ISSN: 0973-7510
E-ISSN: 2581-690X
Foodborne pathogens are the main threat and cause of food poisoning. The majority of food infections have been related to the biofilm formation of foodborne pathogens in the food industry. Shewanella putrefaciens (KX355803, GRD 03), a Gram-negative pathogen isolated from mackerel fish, was identified and recognized as a food spoilage bacterium and a strong biofilm producer. The adhesion or attachment ability of Shewanella putrefaciens was determined on steel, plastic, glass, PVC and wood. NB (Nutrient broth), LB (Luria-Bertani broth), TSB (Tryptic soy broth) and BHI (Brain heart infusion broth) were enriched with glucose and shows optimum for bacterial adhesion. In the microtiter plate method (MTP), the strong attachment was observed at 48 and 72 hours of incubation and significant differences were obtained at p < 0.05. As the incubation period increases, the OD value (Optical density) of samples also increase. Biofilm formation is the major cause cross-contamination, and shows resistance to certain disinfectants, which leads to environmental stress tolerance. This study suggested with optimum biofilm production of isolate from fish by using glucose enriched media on different substrates, also comparing different growth media provide a detailed idea about biofilm-forming ability at different incubation time intervals.
Biofilm, Foodborne pathogen, Optical density, Disinfectant, Substrate
Shewanella putrefaciens play a predominant role in food spoilage processes, mainly correlated with spoilage of frozen fish and meat products.1,2 This pathogen has a potential to live in freshwater habitation.1 Shewanella putrefaciens belongs to Shewanellaceae family. Pseudomonas putrefaciens was classified in 1985 and finally known as Shewanella putrefaciens.3 The directly attached bacterial colonies or communities on a specific surface initiate the formation of biofilm. Biofilm producers are enclosed in an extracellular matrix (EPS) with the ability to manage various phenotypic behaviours like attachment of bacterial cells that leads to bacterial gene transcription. Within biofilm cell communities, bacterial cells communicate with each other via some chemically produced signaling molecules known as Autoinducers (AI), and the process stated as Quorum sensing (QS). Majority of foodborne infectious diseases are originated due to biofilm formation.4 Biofilm formation is a major issue in food processing industries. Cross-contamination is the main reason for biofilm producers, when food moves through some unhygienic surfaces and the surrounding air develops the appearance of foodborne pathogens, leads to biofilm production and contamination. Biofilm process required different stages, involved, attachment of bacterial cells, maturation and dispersion.5 To cope with this bacterial settlement in biofilm, a serious hygiene approach is mandatory, which comprises disinfection and proper cleaning. Nowadays, these biofilm producers have the potentiality to fight against such disinfectants.6,5 Briedier et al.7 discussed about some foodborne pathogens mainly involved in biofilm production and adhesion on different substrates in the food industry.7 Therefore, this present study targets the isolation and identification of foodborne pathogen Shewanella putrefaciens from fish sample and its biofilm formation capacity on different substrates.
Fish samples (Mackerel fish gut portion) were procured from a local fish peddler in Coimbatore, Tamil Nadu. Using a clean and sterile blade, the samples were gently chopped and smashed with peptone water, which is used to enrich the media for bacterial growth (Peptone -10g/mL, Sodium chloride – 5g/mL, HiMedia, India). The unpurified grounded sample was sieved through Whatman filter paper grade No. 1 (110nm, Sigma-Aldrich). Finally, the filtrate was collected and processed for serial dilution. The filtrate was serially diluted from 10-2 to 10-8 dilutions. Each of them were spread plated onto Nutrient agar plate and kept for 24-48 hours of incubation (37°C). The single isolated colonies were subcultured individually on Nutrient agar plate and maintained in glycerol stock at -21°C.
Biochemical assays
For morphological identification Gram staining was performed.8 Different biochemical tests were done and fish pathogen was recognized according to Saeed et al.10 Biochemical tests involve indole production, Methyl-Red test and Voges-Proskauer test (MR-VP), citrate utilization, catalase. oxidase, urease, KOH (Potassium hydroxide) and starch hydrolysis. KOH test an another test to recognize the Gram-negative bacteria. The results were recorded after each assays and further molecular identification was carried out.
DNA sample preparation and 16S rRNA PCR amplification
DNA isolation was performed according to Giacommazzi et al.9 The presence of DNA was proved using 0.8% of Agarose gel electrophoresis. For PCR amplification of DNA, BAC forward and BAC universal primers were used. Primer was purchased from Integrated DNA Technologies (IDT), Bangalore, India. Amplified PCR product was confirmed by 0.8% agarose gel electrophoresis using a 500bp ladder (Helini, India).
BAC Forward- 5’-AGA GTT TGG ATG GCT CAG-3’
BAC Universal Reverse Primer – 5’-CGG TTA CCT TGT TAC GAC-3’
The amplified product was forwarded to Eurofins, Scientific India, Pvt Ltd for 16S rRNA gene sequencing. The organism was identified using phylogenetic analysis and FASTA sequences were obtained, submitted in NCBI, GenBank.
Biofilm formation techniques
Tube assay method
Four different growth media such as NB, LB, TSB, BHI (HiMedia, India) and their glucose enriched forms NB1, LB1, TSB1 and BHI1 were used to enhance the bacterial growth and to check the biofilm-forming ability of isolated fish pathogen S. putrefaciens GRD 03. The appropriate growth temperature of this foodborne pathogen was obtained at 37°C under laboratory conditions.
Media composition : Nutrient broth (NB)- Peptone-5g/L, Sodium chloride- 5g/L, Beef extract- 1.5g/L, Yeast extract- 1.5g/L (pH= 7.4±0.2)
Luria-Bertani (LB) broth- Casein enzymic hydrolysate-10g/L, Yeast extract-5g/L, Sodium chloride-10g/L (Final pH= 7.4±0.2)
Brain Heart Infusion (BHI) broth- Proteose peptone -10g/L, Dextrose-2g/L, Sodium chloride-5g/L, Disodium phosphate-2.5gL (pH = 7.4±0.2)
Tryptic soy broth (TSB)- Pancreatic digest of casein-17g/L, Peptic digest of soybean-3g/L, Glucose-2.5g/L, Dipotassium phosphate-2.5g/L (pH= 7.3±0.2)
Modified forms of media such as NB1, LB1 TSB1 and BHI1 were supplemented with 25g/L of glucose (Himedia, India) to attain the maximum growth of bacteria.
Bacterial culture (100µL) was suspended in 10mL of NB, LB, TSB, BHI, NB1, LB1 TSB1 and BHI1 media, tubes were kept for overnight incubation in a shaking incubator. After incubation, tubes were cleansed with phosphate buffer saline (pH-7, HiMedia) and air-dried and subjected to crystal violet staining (0.1%) and extra stain was washed with distilled water and dried in an inverted position.4
Congo Red Agar plate method (CRA)
The isolated culture was streaked onto sterile CRA agar (HiMedia) plate and subjected for overnight incubation at 37°C. Significant production of black or brown colonies was considered as positive result.4
Microtiter plate method biofilm formation (MTP)
In this method, 96-wells flat bottomed plate was used. Four different incubation periods were optimized such as 24, 48, 72 and 96. Fifteen (15µL) of incubated broth culture and 225µL of different media were added to MTP plate with 96-wells and incubated in a rocking table. The culture filtrate was discarded and washed twice with sterilized water and stained with crystal violet (0.1%), finally, the plate was incubated for 3 minutes at room temperature. Leftover dye was cleansed with distilled water and resuspended the wells with 200µL of glacial acetic acid (33%, HiMedia). The absorbance of MTP was carried out at 490nm.11
Biofilm formation on different substrates using crystal violet binding assay
This method is appropriate for the determination of the biofilm-forming potential of foodborne pathogens on selected substrates such as glass, Polyvinyl chloride (PVC), plastic, wood and steel. The substrates were cleaned and washed with sterile water and detergent and dried. After proper drying substrates were aseptically transferred into different broth media and incubated for 24, 48 and 72 hours at 37°C. After incubation periods, each substrate was removed carefully and subjected to crystal violet staining. Glass, PVC, steel, plastic and wood were washed with sterilized water and cleansed with methanol to fix the excess bacteria and again stained for 3 minutes. Again washed with tap water, and resuspended in 2.5mL of 33% glacial acetic acid (HiMedia). The OD (optical density) was calculated at 630nm 12-14.
Statistical analysis
Experiments were conducted in triplicate values. Statistical analysis was determined by Graphpad Prism 8.0 and the results were calculated as mean ± standard deviation (Mean ± SD). Significant differences of samples were exhibited by one-way-analysis of variance (ANOVA) with a significance level p < 0.05.
Isolation, biochemical and molecular identification of fish pathogen
A total of 10 bacterial strains were isolated from mackerel fish (gut). Among 10 isolates one culture named GRD 03 was recognized as Gram-negative bacteria after morphological examination. Different biochemical tests (Table 1) were performed for GRD 03 strain and found to be positive results for catalase, oxidase, urease and indole production and all the other tests results indicated negative. This Gram-negative pathogen was yellow in colour and the accurate growth temperature was 37°C for 24 to 48 hours of optimum incubation in Nutrient agar.
Table (1):
Biochemical assays performed for the isolated fish pathogen, + Present, – Absent.
Gram staining |
_ |
Indole production |
+ |
MR |
_ |
VP |
_ |
Citrate utilization |
_ |
Catalase |
+ |
Urease |
+ |
Oxidase |
+ |
KOH |
+ |
Starch hydrolysis |
_ |
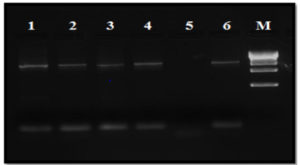
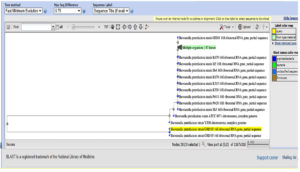
Bacterial DNA was obtained by 0.8% agarose gel electrophoresis and PCR conditions were optimized and amplified product showed at 1500 bp. Amplified product after 16S rRNA sequencing was recognized using BLAST sequence (Fig. 2) and phylogenetic tree (Fig. 3) analysis. GRD 03 shows 94% similarity with Shewanella putrefaciens. The FASTA sequences were deposited in NCBI, GenBank under the accession number KX355803.
Fig. 2. One to 6 Lanes represents genomic DNA isolated from GRD 03 (Shewanella putrefaciens, KX355803) on 0.8% agarose and Lane 5 was left without any addition of sample. M – Denote 500 bp DNA ladder
The phylogenetic tree produces 16S ribosomal RNA partial sequence with 94% similarity with S. putrefaciens. Phenotypic and genotypic identification procedures have been considered as a primary tool for the classification of a bacterium. Due to certain limitations, molecular techniques are also very mandatory to identify a pathogen.15 Certain studies provided some tools for the identification of fish pathogen by combining molecular and 16S rRNA tools with cost-efficiency.16 Another study by Beaz-Hidalgo et al.17 isolated three different Shewanella strains using similar ERIC-PCR profiles.17 16S rNA gene sequencing is not sufficient to confirm the bacterial position in phylogenetic tree, also need gyrB gene also need to be sequence to confirm.18-20 Shewanella putrefaciens is directly involved in various health disorders and can cause Shewanellosis in humans. Few studies have reported that, Shewanella putrefaciens required Sodium chloride for their potential growth and activity.21-24
Biofilm production assays
Foodborne pathogens are the main reason for biofilm formation and food contamination.25 In tube method, S. putrefaciens (GRD 03) showed biofilm formation after 24 hours of incubation. The tube method (Fig. 4) has been considered as an accurate method to detect the biofilm production of pathogens.25 Shewanella putrefaciens resulted strong biofilm production in NB, LB, NB 1, and LB 1 after overnight incubation. Comparatively, BHI and TSB shows better biofilm production on glass tube when compared to BHI1 and TSB1 modified media. Biofilm formation is strongly associated with different behaviors like medium, environment and mainly surface.26 According to McEldowney and Fletcher,27 hydrophobic, electrostatic properties, environmental changes, nutrients and pH were involved in the adhesion potential of subsequent bacteria.27
Fig. 4. Eight tubes with biofilm production in eight media (NB, LB, BHI, TSB, NB 1, LB 1, BHI 1, TSB 1) (from left side to right side tubes)
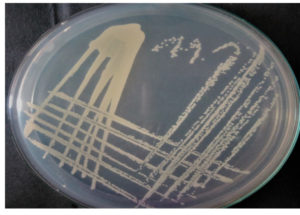
Fig. 5. Indicates the strong biofilm production of Shewanella putrefaciens, GRD 03 KX355803, producing black colour bacterial colonies.
Congo red agar (CRA) assay showed black colonies as a result of biofilm producer Shewanella strain, resulted that GRD 03 was capable of producing biofilm (Fig. 5). Congo red agar method is performed as a basic screening technique for observing biofilm-producing bacteria in the laboratory.28
In microtiter plate (MTP) method, 24, 48, 72 and 96 hours were optimized for the biofilm formation of test organism, and 48, 72 and 96 hours were optimum for strong attachment of biofilm-producing bacteria. But according to absorbance value, 96 hours of incubation resulted better biofilm production. NB, LB, BHI, NB1, LB1, and BHI1 were resulted as efficient for optimum biofilm production at 48, 72 and 96 hours of time intervals (Table 2). BHI and TSB were not significant (p < 0.080) at 24 hours of biofilm formation, but shows significance (p < 0.004) at 48, 72 and 96 hours of incubation time. Nutrient broth and LB broth resulted significance at 24 and 48 hours of intervals (p < 0.001) also shows 48, 72 and 96 hours (p < 0.001) were optimum for S. putrefaciens biofilm production. NB1 was obtained as significant at 48, 72 and 96 hours of incubation (p < 0.002, p < 0.001, p < 0.001 respectively). For LB1 media, 48, 72 and 96 hours were found to be significant and showed p < 0.006, p < 0.00, and p < 0.001. BHI1 and TSB1 appeared as significant at 48, 72 and 96 hours and shows p < 0.004, p < 0.001, and p < 0.002 sequentially. Certain studies revealed that, attachment and biofilm production abilities were directly connected to glucose concentration present in specific media. 29,30.
Table (2):
Biofilm formation of Shewanella putrefaciens at different incubation period using crystal violet binding assay.
Media |
24 hours |
48 hours |
72 hours |
96 hours |
|---|---|---|---|---|
NB |
0.98 ± 0.02c |
1.72 ± 0.07d,,e |
2.10 ± 0.00d,e |
3.11 ± 0.03e,f |
LB |
1.02 ± 0.03c |
1.34 ± 0.13b,c |
1.95 ± 0.06e |
3.32 ± 0.00e,f |
BHI |
0.95 ± 0.05 |
1.66 ± 0.00d,e |
2.00 ± 0.00c,d |
2.99 ± 0.00d,,e,f |
TSB |
0.89 ± 0.00 |
1.01 ± 0.00d,e |
1.99 ± 0.00e |
3.21 ± 0.14e,f |
NB 1 |
1.11 ± 0.00b,c |
1.51 ± 0.06d,e |
2.11 ± 0.00d,e,f |
3.16 ± 0.06d,e,f |
LB 1 |
1.37 ± 0.07c |
1.83 ± 0.23c,d |
2.14 ± 0.00f |
3.44 ± 0.07d,f |
BHI 1 |
0.99 ± 0.00c |
1.07 ± 0.10b,c |
1.93 ± 0.06d,e |
3.00 ± 0.08e,f |
TSB 1 |
0.72 ± 0.07d |
1.22 ± 0.00d,e |
1.71 ± 0.07c,f |
3.11 ± 0.03d,e,f |
Biofilm adhesion capacity of foodborne pathogen on different substrates
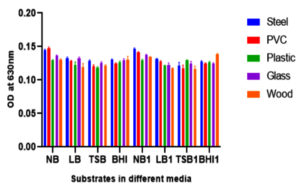
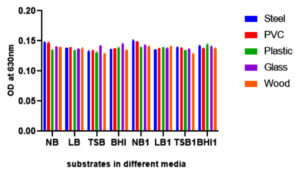
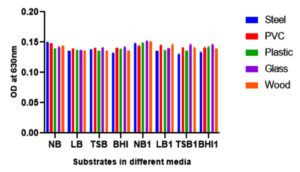
Bacterial biofilm has been recognized as a main health threat in clinical industry. Crystal violet staining method was performed to obtain the biofilm attachment on different substrates. The main detrimental issue to human health is bacterial biofilm formation. These kinds of microorganisms are able to grow on different environmental conditions in extracellular matrix. In this study different substrates like glass, PVC, plastic, steel and wood were used to observe the biofilm formations of foodborne pathogen Shewanella putrefaciens at 24 hours, 48 hours and 72 hours of total incubation periods in different medium (Fig. 6, Fig. 7 and Fig. 8). As incubation time increases, the rate of biofilm formation on substrates also increases. Shewanella putrefaciens also resulted better attachment on steel, PVC, and glass in Nutrient broth enriched (NB1) and nutrient broth media after 24 hours. Wood showed best attachment in Brain heart infusion enriched media at 24 hours of incubation. After 48 hours of incubation, steel resulted as best substrate for biofilm formation in NB and NB1 (glu). As the glucose concentration in the culture media increases, the bacterial growth and biofilm formation on substrates also increase. There are certain factors that trigger biofilm production of pathogenic bacteria such as nutriens, pH, temperature, salt and sugar concentrations etc.30 So here, by increasing sugar concentration in NB may influence the bacterial adhesion on substrates. In 24 hours, S. putrefaciens showed higher attachment on steel, PVC and glass, also biofilm attachment was maximum on PVC and steel in nutrient broth media. The glucose enriched BHI1 was found to be optimum for the bacterial attachment on wood surface after 24 hours of biofilm formation. The OD values were obtained as higher on steel and PVC substrates after 48 and 72 hours of incubation. 72 hours of incubation was recognized as the optimum time period for strong biofilm production and Nutrient broth modified media enhances more biofilm formation on all substrates. The results of this study represented that, the biofilm-forming ability of bacterial culture was significantly different among different substrates. Recent studies indicated that, bacterial communities can develop biofilm on PVC and other plastics (PE and PP) clustered in two different groups and plastic substrate influence the total structural changes in biofilm and also stimulate metabolic functions of biofilm formations.31 Formation, growth and maturation of biofilms were the primary environmental factors to infect microbial community.32 The substrates characteristics like dryness, hydrophobicity, etc. affect the capability of bacterial cells to adhere to the surface.33,34 Steel (hydrophilic) is widely used in food processing, construction of equipment’s and food packaging (canned foods) purposes. In our study, S. putrefaciens shows better biofilm adhesion on hydrophilic substrate like steel.
Fig. 6. Shows biofilm formation of GRD 03 Shewanella putrefaciens on different solid surfaces in eight different media after 24 hours of incubation (Nurient broth (NB), Luria-Bertani (LB), Brain Heart Infusion broth (BHI), Tryptic Soy Broth (TSB) and glucose enriched forms (NB 1, LB 1, BHI 1 and TSB 1)
Fig. 7. Biofilm production of Shewanella putrefaciens GRD 03 on different substrates after 48 hours of incubation in eight different media
In conclusion, this study explored and investigated the biofilm-producing effect of fish pathogen Shewanella putrefaciens isolated from mackerel fish gut. The preliminary techniques of biofilm formation such as tube assay and congo red agar method revealed, the strain to be good biofilm producers. The microtiter plate method showed better biofilm production after 48, 72 and 96 hours of incubation. The bacterial adhesion was checked using different substrates such as glass, steel, PVC, plastic and wood at 24, 48, and 72 hours of different intervals. In this method, S. putrefaciens was exhibited optimum biofilm attachment on steel and PVC. By the addition of glucose in media influences the bacterial growth density and provides good attachment. Glucose enriched media of Nutrient broth, Luria-Bertani broth, Tryptic soy broth and Brain heart infusion broth were ideal for bacterial attachment and growth. Shewanella putrefaciens (GRD 03, KX355803) adhesion ability provides some potential pathogenic nature of foodborne bacteria. Demonstrated results are very crucial for the diagnosis of various pathogenic bacterial infections in food and health industry. Further work is mandatory to investigate to prevent fish pathogenicity in marine and fresh water environment. The concept of this work may be convenient and useful in future to synthesise antimicrobial agents against foodborne pathogen diseases.
ACKNOWLEDGMENTS
The authors would like to express their sincere thanks to Dr. R. Suganthi for providing assistance on different methods. The authors are also grateful to Dr. G.R. Damodaran College of Science, School of Biotechnology for providing a high class infrastructure to complete the research work.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
None.
ETHICS STATEMENT
Not applicable.
AVAILABILITY OF DATA
The datasets generated and analysed during the current study are available from the corresponding author on reasonable request.
- Pazdzior E. Shewanella putrefaciens – A new opportunistic pathogen of freshwater fish. J Vet Res. 2016;60(4):429-434.
Crossref - Borch E, Kant-Muermans ML, Blixt Y. Bacterial spoilage of meat and cured meat products. Int J Food Microbiol. 1996;33(1):103-120.
Crossref - MacDonell MT, Colwell RR. Phylogeny of the Vibrionaceae, and recommendation for two new genera, Listonella and Shewanella. Syst and Appl Microbiol. 1985;6(2):171-182.
Crossref - Hassan A, Usman J, Kaleem F, Omair M, Khalid A, Iqbal M. Evaluation of different detection methods of biofilm formation in the clinical isolates. Braz J Infect Dis. 2011;15(4):305-311.
Crossref - Brooks J D, Flint S H. Biofilms in the food industry: problems and potential solutions. Int J of Food Sci Technol. 2008;43(12):2163-2176.
Crossref - Bridier A, Briandet R, Thomas V, Dubois-Brissonnet F. Resistance of bacterial biofilms to disinfectants: a review. Biofouling. 2011;27(9):1017-1032.
Crossref - Bridier A, Sanchez-Vizuete P, Guilbaud M, Piard JC, Naitali M, Briandet R. Biofilm-associated persistence of food-borne pathogens. Food Microbiol. 2015;45(Pt B):167-178.
Crossref - Gram C. The differential staining of Schizomycetes in tissue sections and in dried preparations. Fortschitte der Medicin. 1884;2(6):185-189. https://scholar.google.com/scholar?hl=en&as_sdt=
0%2C5&q=Gram+C.+The+differential+staining+of+Schizomycetes+in++
tissue+sections+and+in+dried+preparations.+Fortschitte++der+Medicin.+
1884%3B2%286%29%3A185-189&btnG=#:~:text=The%20differential
%20staining%20of%20Schizomycetes%20in%20tissue%20sections%20
and%20in%20dried%20preparations - Giacomazzi S, Leroi F, Joffraud J-J. Comparison of three methods of DNA extraction from cold-smoked salmon and impact of physical treatments. J Appl Microbiol. 2005;98(5):1230-1238.
Crossref - Saeed MO, Alamoudi MM, Al-Harbi AH. A Pseudomonas associated with disease in cultured rabbit fish Siganus rivulatus in the Red Sea. Dis of Aqua Org. 1987;3(3):177-180.
Crossref - Darwish SF, Asfour HA. Investigation of biofilm-forming ability in Staphylococci causing bovine mastitis using phenotypic and genotypic assays. Sci World J. 2013;2013:378492.
Crossref - Adetunji, VO, Isola TO. Crystal violet binding assay for assessment of biofilm formation by Listeria monocytogenes and Listeria spp. on wood, steel and glass surfaces. Global Veterinaria. 2011;6(1):6-10.
- Pawar DM, Rossman ML, Chen J. Role of curli fimbriae in mediating the cells of enterohaemorrhagic Escherichia coli to attach to abiotic surfaces. J Appl Microbiol. 2005;99(2):418-425.
Crossref - Stepanovic S, Cirkovic I, Ranin L, Svabic-Vlahovic M. Biofilm formation by Salmonella spp. and Listeria monocytogenes on plastic surface. Lett Appl Microbiol. 2004;38(5):428-432.
Crossref - Khor WC, Puah SM, Tan JA, Puthucheary SD, Chua KH. Phenotypic and Genetic Diversity of Aeromonas Species Isolated from Fresh Water Lakes in Malaysia. PLoS One. 2015;10(12):e0145933.
Crossref - Sebastião F A, Furlan L R, Hashimoto D T, Pilarski F. Identification of bacterial fish pathogens in Brazil by direct colony PCR and 16S rRNA gene sequencing. Adv Microbiol. 2015;5(06):409.
Crossref - Beaz-Hidalgo R, Agueria D, Latif-Eugenin F, Yeannes MI, Figueras MJ. Molecular characterization of Shewanella and Aeromonas isolates associated with spoilage of Common carp (Cyprinus carpio). FEMS Microbiol Lett. 2015;362(1):1-8.
Crossref - Satomi M, Oikawa H, Yano Y. Shewanella marinintestina sp. nov., Shewanella schlegeliana sp. nov. and Shewanella sairae sp. nov., novel eicosapentaenoic-acid-producing marine bacteria isolated from sea-animal intestines. Int J Syst Evol Microbiol. 2003;53(Pt 2):491-499.
Crossref - Satomi M, Vogel BF, Venkateswaran K, Gram L. Description of Shewanella glacialipiscicola sp. nov. and Shewanella algidipiscicola sp. nov., isolated from marine fish of the Danish Baltic Sea, and proposal that Shewanella affinis is a later heterotypic synonym of Shewanella colwelliana. Int J Syst Evol Microbiol. 2007;57(Pt 2):347-352.
Crossref - Abbott SL, Cheung WK, Janda JM. The genus Aeromonas: biochemical characteristics, atypical reactions, and phenotypic identification schemes. J Clin Microbiol. 2003;41(6):2348-2357.
Crossref - Bowman J P. Genus IV. Methylophaga. Bergey’s manual of systematic bacteriology: Springer, New York, NY, USA. 2005.
- Gilardi G L. Cultural and biochemical aspects for identification of glucose-nonfermenting gram-negative rods. Microbiology Series [MICROBIOL. SER.]. 1985.
- Khashe S, Janda JM. Biochemical and pathogenic properties of Shewanella alga and Shewanella putrefaciens. J Clin Microbiol. 1998;36(3):783-787.
Crossref - Moule AL, Wilkinson SG. Composition of lipopolysaccharides from Alteromonas putrefaciens (Shewanella putrefaciens). Microbiol. 1989;135(1):163-173.
Crossref - Zhao X, Zhao F, Wang, J, Zhong N. Biofilm formation and control strategies of foodborne pathogens: food safety perspectives. RSC advances. 2017;7(58):36670-36683.
Crossref - Donlan RM, Costerton JW. Biofilms: survival mechanisms of clinically relevant microorganisms. Clin Microbiol Rev. 2002;15(2):167-193.
Crossref - McEldowney S, Fletcher M. Variability of the influence of physicochemical factors affecting bacterial adhesion to polystyrene substrata. Appl Environ Microbiol. 1986;52(3):460-465.
Crossref - Rewatkar AR, Wadher BJ. Staphylococcus aureus and Pseudomonas aeruginosa-Biofilm formation Methods. J Pharm Biol Sci. 2013; 8(5): 36-40.
Crossref - Stanley NR, Lazazzera BA. Environmental signals and regulatory pathways that influence biofilm formation. Mol Microbiol. 2004;52(4):917-924.
Crossref - Pan Y, Breidt F Jr, Gorski L. Synergistic effects of sodium chloride, glucose, and temperature on biofilm formation by Listeria monocytogenes serotype 1/2a and 4b strains. Appl Environ Microbiol. 2010;76(5):1433-1441.
Crossref - Pinto M, Langer TM, Huffer T, Hofmann T, Herndl GJ. The composition of bacterial communities associated with plastic biofilms differs between different polymers and stages of biofilm succession. PLoS One. 2019;14(6):e0217165.
Crossref - Miao L, Wang C, Adyel TM, et al. Microbial carbon metabolic functions of biofilms on plastic debris influenced by the substrate types and environmental factors. Environ Int. 2020;143:106007.
Crossref - Van Houdt R, Michiels CW. Biofilm formation and the food industry, a focus on the bacterial outer surface. J Appl Microbiol. 2010;109(4):1117-1131.
Crossref - Lee JS, Bae YM, Lee SY, Lee SY. Biofilm Formation of Staphylococcus aureus on Various Surfaces and Their Resistance to Chlorine Sanitizer. J Food Sci. 2015;80(10):M2279-M2286.
Crossref - Di Ciccio P, Vergara, A, Festino, AR, Paludi D, Zanardi E, Ghidini S, Ianieri A. Biofilm formation by Staphylococcus aureus on food contact surfaces: Relationship with temperature and cell surface hydrophobicity. Food Cont. 2015;50:930-936.
Crossref
© The Author(s) 2022. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.