ISSN: 0973-7510
E-ISSN: 2581-690X
Naringin is a flavanone found in citrus fruits and vegetables which are responsible for the asperity, due to which citrus fruit are unable to consume and from which we are unable to get the valuable nutrition in our body. To overcome this asperity in citrus fruits, present investigation is carried out, which provides a detailed report of isolation and screening of naringinase producing micro-organisms from different samples collected from Kalyan -Karnataka region, India. A sum of around 50 strains were isolated from 20 different samples which showed the positive result for naringinase activity, among 50 strains obtained 42 strains are fungi and remaining 8 were bacteria which showed good to moderate results in preliminary screening by 1% FeCl3 method, further 5 fungi isolates which showed a good naringinase activity in submerged fermentation were assayed by Spectrophotometric method. As a result, KLA-80 showed a maximum result with an activity of 559 U/ml. Further from the identification of the isolate KLA-80 by BLAST analysis found to be having similarity with Aspergillus flavus and the accession number of OQ152018 is obtained.
Aspergillus flavus, Flavanone, Grape fruit, Naringin, Naringinase and Naringenin
Citrus based fruits are popular all over the world with a production of over 100 million tons in every given year.1 In India, North-eastern region is having a rich treasure of various citrus fruits, a numerous reservoir of citrus exists in wild and semi-wild form and it is dispersed in that region without any commercial value.2 It stands at 3rd position after the production of mango and banana in India.3 The citrus fruit processing industries does not increased in India as compared to production due consumer awareness problems and the main problem is the bitterness of juices extracted from it.4 Citrus fruits have a compound flavanone neo-hesperidosides which is known to found in sour orange and other citrus varieties, which contain high level of naringin which is responsible for the bitterness of fruit,5 which is known to be present in grapefruits, buntan orange, natusdaidai.6
Naringin is a one of the major flavanone glycoside obtained from grapefruits and many more citrus fruits, it is relatable to almost all tasteless compounds like hesperidin which found in a sweet orange and lemons.7 The concentration of naringin flavanone in grape fruit is found to be around 0.017-0.025% and the orange peel has a concentration of 0.036% concentration of naringin which is accountable for the essence in making the juice bitter, about 50mg/l of naringin is responsible for imparting bitterness to the juice.8
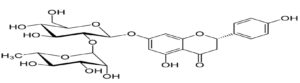
The compound Naringin was first discovered by Devry (1857). It has a chemical formula C15H1205. Though at first it was considered to be a chalkonel it was finally proved to have the constitution of 5: 7: 4′-trihydroxyflavanone (I, R = H)9 ( Figure 1).
Some conventional methods are used to decrease the concentration of naringin namely physical and chemical methods are applied for the removal of bitterness from the juices by Poly (styrene-co-divinylbenzene), resin, beta -D cyclodextrin bound polymer methods.10 The use of microbial enzymes in the textile and other chemical industries in processing is getting recognition worldwide because of their non-toxic and eco-friendly criterion.11 Debittering the citrus juice using the naringinase enzyme is a budding approach, as it makes less damage to the nutritional quality and increases the palatability properties of the juices.
A large number of micro-organisms are isolated for the debittering of the citrus juices; specifically those micro-organisms have a naringinase enzyme which has the capability to degrade the naringin into a non-bitter compound although naringin is not like other common enzymes.12
Kishi (1955) has isolated 96 strains and it is found that Aspergillus niger can produce naringinase in significant quantity.13 Subsequently, other strains of A. niger were also identified for naringinase production such A. niger MTCC 1344.14 A. niger CECT 2088,15 Bacillus cereus K-1,16 Aspergillus sp. SK01,17 were reported as a good producer of naringinase enzyme. Further immobilization of naringinase by chitosan microsphere isolated from Pencillium decumbens was found to retain thermal stability of enzyme and also 88.1±28% of initial activity after 10runs of naringin hydrolysis from fresh grapefruit.18 While Thermogram analysis (TGA) showed that nano-encapsulated enzyme showed highest activity whereas the free enzyme loses its 92% of its weight at 110°C.19 From these studies it is found that immobilization of enzymes have gained importance nowadays in industries as it allows for the preservation enzymatic activity for a longer interval of time and ease in their duplicate uses.20
Other then removal of bitterness from the citrus fruit, rhamnosidase activity of the enzyme in coalescence with arabinosidase and β-glucosidase was used for the enhancement of wine aroma.21 It also used in the metabolites bio-transformation such as antibiotics and steroids.22 Some studies also mentioned that naringinase have the capacity to produce specific fatty acids.23 Rhamnosidase activity of the naringinase was found to have a important role in the softening of the viscous property of the gellan gum.24
In the present investigation, isolation of naringinase producing micro-organisms from different types of samples and determination of the activity of the enzymes is checked along with the identification of the micro-organisms which have a highest activity of enzyme is determined and phylogenetic analysis of selected isolate is done.
Chemicals and reagents
All the chemicals and reagents used in this experimental design were procured from HiMedia and SRL ltd Mumbai, India.
Isolation of naringinase producing micro-organisms
A total of 20 different types of samples were collected from various regions of Kalyan-Karnataka. The samples were serially diluted using sterile distil water. The medium used for the production of naringinase had the composition (g/l) sucrose, 30gm; Sodium nitrate, 4gm; Dipotassium hydrogen phosphate, 1gm; Magnesium sulphate, 0.50gm; Potassium chloride, 0.50gm; Ferrous sulphate, 0.010gm; Agar, 15gm and Naringin, 0.20gm pH at 6 at 30°C. The cultures showing good growth were transferred to selection media(g/l)NH4NO3, 5gm; Kcl, 0.2gm; KH2PO4, 0.4gm; FeSO4.7H2O, 0.01gm; ZnSO4, 0.01gm; MnSO4, 0.01gm; MgSO4, 0.2gm; Agar, 15gm; Naringin, 1gm; After three more transfers, pure culture was obtained which were preserved at 4°C for future purpose.
Primary screening (qualitative assay)
Literature available on screening for naringinase producers is scanty. Kabyashree Phukan, et al.25 has done primary screening by FeCl3 method as prescribed; 24hrs old culture from the naringin PDB broth is used for screening i.e., 0.5ml 1% FeCl3 was added in the 5 test tubes having 3ml of fresh liquid media and 0.5ml of 24hrs old culture i.e. inoculums. Once FeCl3 is added to the media, the reddish brown color media changes to a light brown color, when it is collate with the control. Hence PDB which forms light brown color were showing positive results, and that cultures were used for the production of naringinase enzyme.
The cultures which showing positive results for 1% Fecl3 test (primary screening) were maintained in a pure culture of media containing 0.1% of naringin as a carbon source were maintained.
Selection of media for fermentation
There are 3 different types of media are used for the screening of naringinase with some changes in nitrogen sources to determine which media is suitable for naringinase production as mentioned in below Table 1.
Table (1):
Different media used for fermentation of naringinase
Media 1(g/l) |
Media 2(g/l) |
Media 3(g/l) |
|---|---|---|
Naringin-1gm |
Naringin-1gm |
Naringin-1gm |
NaNO3-2gm |
Peptone-3gm |
Yeast extract-2gm |
MgSo4-0.5gm |
MgSO4-0.5gm |
MgSO4-0.5gm |
Kcl-0.5gm |
Kcl-0.5gm |
FeSO4-0.5gm |
K2HPO4-1gm |
K2HPO4-1gm |
Nacl-0.5gm |
Fecl3-0.1gm |
K2HPO4-1gm |
The 500ml of Erlenmeyer flask containing 100ml of production media was prepared with pH-6 and temperature was maintained at 35°C and it was inoculated with one ml of isolate. The inoculated flasks are kept in an incubator at shaking conditions of 120 rpm for up to 5 days. The samples are withdrawn aseptically at 24 hrs intervals to determine the activity of enzyme.
Inoculum preparation
5 days old culture of Aspergillus flavus KLA-80 was used for the preparation of the inoculum for the fermentation by adding 10ml of distilled water with 0.1% Tween-80 and suspending the spores with a help of sterile loop. The spore suspension was adjusted to desired range (i.e., 1×108) by using haemocytometer.26
Enzyme assay
The naringinase assay was carried out by the method prescribed by Davis.27 The fermented culture was centrifuged at 3,000rpm for 10min at 4°C from that centrifuged culture 0.1ml of supernatant is taken and that supernatant was made to react with 0.9ml of 0.05%(w/v) naringin dissolved in 0.1M sodium acetate buffer(pH 4.0). The reaction mixture was then incubated at 500C for 60min after that 5ml of 90% diethylene glycol is added followed by the addition of 0.1ml of 4N NaOH. The mixture was kept at ambient temperature for 10min for the colour development the resulting yellow colour was measured at 420nm. One unit (U) of naringinase activity was defined as the amount of enzyme that could hydrolyze 1 μ/mol of naringin/ min at the assay conditions.
HPLC analysis
HPLC analysis is performed for the both standard and the isolate. The HPLC system of an Shimadzu HPLC coupled with a RP C18 reverse column (2.5x 150mm, 3.5µm) and a photo diode array detector was used for the analysis with an wavelength set at 280nm. The flow rate of 0.3ml/min was programmed and 5µl of the volume was injected. Naringin in the sample is identified by comparing the retention time of sample with standard.
Molecular characterization of the isolated strain
The selected isolate KLA-80 was undergone identification by lacto-phenol cotton blue test and Aspergillus differentiate media is done to determine the type of fungi. The genomic DNA extracted from the potential isolate KLA-80 using DNA miniprep kit. ITS-rDNA partial gene was successfully amplified using primers ITS4 & ITS5. The sequencing PCR was set up with ABI-BigDye® Terminatorv3.1 Cycle Sequencing Kit.
Phylogenetic tree analysis
The sequence analysis was carried out using Bioinformatics tool BLAST of NCBI. Multiple sequences is aligned used Clustal W. The phylogenetic tree was constructed by neighbour-joining method (NJ) using Mega11 software.28 The obtained sequences are compared to the sequences obtained in the gene bank using BLAST program on the NCBI website. Some of the closely related sequences from the gene bank are used to prepare Phylogenetic tree.
Isolation of the micro-organism
Isolation of micro-organisms from different types of samples were done by serial dilution method, i.e., 1gm of the samples is serially diluted using sterile distilled water, for the bacterial isolation 10-5 and 10-6 is taken so as for fungi isolation 10-3 and 10-4 should be taken. 0.1ml from the dilution test tube is taken on above mentioned media which is supplement with 0.1% of Naringin. The culture which showing good results are transferred to the fresh media after 3 more transfers, the pure culture of the micro-organism is obtained. The pure culture was maintained in the Naringin Agar media for the further investigation.
Primary screening
As mentioned above primary screening was done using Naringin PDB media i.e., the isolated colonies on the selected media is transferred to Naringin PDB media and allowed it to grown for 3 days and primary screening method was performed by using 1%Fecl3 method i.e., if the strain produces naringin enzyme then it hydrolyses naringin present in the media and the end product of the reaction is naringenin (Figure 2). Naringenin reacts with Fecl3 and gives a reddish brown colour. Therefore, the strain which shows reddish brown colour appearance was determined as naringinase producing strains and which does not develop brown color is considered as negative result for naringinase production. In the primary screening procedure 50 strains are showed vibrant results in which 42 strains are fungi and remaining 8 isolates are bacteria. As there is no proper evidences are available to confirm these isolates as naringinase producers so further secondary screening is carried out for all the 50 isolates (Table 2).
Table (2):
Different isolates showing naringinase activity.
| Fungi | ||
|---|---|---|
| No. | Isolate | Activity (u/mL) |
| 1. | KLA 5 | 376 U/mL |
| 2. | KLA 12 | 457 U/mL |
| 3. | KLA 18 | 533 U/mL |
| 4. | KLA 19 | 469U/mL |
| 5. | KLA 13 | 386 U/mL |
| 6. | KLA 28 | 427 U/mL |
| 7. | KLA 33 | 386 U/mL |
| 8. | KLA 36 | 438 U/mL |
| 9. | KLA 37 | 345 U/mL |
| 10. | KLA 38 | 251 U/mL |
| 11. | KLA 45 | 298 U/mL |
| 12. | KLA 49 | 202 U/mL |
| 13. | KLA 57 | 318 U/mL |
| 14. | KLA 59 | 298 U/mL |
| 15. | KLA 63 | 274 U/mL |
| 16. | KLA 65 | 201 U/mL |
| 17. | KLA 72 | 380 U/mL |
| 18. | KLA 80 | 559 U/mL |
| 19. | KLA 81 | 379 U/mL |
| 20. | KLA 88 | 315 U/mL |
| 21. | KLA 92 | 387 U/mL |
| 22. | KLA 96 | 284 U/mL |
| 23. | KLA 84 | 217 U/mL |
| 24. | KLA 93 | 284 U/mL |
| 25. | KLA 103 | 216 U/mL |
| 26. | KLA 113 | 317 U/mL |
| 27. | KLA 120 | 368 U/mL |
| 28. | KLA 123 | 400 U/mL |
| 29. | KLA 128 | 489 U/mL |
| 30. | KLA 138 | 298 U/mL |
| 31. | KLA 140 | 285 U/mL |
| 32. | KLA 141 | 326 U/mL |
| 33. | KLA 158 | 373 U/mL |
| 34. | KLA 160 | 368 U/mL |
| 35. | KLA 163 | 391 U/mL |
| 36. | KLA 166 | 287 U/mL |
| 37. | KLA 169 | 239 U/mL |
| 38. | KLA 173 | 261 U/mL |
| 39. | KLA 177 | 374 U/mL |
| 40. | KLA 197 | 315 U/mL |
| 41. | KLA 198 | 254 U/mL |
| 42. | KLA 183 | 267 U/mL |
| Bacteria | ||
| No. | Isolate | Activity (u/mL) |
| 1. | KLA 53 | 102 U/mL |
| 2. | KLA 78 | 80 U/mL |
| 3. | KLA 109 | 122 U/mL |
| 4. | KLA 118 | 108 U/mL |
| 5. | KLA 135 | 124 U/mL |
| 6. | KLA 143 | 198 U/mL |
| 7. | KLA 185 | 87 U/mL |
| 8. | KLA 186 | 124 U/mL |
Screening of selection media
Three different types of media are used to determine the activity of the enzyme out of which Media-1 have NaNo3 acts as a in-organic nitrogen source along with Fecl3 which helps in the oxidation-reduction process for the production of enzyme, whereas in the Media-2 Peptone and in Media-3Yeast extract is used as nitrogen source along with naringin as a sole carbon source in all the 3 media. After fermentation Media-1 has showed highest activity when compared other two media. So Media-1 is used for the fermentation of naringinase.
Production of naringinase under sub-merged fermentation
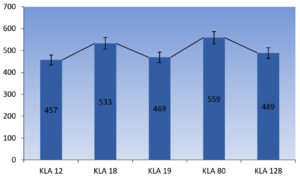
Submerged fermentation was done for the all 50 isolates in the media-1, which is found to be good naringinase producer among the 3 different media. The media- 1 contains a composition of Naringin, 1g; NaNO3, 2g; KH2PO4, 1g; Kcl,0.5g; MgSO4, 0.5g; Fecl3, 0.1g. Among all 50 isolates 5 isolates showed good results. Out of which KLA-80 showed a highest activity of 559 u/ml as shown in Figure 3. The activity of enzyme is expressed in u/ml. Further identification and sequencing and biochemical properties of KLA-80 have been done to identify the specific micro-organism (Figure 3).
HPLC analysis
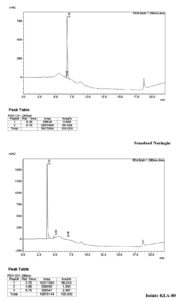
The HPLC analysis was done for the determination of naringinase activity in the micro-organisms. The sample along with standard was analyzed for the presence of Naringin in the sample. Both the sample and the standard were detected at 280nm and the results were analyzed. In which it is found that sample retention time 3.75 was decreased in compare to standard retention time of 6.76. From this it is found that the isolate KLA-80 has a naringinase activity (Figure 4).
Identification of microbial isolate
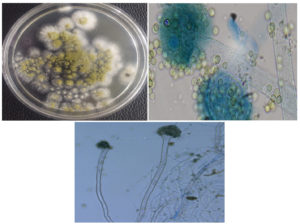
The microbial isolate KLA-80 was described as a fungus on the basis of cultural characteristics and microscopic observations. Colonies on MEA at 25°C citrine green, velvety, fast growing, reverse dull olive green. Conidial heads loose on in compact columnar, splitted into columns. Conidiophores straight, colourless, smooth walled, width gradually increasing towards length, foot cell present, upto 675.4× 14.0µm. Vesicles sub-globose to flask shaped, olivaceous from, fertile all over, upto 32.8×31.3 µm. Sterigmata uni to biseriate, primaries ampulliform, smooth walled, olivaceous brown, upto 15.6×5.5 µm, Secondary-ampulliform, olivaceous brown, smooth walled, upto 6.3×3.4 µm. Conidia produced in long chain, globose to subglobose, sub-olivaceous too livaceous, smooth to roughed walled, up to 6.8 × 6.6µm. Sclerotia, Chlamydospores absent and Teleomorph absent (Figure 5).
Figure 5. a) KLA-80 grow on PDA media containing 0.1% Naringin, b) Lacto-phenol cotton staining of KLA-80(Radical Microscope), c) Digital Microscopic image of KLA-80
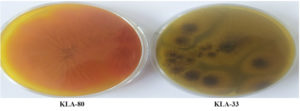
Aspergillus differentiate media is done to determine the type of Aspergillus based on their growth recommended by Dehghan et al.29 The Aspergillus flavus (KLA-80) appears green on the upper layer and orange on the reverse whereas Aspergillus niger (KLA-33) shows black on the upper layer and on the reverse also, from this we can easily differentiate Aspergillus flavus from Aspergillus niger (Figure 6).
Phylogenetic tree
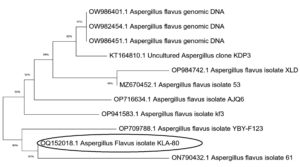
The phylogenetic analyses of the selected naringinase producing fungi are closely related to the Aspergillus genera. In this study the isolated culture showed 100% sequence similarity with Aspergillus flavus based on nucleotide homology and analysis and accession number of OQ152018 is obtained for the given culture (Figure 7).
Enzymes extracted from the micro-organisms are gaining interest because of their widespread uses in the industries due to their catalytic activity, stability, and ease in the production and optimization than plants and animal enzymes.30 Submerged fermentation is a technique which is quite fit for bacteria and fungi as they requires a high moisture content due to which purification of product is simpler in Smf method.31 The present research was aimed to produce naringinase using sub-merged fermentation was employed in which 5 fungal isolates showed a good naringinase activity as shown in Figure 3. There are numerous work was done on the naringinase activity isolated from the different sources. Aspergillus flavus isolated from citrus fruit showed a highest activity of 449.58ug/dry matter on the eighth day at 50mg/L concentration of naringin and increase in concentration results in the decrease in enzyme activity.32 Under solid state fermentation. A. oryzae JMU316 which was isolated from a decayed ‘Guanxi pomelo’ has showed activity in the presence peptone as a nitrogen source with a activity of 408.28IU/ml.33 Fungi at 30°C showed a growth with an initial pH of 7 using rutin and di-ammonium hydrogen phosphate as carbon and nitrogen sources 8.0 and 9.0 g/l, respectively.34 In the present study, there were 5 fungal isolates are obtained from the soil which showed a good naringinase activity under Smf. From this research it is found that soil is a good source of Aspergillus flavus, which is a best source for naringinase production. Fungi is known to be one of the best sources for naringinase production as compared to bacteria, various sources such as soil, peels of citrus fruit are found to be a home for fungi as they have a compatible environment for their growth.
From the above investigation it was found that the isolate KLA-80 is showing good naringinase activity; from this, it is concluded that Aspergillus flavus is found to be a good naringinase producer and suitable for industrial production of naringinase when compared to other strains, as it showed a good naringinase activity in submerged fermentation. Further, this isolate is used for the optimization process to enhance the activity of the naringinase enzyme.
ACKNOWLEDGMENTS
The authors would like to thank Karnataka Fund for Infrastructure Strengthening in Science and Technology in Higher Educational Institutions (K-FIST) and Karnataka Science and Technology Promotion Society (KSTePS). Department of Science and Technology, Karnataka, in helping to develop lab infrastructure and providing financial support to carry out this research work.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
This study was supported by K-FIST L2 (GRD-756) and KStePS (LIF-09:2021-2022).
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
Not applicable.
- Hasan S, Haque ME, Afrad MSI, Alam MZ, Hoque MZ, Islam MR. Influences of Socio-economic Factors on Lemon Pest Management Practices in Tangail District of Bangladesh. South Asian Journal of Social Studies and Economics. 2021;59-67.
Crossref - Konno A, Miyawaki M, Misaki M, Yasumatsu K. Bitterness Reduction of Citrus Fruits by β-Cyclodextrin. Agric Biol Chem. 1981;45(10):2341-2342.
Crossref - Ghosh D, Bhose S, Manimekalai R, Gowda S. Molecular detection of Candidatus Phytoplasma spp. causing witches’ broom disease of acid lime (Citrus aurantifolia) in India. J Plant Biochem Biotechnol. 2013;22(3):343-347.
Crossref - Hazarika TK. Citrus genetic diversity of north-east India, their distribution, ecogeography and ecobiology. Genet Resour Crop Evol. 2012;59(6):1267-1280.
Crossref - Hasegawa S, Berhow MA, Fong CH. Analysis of Bitter Principles in Citrus. Modern Methods of Plant Analysis. Fruit Analysis. 1996;18.
Crossref - Mahawar MK, Bibwe B, Jalgaonkar K, Ghodki BM. Mass modeling of kinnow mandarin based on some physical attributes. J Food Process Eng. 2019;42(5):e13079.
Crossref - Puri M, Marwaha SS, Kothari RM, Kennedy JF. Biochemical Basis of Bitterness in Citrus Fruit Juices and Biotech Approaches for Debittering. Crit Rev Biotechnol. 2008;16(2):145-155.
Crossref - Pavithra M, Belur PD, Saidutta MB. Production of naringinase by a new soil isolate Serretia sp, effect of different carbon and nitrogen sources. Research Journal of Biotechnology. 2012;7(4):208-211.
- Pavithra M, Belur PD, Saidutta MB. Production of naringinase from a new soil isolate, bacillus methylotrophicus: isolation, optimization and scale-up studies. Prep Biochem Biotechnol. 2014;44(2):146-163.
Crossref - Rangswami S, Seshadr TR, Veerarahaviah J. Constitution of naringin. The Position of the Sugar Group. Department of chemistry and chemical technology, Andhra University. 1939.
- Ribeiro MH. Naringinases: Occurrence, Characteristics, and Applications. Appl Microbiol Biotechnol.2011;90(6):1883-1895.
Crossref - Kaur H, Gill PK. Microbial enzymes in food and beverages processing. Engineering Tools in the Beverage Industry. 2019.
- Puri M, Karla S. Purification and characterization of naringinase from a newly isolated strain of Aspergillus niger 1344 for the transformation of flavonoids. World J Microbiol Biotechnol. 2005;21:753-758.
Crossref - Kishi K. Production of naringinase from Aspergillus niger. Kagaku to Kogyo (Chem Ind Jpn) 29:140.1955.
- Production of Prunin and Naringenin by Using Naringinase from Aspergillus oryzae NYO2 and Their Neuroprotective Properties and Debitterization. (n.d.).
Crossref - Pegu Bk, Kardong D, Gogoi D. Isolation and molecular characterization of Bacillus cereus, a new naringinase producer. IJAIR. 2019;8(1). https://www.researchgate.net/profile/Bhaba-Pegu/publication/336085824_Isolation_and_molecular_chracterization_of_Bacillus_cereus_a_new_naringinase_producer/links/5d8dc5db458515202b6d09fd/Isolation-and-molecular-chracterization-of-Bacillus-cereus-a-new-naringinase-producer.pdf. Accessed January 2023.
- Gupta AK, Yumnam M, Medhi M, Koch P, Chakraborty S, Mishra P. Isolation and characterization of naringinase enzyme and its application in debittering of Pomelo juice (Citrus grandis): A comparative study with macroporous resin. J Food Process Preserv. 2021;45(5).
Crossref - Bodakowska-Boczniewicz J, Garncarek Z. Immobilization of Naringinase from Penicillium decumbens on Chitosan Microspheres for Debittering Grapefruit Juice. Molecules. 2019;24(23):4234.
Crossref - Housseiny MM, Aboelmagd HI. Nano-encapsulation of naringinase produced by Trichoderma longibrachiatum ATCC18648 on thermally stable biopolymers for citrus juice debittering. J Microbiol. 2019;57(6):521-531.
Crossref - Kumar S, Gautam P, Sharma M. Source, Production, Immobilization and Industrial Applications of Debittering Enzyme Naringinase: Review. Webology. 2021;18(1):1704-1722.
Crossref - Gallego MV, Pinaga F, Ramon D, Valles S. Purification and Characterization of an α-L-Rhamnosidase from Aspergillus terreus of Interest in Winemaking. J Food Sci. 2001;66(2):204-209.
Crossref - Ribeiro MH. Naringinases: occurrence, characteristics, and applications. Appl Microbiol Biotechnol. 2011;90(6):1883-1895.
Crossref - Saerens K, Van Bogaert I, Soetaert W, Vandamme E. Production of glucolipids and specialty fatty acids from sophorolipids byPenicillium decumbensnaringinase: Optimization and kinetics. Biotechnol J. 2009;4(4):517-524.
Crossref - Kaur A, Singh S, Singh RS, Schwarz WH, Puri M. Hydrolysis of citrus peel naringin by recombinant a-L-rhamnosidase from Clostridium stercorarium. J Chem Technol Biotechnol. 2010;85(10):1419-1422.
Crossref - Phukan K, Kardong D. Isolation of naringinase producing soil bacteria from Psidium guajava L. and Terminalia chebula Retz and its enzymatic activity. AIMS Molecular Science. 2020;7(3):292-304.
Crossref - Lingappa K, Babu V. Production of lovastatin by solid state fermentation of crabs (Ceratonia siliqua) pods using aspergillus terrus KLVB 28-2015.
- Davis DW. Determination of Flavonones in Citrus Juice. Anal Chem. 1947;19(7):46-48.
Crossref - Kumar S, Stecher G, Tamura K. MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol. 2016;33(7):1870-1874.
Crossref - Dehghan P, Zaini F, Rezai S, Jebali A, Kordbacheh P, Mahmoudi M. Detection of Aflr Gene and Toxigenicity of Aspergillus flavus Group Isolated from Patients with Fungal Sinusitis. Iranian J Publ Health. 2008;37(3):134-141
- Saqib-Farooq, Ganaic TA, Mir S, Shah MA. Exogenous enzyme. Food biopolymers: Structural, Functional and Nutraceutical Properties. 2021:319-338.
Crossref - Osemwegie OO, Adetunji CO, Ayeni EA, et al. Exopolysaccharides from bacteria and fungi : Current status and perspectives in Africa. 2020;6(6):e04205.
Crossref - Srikantha K, Kapilan R, Seevaratnam V. Characterization of best naringinase producing fungus Isolated from the citrus fruits. Int J Biol Res. 2016;4(2):83-87.
Crossref - Chen D-x, Niu T-g, Cai H-n. Optimizing culture medium for debittering constitutive enzyme naringinase production by Aspergillus oryzae JMU316. Afr J Biotechnol. 2010;9(31):4970-4978.
- Mamma D, Kalogeris E, Hatzinikolaou DG, et al. Biochemical Characterization of the Multi-enzyme System Produced by Penicillium decumbens Grown on Rutin. Food Biotechnol. 2004;18(1):1-18.
Crossref
© The Author(s) 2023. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.