum–L. acidophilus probiotic formulation using colorectal cancer Apc Min/+ mouse model. Appl Microbiol Biotechnol. 2017;101(5):1999-2019.
Crossref - Si W, Liang H, Bugno J, et al. Lactobacillus rhamnosus GG induces cGAS/STING- dependent type I interferon and improves response to immune checkpoint blockade. Gut. 2022;71(3):521-533.
Crossref - Rahne M, Basic A, Almstahl A. Explorative study on Lactobacillus species and their acid-producing capacity and anti-microbial activity in head and neck cancer patients. Clin Exp Dent Res. 2021;7(5): 924-933.
Crossref - Zaharuddin L, Mokhtar N, Nawawi K, Ali RAR. A randomized double-blind placebo-controlled trial of probiotics in post-surgical colorectal cancer. BMC Gastroenterol. 2019;19(1):131.
Crossref - Utz V, Visnuk D, Perdigon G, de LeBlanc AdeM. Milk fermented by Lactobacillus casei CRL431 administered as an immune adjuvant in models of breast cancer and metastasis under chemotherapy. Appl Microbiol Biotechnol. 2021;105(1):327-340.
Crossref - Sharma M, Chandel D, Shukla G. Antigenotoxicity and cytotoxic potentials of metabiotics extracted from isolated probiotic Lactobacillus rhamnosus MD 14 on Caco-2 and HT-29 human colon cancer cells. Nutr Cancer. 2020;72(1):110-119.
Crossref - Gao L, Sun Z, Yu H, Jing J-X, Xu Z-Q. Inhibiting effect and mechanism of Lactobacillus E6-1 on oral cancer cell line Cal-27. Shanghai Journal of Stomatology. 2019;28(6):605-609.
- Sun M, Liu W, Song Y, Tuo Y, Mu G, Ma F. The Effects of Lactobacillus plantarum-12 crude exopolysaccharides on the cell proliferation and apoptosis of human colon cancer (HT-29) cells. Probiotics Antimicrob Proteins. 2021;13(2):413-421.
Crossref - Zinatizadeh N, Khalili F, Fallah P, Farid M, Geravand M, Yaslianifard S. Potential preventive effect of lactobacillus acidophilus and lactobacillus plantarum in patients with polyps or colorectal cancer. Arq Gastroenterol. 2018;55(4):407-411.
Crossref - Rajoka MR, Zhao H, Lu Y, et al. Anticancer potential against cervix cancer (HeLa) cell line of probiotic Lactobacillus casei and Lactobacillus paracasei strains isolated from human breast milk. Food & Function. 2018;9(5):2705-2715.
Crossref - Qu T, Yang L, Wang Y, Jiang B, Shen M, Ren D. Reduction of serum cholesterol and its mechanism by Lactobacillus plantarum H6 screened from local fermented food products. Food & Function. 2020;11(2):1397-1409.
Crossref - Nami Y, Bakhshayesh RV, Manafi M, Hejazi MA. Hypocholesterolaemic activity of a novel autochthonous potential probiotic Lactobacillus plantarum YS5 isolated from yogurt. LWT. 2019;111:876-882.
Crossref - Markowiak P, Slizewska K. Effects of Probiotics, Prebiotics, and Synbiotics on Human Health. Nutrients. 2017; 9(9):1-30.
Crossref - Lee N, Shin M, Youn G, et al. Lactobacillus attenuates progression of nonalcoholic fatty liver disease by lowering cholesterol and steato-sis. Clin Mol Hepatol. 2021;27(1):110-124.
Crossref - Park S, Kang J, Choi S, et al. Cholesterol-lowering effect of Lactobacillus rhamnosus BFE5264 and its influence on the gut microbiome and propionate level in a murine model. PLoS ONE. 2018;13(11):e0208294.
Crossref - Kim D, Jeong D, Kang I, Kim H, Song K-Y, Seo K-H. Dual function of Lactobacillus kefiri DH5 in preventing high-fat-diet-induced obesity: direct reduction of cholesterol and upregulation of PPAR-a in adipose tissue. Mol Nutr Food Res. 2017;61(11):1700252.
Crossref - Lew L-C, Choi S-B, Khoo B-Y, Sreenivasan S, Ong K-L,Liong M-T. Lactobacillus plantarum DR7 reduces cholesterol via phosphorylation of AMPK that down-regulated the mRNA expression of HMG-CoA reductase. Korean J Food Sci Anim Resour. 2018;38(2):350-361.
Crossref - Yamasaki M, Minesaki M, Iwakiri A, et al. Lactobacillus plantarum 06CC2 reduces hepatic cholesterol levels and modulates bile acid deconjugation in Balb/c mice fed a high-cholesterol diet. Food Sci Nutr. 2020;8(11):6164-6173.
Crossref
© The Author(s) 2022. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.