ISSN: 0973-7510
E-ISSN: 2581-690X
In the present study the freshwater algae Chlorosarcinopsis eremi was isolated from the water sample of Kovai Kutralum. The pure culture of the algae was obtained using serial dilution technique followed by streak plate method. The species was observed under light microscopy and identified with the help of algal manuals and also by sequencing 18S rRNA genes. The growth of the algae, C. eremi was characterized in different growth media. The different growth media’s include Bold’s Basal medium, Modified Bourrelly medium, BG 11 medium, Half strength Chu10 medium and Modified Hoagland’s medium. The growth was measured by observed optical density of the biomass and chlorophyll “a” content. The experiment was carried out three times and mean values and standard deviation were calculated. From the results it is concluded that growth of C. eremi was maximum in Bold’s basal medium when compared with other media tested.
Chlorosarcinopsis eremi; 18SrRNA; Growth characteristic; Bold’s basal medium.0
Microalgae are highly diversified and found distributed in both freshwater and marine water. They exist with diverse morphology and in a wide variety of habitats. These diversified microalgae are also rich in secondary metabolites. These Microalgal secondary metabolites are proved to have bacteriostatic, bactericidal, antifungal, antiviral and antitumor activity (Justo et al., 2001). But Isolation and purification of such pharmacologically important secondary metabolites are found difficult due to their low quantity of algal biomass. Formulation of a suitable culture medium is prerequisite in achieving high biomass production. Many commercial media are available, since than is not enough to meet the requirement. Hence much research is needed in this area of biomass production. With this as aim the present study is focused on the isolation and growth characteristic of pharmacologically important microalgae Chlorosarcinopsis eremi in different growth media for its better biomass production.
C. eremi was previously reported to have the presence of Alkaloids, Anthraquinones, Cardiac glycosides, Flavonoids, reducing sugars, Saponins and Terpenoids. It was also found to contain antibacterial activity against Escherichia coli, Klebsiella sp, Pseudomonas sp, Salmonella, Staphylococcus aureus, Proteus sp, Methicillin-Resistant Staphylococcus aureus, Serratia sp and Bacillus sp (Dhanalakshmi and Angayarkanni, 2013).
Collection, Isolation and identification of microalgae
The water samples were collected from Kovai Kutralam, Siruvani hill ranges of Western Ghats, Coimbatore, TamilNadu, India. The collected water samples were centrifuged at 5000rpm for 10 minutes. The pellets were subjected to serial dilution (Shivhare et al., 2014) followed by streak plate method. The purity of each colony was observed under light microscopy and identified with the help of algal manuals (Prescott, 1954; Watanabe, 1983).
PCR amplification and sequencing of the 18S rRNA genes
The DNA of the algae was isolated using CTAB method. The ITS genes were amplified by polymerase chain reaction (PCR) using primers ITS2 FP: 5’ AGGAGAAGTCGTAACAAGGT’3 ITS2 RP:5’ TCCTCCGCTTATTGATATGC 3’. PCR product obtained was gel purified and quantitated and was sequenced. The sequence was analyzed and consensus sequence generated from forward and reverse sequences using aligner software. This sequence was subjected to BLAST.
Estimation of algal biomass of C. eremi in different growth media
The growth of Chlorosarcinopsis sp was measured directly by analysing the biomass of the culture. The isolated algal culture (1mL) previously grown in Bold’s Basal medium was inoculated in 100 mL conical flask with 25ml of the medium (Modified Bourelly’s medium, Modified Hoagland’s medium, Bold’s basal medium, half strength Chu 10 medium and BG-11 medium) (Guillard, 2005). The culture flasks were incubated at 25 ± 1ºC under 1.2 ± 0.2 lux light intensity with 12:12 h light and dark cycle for a period of 5 days. The growth of the algal biomass was assessed by means of optical density read at 540 nm using UV-Vis spectrophotometer (Miron et al., 2003). Growth pattern was checked at every 24h interval. Growth rate and division times were calculated as mentioned earlier. The experiment was carried out three times and mean values and standard deviation were calculated.
Estimation of chlorophyll ‘a’ content of C. eremi in different growth media
The algal culture of 1ml was inoculated uniformly in different growth media. The inoculated culture flasks were incubated at 25 ± 1ºC under 1.2 ± 0.2 lux light intensity with 12:12 h photoperiod of 5 days. Homogenous algal suspension (5ml) was centrifuged at 5000×g for 10 minutes. The pellets were washed twice in distilled water. After washing the pellets were resuspended in 4ml of methanol (80%) and vortexed thoroughly. The test tubes were incubated for 1 h at 60°C in water bath with occasional shaking. The tubes were then cooled and the contents were centrifuged at 5000×g for 5 minutes. The supernatant was transferred to another tube and once again 4 ml of the solvent was added to pellet and the process was repeated. The supernatant was pooled and made up to 10mL with methanol (to compensate the solvent loss during heating). The absorbance was read at 663nm in a spectrophotometer against methanol blank (Mac kinney et al., 1945).
Chlorophyll a = A663 × 12.63 × volume of sample / Volume of methanol mg/ml-1
A663 =Absorbance at 663
12.63 = Correction factor
The experiment was carried out three times, mean values and standard deviations were calculated statistically.
The organism isolated from Kovai Kutralam, Siruvani hill ranges, Western Ghats, Coimbatore, TamilNadu, India was identified as C. eremi (Chantanachat, S and Bold, H.C), is a multicellular representative of Chlorococcales under the light microscope and with the help of algal manuals. The ITS gene sequences (559bp) were subjected to BLAST search (fig 1 and 2) and proved to have high affinity to Chlorosarcinopsis eremi (93%). The phylogenetic tree was constructed based on neighbour joining method. The partial nucleotide sequence has been submitted in NCBI GenBank nucleotide sequence database with the accession number KM114872.
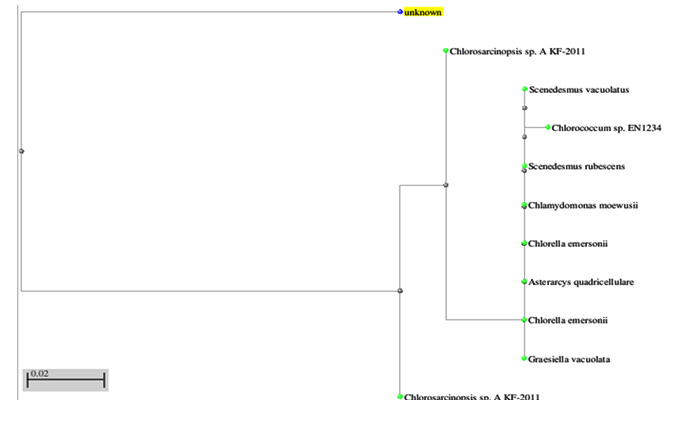
Fig. 1. Phylogenetic tree constructed for 18SrRNA gene sequence of Chlorosarcinopsis eremi.
 Fig 2. 18SrRNA sequence of Chlorosarcinopsis eremi
Fig 2. 18SrRNA sequence of Chlorosarcinopsis eremiC. eremi was grown in five different growth medium for the growth characteristic study. The growth characteristic study was carried out for a period of five days. The biomass was estimated by measuring the optical density under UV/VIS spectrophotometer at 540nm. Among the five different media tested the algal biomass was found to be maximum in bold’s basal medium.
The chlorophyll ‘a’ content of C. eremi was also studied as a parameter for growth characterization. The optical density of chlorophyll ‘a’ content were measured at 663nm using UV/VIS spectrophotometer. The growth characteristic study was carried out in five different growth media for a period of five days. The chlorophyll ‘a’ content of C. eremi was also found to be maximum in Bold’s basal medium when compared with other medium tested (Table 2). Similar results were observed previously by AL-Shatri et al. (2013). He reported Bold’s basal medium as optimal growth media for Scenedesmus dimorphus. Similarly Illavarasi et al., 2011, reported the growth and total chlorophyll content of Scenedesmus sp was found to be significant in Bold’ basal medium.
Table (1):
Estimation of algal biomass (OD540) of Chlorosarcinopsis eremi in different growth media.
Day of observation |
Bold’s Basal medium(mg/mL) |
Modified Bourrelly medium (mg/mL) |
BG 11 medium (mg/mL) |
Half strength Chu10 medium (mg/mL) |
Modified Hoagland’s medium (mg/mL) |
|---|---|---|---|---|---|
1 |
0.029 ± 0.002 |
0.026 ± 0.000 |
0.024 ± 0.000 |
0.021 ± 0.002 |
0.021 ± 0.000 |
2 |
0.06 ± 0.001 |
0.03 ± 0.002 |
0.03 ± 0.001 |
0.04 ± 0.002 |
0.03 ± 0.001 |
3 |
0.08 ± 0.002 |
0.03 ± 0.000 |
0.03 ± 0.001 |
0.05 ± 0.001 |
0.03 ± 0.002 |
4 |
0.05 ± 0.002 |
0.01 ± 0.000 |
0.03 ± 0.002 |
0.05 ± 0.001 |
0.01 ± 0.002 |
5 |
0.05 ± 0.001 |
0.01 ± 0.002 |
0.03 ± 0.010 |
0.04 ± 0.001 |
0.01 ± 0.000 |
Specific growth rate |
1.220588 |
1.1 |
1.151515 |
1.204082 |
1.09375 |
Data are given as mean ± standard deviation of triplicate experimental culture
Table (2):
Estimation of Chlorophyll ‘a’content (mg/ml) of Chlorosarcinopsis eremi from different growth media.
Day of observation |
Bold’s Basal medium(mg/mL) |
Modified Bourrelly medium (mg/mL) |
BG 11 medium (mg/mL) |
Half strength Chu10 medium(mg/mL) |
Modified Hoagland’s medium(mg/mL) |
|---|---|---|---|---|---|
1 |
1.596 ± 0.005 |
1.556 ± 0.005 |
1.576 ± 0.001 |
1.508 ± 0.001 |
1.502 ± 0.001 |
2 |
1.84 6± 0.002 |
1.742 ± 0.002 |
1.75 3 ± 0.002 |
1.792 ± 0.000 |
1.734 ± 0.003 |
3 |
2.301± 0.001 |
2.09 4± 0.007 |
2.09 4± 0.001 |
2.203 ± 0.001 |
2.020 ± 0.001 |
4 |
2.281± 0.000 |
1.00 2± 0.001 |
2.12 8± 0.001 |
2.214 ± 0.001 |
1.003 ± 0.001 |
5 |
1.914± 0.001 |
1.00± 0.00 |
1.50 6± 0.001 |
1.763 ± 0.003 |
1.114 ± 0.001 |
Data are given as mean ± standard deviation of triplicate experimental culture
From the present study it was concluded that the optimal growth medium for the culture of Chlorosarcinopsis eremi is Bold’s Basal medium both in case of biomass and Chlorophyll ‘a’ content. The other four growth media tested, Modified Bourrelly medium, BG 11 medium, Half strength Chu10 medium and Modified Hoagland’s medium did not support for the biomass production of the algae C. eremi .From the result it is also inferred that the presence of optimal concentration of NaCl, 10-fold increase of boric acid and calcium chloride in Bold’s basal medium may improved the growth of the algae.
- Al-Shatri AHA, Ali E, Al-Shorgani NKN and Kalil MS. Growth of Scenedesmus dimorphus in different algal media and pH profile due to secreted metabolites. African Journal of Biotechnology, 2014; 13: 1714-1720.
- Chantanachat S and Bold HC. Phycological studies II. Some algae from arid soils. University of Texas Publications, 1962; 6218: 1-74.
- Dhanalakshmi M and Angayarkanni J. Phytochemistry and antibacterial activity of Chlorosarcinopsis sp. International journal of scientific and Technology Research, 2013; 2(10): 315- 321.
- Guillard RRL. Purification methods for microalgae In: Algal Culturing Techniques, Andersen, R.A. (ed.), Elsevier Academic Press, Amsterdam, 2005; 117-132pp.
- Illavarasi A, Mubarakali D, Praveenkumar R, Baldev E and Thajuddin N. Otimization of various growth media to freshwater microalgae for biomass production. Biotechnology, 2011; 10(6): 540-545.
- Justo, GZ, Silva MR and Queiroz MLS, Effects of green algae chlorella vulgaris on the response of the host hematopoietic system to intraperitoneal ehrlich ascites tumour transplantation in mice [J]. Immunopharm. Immunotoxicol, 2001; 23: 199-131
- Mackinney G. Absorption of light by chlorophyll solution. Journal of Biological Chemistry, 1945; 148: 314-322.
- Miron AS, Garcia MC, Gomez AC, Camacho FG, Grima EM and Chisti Y. Shear stress tolerance and biochemical characterization of Phaeodactyylum tricornutum in quasi steady-state continuous culture in outdoor photobioreactors. Biochemical Engineering Journal, 2003; 16: 287-297.
- Prescott GW. How to know freshwater algae. Dubuque Brown Co, Iowa, 1954; 272pp.
- Shivhare AKS, Mishra VK, Sethi and Bhadoria AKS. Growth rate, biochemical and biomass analysis of Monoraphidium Minutum Algae in upper lake Bhopal (M.P). International Journal of Green and Herbal Chemistry, 2014; 3: 434-441.
- Watanabe S, New and interesting green algae from soils of some Asian and Oceanian regions. Arch. Protistenkdr 1983; 127: 223–70.
© The Author(s) 2018. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


