ISSN: 0973-7510
E-ISSN: 2581-690X
Staphylococcus aureus, a major gram positive bacterial pathogen that causes a wide spectrum of clinical infections, ranging from localized soft-tissue infections to life-threatening bacteremia and endocarditis. S. aureus infects tissues when the skin or mucosal barriers have been breached which leads to many different types of infections, including boils, carbuncles (a collection of boils) and abscesses. The present study envisaged the incidence of S. aureus in skin and soft tissue infection (SSTI), its susceptibility pattern to different antibiotics and molecular characterization isolated from three different hospitals of Odisha. Out of 1702 samples collected 1622(95.29%) were culture positive for different bacterial pathogens from which S. aureus was 356 (20.9%). The incidence rate of S. aureus among male and female group of patients was 57.5% and 42.2% respectively. The isolated S. aureus were resistant to most of the antibiotics such as azithromycin, penicillin, doxycycline, ciprofloxacin, tetracycline, gentamicin, ofloxacin, chloramphenicol, ampicillin and oxacillin. Further methicillin resistant S. aureus (MRSA)-95.8% were also identified from theS. aureus strains which were multi drug resistant and few were resistant to vancomycin (5%). The MRSA strains were confirmed genotypically by amplification of methicillin resistant (mecA- 63.3%) gene. Identification and antibiogram profile of S. aureus is highly essential for the treatment of sepsis patients in Odisha.
Staphylococcus aureus, MRSA, Sepsis, Odisha.
Staphylococcus aureus is able to cause a large diversity of lethal infections in human because of a wide range of virulence factor that includes various toxin and enzymes1. It has emerged as one of the most important human pathogen and has become a leading cause of hospital and community acquired infection2.The introduction of penicillin greatly improved the prognosis of patients with severe staphylococcus infection, but after a few years resistance appeared owing to production of b- lactamase. Methicillin was designed to resist b- lactamase degradation, but methicillin resistant Staphylococcus aureus (MRSA)strain that were resistant to all of â- lactam antibiotics were identified soon after methicillin was introduced3.
Sepsis causes millions of deaths globally each year.4In various sepsis cases skin and soft tissue infection (SSTI) caused by Staphylococcus aureus is common in hospitals. Approximately 7% to 10% of hospitalized patients were affected by SSTIs, and they are very common in the emergency care setting5. S. aureus associated with SSTIs frequently begin as minor boils or abscesses and may progress to severe infections involving muscle or bone and may disseminate to the lungs or heart valves (i.e. endocarditis)6. In the late 1990s, MRSA strains emerged in the community setting, without established high risk factor called community- associated (CA)-MRSA7,8. SSTI due to CA-MRSA were more frequent among male9 and ranged from 16% to 44% of cases globally 10.The incidence of MRSA varied from 25 per cent in western part of India11to 50 per cent in South India12. Most SSTI are treated in outpatient settings with empiric antimicrobial therapy. Due to relative resistance of antibiotics which is difficult to treat and more virulent strains with capacity to adapt quickly to changing environment make the pathogens acquired in the hospital a matter of concern.13 Various studies across the globe have been consistent enough to show a predictable bacterial profile in the wound infections and few studies have attempted to estimate the number of Staphylococcus aureus- SSTIs; which makes an important observation for a clinician who intends to start empirical treatment to his patients, while laboratory culture reports are awaited.13
So far there is scanty report available on MRSA related to septic patients from Odisha. The present study has been envisaged to document the incidence of S. aureus (MRSA) strains in skin and soft tissue infections of septic patients from three different hospitals from this state.
Study site
The skin and soft tissue infection like cellulitis, abscesses and soft tissue infection were included from the OPD and IPD of Capital Hospital, Bhubaneswar, District Headquarter hospital, Khurda and SCB Medical College and Hospital, Cuttack from March, 2015 to Feburary, 2018. The inform consent were taken from patients/ their attendants before collection of samples. The pus samples were collected by sterile swabs in sterile vials in cold condition bearing the patients name, age, sex and were transported to microbiology division of Regional Medical Research Centre (RMRC), Bhubaneswar.
Bacteriological analysis
The samples were immediately inoculated on MacConkey, blood Agar and nutrient Agar (Hi Media, Mumbai) plates and incubated at 37°C for 24hrs. Blood Agar plates were incubated in aerobic and anaerobic conditions. Significant colonies with beta hemolysis in blood agar plates were picked up for gram’s staining and biochemical tests were done for confirmation of bacterial pathogens. Identification of S. aureus isolates was done based on colony morphology and biochemical tests like catalase, coagulase test (slide and tube), etc. Pure isolates of S. aureus were further confirmed by manitol salt agar test and chromogenic agar media (Hi Media, Mumbai) was used for the rapid detection of MRSA strains14.
Antimicrobial susceptibility
Antibiotic susceptibility analysis was performed by modified Kirby Bauer’s Disk diffusion technique (1966) with commercially available antibiotic disc (Hi Media, Mumbai). Characterization of stains as susceptible, intermediately resistant or resistant was done as based on the size of the inhibition zone according to the manufacturer’s instruction which matched the interpretive criteria recommended by WHO15. The antibiotics used were penicillin(P,10 µg), azithromycin(AZM, 15 µg), ciprofloxacin(CIP,5 µg), tetracycline(T,30 µg), gentamicin (G, 10µg), doxycycline (DO,30 µg), vancomycin (VA,30 µg), ofloxacin(OF,5 µg), chloramphenicol(C,30 µg), ampicillin(A, 10 µg), cefoxitin (CX,30 µg) and oxacillin(OX,1 µg).
MIC by E-Test
The E-Test (Hi Media Laboratories Pvt, Ltd, Mumbai, India) for determining oxacillin MICs were performed on Muller-Hilton agar plate with 2%NaCl as per instruction and plates were incubated at 35°C for 24hrs. Similar, procedure was followed for MIC of vancomycin and reading was taken after 24hrs of incubation as per the criteria provided by the CLSI guidelines (CLSI 2005).
The detail is as follows:
MIC of oxacillin>4µg/ml- resistant strains (MRSA);
MIC<2µg/ml- sensitive Staphylococcus aureus (MSSA).
MIC of vancomycin>16µg/ml- resistant Staphylococcus aureus (VRSA);
MIC (4-8µg/ml)-intermediate Staphylococcus aureus (VISA);
MIC<2µg/ml-sensitive Staphylococcus aureus (VSSA) strains.
PCR amplification
Polymerase Chain Reaction was performed for molecular confirmation of MRSA strains by amplification of methicillin resistant (mecA) gene in a thermal cycler as described by Stegger et al, (2012)16. DNA amplification was carried out for 30 cycles according to the following protocol: denaturation at 94°C for 30s, annealing at 55°C for 30s, and extension at 72°C for 1 min. with a final extension at 72°C for 5 min. The PCR products were analysed in 2% agarose gel stained with ethidium bromide and visualized under UV light.
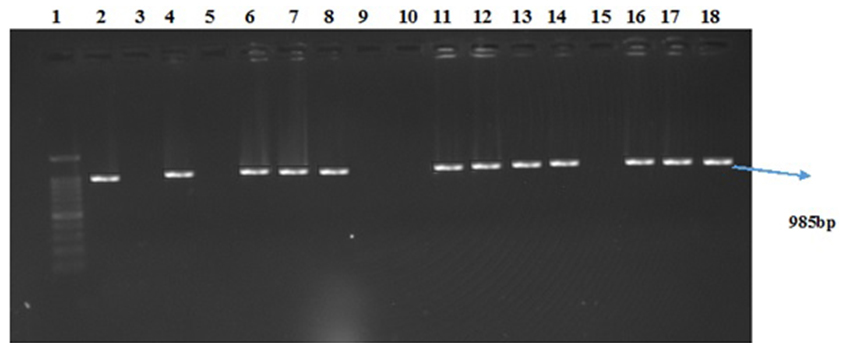 Fig. 1. Detection of the mecA gene (985bp) by polymerase chain reaction assay by 1.8% agarose gel electrophoresis. Lane 1: 100bp molecular weight marker; lanes 2: positive control (standard S. aureus) strain and Lane 3: negative control, Lane 4 -18: Samples
Fig. 1. Detection of the mecA gene (985bp) by polymerase chain reaction assay by 1.8% agarose gel electrophoresis. Lane 1: 100bp molecular weight marker; lanes 2: positive control (standard S. aureus) strain and Lane 3: negative control, Lane 4 -18: SamplesOut of 1702 samples collected,1622 (95.29%) were culture positive for different bacterial pathogens from which S. aureus was 365 (20.9%).S. aureus isolated from different categories were, soft tissue infection-195(12.02%), abscess- 33(2.03%) and cellulitis-137(8.5%). MRSA strains isolates was 350 (95.8%) from the total S. aureus isolates (Table 1).
Table (1):
Prevalence of Staphylococcus aureus from different SSTI patients.
Category of SSTI cases |
Total cases |
Culture+ve |
S .aureus |
MRSA |
|---|---|---|---|---|
Soft tissue Infection |
510(30.08%) |
501(29.4%) |
195(12.02%) |
190(52%) |
Abscess |
252(14.8%) |
216(12.7%) |
33(2.03%) |
30(8.2%) |
Cellulitis |
940(55.23%) |
905(53.2%) |
137 (8.5%) |
130(35.6%) |
Total |
1702 |
1622(95.29%) |
365 (20.9%) |
350(95.8%) |
In this study the clinical characterization for the positive cases of S. aureus in soft tissue infection was cellulitis with inflammation, wound infection, scalded skin, and folliculitis; in abscess cases infection of sebaceous cyst with pus discharge was observed. Most of the patients were from the community attending the OPD of the hospitals and few were indoor patients admitted in case of severe condition. Maximum Staphylococcus aureus infected cases were seen in the age group of 30-40 years in both male-90 (24.6%) and female- 73(20%) followed by age group 50-60 years then 40-50 years . Least cases were found in age group 18 -30 years. (Table 2). Most of the infections were associated with single pathogens and few were with multiple pathogens. Poly microbial infection was isolated from 362 (17.2%) cases but the coexistence of Staphylococcus aureus with different gram negative bacteria were seen in few cases.
Table (2):
Age group wise distribution of S. aureus positive cases.
| Age | Gender | |
|---|---|---|
| Male | Female | |
| >18 – <30 | 6 (1.6%) | 9(2.5%) |
| >30- <40 | 90 (24.6%) | 73(20%) |
| >40 – <50 | 39(10.%) | 13(3.5%) |
| >50 – <60 | 76(20.8%) | 59(16.2%) |
| Total | 211(57.8%) | 154(42.2%) |
Staphylococcus aureus were resistant to most of the antibiotics such as azithromycin, penicillin, doxycycline, ciprofloxacin, tetracycline, gentamicin, ofloxacin, chloramphenicol, ampicillin and oxacillin. Ninety percent of the Staphylococcus aureus isolated were methicillin resistant S. aureus (MRSA) strains as they were resistant to oxacillin as per CLSI guidelines and vancomycin resistant strain were 20.1% (Table-3). The MIC test for oxacillin and vancomycin was performed on 200 MRSA confirmed strains which showed 120(60%) were resistant for oxacillin and 80(40%) were sensitive. Out of total MRSA strain (200), 90(45%) of the MRSA strains were sensitive to vancomycin, 100(50%)-intermediate and 10(5%) were resistant. PCR amplification on 260 S. aureus positive strains revealed that 164(63.3%) were positive for mecA gene.
Table (3):
Resistance and sensitivity Profile (%) of S.aureus to different antibiotics.
| Antibiotics | Strain (MRSA n=721) (MSSA n=77) |
Resistant (%) | Sensitive (%) | P value |
|---|---|---|---|---|
| Azithromycin | MRSA | 65 | 35 | 2E-06 |
| MSSA | 14.7 | 85.3 | ||
| Penicillin | MRSA | 97.4 | 2.6 | – |
| MSSA | 23.6 | 76.4 | ||
| Ciprofloxacin | MRSA | 86.1 | 13.9 | – |
| MSSA | 47.3 | 52.7 | ||
| Chloramphenicol | MRSA | 59 | 41 | 0.0011 |
| MSSA | 27.3 | 72.7 | ||
| Doxycycline | MRSA | 31.2 | 68.8 | 0.00497 |
| MSSA | 4.1 | 95.5 | ||
| Ofloxacin | MRSA | 76.1 | 23.9 | 2.6E-05 |
| MSSA | 38.3 | 61.7 | ||
| Tetracycline | MRSA | 61.5 | 38.5 | 1.4E-05 |
| MSSA | 14.8 | 85.2 | ||
| Vancomycin | MRSA | 20.1 | 79.9 | – |
| MSSA | 100 | 0 | ||
| Ampicillin | MRSA | 65.9 | 34.1 | 1E-06 |
| MSSA | 16.5 | 83.5 | ||
| Gentamicin | MRSA | 56.6 | 43.4 | 1E-06 |
| MSSA | 4.1 | 95.9 |
MRSA and MSSA- methicillin resistant and sensitive S. aureus(P<0.005 is Significant)
Staphylococcus aureus is an important human pathogen and is implicated in a wide variety of infections such as skin and soft-tissue infections (SSTIs), endovascular infections, pneumonia, septic arthritis, endocarditis, osteomyelitis, foreign-body infections, and sepsis.17 In most of the sepsis case it was seen that S. aureus infection starts from small skin sites and further progresses to severe infections involving muscles or bone and may disseminate to lungs or heart. The cause of transmission of S. aureus infection could be by direct contact with infected patients, colonized subjects or a contaminated environment as most of the S. aureus were isolated generally from OPD patients. Pain and fever were invariably present in infection involving lower extremities; and in blood infection an increase in WBC count was observed. In most of the skin tissue infection, abscesses and ulcer cases the recurrence of lesions was frequent as observed in our study.
Effective antibacterial therapy may improve cure rate especially among patients with larger/severe infection. Over the past 20 years, the incidences of both community-acquired (CA) and hospital-acquired (HA) S. aureus infections have increased, while antibiotic treatment is increasingly hampered by the spread of S. aureus strains that are resistant to multiple antibiotics, including methicillin.18As per our study the infection rate was more in male population than the female which was similar to the research carried out in United States19.The predominance of mono-microbial infections from chronic wound infections was observed which was similar to the findings reported from Banaras Hindu University, Varanasi20. Incidence of MRSA strains isolates was 350 (95.8%) from the total S. aureus isolates and coagulase negative S. aureus was10.2%. Similar types of findings were seen in clinical cases in Iraq hospitals where MRSA percentage isolated was 95.2% and coagulase negative S. aureus (CoNS) was 4.7% 21.
According to the antibiotic status of this study doxycycline and vancomycin were highly sensitive drugs against MRSA isolates but were resistant to most of the other antibiotics. All of the MSSA strains were sensitive to azithromycin, penicillin, chloramphenicol, doxycycline, tetracycline, gentamicin, ofloxacin, vancomycin, ampicillin, cefoxitin and oxacillin and showed least sensitive to ciprofloxacin. The MIC results confirmed the presence of MRSA and VRSA strains as these were resistant to both oxacillin and vancomycin, similar results were reported in an Indian teaching Hospital from Odisha by Dubey et al., 201314.But MRSA strain which were 100% sensitive to vancomycin was reported from a surveillance study done in different hospitals of North and south India22. The resistance value that were observed in the S. aureus isolated strains by phenotypic methods was compared by PCR amplification for MRSA detection and it interpreted similar results.
The emergence and spread of antibiotic resistance remains a global public health concern. So a core function of all clinical laboratories have to determine the antibiotic susceptibility pattern of bacterial isolates which would guide the clinicians in treating the patient infected with MRSA. High clinical suspicion and appropriate microbiological tests are also essential for early diagnosis and proper administration of antibiotics to reduce morbidity rate. So, continuous surveillance on resistance pattern of MRSA isolates and molecular study of toxic genes are essential for the understanding of new and emerging trends of this infection. Thus, this type of study should be encouraged in different hospitals of Odisha.
This project was supported by the funding from ICMR, New Delhi,(No.80/889/2014-ECD-I) which is highly acknowledged. We our thankful to the Director, Regional Medical Research Centre, Bhubaneswar for providing all the facilities to carry out the research work. We are also thankful to the chief medical officer and the staffs of District Headquarter Hospital, Khurda; the director and the staffs of Capital Hospital, Bhubaneswar, and the superintend and the staffs of surgery department of SCB Medical College and Hospital, Cuttack for their help during sample collection.
- Bal, A.M., Gould, I.M. Antibiotic resistance in Staphylococcus aureus and its relevance in therapy. Expert Opin Pharmacother. 2005; 6(13):2257-69.
- Shittu, A.O., Lin, J. Antimicrobial susceptibility patterns and characterization of clinical isolates of Staphylococcus aureus in KwaZulu-Natal province, South Africa. BMC Infect Dis. 2006; 28; 6:125.
- Ji, Y.(ed): Methicillin Resistant Staphylococcus aureus (MRSA) protocols, 2007; pp1-9.
- Dellinger, R.P., Levy, M.M., Carlet, J.M., Bion, J., Parker, M.M., Jaeschke, R., Reinhart, K., Angus, D.C., Brun-Buisson, C., Beale, R., Calandra, T., Dhainaut, J.F., Gerlach, H., Harvey, M., Marini, J.J., Marshall, J., Ranieri, M., Ramsay, G., Sevransky, J., Thompson, B.T., Townsend, S., Vender, J.S., Zimmerman, J.L., Vincent, J.L. Surviving Sepsis Campaign: international guidelines for management of severe sepsis and septic shock: 2008. Intensive care medicine, 2008;34(1): 17–60.
- McCaig, L.F., McDonald, L.C., Mandal, S., Jernigan, D.B. Staphylococcus aureus–associated Skin and Soft Tissue Infections in Ambulatory Care. Emerg Infect Dis. 2006; 12(11): 1715–1723.
- Ki, V., Rotstein, C. Bacterial skin and soft tissue infections in adults: A review of their epidemiology, pathogenesis, diagnosis, treatment and site of care. Can J Infect Dis Med Microbiol. 2008; 19(2):173-84.
- Centers for Disease Control and Prevention. Four pediatric deaths from community-acquired methicillin-resistant Staphylococcus aureus—Minnesota and North Dakota, 1997–1999. JAMA.1999;282:1123–5.
- Chambers, H.F. The changing epidemiology of Staphylococcus aureus? Emerg Infect Dis. 2001; 7:178–82.
- Ruhe, J.J., Smith, N., Bradsher, R.W., Menon, A. Community-onset methicillin-resistant Staphylococcus aureus skin and soft-tissue infections: impact of antimicrobial therapy on outcome. Clin Infect Dis 2007; 44:777–84.
- King, M.D., Humphrey, B.J., Wang, Y.F., Kourbatova, E.V., Ray, S.M., Blumberg, H.M. Emergence of community-acquired methicillin-resistant Staphylococcus aureus USA 300 clone as the predominant cause of skin and soft-tissue infections. Ann Intern Med, 2006; 144: 309–17.
- Patel, A.K., Patel, K.K., Patel, K.R., Shah, S., Dileep, P. Time trends in the epidemiology of microbial infections at a tertiary care center in west India over last 5 years. J Assoc Physicians India 2010; 58(Suppl): 37-40.
- Gopalakrishnan, R., Sureshkumar, D. Changing trends in antimicrobial susceptibility and hospital acquired infections over an 8 year period in a tertiary care hospital in relation to introduction of an infection control programme. J Assoc Physicians India, 2010; 58(Suppl): 25-31.
- Karia, J.B., Gadekar, H.B., Lakhani, S.J. Study of bacterial profile of pus culture in Dhiraj general hospital. Indian journal of Applied Basic Medical Science., 2013; 15[A] 19: Article-12.
- Dubey, D., Rath, S., Sahu, M.C., Pattnaik, L., Debata, N.K., Padhy, R.N. Surveillance of infection status of drug resistant Staphylococcus aureusin an Indian teaching Hospital. Asian Pac J Trop Dis, 2013; 3(2): 133-42
- Bauer, A.W., Kirby, W.M.M., Sherries, J.C., Turek, M. Antibiotic susceptibility testing by a standardized single disc method. American Journal of Clinical Pathogens. 1966; 45:493-96.
- Stegger, M., Andersen, P.S., Kearns, A., Pichon, B., Holmes, M.A., Edwards, G., Laurent, F., Teale, C., Skov, R., Larsen, A.R. Rapid detection, differentiation and typing of methicillin-resistant Staphylococcus aureus harbouring either mecA or the new mecA homologue mecA(LGA251). Clin Microbiol Infect. 2012; 18(4):395-400.
- David, M.Z., Daum, R.S. Community-associated methicillin-resistant Staphylococcus aureus: epidemiology and clinical consequences of an emerging epidemic. ClinMicrobiol Rev. 2010; 23(3):616-87.
- Ghebremedhin, B., Olugbosi, M.O., Raji, A.M., Layer, F., Bakare, R.A., König, B., König, W. Emergence of a community-associated methicillin-resistant Staphylococcus aureus strain with a unique resistance profile in Southwest Nigeria. J ClinMicrobiol. 2009; 47(9):2975-80.
- Chaberny, I.F., Ziesing, S., Mattner, F., Barwolff, S., Brandt, C., Eckmanns, T., Rüden, H., Sohr, D., Weist, K., Gastmeier, P. The burden of MRSA in 4 German university hospitals. Int J Hyg Environ Health 2005; 208: 447-453.
- Basu, S., Panray, T.R., Singh,T.B., Gulati, A.K., Shukla, V.K. A prospective descriptive study to identify the microbiological profile of chronic wounds in outpatients. Octomy Wound Manage. 2009; 55(1):14-20.
- Safi al- kadmy, I.M. A genetic study to differential HA/CA MRSA isolated from clinical cases in Iraq hospitals. Mintage journal of Pharmaceutical & Medical Sciences. 2013; 57-62.
- Joshi, S., Ray, P., Manchanda, V., Bajaj, J., Chitnis,D.S., Gautam, V., Goswami, P.,Gupta, V.,Harih,B.N., Kagal,A., Kapil,A., Rao, R., Rodrigues, C., Sardana,R., Devi,K.S., Sharma,A., Balaji,A. Methicillin resistant Staphylococcus aureus (MRSA) in India: prevalence & susceptibility pattern. Indian J Med Res. 2013; 137(2):363-9.
© The Author(s) 2018. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


