ISSN: 0973-7510
E-ISSN: 2581-690X
Urinary tract infection (UTI) is the leading cause of death among patients and majority of people visiting the hospital worldwide. In the current quantitative study, urine samples were obtained using sterile method and 100 isolates, including 60 Escherichia coli, 28 Klebsiella sp., and 12 Proteus sp., were investigated in a laboratory. Current study showed that E. coli 60/100 (60%) was the most common cause of UTI followed by Klebsiella sp. 28/100 (28%) and Proteus sp. 12/100 (12%). Cefotaxime (86.67% sensitive) was found to be the best drug for treating infections with E. coli, ceftazidime (100% sensitive) for infections with Klebsiella sp., and imipenem and levofloxacin (100% sensitive) for infections with Proteus sp. Overall, 51% of the isolated strains showed high multidrug resistance (MDR). Because of the alarming increase in extensively drug-resistant and MDR uropathogens, which is a concern in public health, the rational use of antimicrobial therapy should be implemented.
UTI, MDR, Uropathogenic, E.coli, Dhaka
Increasing antimicrobial resistant (AMR) bacteria are becoming a significant threat to public health. In recent years, multidrug-resistant (MDR), extensively drug-resistant (XDR), and pan-drug-resistant (PDR) bacteria have been explored.1-3 MDR is defined as acquired non-susceptibility to at least one agent in three or more antimicrobial categories, while XDR exhibits non-susceptibility to at least one agent in two or fewer antimicrobial categories (i.e., bacterial isolates remain susceptible to only one or two antimicrobial categories).
Urinary tract infection (UTI) is becoming complex due to the above-mentioned resistant bacteria. UTI is a common infection in Bangladesh as well as in other countries of the world, caused by colonization of pathogenic and opportunistic microbes.4,5 In Bangladesh and other developing countries, high increase in patients with UTI is found due to poor hygiene, catheterization, intercourse, pregnancy etc.5,6 Although antibiotics are used for preventing UTIs, several studies indicated that many uropathogens are becoming resistant to a wide range of antibiotics due to underuse, abuse, and overuse.7
Escherichia coli is a facultative anaerobe belonging to Enterobacteriaceae family, and uropathogenic E. coli (UPEC) strains form colonies in the large intestines of warm-blooded animals, including humans. UPEC strains have been identified to cause majority of community acquired UTIs and the causal agent in 90% of all UTI cases in ambulatory individuals.8
Klebsiella sp. is an emerging persistent bacteria that cause UTI, posing public health problems worldwide. In recent years, there is increasing prevalence in some antibiotic resistant bacteria, especially members of Klebsiella genus.9-11
Proteus sp. also belongs to Enterobacteriaceae family and is distinguishable from other genera by their swarming growth across agar surface. They are widely found in the environment playing a role in decomposing organic matter. Proteus sp. are often associated with UTIs in individuals with structural or functional abnormalities, including patients undergoing catheterization.12-14
Due to increasing MDR uropathogens, this study aimed to isolate, characterize, and determine the prevalence of MDR E. coli, Klebsiella sp., and Proteus sp. in the urine sample of patients with UTI.
Sample Collection
Four hundred specimens were collected in a tertiary care hospital. Using a sterile method, clean midstream urine samples were collected in glass containers. Samples were then transported in an icebox to a microbiology laboratory while maintaining the physio-chemical parameters.15
Demographic Data of Patients
To maintain homogeneity of variance in the study, equal number of positive samples were included in each age and gender group (Table 1). Young females and males from the age group 15–30 years old and older individuals from 30 years of age and above participated in the study. Underlying diseases were not observed in the participants.
Table (1):
Demographic data of patients.
Age and Gender |
Frequency |
|---|---|
Young Female( 15-30 years) |
25 |
Elder Female (> 30 years) |
25 |
Young Male( 15-30 years) |
25 |
Elder Male (> 30 years) |
25 |
Isolation of Uropathogens
Using calibrated sterile inoculating loops, 0.01 mL of each urine sample was plated on Mac Conkey agar, blood agar, cystine–lactose–electrolyte deficient (CLED) agar, and HIChrome agar media plates for detecting E. coli and Klebsiella sp. and on xylose lysine deoxycholate (XLD) agar for detecting Proteus sp. Inoculated petri plates were then incubated at 37 °C for 24–48 h. The number of colonies (CFU/mL) was counted for UTI diagnosis. Samples showing >105CFU/mL after incubation were positive indicator for UTI. Isolated colonies were identified based on biochemical tests.15,16
Antimicrobial Susceptibility
Antibacterial susceptibility testing was performed using Kirby–Bauer disk diffusion method against different antibiotics, including amoxycillin (AMC), amikacin (AMK), azithromycin (AZM), cefixime (CFX), ciprofloxacin (CIP), ceftazidime (CAZ), ceftriaxone (CRO), cefepime (CEFE), cefotaxime (CEFO), gentamycin (GEN), nitrofurantoin (NIT), levofloxacin (LEVO), imipenem (IMI), and sulfamethoxazole (SXT).14-17
The misuse and abuse of antibiotics are the major cause of drug resistance, while some pathogens develop innate resistance by environmental adaptation. Resistance develops also due to selective pressure of antibiotics.18,19
This study aimed to investigate the prevalence of AMR uropathogens isolated from patients with UTI.
A total of 60 E. coli, 28 Klebsiella sp., and 12 Proteus sp. were identified to observe the antibiotic susceptibility having significant growth (Table 2). Previous studies demonstrated high abundance of E. coli ranging from 53.7%20 to 61.45%21,22 among uropathogenic isolates.
Table (2):
Prevalence of organism in each patient group.
Organism |
Young Female |
Elder Female |
Young male |
Elder Male |
Total |
|---|---|---|---|---|---|
E. coli |
15/25(60%) |
15/25(60%) |
15/25(60%) |
15/25(60%) |
60/100(60%) |
Klebsiella sp |
7/25(28%) |
7/25(28%) |
7/25(28%) |
7/25(28%) |
28/100(28%) |
Proteus sp |
3/25(12%) |
3/25(12%) |
3/25(12%) |
3/25(12%) |
12/100(12%) |
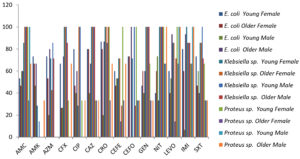
We observed that 13.33% and 60% of the E. coli strains were resistant to CEFO and NIT, respectively (Figure 1), Klebsiella sp. strains showed no resistance to CAZ, while 64.52% were resistant to AMK (Figure 1); Proteus sp. strains showed susceptibility to IMI and LEVO, whereas 100% resistance to AZM (Figure 1 and Table 3).
Table (3):
Antibiotic resistance of bacterial Isolates.
Organism |
AMC |
AMK |
AZM |
CFX |
CIP |
CAZ |
CRO |
CEFE |
CEFO |
GEN |
NIT |
LEVO |
IMI |
SXT |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
E. coli (%) |
50 |
35 |
28.33 |
40 |
38.33 |
28.33 |
16.66 |
48.33 |
13.33 |
43.33 |
60 |
40 |
18.33 |
50 |
Klebsiella (%) |
23.57 |
64.52 |
46.43 |
25.00 |
42.86 |
0 |
23.33 |
37.62 |
57.14 |
25.00 |
23.33 |
20.48 |
28.57 |
32.14 |
Proteus (%) |
16.66 |
83.33 |
100 |
75 |
83.33 |
75 |
50 |
50 |
41.66 |
66.67 |
16.67 |
0 |
0 |
33.33 |
Figure 1. Antibiotic Sensitivity of E. coli, Proteus sp. & Klebsiella sp. in different age and sex groups.
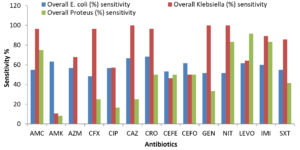
CEFO (86.67% sensitive) was found to be the most effective drug for treating infections with E. coli, CAZ (100% sensitive) against Klebsiella sp., and IMI and LEVO (100% sensitive) against Proteus sp. Previous studies found 91.3% sensitivity to meropenem (Figure 2).22 Akter et al. showed that carbapenems, aminoglycoside, and piperacillin-tazobactam were the most effective drugs against the E. coli strains; however, progression of drug resistance in these isolates was observed within 4 years.6
Fifteen (60%) MDR bacteria were found in the samples of older males, 13 (52%) in the samples of young males, eight (32%) in the samples of young females, and 15 (60%) in the samples of older females.
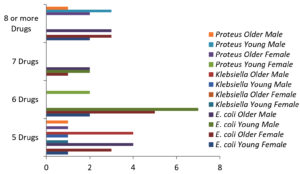
Among the E. coli isolates from older female samples, four isolates were resistant to five drugs, including AMC, AZM, CFX, CIP, and NIT; eight isolates were resistant to at least six drugs, including AMC, AZM, CRO, CEFO, GEN, IMI, CEFE, AMK, NIT, CFX, CAZ, LEVO, and SXT; ten isolates were resistant to seven drugs, including CAZ, CEFE, AMK, AZM, GEN, LEVO, SXT, AMC, CFX, CIP, CEFO, IMI, CRO, and NIT; and eleven isolates were resistant to eight or more drugs, including AMK, CAZ, SXT, AMC, CEFE, GEN, NIT, CIP, CRO, CEFO, LEVO, and IMI.
Among E. coli isolates from young female samples, four isolates were resistant to five drugs, including AMC, AZM, CFX, CIP, CRO, CEFO, NIT, and LEVO; at least six isolates showed resistance to six drugs, including AMC, AMK, AZM, CIP, CAZ, CRO, CEFE, CEFO, NIT, LEVO, IMI, and SXT; four isolates were resistant to seven drugs in all antibiotics except CFX and GEN; and thirteen isolates were resistant to eight or more antibiotics.
Among E. coli isolates from young male samples, two isolates were resistant to five drugs, including AMC, AMK, AZM, CFX, CRO, NIT, LEVO, and SXT, 10 isolates were resistant to at least six drugs except CAZ; eight isolates were resistant to seven drugs, including AMC, AMK, CIP, CEFE, CEFO, NIT, IMI, and SXT; and two isolates were resistant to eight or more drugs.
Among E. coli isolates in older male samples, one isolate was resistant to five drugs, including CFX, CAZ, CEFE, NIT, and LEVO; five isolates were resistant to at least six drugs in all antibiotics except GEN; twelve isolates were resistant to seven drugs in all tested drugs, and eleven isolates were resistant to eight or more drugs.
Among Klebsiella sp. isolates from older female samples, one isolate was resistant to five drugs, three isolates were resistant to at least six drugs in all antibiotics except GEN, two isolates were resistant to seven drugs, and eight isolates were resistant to eight or more drugs. Among Klebsiella sp. isolates from the samples of young females, two isolates were resistant to five drugs, one isolate was resistant to at least six drugs, no isolate was resistant to seven drugs, and eight isolates were resistant to eight or more drugs in all drugs. Among Klebsiella isolates from young male samples, one isolate was resistant to five drugs, six isolates were resistant to at least six drugs in all drugs, two isolates were resistant to seven drugs, and three isolates were resistant to eight or more drugs. Among Klebsiella isolates from older male samples, four isolates were resistant to five drugs, five isolates were resistant to at least six drugs in all drugs, no isolate was resistant to seven drugs, and six isolates were resistant to eight or more drugs in all drugs.
Among Proteus sp. isolates from older female samples, two isolates were resistant to five drugs, no isolate was resistant to at least six drugs, three isolates were resistant to seven drugs, and two isolates were resistant to eight or more drugs in all drugs. Among Proteus sp. isolates from young female samples, one isolate was resistant to five drugs no isolate was resistant to at least six drugs, one isolate was resistant to seven drugs, and six isolates were resistant to eight or more drugs in all drugs. Among Proteus sp. isolates from young male samples, no isolate was resistant to five drugs, two isolates were resistant to at least six drugs, one isolate was resistant to seven drugs, and five isolates were resistant to eight or more drugs in all drugs. Among Proteus isolates in older male samples, one isolate was resistant to five, six, and seven drugs, and four isolates were resistant to eight or more drugs in all drugs (Figure 3, Table 4). MDR cases were reported to be 54.2% by Asaduzzaman et al.,23 while 70.67% by Begum et al.15 In a previous study, E. coli was the most predominant isolate (72.4%),18 and similar results were observed in several other studies.6 Nevertheless, in the current study, 68.62% E. coli isolates showed MDR (Table 4).
Table (4):
MDR prevalence among different groups of patients.
| Organism | Patient groups | 5 Drugs | 6 Drugs | 7 Drugs | 8 or more drugs |
|---|---|---|---|---|---|
| E. coli | Young Female | 1 | 2 | 0 | 2 |
| Elder Female | 3 | 5 | 1 | 3 | |
| Young Male | 0 | 7 | 2 | 0 | |
| Elder Male | 4 | 0 | 2 | 3 | |
| Klebsiella sp. | Young Female | 1 | 0 | 0 | 0 |
| Elder Female | 0 | 0 | 0 | 0 | |
| Young Male | 1 | 0 | 0 | 0 | |
| Elder Male | 4 | 0 | 0 | 0 | |
| Proteus sp. | Young Female | 0 | 2 | 0 | 0 |
| Elder Female | 1 | 0 | 0 | 2 | |
| Young Male | 0 | 0 | 0 | 3 | |
| Elder Male | 1 | 0 | 0 | 1 |
The current study revealed that MDR cases are increasing continuously. Within the last couple of years, highly effective antibiotics were almost useless against newly identified uropathogenic strains due to drug resistance. By controlling the abuse of antibiotics and reducing selective pressure from available antibiotics, it may be possible to reduce the rise in resistant bacteria. Health care professionals need to be aware of the current situation of antibiotic resistance and focus on prescribing antibiotics after obtaining culture and sensitivity reports.
ACKNOWLEDGMENTS
None.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
None.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
This study was approved by the Institutional Ethical Approval Committee (IEAC), Primeasia University, India with reference number PAU/IEAC/22/108.
INFORMED CONSENT
Written informed consent was obtained from the participants before enrolling in the study.
- Basak S, Singh P, Rajurkar M. Multidrug Resistant and Extensively Drug Resistant Bacteria: A Study. J Pathog. 2016;1-5.
Crossref - Pattnaik D, Panda SS, Singh N, Sahoo S, Mohapatra I, Jena J. Multidrug resistant, extensively drug resistant and pan drug resistant gram negative bacteria at a tertiary care centre in Bhubaneswar. Int J Community Med Public Health. 2019;6(2):567-572.
Crossref - Abony M, Alam KM, Banik A, Khatun MF, Jannat B, Datta S. Isolation, identification of antibiotic producing microorganism from actinomycetaceae family from soil samples of Dhaka and Comilla. Journal of Primeasia University. 2018;2(1):11-18.
- Alam J, Juliana FM, Rahimgir M, Hossain MN, Fatema B, Asaduzzaman M. Resistance pattern of ciprofloxacin against common uropathogens in selected area of Dhaka city, Bangladesh. IOSR Journal of Nursing and Health Science. 2017;6 (5):52-57.
Crossref - Lisa LA, Paul DK, Biswas SK, Barman NC, Barman SK. Drug Resistance Profiles of Potential Gram Negative Rods Isolated from Urinary Tract Infected (UTI) Patients of Bangladesh with Four South Asian Countries. Int J Pharma Sci. 2015;5(4):1160-1166.
- Akter T, Hossain MJ, Khan MS, et al. Isolation, identification and antimicrobial susceptibility pattern analysis of E. coli isolated from clinical samples of Bangladesh. Asian J Biomed and Pharma Sci. 2016;6(54):13-16. https://www.researchgate.net/publication/300169811
- Alam MJ, Mousumi SJ, Rana R, et al. Ceftriaxone resistance patterns of uropathogens isolated from urinary tract infection patients in selected areas of Dhaka city, Bangladesh. IOSR J Nursing Health Sci. 2017;6(5):28-34.
Crossref - Kaper JB, Nataro JP, Mobley HLT. Pathogenic Escherichia coli. Nat Rev Microbiol. 2004;2(2):123-140.
Crossref - Martin RM, Bachman MA. Colonization, infection, and the accessory genome of Klebsiella pneumonia. Front Cell Infect Microbiol. 2018;8:4.
Crossref - Cristea OM, Avramescu CS, Balasoiu M, Popescu FD, Popescu F, Amzoiu MO. Urinary tract infection with Klebsiella pneumoniae in Patients with Chronic Kidney Disease. Curr Health Sci J. 2017;43(2):137-148.
Crossref - AL Khikani FH, Abadi RM, Ayit AS. Emerging Carbapenemase Klebsiella oxytoca with Multidrug Resistance Implicated in Urinary Tract Infection. Biomed Biotechnol Res J. 2020;4(2):148-151.
Crossref - Alam MF, Abony M, Sikdar MSS, et al. Comparative Antibiogram of Escherichia coli Isolated from the Urinary Tract Infection in Patients of Different Age and Sex Groups in Chittagong. J Primeasia Univ. 2019;3(1):11-13. https://www.researchgate.net/publication/349120558
- Rozalski A, Sidorczyk Z, Kotelko K. Potential K virulence factors of Proteus bacilli. Microbiol Mol. Biol Rev. 1997;61(1):65-89.
Crossref - Schaffer JN, Pearson MM. Proteus mirabilis and Urinary tract infections. Microbiol Spectr. 2015;3(5):10.1128.
Crossref - Begum NS, Shamsuzzaman SM. Emergence of multidrug resistant and extensively drug resistant community acquired uropathogens in Dhaka city, Bangladesh. Bangladesh J Med Microbiol. 2015;9(2):7-12.
Crossref - Odoki M, Aliero AA, Tibyangye J, et al. Prevalence of Bacterial Urinary Tract Infections and Associated Factors among Patients Attending Hospitals in Bushenyi District, Uganda. Int J Microbiol. 2019;219:4246780.
Crossref - Johnson JR, Stamm WE. Urinary tract infections in women:diagnosis and treatment. Ann Intern Med. 1989;111(11):906-917.
Crossref - Asma R, Alam MJ, Rahimgir M, et al. Prevalence of Multidrug-Resistant, Extensively Drug-Resistant and Pandrug-Resistant Uropathogens Isolated From Urinary Tract Infection Cases in Dhaka, Bangladesh. Avicenna J Clin Microbiol Infect. 2019;6(2):44-48.
Crossref - Magiorakos AP, Srinivasan A, Carey RB, et al. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: an international expert proposal for interim standard definitions for acquired resistance. Clin Microbiol Infect. 2012;18(3):268-281.
Crossref - Pardeshi P. Prevalence of urinary tract infections and current scenario of antibiotic susceptibility pattern of bacteria causing UTI. Indian J Microbiol Res. 2018;5(3):334-338.
Crossref - Biswas R, Rabbani R, Ahmed HS, Sarker MAS, Zafrin N, Rahman MM. Antibiotic sensitivity pattern of urinary tract infection at a tertiary care hospital. Bangladesh Crit Care J. 2014;2(1): 21-24.
Crossref - Prakash D, Saxena SR. Prevalence and antimicrobial susceptibility pattern of Escherichia coli in hospital acquired and community acquired patients related to urinary tract infection in India. J Appl Pharm Sci. 2013;3(8):124-32.
Crossref - Asaduzzaman MH, Hasan MZ, Khatun MM, et al. Resistance pattern of levofloxacin against uropathogens causing urinary tract infection in selected areas of Dhaka city. bangladesh. J Biol Agric Health. 2018;8(4):74-81. https://www.iiste.org/Journals/index.php/JBAH/article/view/41105/42268
© The Author(s) 2022. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.