ISSN: 0973-7510
E-ISSN: 2581-690X
Hyperglycemia is a condition in which the blood glucose levels exceed normal limits. Ethanol extract of Jamaican cherry leaves (Muntingia calabura L.) contains active components that have the potential to lower blood sugar levels and heal pancreatic cell damage in rats. Gut microbiota imbalance can cause immune system abnormalities, illnesses, and metabolic disorders, including diabetes. The aim of the present study was to determine whether an ethanol extract of Jamaican cherry leaves can repair pancreatic cell damage, as well as influence the total coliforms and lactic acid bacteria in hyperglycemic mice. In this study, 25 mice were randomly placed into five groups, which were then provided with food and drinking water. Alloxan in a dose of 160 mg/kg bw was administered to the positive control group and three treatment groups. Each of the three groups was then treated with the ethanol extract of Jamaican cherry leaves at doses of 300, 400, or 500 mg/kg bw for 14 days. The groups that received the extract were able to repair the pancreatic damage considerably by increasing the number of normal pancreatic cells. This was supported by histological observations. The total abundance of lactic acid bacteria in hyperglycemic mice was 1.08 ± 45 x 1010 CFU/g, which was substantially lower than that in mice treated with the extract. The overall coliform abundance in hyperglycemic mice was 4.37 ± 41 x 1010 CFU/g, which was higher than that in mice supplemented with the extract. Therefore, the administration of Jamaican cherry leaf ethanol extract on a regular basis can increase the abundance of cecum microbiota in hyperglycemic mice. Furthermore, this extract can be used as a natural alternative treatment for recovering pancreatic cell damage.
Alloxan, Gut Microbiota, Hyperglycemia, Muntingia Calabura, Pancreatic Cells
Poor glucose metabolism has long been associated with acute and life-threatening diseases. Transient hyperglycemia, also known as stress hyperglycemia or stress-induced hyperglycemia, is mainly detected in individuals without diabetes.1 Hyperglycemia is often caused by damaged pancreatic b cells that cannot produce insulin or damaged insulin receptors.2 Untreated hyperglycemia can lead to a variety of potentially life-threatening effects, including damage to the eyes, kidneys, nerves, heart, and peripheral vascular system. Thus, proper and efficient treatment of hyperglycemia is essential to avoid complications and improve patient outcomes.3
Alloxan is commonly used in animal experiments to induce diabetes. The cytotoxic effect of this diabetogenic agent is mediated by reactive oxygen species (ROS). With the production of superoxide radicals, alloxan and its reduction product, dialuric acid, initiate a redox cycle. The activity of ROS, along with a high increase in cytosolic calcium concentration, results in the rapid death of b cells.4 Ingredients high in antioxidants provide protection against oxidative stress-induced damage during diabetes complications.5-6 The glucose-lowering effect of natural ingredients may be significant in b cell regeneration process.7
Hyperglycemic conditions not only affect metabolism but also influence the microbiome profile. Oral bacteria contribute to hyperglycemia by increasing insulin resistance, which results in uncontrolled blood glucose levels.8 Berberine, an isoquinoline alkaloid derived from Ctenopharyngodon idellus, is beneficial in the treatment of diabetes in animals because of its strong hypoglycemic (glucose-lowering) and hypolipidemic (lipid-lowering) properties. It also aids in the management of bacterial intestinal disorders. Berberine may influence blood glucose levels through structural regulation of the gut flora.9 Furthermore, flavonoids ameliorate the pathogenesis of diabetes and its complications by regulating glucose metabolism, hepatic enzyme activity, and lipid profile.10 Jamaican cherry (Muntingia calabura L.) leaf may be used as a feed supplement because it contains antioxidants such as flavonoids, tannins, saponins, triterpenes, and polyphenols.11 Furthermore, M. calabura fractions were found to contain secondary metabolites such as alkaloids, which act as hepatoprotectors.12 However, there is currently no evidence that Jamaican cherry leaf extract can reverse pancreatic cell damage or influence the gut microbiota in hyperglycemic rats. Therefore, the present study investigated whether an ethanol extract of Jamaican cherry leaves can repair damaged pancreatic cells, as well as influence the abundance of the total coliforms and lactic acid bacteria (LAB) in hyperglycemic rats.
Preparation of Jamaican Cherry Leaf Extract
Dark-green Jamaican cherry leaves were chosen, weighed, and thoroughly cleaned. They were first dried for 24 h, then sliced into small pieces and baked at 70°C for 24 h. The dried leaves were weighed and pureed in a blender until they reached a powder-like consistency. The pureed leaves were then macerated three times in 1 L 96% ethanol for 24 h. The macerate was filtered using a Buchner funnel to obtain a filtrate. The filtrate was concentrated with a rotary evaporator at 50°C to produce a thick extract, which was then baked to set a shape and thickened with 1% Na-CMC.
Total Flavonoid Extract (Muntingia calabura L.)
Flavonoid content was calculated using the absorbance curve of a standard quercetin solution. The standard solution was prepared by dissolving 25 mg of standard quercetin in 25 mL ethanol to obtain a concentration of 1000 ppm. A 1 mL stock solution was pipetted into a 10 mL volume. Several concentration series were produced from a standard solution of 100 ppm quercetin, notably, 4, 6, 8, 10, and 12 ppm. Subsequently, 3 mL of ethanol, 0.2 mL of 10% AlCl3, and 0.2 mL of 1 M potassium acetate were added to 5 mL of each quercetin solution. The samples were then incubated at room temperature for 30 min. A UV-Vis spectrophotometer was used to measure the absorbance of the mixture in a wavelength of 440 nm. Total flavonoid content was determined using quercetin as a reference.13-14 The leaf extract was weighed and dissolved in 25 mL ethanol. The same steps used in the preparation and measurement of the normal quercetin solutions were repeated for the sample solutions. The measurements were performed three times.
Experimental Animal Maintenance
Twenty-five male mice (Mus musculus L.) aged 2-3 months with a weight of 30-40 g were used in this study. The mice were procured from Lampung Veterinary Center. This study was conducted in compliance with the ethical principle of replacement, reduction, and refinement (3R) concept. Mice were allowed to acclimatize for one week in a new cage habitat that used rice husks as a base. During acclimation, mice were provided with drinking water and food pellets as their regular diet. The typical amount of feed for mice was 3-4 g/animal/day. 15
Alloxan Administration to Induce Hyperglycemia
Administering a dose of 160 mg/kg bw alloxan may cause hyperglycemia in mice.16 Therefore, the present study used this particular dose of alloxan to induce diabetes in mice. First, mice were made to fast for 6-8 h. After fasting, the blood glucose levels of the mice were assessed using a glucometer, and their starting body weight was measured using a digital scale. Then, their tails were damaged with a needle, and alloxan was injected subcutaneously at the nape of the neck approximately two hours after the tail wound began to dry.
Alloxan was dissolved in 0.3 mL of aqua pro injection before being injected into mice. After 24 h of alloxan administration, 0.3 mL of 5% sugar water was given orally to prevent hyperglycemia. Blood sugar levels were examined five days after alloxan injection. The mice used in this study had blood glucose levels higher than 125 mg/dL, which is considered hyperglycemic by the International Diabetes Federation. Mice that had a rise in blood glucose levels were administered Jamaican cherry leaf extract orally for 14 days.
Preparation of Ethanol Extract from Jamaican Cherry Leaves
Ethanol extract of Jamaican cherry leaves was administered to mice in doses of 300, 400, and 500 mg/kg bw for 14 days. Jamaican cherry leaf extract at a dose of 400 mg/kg bw is found to be beneficial in decreasing blood sugar levels.17
Surgery on Test Animals and Organ Sampling
Mice were dissected and anesthetized with chloroform on the 14th day or at the end of the study period. This was accomplished by placing the experimental animals in glass jars previously filled with cotton soaked in chloroform. Anesthesia was administered by making each mouse inhale 0.67 mL of chloroform for 60s, which was measured using a stopwatch.18 After the mice stopped moving, surgery was performed vertically on the ventral region of their bodies. The pancreas was removed when the stomach was opened. The removed pancreas was immediately fixed in a 10% formalin solution, and to obtain excellent fixation results, the ratio of specimen volume to formalin solution was kept as 1:10. For histological examination of the pancreas, it was transferred to the Laboratory of Anatomy, Histology, and Anatomical Pathology of the Faculty of Medicine. Meanwhile, the removed intestinal organs were transferred to the Microbiology Laboratory of the Faculty of Mathematics and Natural Sciences.
Preparation of Histological Sections of Mouse Pancreas
Based on the procedure used by Sari & Noverda19 histological sections of mouse pancreas were prepared using hematoxylin-eosin staining. The entire process comprise material selection, fixation, dehydration, purification, paraffin infiltration, embedding, cutting, pasting, deparaffinization, staining, closure, and labeling.
Assessing the Level of Pancreatic Damage
The level of pancreatic damage in mice was observed by detecting tissue damage in the histological sections of mouse pancreas throughout the treatment group and then grading the degree of damage.20 Furthermore, the pancreatic damage was evaluated by determining the area of the islets of Langerhans in each treatment group using the gravimetric approach.21 The degree of damage was graded according to the scoreboard provided in Table 1.
Tests for calculating the Langerhans area = [ The pancreatic langerhans
replica’s weigh / 1cm x 1cm paper weight ] × 1cm2
Table (1):
Cell damage appearance pancreas scoring.
Score |
Information |
|---|---|
0 |
There was no damage to the cells or the structure of the Langerhans islets; the structure and size were normal. |
1 |
Total necrosis of pancreatic cells in quarter, with cell degeneration seen as cytoplasmic vacuoles. |
2 |
Total necrosis of pancreatic cells in a half, the presence of necrotic cells throughout the field of vision, karyorrhexis of cells (nuclear fragmentation). |
3 |
Total pancreatic cell necrosis is ¾, with the presence of inflammatory cells in the pancreatic interstitial space. |
4 |
All pancreatic cells are necrotic. |
Total Coliform and Lactic Acid Bacteria in Mice Intestines (Mus musculus L.)
Microbial analysis involves calculating the total quantities of LAB and coliforms. Total plate count was calculated using the dilution approach. The sample of cecum contents was diluted to 10-8 in stages. Total LAB was calculated by inoculating 1 mL of the sample on deMan Rogosa Sharpe agar medium with dilution factors of 10-6, 10-7, and 10-8. For total coliforms, the samples were aerobically inoculated on Endo agar medium. All samples were incubated at 37°C for 24 h. The total number of bacteria was estimated by dividing the product of the number of colonies and the dilution factor by the number of colonies on each medium.22
Total Flavonoid Content of Different Concentrations of Jamaica Cherry Leaf Ethanol Extract
Flavonoids are an important class of natural products that are abundant in fruits, vegetables, and some drinks. They are a type of plant secondary metabolite with a polyphenolic structure and can be found in various regions of the plant. The total flavonoid content in the ethanol extract of Jamaican cherry leaves increased with increasing treatment dose (Table 2).
Table (2):
Total flavonoid which measured using spectrophotometer at l 440 nm.
No |
Treatment Dose |
Cherry Leaf Ethanol Extract |
Total flavonoid levels (mg QE/g extract) |
|---|---|---|---|
1 |
Dose 300mg/ kgbw |
1,029 g |
799,65 |
2 |
Dose 400mg/ kgbw |
1,372 g |
1006,20 |
3 |
Dose 500mg/ kgbw |
1,715 g |
1332,76 |
Note: The term QE/g (quercetin equivalent) refers to the number of milligrams of quercetin in one gram of extract. *kgbw; kilograms body weight.
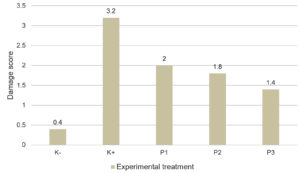
Damage Score for Mouse Pancreases
Figure 1 depicts the damage score results for injured mouse pancreases. The histopathological grading data reflected the extent of pancreatic damage in each group after alloxan injection and subsequent administration of cherry leaf ethanol extract.
Figure 1. Damage score of pancreatic Langerhans under microscope observation (400x)
Description: K-: Negative control, K+ : Positive control, P1 : Dose 300 mg/kgbw, P2 : Dose 400 mg/kgbw, P3 : Dose 500 mg/kgbw
The average pancreatic damage score of negative control group (K-) was 0.4 ± 0.31. Compared with the other treatment groups, the K- group had the least damage. Meanwhile, the mean score (3.20 ± 44) in the positive control group (K+) revealed that the majority of the cells had been damaged. This indicated that the K+ group with alloxan injection was the most adversely affected group among all the treatment groups. In comparison to K+, groups P1, P2, and P3 that were administered the ethanol extract of cherry leaves exhibited less damage.
According to the post hoc Mann–Whitney test (p = 0.006), P3 (500 mg/kg bw/day) was the optimum dosage for repairing pancreatic cell damage in mice. However, the injured pancreases in this study were not completely healed. This is because the statistical tests used for the P1, P2, and P3 implementations show no meaningful formation, and the doses used to heal pancreatic cell damage is considered insufficient.
Average Area of the Islets of Langerhans
Table 3 shows the average area of the islets of Langerhans in the K-, K+, P1, P2, and P3 groups.
Table 3 shows the average value of the area of Langerhans Island in the K-, K+, P1, P2, and P3 groups based on the study’s findings.
Average area of the islets of Langerhans in P1, P2, and P3 groups (mice with alloxan injection and administered with ethanol extract of cherry leaves in a dose of 300, 400, or 500 mg/kg bw) was larger than that in the K+ group (mice with only alloxan injection). The K- group had the largest average Langerhans islets area, while K+ had the smallest. Table 3 indicates that higher the extract dose used, larger the area of Langerhans islets recovered.
Table (3):
The average size of the island of Langerhans.
Group |
Area of Langerhans Island (μm) (Mean ± SD) |
|---|---|
K- |
47790,85 ± 9085,34 |
K+ |
34125,32 ± 5719,44 |
P1 |
38670,87 ± 8258,62 |
P2 |
39456,66 ± 1449,77 |
P3 |
43840,17 ± 1491,10 |
Histopathological Picture of the Mice’s Pancreas in Each Treatment Group
Negative Control Mice Group (K-)
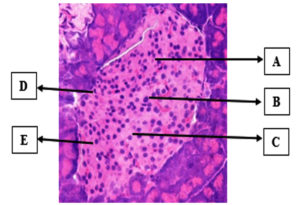
Figure 2 illustrates the histopathological section of the K- group that was not administered alloxan. In comparison to the other treatment groups, the K- group demonstrated the best or normal pancreatic condition. Although they were not exposed to alloxan, necrotic cells with an average damage of 0.4 ± 0.44 were discovered in some mice pancreases. This was caused by apoptosis, a process of cell destruction that can occur in healthy cells as well.
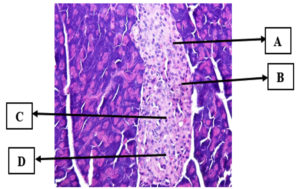
Figure 2. Pancreatic histopathology at 400x magnification (Hematoxylin-Eosin staining). (A) beta cells, (B) alpha cells, (C) acinar cells, (D) blood arteries, and (E) intercellular gaps are depicted (Personal Documentation)
Positive Control Mice Group (K+)
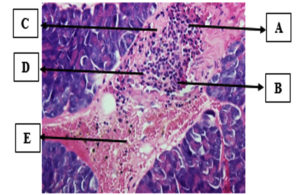
The K+ group of mice was stimulated with a dosage of 160 mg/kg bw/day alloxan without providing any treatment until the completion of the trial (Figure 3). The positive control group (K+) served as a reference for the histopathological image of a damaged pancreas. The pancreatic islets of Langerhans were substantially smaller in the K+ histopathological image than in the K- image.
Figure 3. Histopathology of the pancreas in the positive control (Hematoxylin-Eosin staining). (A) Beta cells, (B) cell necrosis, (C) inflammatory cells, (D) cell nucleus undergoes karyorrhexis (nuclear fragmentation) (D). Cell necrosis, pyknosis (nuclear shrinkage), and (E) cell degeneration in the form of cytoplasmic vacuoles (Personal documentation)
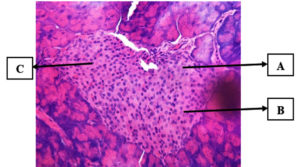
Administration of 300 mg/kg bw of Leaf Ethanol Extract (P1)
Alloxan was administered to a group of mice, followed by an administration of the ethanol extract of kersen leaves at a dose of 300 mg/kg bw (P1). This group had a higher mean damage score of 2.0 ± 0.70 than the other therapy groups, P2 and P3. Figure 4 shows the pancreatic histopathology in group P1. Compared with the positive control group, the P1 group showed an improvement; however, in the P1 group, the pancreas seemed smaller in size and β cells were less evident than in the K- group. The islets of Langerhans underwent cell renewal, resulting in a considerable increase in their area. The efficiency of cherry leaf ethanol extract in regenerating and repairing cells damaged by harmful chemicals such as alloxan was clearly demonstrated in the P1 group.
Figure 4. Group P1 pancreatic histology (hematoxylin-eosin staining). Description: (A). cells have grown in size (B). fewer cell clusters and more connective tissue (C). Pyknosis results from nuclear necrosis of cells (shrinkage of the nucleus). (D). Nuclear necrosis causes karyorrhexis (nuclear fragmentation) (Personal Documentation)
Administration of 400 mg/kg bw of Leaf Ethanol Extract (P2)
This group included mice that had been injected with alloxan and were subsequently administered the extract at a dose of 400 mg/kg bw/day (P2) for 14 days. It had a damage score of 1.8 ± 0.44. The histopathological scores in the P2 group were lower than those in the P1 and K+ groups. Figure 5 shows the pancreatic histological image of the P2 group.
Figure 5. Group P2 pancreatic histology (Hematoxylin-Eosin staining). (A) Cell necrosis, cell nucleus karyorrhexis (nuclear fragmentation), (B) decreased cell clusters and increased connective tissue (C). Necrosis of the cell nucleus, pyknosis (nuclear shrinkage) (D). Hypertrophied cells (Personal Documentation)
Langerhans islets damage was still observed in the P2 group because of necrosis of the cell nuclei undergoing karyorrhexis (nuclear fragmentation) and pyknosis (nuclear shrinkage), a decrease in the number of cell clusters, and an increase in the amount of connective tissue in the center of the Langerhans islets. Islets of Langerhans are still not normal. The P2 group underwent Langerhans repair, with regenerated cells defined by cell colonies and a more homogenous cell distribution.
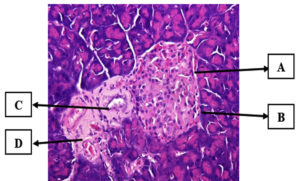
Administration of 500 mg/kg bw of Leaf Ethanol Extract (P3)
This group had a nearly normal histological image of the pancreas, and the islets of Langerhans began to improve microscopically, as shown in Figure 6. This improvement is assumed to be associated with the increase in extract dosage. The bioactive components in the ethanol extract of the cherry leaves were most likely responsible for the improvement observed in this group.
Figure 6. Group P3 pancreatic histology (Hematoxylin-Eosin staining). (A) Cell necrosis, cell nucleus undergoes karyolysis (nucleus destruction), (B) Normal pancreatic beta cells, (C) Intercellular cavity
Lactic Acid and Total Coliform Bacteria
In hyperglycemic mice, an ethanol extract of cherry leaves exhibited a favorable count of coliform bacteria in the cecum. Table 4 shows the total coliform count.
Table (4):
Total coliform counted using colony counter.
No |
Group |
Total Coliform (Log 10 CFU/g) |
|---|---|---|
1 |
K- |
2,08 ± 0,81a |
2 |
K+ |
4,37 ± 1,00b |
3 |
P1 |
2,74 ± 1,00a |
4 |
P2 |
2,51 ± 0,58a |
5 |
P3 |
2,40 ± 0,43a |
In the negative control group (K-), the total coliform count was 2.08 ± 0.81. There was a substantial difference in the number of coliforms in the cecum of the P3 and K+ groups. The total coliform count in the cecum of K+ mice was 4.37 ± 1.00, which was greater and substantially different from that of negative control mice (K-). The total coliform count in the cecum was 2.40 ± 0.43 in the P3 group. This was not substantially different from that in the K- group mice, which were only provided with food and water until the end of the trial. Furthermore, the overall quantity of coliform bacteria was reduced after the administration of the cherry leaf ethanol extract (Table 4).
Table (5):
Lactic acid bacteria (LAB) counted using colony counter.
No |
Group |
Total LAB (Log 10 CFU/g) |
|---|---|---|
1 |
K- |
3,61 ± 1,92a |
2 |
K+ |
1,08 ± 1,11b |
3 |
P1 |
2,86 ± 1,89a |
4 |
P2 |
2,93 ± 1,52a |
5 |
P3 |
3,51 ± 2,06a |
Table 5 shows how the application of the extract to the test animals at various dosages changed the total LAB. Hyperglycemic mice treated with cherry leaf ethanol extract exhibited an effective increase in LAB colonization: a higher dosage corresponded to a higher LAB population. There was a considerable difference in LAB population between the P3 and K+ groups. Administering an ethanol extract of cherry leaves can cause normal LAB colonization in the cecum of mice, as is evident by the lack of a significant difference between the total abundance of cecum LAB in the P3 and K- groups.
Glycemic homeostasis is the balance or regulation of blood glucose levels. Diabetes, naturally and significantly, impairs this condition. As its complications increase, it causes many problems, including retinopathy, nephropathy, and neuropathy, which are collectively known as chronic conditions. They are the primary players in comorbidity and ultimately death that are frequently associated with diabetes.23 Experimental animals are frequently fed an alloxan-laced diet to intentionally induce hyperglycemia.
Alloxan has two separate pathogenic effects: it selectively decreases glucose-induced insulin release by inhibiting glucokinase, the glucose sensor of β cells, and it induces insulin-dependent diabetes by generating ROS, culminating in the selective necrosis of β cells.24 For normal biological activities, ROS act as cell signaling molecules. However, the production of excessive ROS can damage various cellular organelles and impede their functions, eventually disrupting normal physiology. An imbalance between ROS generation and antioxidant defenses that protect cells has been linked to the etiology of several disorders, including cancer, asthma, pulmonary hypertension, and retinopathy.25
To compensate for the ROS generated in the present experiment, an antioxidant-rich ethanol extract was provided. Muntingia calabura, often known as the strawberry tree or Jamaican cherry, is a cherry tree that contains many flavonoids, which are naturally abundant antioxidants. This study demonstrated that the flavonoid content of Jamaican cherry leaves is higher than that of other plants (Table 6).
Table (6):
This research compared the falovonoid content of several plants.
Sources |
Solvent |
Extract Weight |
Flavonoid Level |
References |
|---|---|---|---|---|
Sirsak leave (Annona muricata L.) |
Etanol |
0,050g |
28,27 mg QE/g |
(Mukhriani et al., 2015) |
Sambiloto leave (Andrographis paniculata) |
Etanol |
1g |
34,469 mg QE/g |
(Kumala, 2017) |
Samsit leave (Gynura segetum) |
Etanol |
0,050g |
26,26 mg QE/g |
(Lau et al., 2019) |
Kersen leave (Muntingia calabura L.) |
Etanol |
0,025g |
19,428 mg QE/g |
(This study) |
Given the growing number of hyperglycemia patients and the frequent ineffectiveness of treatments, the priority is to develop new and more effective treatments based on natural compounds with high biological activity, such as flavonoids—plant compounds that are an important component of the human diet.26
Alloxan at a dose of 160 mg/kg bw (K+) damaged the islets of Langerhans, which are responsible for regulating blood glucose levels by releasing insulin. When these cells stop functioning, blood glucose levels increase, leading to hyperglycemia. The administration of cherry leaf ethanol extract not only inhibited ROS, the primary source of β cell destruction, but also stimulated cell repair. Furthermore, these natural chemicals protect cells against DNA damage caused by different carcinogens, including ROS, owing to their antioxidant properties.27 Thus, they reduce DNA damage and prevent genetic instability that leads to β cell destruction.28 Consequently, we observed a recovery of damaged β cells to their normal conditions after treatment with the ethanol extract of cherry leaves at 300, 400, and 500 mg/kg bw.
Hyperglycemia is caused by persistently increased glucose concentrations above the physiological range. It is widely established that type 2 diabetes-related hyperglycemia alters the gut microbiota.29 Thus, the present study aimed to determine whether an ethanol extract of Jamaican cherry leaves can repair pancreatic cell damage, as well as influence the total population of coliforms and LAB in hyperglycemic mice. Although this was a preliminary study, the results demonstrated an increase in the small intestine microbiome population, particularly in LAB, after the administration of cherry leaf ethanol extract (Table 5). The surge in LAB population moderated the growth of the overall coliform population (Table 4). This demonstrates that administering antioxidants as part of a regular diet not only avoids additional damage to pancreatic β cells but can also balance the good microbiome population and reduce pathogen overgrowth. This is consistent with the study by Riaz Rajoka et al.30 They reported that prolonged exposure to ROS can result in microbial dysbiosis, which can lead to various intestinal disorders including intestinal damage, colorectal malignancies, enteric infections, and inflammatory bowel disease. Food-derived antioxidant chemicals have recently been proven to protect individuals from intestinal oxidative stress by changing the composition of beneficial bacterial species in the gut.
In hyperglycemic mice with alloxan administration, an ethanol extract of cherry (Muntingia calabura L.) leaves improved the histological structure of the pancreas (cells in the islets of Langerhans). The histological image of the pancreas of healthy mice was not substantially different from that of the treatment group administered a dose of 500 mg/kg bw of the extract. Flavonoids, which are antioxidant substances found in cherry leaves, are hypothesized to activate the anti-ROS activity of alloxan. They balance the gut microbiota by reducing the increase in total coliforms and increasing LAB in hyperglycemic mice.
ACKNOWLEDGMENTS
The authors would like to express gratitude to everyone who helped, supported and contributed to this research.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
HB, AA and RPL conceptualized the study and applied methodology. ER and HB performed the experiments. HB, AA and RPL perform the evaluation and obtained the experimental results. AA and RPL drafted and reviewed the manuscript. All authors read and approved the final manuscript for publication.
FUNDING
None.
ETHICS STATEMENT
This study was approved by the Faculty of Medicine, University of Lampung, Indonesia with reference number 917/UN26.18/PP.05.02.00/2022.
DATA AVAILABILITY
The datasets generated and/or analysed during the current study are available from the corresponding author on reasonable request.
- Yang X, Zhang R, Jin T, et al. Stress Hyperglycemia Is Independently Associated with Persistent Organ Failure in Acute Pancreatitis. Dig Dis Sci. Published online 2021.
Crossref - Giri B, Dey S, Das T, Sarkar M, Banerjee J, Dash SK. Chronic Hyperglycemia Mediated Physiological Alteration and Metabolic Distortion Leads to Organ Dysfunction, Infection, Cancer Progression and Other Pathophysiological Consequences: An update on glucose toxicity. Biomed Pharmacother. 2018;107:306-328.
Crossref - Mouri Mi, Badireddy M. Hyperglycemia. StatPearls; 2022. https://www.ncbi.nlm.nih.gov/books/NBK430900/
- Szkudelski T. The mechanism of alloxan and streptozotocin action in B cells of the rat pancreas. Physiol Res. 2001;50(6):537-546.
- Chukwunonso Obi B, Chinwuba Okoye T, Okpashi VE, Nonye Igwe C, Olisah Alumanah E. Comparative Study of the Antioxidant Effects of Metformin, Glibenclamide, and Repaglinide in Alloxan-Induced Diabetic Rats. J Diabetes Res. 2016;2016:1635361.
Crossref - Abdulgani N, Trisnawati I, Hidayati D, Aisyatussoffi N, Arifiyanto A. Snakehead (Channa striata) Extracts Treatment towards Hyperglycemic Mice (Mus musculus) Blood Glucose Levels and Pancreatic Histology Structure. J Appl Environ Biol Sci. 2014;4(5):1-6.
- Song I, Patel O, Himpe E, Muller CJF, Bouwens L. Beta Cell Mass Restoration in Alloxan-Diabetic Mice Treated with EGF and Gastrin. PLoS One. 2015;10(10):e0140148.
Crossref - Wei Y-S, Hsiao Y-C, Su G-W, et al. Identification of Hyperglycemia-Associated Microbiota Alterations in Saliva and Gingival Sulcus. Arch Biochem Biophys. 2020;682:108278.
Crossref - Pan H, Li Z, Xie J, et al. Berberine Influences Blood Glucose via Modulating the Gut Microbiome in Grass Carp. Front Microbiol. 2019;10.
Crossref - Al-Ishaq RK, Abotaleb M, Kubatka P, Kajo K, Büsselberg D. Flavonoids and Their Anti-Diabetic Effects: Cellular Mechanisms and Effects to Improve Blood Sugar Levels. Biomolecules. 2019;9(9):430.
Crossref - Uyun S, Damayanti AA, Azhar F. The Effect of Cherry Leaves Extract (Muntingia calabura) on Growth Performance of White Shrimp (Litopenaeus vannamei). J Biol Trop. 2021;21(1):262-270.
Crossref - Pratiwi ED, Dewi NP. Screening of Phytochemical Secondary Metabolites of Muntingia Calabura: a Potential as Hepatoprotector. J Fundam Appl Pharm Sci. 2022;2(2):59-65.
Crossref - Chen C-H. Xenobiotic Metabolic Enzymes: Bioactivation and Antioxidant Defense. Springer Nature Switzerland AG; 2020.
Crossref - Chang CC, Yang MH, Wen HM, Chern JC. Estimation of Total Flavonoid Content in Propolis by Two Complementary Colometric Methods. J Food Drug Anal. 2002;10(3):178-182.
Crossref - Mutiarahmi CN, Hartady T, Lesmana R. Use of Mice as Experimental Animals in Laboratories That Refer to The Principles of Animal Welfare: a Literature Review. Indones Med Veterinus. 2021;10(1):134-145.
Crossref - Nurfitri WA, Widiastuti EL, Nurcahyani E. Efek Ekstrak Metanol Daun Jeruju (Acanthus Ilicifolius L.) Serta Buah Jeruju dan Taurin Dalam Menurunkan Kadar Glukosa Darah dan Kolesterol Serta Fertilitas Mencit Jantan (Mus Musculus) yang Diinduksi Aloksan. Obat Indones Ke 55. Published online 2019. http://repository.lppm.unila.ac.id/12751/
- Aligita W, Susilawati E, Sukmawati IK, Holidayanti L, Riswanti J. Antidiabetic activities of Muntingia calabura L. leaves water extract in type 2 diabetes mellitus animal models. Indones Biomed J. 2018;10(2):165-170.
Crossref - Ifada R, Hermayanti D, NurHasan YT. Pengaruhekstrak Daun Lidah Mertua (Sansevieria trifasciata var. laurentii ) Terhadap Kadar Hemoglobin Tikus Puith Jantan (Rattus Norvegicus strain Wistar) Yang Diinduksi Timbal Per-Oral. Saintika Med. 2018;12(2):68.
Crossref - Sari AK, Noverda A. Penetapan Kadar Fenolik Total dan Flavonoid Total Ekstrak Beras Hitam (Oryza Sativa L) dari Kalimantan Selatan. J Ilm Ibnu Sina. 2017;2(2):327-335.
- Dharma IGBS, Berata IK. Studi Histopatologi Pankreas Tikus Putih (Rattus novergicus) yang Diberi Deksametason dan Suplementasi Vitamin E. Indones Med Veterinus. 2015;4(3):257-266.
- Irwan A, Wicaksono F. Perbandingan pengukuran luas daun kedelai dengan metode gravimetri , regresi dan scanner Comparations of soybean ’s leaf area measurement using gravimetry , regression , and scanning. J Kultiv. 2017;16(3):425-429.
- Bintari NWD, Parwati PA. Suplementasi Soya Yogurt Untuk Perbaikan Kelimpahan Mikroba Sekum (Bakteri Asam Laktat Dan Escherichia coli) Tikus Diabetes Soya Yogurt. Metamorf J Biol Sci. 2021;8(1):35.
Crossref - Ighodaro OM, Adeosun AM, Akinloye OA. Alloxan-Induced Diabetes, a Common Model for Evaluating The Glycemic-Control Potential of Therapeutic Compounds and Plants Extracts in Experimental Studies. Medicina (B Aires). 2017;53(6):365-374.
Crossref - Lenzen S. The mechanisms of Alloxan- and Streptozotocin-Induced Diabetes. Diabetologia. 2008;51(2):216-226.
Crossref - Auten RL, Davis JM. Oxygen Toxicity and Reactive Oxygen Species: The Devil Is in the Details. Pediatr Res. 2009;66(2):121-127.
Crossref - Korga-Plewko A, Michalczyk M, Adamczuk G, et al. Apigenin and Hesperidin Downregulate DNA Repair Genes in MCF-7 Breast Cancer Cells and Augment Doxorubicin Toxicity. Molecules. 2020;25(19):4421.
Crossref - Arifiyanto A, Setyaningrum E, Nukmal N, Aeny TN. Short Communication: In vitro Antimicrobial and Antimalarial Screening of a Crude Extract of Streptomyces sp. AB8 Isolated from Lapindo Mud Volcano Area, Sidoarjo, Indonesia. Biodiversitas. 2021;22(7):2817-2823.
Crossref - Dumanović J, Nepovimova E, Natić M, Kuča K, Jaćević V. The Significance of Reactive Oxygen Species and Antioxidant Defense System in Plants: A Concise Overview. Front Plant Sci. 2021;11.
Crossref - Kesh K, Mendez R, Abdelrahman L, Banerjee S, Banerjee S. Type 2 Diabetes Induced Microbiome Dysbiosis Is Associated with Therapy Resistance in Pancreatic Adenocarcinoma. Microb Cell Fact. 2020;19(1):75.
Crossref - Riaz Rajoka MS, Thirumdas R, Mehwish HM, et al. Role of Food Antioxidants in Modulating Gut Microbial Communities: Novel Understandings in Intestinal Oxidative Stress Damage and Their Impact on Host Health. Antioxidants (Basel, Switzerland). 2021;10(10):1563.
Crossref
© The Author(s) 2022. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.