ISSN: 0973-7510
E-ISSN: 2581-690X
The bacterium Bacillus thuringiensis widely used as bio-pesticide. The specific toxic activity to insects and another microbe(s) regarding the existing of crystals that have different morphologies, sizes, numbers and compositions, as Bt strains the crystals have more different proteins, which contain encoded Cry distinct genes. In this work we isolated 140 Bacillus thuringiensis isolates from 68 soil samples from 19 local areas in Punjab. Colonies examination was done by microscope for toxin production. Bacillus thuringiensis isolates showed a typical colony morphology, predominantly off white to creamish with irregular margins. Based on certain biochemical characterstics they were confirmed to be strains of Bacillus thuringiensis. Bt strains harboring crystalline inclusions that might have potential for use as bioinsecticidal agent was done under phase contrast microscope and Scanning electron microscope. Based on preliminary screening with phase contrast microscopy, its parasporal crystals have unsual shapes. Bt isolates were classified into bi-pyramidal crystals, pyramidal, cuboidal, amorphous and round crystal protein inclusions. After preliminary screening with phase contrast microscopy, Scanning electron microscopy confirmed the surface view of rod shaped bacterial cells found in chains and cluster arrangement. Bt isolated tended to have sub-terminal spores.
Bacillus thuringiensis, crystal, phase contrast, scanning electron microscopy.
One of the few alternatives to chemical compounds for biological control is Bacillus thuringiensis (Bt). This facultative aerobic, Gram-positive, spore-forming saprophyte soil bacterium, has been successfully used as a biological insecticide over the last 6 decades and constitutes 95% of all commercial bio-insecticides, due to its high specificity, safety and effectiveness in the control of wide spectrum of human disease vectors and agriculture-pests (Schunemann etal 2014).
Bacterial spores are presently a subject of intense study, including cytological investigation, which makes it possible to establish connections between the biological and physicochemical properties of the spores and the features of their specific surface structures. Spores are the best studied of the dormant forms (DF), which are used for the propagation and preservation of a species (Oestergaard et al 2007). Spore-formation enables Bt to survive in harsh environments resulting in a ubiquitous distribution. Bt has been isolated from soil, aquatic environments including sewage, dead insects and their breeding sites, herbivore faeces, stored grains, phylloplane and forest. Bt bacteria has a remarkable characteristic that can produce insecticidal crystal proteins killing insects during sporulation phase. During sporulation, Bt produces one or more parasporal insecticidal proteinaceous crystals (ICPs), comprised of one or more crystal (Cry) and cytolytic (Cyt) proteins, recognized as ´-endotoxins (Palma et al 2014).
Colony morphology can help to distinguish B. thuringiensis colonies from other Bacillus species. The organism forms white, rough clonies, which spread out and can expand over the plate very quickly. B. thuringiensis strains have unswollen and ellipsoidal spores that lie in the subterminal position. The morphology, size, and number of parasporal inclusions may vary among B. thuringiensis strains. However, four distinct crystal morphologies are apparent: the typical bipyramidal crystal, related to Cry 1 proteins, cuboidal inclusions related to Cry 2 proteins and usually associated with bipyramidal crystals, amorphous and composite crystals releated to Cry 4 and Cyt proteins and flat, square crystals, related to Cry 3 protein (Lopez-Meza and Ibarra, 1996). Spherical and irregular pointed crystal morphologies can also be observed in B. thuringiensis strains.
Observing parasporal crystal proteins during sporulation of Bt strain by SEM is the most direct and effective method to identify Bt bacteria. The principle behind scanning electron microscope (SEM) is focused beam of high-energy electrons to generate a variety of signals at the surface of solid specimens. The signals which arise from electron- sample interactions reveal information about the sample including external morphology (texture), chemical composition, and crystalline structure and orientation of materials making up the sample.
The present work deals with isolation, biochemical characterization and crystal morphological study of the spore. Spores of bacteria of the Bacillus cereus–B. thuringiensis group were chosen for the morphological analysis of their surface structures by a variety of electron microscopic techniques. B. thuringiensis, the species related to B. cereus, is the best studied entomopathogenic bacterium, which is used for production of efficient, environmentally safe preparations against insect pests. In some cases, insecticidal activity of B. thuringiensis results from combined action of spores and crystalline proteinaceous toxins. In this paper, we described the observing protocol for identifying the spores and crystals of Bt isolates by using scanning electron microscope in our laboratory in detail.
Soil sample collection and Bt isolation
One hundred and forty soil samples were collected from different regions across the Punjab state The soil surface was scraped with sterile spatula; about 10 g were taken from a depth of 2 to 5 cm, and stored in sterile cups. Samples were kept in the laboratory at 4°C until their processing. Bt isolation from soil samples was carried out by the selective sodium acetate heat pasteurization method as previously described (Martin and Travers 1989; Xavier et al 2007b). Briefly, 5 g of the sample was added to 10 ml of Luria Bertani (LB) broth containing 0.25 M sodium acetate. Presumably, sodium acetate selectively inhibits the germination of B. thuringiensis spores. Inoculated flasks were then incubated on a rotary shaker (200 rpm) for 4 h at 30°C. After incubation, 1 ml aliquot of thoroughly mixed culture broth was transferred to a pre-warmed 6 ml glass tube and heated at 80°C for 10 min, and then serially diluted aliquots were placed on LB agar and incubated overnight at 30°C. Bt colonies that were white, spread out and seems to fried egg on plate (Ammons et al 2005) were examined by phase contrast microscopy.
Phase contrast microscopy
Bt colonies were suspended in sterile distilled water as a wet mount and examined with phase contrast microscope (100X oil immersion objective) for the presence of parasporal crystals. Bt colonies were then classified into different groups based on their crystal shape (Lopez-Pazos et al 2009). Potential Bt candidates were subcultured and purified on nutrient agar plates and then stored as stock culture in sterile liquid Nutrient Broth containing 50% glycerol at “20°C (Hernandez et al 2005).
Scanning electron microscopy
In order to obtain the spore-crystal mixture, Bt isolates were grown in nutrient agar medium for 5 days at 30°C, until lysis. The samples were immersed in 2.5% glutaraldehyde and kept at 4° C for 24 hrs, rinsed thrice with 0.1 M caco buffer. Then, 1% osmium tetraoxide was added and dehydrated by passage through graded aqueous ethyl alcohol series (30, 50, 70, 90 and 95%), placed in 100% ethanol at room temperature for few minutes. It was then dried with a critical point dryer unit mounted on aluminum stubs with silver glue and coated with gold-palladium using anion sputtering unit. The samples were then examined under . Samples were examined and photographed with a FEI-Inspect S50, scanning electron microscope operating at a voltage of 1500 kV at 24,000× magnifications (Kati et al 2007), SEM unit at EMN lab, Punjab Agricultural University, Ludhiana.
Localities and collection of samples
A total of sixty eight (68) soil samples were collected from 19 different regions of Punjab (Table 1). The regions are representative of diverse agro climatic zones and geographical areas of the state. Using selective acetate method, 140 isolates were randomly picked up from agar plates seeded with soil samples obtained from nineteen different locations. This method eliminates most of the spore formers and non-spore formers in the soil samples. The spores of the unwanted bacterial species germinate on acetate buffered Luria broth while preventing the desired spores of B. thuringiensis from germinating. The unwanted bacteria that enter the vegetative stage are killed by controlled heat treatment. The inability of Bt species to germinate in the presence of acetate buffer allows the use of this trait to screen for this organism in the environmental samples.
Table (1):
Sampling sites and crystal positive isolates.
Sr. No. |
Location |
No. of soil samples withdrawn |
Number of crystal forming isolates |
|---|---|---|---|
1 |
Abohar |
7 |
14 |
2 |
Bathinda |
4 |
4 |
3 |
Kotkapura |
3 |
6 |
4 |
Patiala |
3 |
6 |
5 |
Nawanshehar |
5 |
12 |
6 |
Gurdaspur |
3 |
4 |
7 |
Ropar |
3 |
2 |
8 |
Samrala |
2 |
7 |
9 |
Faridkot |
3 |
9 |
10 |
Ferozepur |
3 |
8 |
11 |
Ludhiana |
9 |
17 |
12 |
Muktsar |
2 |
3 |
13 |
Khanna |
4 |
8 |
14 |
Sangrur |
3 |
9 |
15 |
Malout |
2 |
6 |
16 |
Nabha |
3 |
2 |
17 |
Barnala |
3 |
2 |
18 |
Jalandhar |
3 |
3 |
19 |
Hoshiarpur |
3 |
4 |
Morphological and Physiological Identification of the isolated Bacilli
Morphological observations with the crystal positive bacilli showed the vegetative cells as native, gram positive rods with dimensions of 1. 1-1. 3 × 2.5-4.5µM. All the strains tested along with the standard showed the typical colony morphology which was predominantly off-white to creamish in colour with irregular margins. A firm pellicle formation under stationary conditions was observed in these isolates which did not disperse but sank to the bottom when the tube was gently tilted. This characteristic was observed to differentiate B. thuringiensis from other Bacilli by Angus (1954).
Crystal protein morphology of Bacillus thuringiensis
After screening the task is to identify strains harboring crystalline inclusions that might have potential for use as bioinsecticidal agent. Cultures were analyzed and viewed under Phase-contrasts microscope under 100X magnification to observe the presence of parasporal inclusions. Parasporal cells with shining crystals confirmed the presence of Bacillus thuringiensis (Fig.1). Some biotypes of Bt have unique crystal shapes which can be observed under phase contrast microscope (Table 3). Although crystal number varied from isolate to isolate, all showed the presence of bipyramidal, cuboidal, round coleopteran specific crystals. These isolates were confirmed as B.thuringiensis on Hi-chrome Bacillus agar medium.
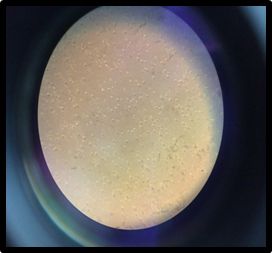 Fig. 1. Phase contrast microscopy of crystalline inclusions of Bacillus thuringiensis
Fig. 1. Phase contrast microscopy of crystalline inclusions of Bacillus thuringiensis
Table (2):
thuringiensis isolates from Punjab.
Sr. no. |
Type of crystals |
Isolates |
Total no of isolates |
|---|---|---|---|
1 |
Bi-pyramidal |
Bt-7, Bt-9, Bt-10, Bt-11, Bt-14,Bt-16, Bt-20, Bt-21, Bt-23, Bt-27, Bt-29, Bt-33, Bt-34, Bt-35, Bt-37, Bt-39, Bt-41, Bt-42, Bt-43, Bt-44, Bt-45, Bt-47, Bt-59, Bt-63, Bt-67, Bt-71, Bt-72, Bt-74, Bt-76, Bt-79, Bt-81, Bt-83, Bt-88, Bt-92, Bt-94, Bt-95, Bt-98, Bt-99, Bt-101, Bt-104, Bt-105, Bt-106, Bt-109, Bt-111, Bt-114, Bt-115, Bt-119, Bt-120, Bt-125, Bt-131, Bt-139, Bt-140 |
52 |
2 |
Pyramidal |
Bt-3, Bt-5, Bt-15, Bt-19, Bt-24, Bt-31, Bt-36, Bt-38,Bt-48,Bt-50, Bt-51, Bt-52, Bt-53,Bt-54, Bt-66, Bt-70, Bt-77, Bt-78, Bt-80, Bt-84, Bt-87, Bt-91, Bt-93, Bt-110, Bt-118, Bt-121, Bt-122, Bt-123, Bt-129, Bt-135, Bt-138 |
31 |
3 |
Cuboidal |
Bt-1, Bt-6, Bt-12, Bt-17, Bt-18, Bt-25, Bt-49, Bt-55, Bt-56, Bt-57, Bt-64, Bt-82, Bt-85, Bt-96, Bt-97, Bt-112, Bt-117, Bt-124, Bt-128, Bt-132, Bt-136 |
21 |
4 |
Amorphous |
Bt-4, Bt-8, Bt-22, Bt-30, Bt-32, Bt-46, Bt-58, Bt-61, Bt-62, Bt-68, Bt-69, Bt-86, Bt-100, Bt-103, Bt-113, Bt-126, Bt-130, Bt-134, |
18 |
5 |
Round |
Bt-2, Bt-13, Bt-26, Bt-28, Bt-40, Bt-60, Bt-65, Bt-73, Bt-75, Bt-89, Bt-90, Bt-102, Bt-107, Bt-108, Bt-116, Bt-127, Bt-133, Bt-137, |
18 |
Scanning electron microscopy of crystalline inclusions of Bacillus thuringiensis isolate
After preliminary screening with phase contrast microscopy, Scanning electron microscopy confirmed the surface view of rod shaped bacterial cells found in chains and cluster arrangement. Bt isolates tended to have sub-terminal spores. Based on the diversity of crystal morphology, Bt isolates showed crystal architectures of irregular, bipyramidal , spherical, marginal and rhomboidal (Fig 2). This is similar to a study of El-kersh et al 2012 which showed Bt isolates with spherical crystals, while, irregular, bipyramidal, and attached crystal to the spores. The differences in the crystal protein morphology distribution might be due to genetic variation caused by the difference in the environmental conditions or to habitat effects (Al-Momani et al 2004).
 Fig. 2. Scanning electron micrograph of the spore (S) and crystal proteins(C) from Bacillus thuringiensis isolate (a) parasporal bodies, (b) bipyramidal crystalline inclusions, (c) different crystalline inclusions – cuboidal, spherical, amorphous
Fig. 2. Scanning electron micrograph of the spore (S) and crystal proteins(C) from Bacillus thuringiensis isolate (a) parasporal bodies, (b) bipyramidal crystalline inclusions, (c) different crystalline inclusions – cuboidal, spherical, amorphousACKNOWLEDGMENTS
The authors express their sincere thanks to Confederation of Indian Industry (CII), Science Engineering Research Board (SERB) and Sampuran Agriventures Pvt. Ltd., Chandigarh for the financial grant received under Primeminister’s Fellowship Scheme for Doctoral research.
- Almomani, F., Obeidat, M., Saadoun, I., Meqdam, M. Serotyping of Bacillus thuringiensis isolates, their distribution in different Jordanian habitats and pathogenicity in Drosophila melanogaster. World J Microbiol Biotechnol, 2004; 20: 749–53.
- Ammons, D., Rampersad, J. and Khan, A. Usefulness of staining parasporal bodies when screening for Bacillus thuringiensis. J Inverteb Pathol, 2002; 79: 203-04.
- Angus, T.A. A bacterial toxin paralyzing silkworm larvae. Nature 1954; 173: 545–46.
- El-Kersh, T. A., Al-sheikh, Y. A., Al-Akeel, R. A., Alsayed , A. A. Isolation and characterization of native Bacillus thuringiensis isolates from Saudi Arabia. Afr J Biotechnol, 2012; 11(8):1924–38.
- Hernandez, C. S., Andrew, R., Bel, Y., Ferre, J. Isolation and toxicity of Bacillus thuringiensis from potato growing areas in Bolivia. J Invert Pathol, 2005; 88(1): 8-16.
- Kati, H., Sezen, K., Nalcacioglu, R., Demirbag, Z. A Highly Pathogenic Strain of Bacillus thuringiensis serovar kurstaki in Pepidopteran Pests. J Microbiol, 2007; 45: 553-57.
- Lopez-Meza, J. E., Ibarra, J. E. Characterization of a novel strain of Bacillus thuringiensis. Appl Environ Microbiol 1996; 62: 1306-10.
- Martin, P. A. W., Travers, R. S. Worldwide abundance and distribution of Bacillus thuringiensis isolates. Appl Environ Microbiol, 1989; 55: 2437–42.
- Oestergaard, J., Ehlers, R., Martínez-Ramírez, A. C., Real, M. R. (2005) Binding of Cyt1Aa and Cry11Aa
- Toxins of Bacillus thuringiensis serovar israelensisto brush border membrane vesicles of Tipula paludosa (Diptera: Nematocera) and subsequent pore formation. Appl Environ Microbiol 73: 3623–29.
- Palma, L., Muñoz, D., Berry, C., Murillo, J., Caballero, P. Bacillus thuringiensistoxins: An overview of their biocidal activity Toxins, 2014; 6: 3296–325. doi: 10.3390/toxins6123296.
- López-Pazos, S. A., Martínez, J. W., Castillo, A. X., Salamanca, J. A. C. “Presence and significance of Bacillus thuringiensis cry proteins associated with the andean weevil premnotrypes vorax (Coleoptera: Curculionidae),” Revista de Biologia Tropical, 2009; 57(4): 1235–43.
- Schünemann, R., Knaak, N., Fiuza, L. M. Mode of action and specificity of Bacillus thuringiensis toxins in the control of caterpillars and stink bugs in soybean culture. ISRN Microbiol. 2014; 2: 1–12.
© The Author(s) 2018. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


