ISSN: 0973-7510
E-ISSN: 2581-690X
Urinary tract infection (UTI) is considered as one of the highly prevalent infections worldwide. Regarding its prevalence, UTI is more common in women. The outcome of the research is expected to predict the severity of the urinary tract infection. The study of prevalence and antibiotic susceptibility helps to provide data on the occurrence of uropathogens in different age groups and helps to treat the infection with effective antibiotics to prevent further progression of infection. The study has been carried out to identify and investigate the prevalence of uropathogens and to determine their antibiotic susceptibility potential. Overall, 713 urine samples were collected, MacConkey agar, Blood agar, CLED agar and Luria-Bertani agar were used for isolation of uropathogens by calibrated loop streak method and incubated at 37°C. Antibiotic susceptibility was carried out using the Kirby Disc diffusion method. A total of 665 showed positive bacteriuria. E. coli (37.3%) and K. pneumoniae (32.03%) were the most common uropathogens followed by E. fergusonii (17.9%), P. aeruginosa (8.4%), and A. baumannii (4.4%). The study found that above 70% of antibiotic susceptibility was observed in fluoroquinolones followed by imipenem, while all isolates showed high resistance to ampicillin. The organisms E. coli, E. fergusonii, and P. aeruginosa exhibited the least susceptibility (>50%) for third-generation cephalosporins. P. aeruginosa and A. baumannii showed no susceptibility to nitrofurantoin, whereas other isolates were least susceptible. The study concludes that K. pneumoniae and E. coli were highly prevalent within the study group and that fluoroquinolone and imipenem can be effective for treatment.
Antibiotic Susceptibility, Bacteriuria, Fluoroquinolones, Prevalence, Uropathogens
UTIs or Urinary Tract Infections are considered as second most infectious disease worldwide. It is the occurrence of microorganisms that cause the invasion of tissues of the urinary tract and adjacent areas. UTIs are the most common outpatient infections, which has incidence rate of 50-60% in adult women.1 The conditions of UTI include acute, chronic, uncomplicated, complicated, symptomatic, asymptomatic and recurrent.2 It is estimated that from 1990 until 2019, the total number of people who encountered UTIs elevated from around 252.2 million to more than 404.6 million, a rise in almost 152.4 million cases.3 Women are more frequently affected with UTI than men. The female anatomy further contributes as a risk factor for developing UTI’s. The shorter urethra in women shortens the distance for the bacteria to reach other adjacent urinary tract areas.4 About 40% women and 12% of men suffer with UTI infection at least one time in their lifetime.5
Bacteria, both Gram-negative and Gram-positive as well as certain fungi are causative agents of UTI. It is reported that more than 95% of UTI are caused by bacteria.6 Gram-negative pathogens, those belongs to Enterobacteriaceae family accounts for UTI. Majority of the Gram-negative bacteria which include Pseudomonas spp., Klebsiella spp., Escherichia coli, Proteus spp. and Enterobacter spp.7 are responsible for causing UTI. Colonization with Citrobacter, Enterobacter, Pseudomonas aeruginosa, S. epidermidis, E. faecalis and E. coli results in health-care-associated infection.7 UTI is generally treated with wide range of broad-spectrum antibiotics. For the treatment of uncomplicated UTIs β-lactams, nitrofurantoin, and trimethoprim antibiotics are recommended in many countries, but fluoroquinolones have exhibited effectual results for complicated and uncomplicated UTIs in several countries.8
The inappropriate and overuse of antibiotics has led to the emergence of antibiotic resistance in pathogens. In hospitals and communities, the emergence and dissemination of resistance in Gram-negative bacteria, imposed therapeutic challenges. Therefore, in this study isolation, characterization and identification of the uropathogens were carried out and their antibiotic susceptibility was determined. Our findings may lead to good treatment outcomes and minimize the risk of resistance development which is public health problem worldwide.
Study layout
The present study was carried out in the Department of Biotechnology and Microbiology, Karnatak University, Dharwad (Karnataka, India), between June 2021 to January 2023, 713 urine samples were collected from UTI subjects visiting the hospitals regularly. Female subjects whose age is between 16-70 years and whose clinical symptoms dysuria, frequency, urgency, flank pain, and hematuria was considered for the study. Females under the age of 16 and over the age of 70 years, pregnant women and subjects with underlying renal disease such as calculi, failure or malignancy, male subjects and female patients with recurrent UTI, and recent antibiotic therapy (within the past two weeks) were excluded from this study. All patients were informed in detail and written consent to participate in this study was obtained.
Collection, processing and culturing of urine samples
Patients were asked to collect mid-stream urine in a sterile container. Initial processing of samples included centrifugation at 1500 rpm for 5 min, the supernatant was disposed and drop of sediment was examined for the presence of leukocytes. The presence of urinary pus cells ≥5 high power field microscope (HPF) 400-fold magnification, indicates the infection. MacConkey agar, Blood agar, Luria-Bertani agar and Cystine-Lactose-Electrolyte-Deficient (CLED) agar were used for the isolation of uropathogens. The calibrated loop streak method was used to inoculate samples (0.01 ml). Further incubation of culture plates was done at 37°C for 18-24 hrs
Phenotypic and biochemical characterization of isolates
Isolates, which were grown on MacConkey agar, Blood agar, Cystine-Lactose-Electrolyte-Deficient agar and Luria-Bertani agar were observed for the colony morphology such as elevation, margin, surface and colors. Further, selected colonies were subjected for biochemical tests, and Gram’s nature. A series of biochemical tests were performed for selected colonies such as Indole, production, MR-VP, Citrate utilization, Mannitol motility, TSI, Catalase, Oxidase and Urease test.
Genotypic characterization of isolates
The representative isolates were selected for 16S rRNA sequencing. Chromosomal DNA was extracted by using spin column kit. The bacterial 16S rRNA gene (1500 bp) was amplified using polymerase chain reaction in a thermal cycler by using 16S rRNA primer; 27 F 5′- AGAGTTTGATCMTGGCTCAG-3′ and 1492 R 5′- TACGGYTACCTTGTTACGACTT- 3′ . The conditions of the thermal cycler were denaturation at 95°C for 2 min in one cycle and final denaturation at 94°C for 30 s, annealing at 52°C for 30 s and extension at 72°C for 90 s. The number of cycles for all three steps was 30, with a final extension at 72°C for 10 min in one cycle. Purification of the amplicons was done by Exonuclease I-Shrimp Alkaline Phosphatase. Purified amplicons were sequenced by the Sanger method. Further, the obtained nucleotide sequences were edited using CHROMASLITE (version 1.5). Later, the sequences were blast-searched for the closest resemblance of the isolates on NCBI server. The phylogeny representative colony strains were constructed with those of clinical pathogens which were already submitted to the NCBI, with decent similarity percentage homology to those strains that are known to cause infection. The phylogenetic tree was constructed by the neighbor-joining method using a bootstrap test (1000 replicates), and the evolutionary distance was computed using MEGA X software. The sequences of the organisms were deposited in the GenBank with accession numbers KU_NI01 (OR478763), KU_NI02 (OQ373378), KU_NI03 (OQ373379), KU_NI04 (OQ373380), KU_NI05 (OQ373381)
Antimicrobial susceptibility testing
Antimicrobial susceptibility testing was performed for the selected bacterial isolates identified from urine cultures using the Kirby-Bauer disk diffusion method9 on Mueller-Hinton agar. The antibiotics used in this study include Ampicillin, Amoxyclav, Nitrofurantoin, Ciprofloxacin, Aztreonam, Imipenem, Piperacillin, Cotrimoxazole, Gentamicin, Cefuroxime, Cefepime, Cefalexin, Cefotaxime, Ceftazidime, Ceftriaxone and Norfloxacin. Antibiotic discs were obtained from Himedia Pvt. Ltd Mumbai, Maharashtra. The bacterial isolates to be tested are inoculated in nutrient broth and incubated at 37°C for 3-5 hrs and adjusted the turbidity of the suspension to the density of 0.5 McFarland standards. A sterile, dry cotton swab was used for uniform distribution of suspension on the Mueller-Hinton agar plate. The inoculums were allowed to dry for 5-15 minutes with the lid in place. Antibiotic discs were placed using sterile forceps. After incubating the plates at 37°C for 24 hrs, results were interpreted according to Clinical laboratory standard institute guidelines.10 E. coli ATCC 4157 was used as the control strain. The zone of inhibition in diameter was measured in mm.
Statistical analysis
Statistical analysis viz., the mean and SD of the data was obtained with Statistical Packages for Social Sciences-15 (SPSS Inc, Chicago, IL, USA). The Pearson’s correlation was performed and P-value was calculated using ANOVA, a value <0.05 was considered as significant.
Incidence of urinary tract infection among female patients
A total of 713 outpatients were examined with symptoms of UTI. Of the total urine samples 665 samples showed positive growth on different media. The major incidence was observed in the age group of 16-30 years, 165 (24.8%) with a mean age of 23.62±3.98 years, followed by 51-60 yrs, 147 (22.1%) with a mean age of 54.98±2.53 years.
Clinical symptoms associated with urinary tract infection
Clinical symptoms of UTI occur as a result of a chain of host-pathogen interactions which leads to pathogenesis.11 Clinical symptoms and their association with UTI are shown in Table 1. Of the total subjects, 428 (64.4%) and 409 (61.5%) showed a suprapubic pain and dysuria, respectively. Statistical analysis showed significant relation between fever, flank pain and suprapubic pain and UTI (P < 0.005) (Table 1).
Table (1):
Clinical symptoms of the cystitis or pyelonephritis of patients and their association with positivity of uropathogens
| Clinical symptoms | The frequency (%) of occurrence | The prevalence (%) of UTI | P-value | ||
|---|---|---|---|---|---|
| Positive | Negative | ||||
| Fever | Yes | 325 (45.6) | 316 (47.5) | 9 (18.8) | 0.0001* |
| No | 388 (54.4) | 349 (52.5) | 39 (81.2) | ||
| Dysuria | Yes | 415 (58.2) | 385 (57.9) | 30 (62.5) | 0.9992 |
| No | 298 (41.8) | 280 (42.1) | 18 (37.5) | ||
| Suprapubic pain | Yes | 435 (61) | 428 (64.4) | 7 (14.6) | 0.0001* |
| No | 278 (39) | 237 (35.6) | 41 (85.4) | ||
| Frequency | Yes | 375 (52.6) | 352 (52.9) | 23 (47.9) | 0.983 |
| No | 338 (47.4) | 313 (47.1) | 25 (52.1) | ||
| Flank pain | Yes | 426 (59.7) | 409 (61.5) | 17 (35.4) | 0.0001* |
| No | 287 (40.3) | 256 (38.5) | 31 (64.6) | ||
| Urgency | Yes | 377 (52.9) | 353 (53.1) | 24 (50) | 0.0516 |
| No | 336 (47.) | 312 (46.9) | 24 (50) | ||
P < 0.05 statistically significant*
Identification of uropathogens isolated from UTI positive patients
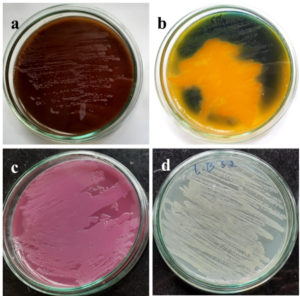
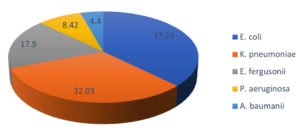
The isolates grown on different media (Figure 1) were subjected to colony and biochemical characterization (Table 2 and Table 3). Strains were indicated as KU_NI01, KU_NI02, KU_NI03, KU_NI04, KU_NI05. All isolates were Gram-negative rods. KU_NI01 and KU_NI03 strains were positive for indole production, MR test, catalase and for TSI test A/A reaction was observed, whereas KU_NI02, KU_NI04, KU_NI05 strains were positive for citrate, catalase. Only the KU_NI04 strain was positive for oxidase and an alkaline slant was observed in the TSI test. The Alk/Alk and A/A reaction for the TSI test was observed for KU_NI02 and KU_NI05 respectively (Table 3). The phylogenetic tree shows the evidence for identifying the isolates and is represented in Figure 2. The identified uropathogens were Escherichia coli (KU_NI01), Acinetobacter baumannii (KU_NI02), Escherichia fergusonii (KU_NI03), Pseudomonas aeruginosa (KU_NI04) and Klebsiella pneumoniae (KU_NI05). The overall frequency of uropathogens was observed as E. coli (37.3%), K. pneumoniae (32.03%), E. fergusonii (17.9%), P. aeruginosa (8.4%), A. baumannii (4.4%) respectively
(Figure 3). The prevalence of uropathogens by age group is depicted in Table 4.
Table (2):
Colony characterization of uropathogens on different culture media
Uropathogens |
Observation on Blood agar |
Observation on Macconkey agar |
Observation on CLED agar |
Observation on Luria-Bertani agar |
|---|---|---|---|---|
E. coli |
Circular, grey and moist colonies |
Dry, pink colonies surrounded by dark pink area of precipitated bile salts, Lactose fermenter, |
Opaque yellow colonies with slightly deeper yellow center |
Smooth, greyish white, opaque colonies |
K. pneumoniae |
Large, round and mucoid grey colonies, gamma hemolysis |
Large, pink mucoid colonies, Lactose fermenter |
Large, round yellowish mucoid colonies |
Large, round, creamy mucoid colonies |
E. fergusonii |
Medium size, round mucoid colonies |
Pink, smooth and slightly mucoid colonies |
Round, yellow mucoid colonies |
Circular, smooth opaque colonies |
P. aeruginosa |
Small, flat and round colonies, beta hemolysis |
Colourless, flat, colorless colonies, Non lactose fermenter |
Round, flat, blue to greenish color |
Round, irregular greenish colonies due to the pigment production |
A. baumanii |
Small to medium sized, round and pale colonies |
Pale or colorless colonies with non-mucoid texture |
Small to medium sized, colorless colonies |
Small to medium sized, round colorless colonies |
Table (3):
Biochemical characterization of uropathogens
Uropathogens |
Gram-staining |
Indole |
MR test |
VP test |
Citrate |
Catalase |
Urease |
Oxidase |
TSI |
|---|---|---|---|---|---|---|---|---|---|
E. coli |
– |
+ |
+ |
– |
– |
+ |
– |
– |
A/A,G |
K. pneumoniae |
– |
– |
– |
– |
+ |
+ |
– |
– |
A/A,G |
E . fergusonii |
– |
+ |
+ |
– |
– |
+ |
– |
– |
A/A,G |
P. aeruginosa |
– |
– |
– |
– |
+ |
+ |
– |
+ |
Alk/Alk |
A. baumanii |
– |
– |
– |
– |
+ |
+ |
– |
– |
Alk/Alk |
Table (4):
Age wise distribution of uropathogens among UTI positive female patients
| Age group, frequency (%) | |||||
|---|---|---|---|---|---|
| Uropathogens | 16-30 yrs | 31-40 yrs | 41-50 yrs | 51-60 yrs | 61-70 yrs |
| E. coli = 248 | 53 (21.4) | 38 (15.3) | 41 (16.53) | 56 (22.6) | 60 (24.2) |
| E. fergusonii = 119 | 27 (22.7) | 19 (16) | 32 (26.9) | 25 (21) | 16 (13.4) |
| K. pneumoniae = 213 | 37 (17.4) | 43 (20.2) | 54 (25.4) | 46 (21.6) | 33 (15.5) |
| P. aeruginosa = 56 | 13 (23.2) | 7 (12.5) | 12 (21.4) | 6 (10.7) | 18 (32.2) |
| A. baumanii = 29 | 0 | 2 (6.9) | 9 (31) | 6 (20.7) | 12 (41.4) |
Figure 1. Growth of uropathogens from urine samples of UTI infected female subjects on different culture media using caliberated loop streak method. a. Blood agar; b. CLED agar; c. MacConkey agar; d. Luria-Bertani agar
Figure 2. Phylogenetic tree of bacterial isolates, constructed through neighbor-joining method with bootstrap values as percentage at the nodes
Risk factors for urinary tract infection
Various risk factors were considered for the study, which are responsible for the progression of the UTI such as age, catheter use, sexual activity, menopause, history of UTI and place of residence. Statistical analysis revealed that sexual activity, catheter use, menopause and history of UTI had significant association with UTI (Table 5).
Table (5):
Association of the various risk factors for the prevalence of UTI with positivity of uropathogens
| Risk factors | The frequency (%) of occurrence | The prevalence (%) of UTI | P-value | ||
|---|---|---|---|---|---|
| Positive | Negative | ||||
| Catheter use | Yes | 223 (31.3) | 197 (29.6) | 26 (54.2) | 0.0001* |
| No | 490 (68.7) | 468 (70.4) | 22 (45.8) | ||
| Sexual activity | Yes | 422 (59.2) | 385 (57.9) | 37 (77.1) | 0.0001* |
| No | 291 (40.8) | 280 (42.1) | 11 (22.9) | ||
| Menopause | Yes | 288 (61) | 272 (40.9) | 16 (33.3) | 0.0001* |
| No | 425 (39) | 393 (59.1) | 32 (66.7) | ||
| History of UTI | Yes | 418 (58.6) | 376 (56.5) | 42 (87.5) | 0.0001* |
| No | 295 (41.4) | 289 (43.5) | 6 (12.5) | ||
| Place of residency | Urban | 449 (63) | 412 (62) | 37 (77.1) | 0.1106 |
| Rural | 264 (37) | 253 (38) | 11 (22.9) | 0.0001* | |
| Age | 16-30 | 175 (24.4) | 165 (24.8) | 9 (18.8) | 0.372 |
| 31-40 | 136 (19.1) | 128 (19.2) | 8 (16.7) | 0.439 | |
| 41-50 | 128 (18) | 117 (17.6) | 11 (22.9) | 0.601 | |
| 51-60 | 159 (22) | 147 (22.1) | 12 (25) | 0.605 | |
| 61-70 | 116 (16.3) | 108 (16.2) | 8 (16.7) | 0.508 | |
P < 0.05 statistically significant*
Antibiotic susceptibility pattern
Antibiotic susceptibility test was carried out for five different isolates using disc diffusion method, a total of sixteen antibiotics were considered for the study. Zone of inhibition was observed (Figure 4). The antibiotic susceptibility rate of selected uropathogens is presented in Table 6. The susceptibility rate of the E. coli was >50% for fluoroquinolones and least for ampicillin, cotrimoxazole and third-generation cephalosporins i.e. <10%. E. fergusonii showed more than 50% susceptibility to nitrofurantoin whereas P. aeruginosa had 100% resistance. Fluoroquinolones were effective against A. baumannii, K. pneumoniae, and P. aeruginosa. Except for E. coli and A. baumannii other isolates showed high susceptibility >70% for gentamicin. Only A. baumannii was susceptible for cefepime 68.32% whereas other isolates including A. baumanii were less than 57% susceptible for third generation cephalosporins. Among all the isolates E. fergusonii and P. aeruginosa were least sensitive i.e. less than 25%. Imipenem had good activity against A. baumanii and P. aeruginosa. E. fergusonii and K. pneumoniae were highly sensitive to amoxyclav (>75%) whereas other isolates susceptibility rate was low.
Table (6):
Antimicrobial susceptibility pattern of Gram-negative uropathogens
| Class of Antimicrobials | Susceptibility rate (%) | |||||
|---|---|---|---|---|---|---|
| Antimicrobial agents | E. coli(n=248) | E. fergusonii(n=119) | K. pneumoniae(n=213) | P. aeruginosa(n=56) | A. baumanii(n=29) | |
| Ampicillin | 13 (5.2) | 10 (8.4) | 17 (8) | 9 (16.07) | 7 (24.1) | |
| Penicillin | Piperacillin | 41 (16.5) | 36 (30.3) | 110 (51.6) | 38 (67.9) | 22 (75.9) |
| Amoxicillin/ | 32 (12.9) | 90 (75.6) | 174 (81.7) | 14 (25) | 19 (55.2) | |
| Clavulanic acid | ||||||
| Ceftazidime | 47 (19) | 40 (33.6) | 121 (56.8) | 11 (19.6) | 12 (41.4) | |
| Cefepime | 62 (25) | 51 (43) | 124 (58.2) | 17 (30.4) | 20 (69) | |
| Cephalosporins | Cefuroxime | 16 (6.5) | 21 (17.6) | 88 (41.3) | 21 (37.5) | 16 (55.2) |
| Ceftriaxone | 23 (9.3) | 38 (31.9) | 84 (39.4) | 27 (48.2) | 12 (41.4) | |
| Cephalexin | 12 (4.8) | 27 (22.7) | 78 (36.6) | 13 (23.2) | 10 (34.5) | |
| Cefotaxime | 37 (14.9) | 32 (26.9) | 112 (52.6) | 24 (42.9) | 15 (51.7) | |
| Fluoroquinolones | Ciprofloxacin | 137 (55.2) | 52 (43.7) | 178 (83.6) | 47 (83.9) | 22 (75.9) |
| Norfloxacin | 139 (56) | 57 (47.9) | 141 (66.2) | 40 (71.4) | 23 (79.3) | |
| others | Nitrofurantoin | 70 (28.2) | 70 (58.8) | 74 (34.7) | 56 (0) | 29 (0) |
| Aztreonam | 52 (21) | 45 (37.8) | 115 (54) | 45 (80.4) | 16 (55.2) | |
| Imipenem | 123 (49.6) | 61 (51.3) | 158 (74.2) | 43 (76.8) | 25 (86.2) | |
| Gentamicin | 112 (45.2) | 86 (72.3) | 165(77.5) | 44 (78.6) | 18 (62.06) | |
| Cotrimoxazole | 19 (7.7) | 15 (12.6) | 73 (34.3) | 18 (32.1) | 16 (55.2) | |
P value – <0.01 for all antibotics tested
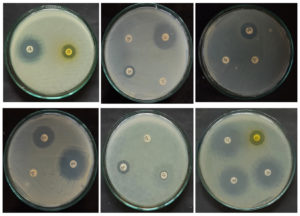 Figure 4a. Antimicrobial susceptibility of E. coli for different antibiotics
Figure 4a. Antimicrobial susceptibility of E. coli for different antibiotics
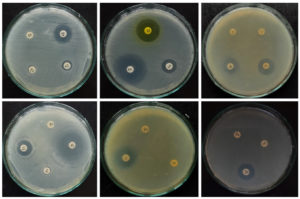 Figure 4b. Antimicrobial susceptibility of E. fergusonii for different antibiotics
Figure 4b. Antimicrobial susceptibility of E. fergusonii for different antibiotics
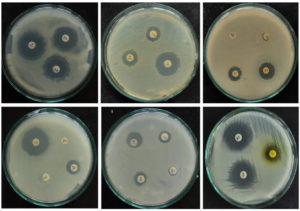 Figure 4c. Antimicrobial susceptibility of K. pneumoniae for different antibiotics
Figure 4c. Antimicrobial susceptibility of K. pneumoniae for different antibiotics
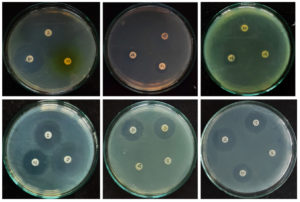 Figure 4d. Antimicrobial susceptibility of P. aeruginosa for different antibiotics
Figure 4d. Antimicrobial susceptibility of P. aeruginosa for different antibiotics
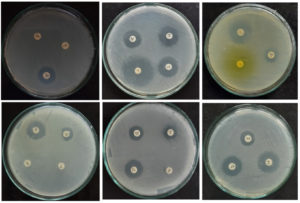 Figure 4e. Antimicrobial susceptibility of A. baumanii for different antibiotics
Figure 4e. Antimicrobial susceptibility of A. baumanii for different antibiotics
Figure 4. Antimicrobial susceptibility pattern of bacterial isolates isolated from urine culture of female patients attending hospitals in Dharwad region
According to the current scenario new challenges have arisen for the treatment of urinary tract infections; this is due to the frequent usage of antibiotics which makes the uropathogens resistant to antibiotics. The emergence of drug resistance in Gram-negative uropathogens is the main global concern that leads to poor clinical outcomes which include failure of treatment, occurrence of bacteraemia, and requirement for intravenous therapy.12 Gram-negative are responsible for approximately 30% of healthcare-associated infections (HAIs), and these bacteria predominate (45%) in cases of UTI.7 The prevalence rate observed in the present study is 93.3%. In our study the occurrence of UTI was more among subjects aged between 16 to 30 years (24.8%) and between 51 to 60 years (22.1%), establishing evidence to earlier reports.13 Due to the variations in the hormonal and physical development, incidence of UTIs is high in women.14 Also, the anatomical structure of the urethra which is shorter than male urethra. This shortness of the urethra allows pathogens to travel easily to the bladder.15,16 In our study the occurrence of infection was high in sexually active females and it is consistent with previous studies.17 In sexually active females, frequent coitus was one of the significant risk factors and also due to the anatomy of the urinary tract, while urination at post sexual intercourse were proven effective for the prevention of UTI.18 Further, deficiency of vitamin D in women of reproductive age is also linked with UTIs. Cathelicidin, an antimicrobial peptide secreted that bladder epithelial cells protects the lower urinary tract from infection which is induced by vitamin D.19 This finding suggested by maintaining proper hygiene in sexually active females can reduce the risk of developing UTIs. Also, women in the menopause condition have a great chance of encountering with UTI. Estrogen, maintains vaginal microflora i.e., Lactobacillus, a predominant bacterial genus reduces vaginal pH, due to which colonization of Enterobacteriaceae is prevented. As there is reduction of estrogen production in menopause condition, it leads to the development of UTI.20
Clinical symptoms such as fever, dysuria, urgency, flank pain and suprapubic pain were considered in our study and are similar to report.11 In our study the occurrence of UTI was more among subjects aged between 16 to 30 years (24.8%) and between 51 to 60 years (22.1%), establishing evidence to earlier reports.13 The present study determines the study of etiological agents causing UTI and their susceptibility to the drugs, which help in the treatment of the infection. The overall incidence rate of UTI in females was 93.3% which is consistent with study21 in which the frequency was higher in female subjects with UTI. However, the remaining cases (6.7%) showed mixed bacteriuria the suspected cases. The Gram-negative rods were the most frequent pathogens observed in our study which is consistent with previous studies.22,23 They possess capsule (K antigen) and LPS (O antigen) as virulence factors that play role in pathogenesis.7 In the present study E. coli (37.3%) was most occurring, followed, by K. pneumoniae (32.03%), P. aeruginosa (8.4%) and A. baumannii (4.4%), similar findings were also reported from other studies.24-26 The bacteria, E. fergusonii (17.9%) was also found among UTI subjects in our study contrast to previous studies,21,23,24 whereas it was reported in other study.27 The phylogenetic tree was constructed using the Neighbor-joining method, the evolutionary distances were computed using Tajima-Nei method. The sequence readings of all the isolates depicted above 80% similarity with E. coli, E. fergusonii, K. pneumoniae, P. aeruginosa and A. baumannii that had been previously reported as clinical isolates (Figure 3). In our study E. coli showed sensitivity to ciprofloxacin (55.2%) which was higher than 26.4% reported from other study28 and high sensitivity was shown for norfloxacin (56%) compared to previous study25 (31.3%). Least sensitivity was shown to Gentamicin (45.2%), imipenem (49.6%), amoxicillin/clavulanic acid (12.91%), cotrimoxazole (7.7%) which is deviated from other studies28,29 except Escherichia sp. all three isolates showed high sensitivity to norfloxacin (>70%) when compared to the previous study.12 E. fergusonii showed moderate susceptibility for imipenem (51.2%) and less susceptibility for ciprofloxacin (43.7%), whereas good susceptibility was shown for gentamicin (72.3%) not deviating from previous study27 except for gentamicin which showed poor sensitivity. This isolate also showed high resistance for ampicillin (91.6%), cefotaxime (73.1%), and intermediate resistance for ceftazidime (66.4%) similar findings were reported in the study.27
P. aeruginosa was 100% resistant to nitrofurantoin, highly sensitive to ciprofloxacin (83.9%) followed by gentamicin (78.6%), imipenem (76.8%) and norfloxacin (71.4%), the susceptibility rates of imipenem and gentamicin is marginally less than other studies29,30 whereas similar result was shown by nitrofurantoin and comparatively ciprofloxacin, norfloxacin exhibited high sensitivity in our study that was contrast to other reports.21,23,29 In our study P. aeruginosa showed only (19.6%) resistance for aztreonam which is least compared to resistance rate 83.09% and 50% respectively in other studies.21,29 The emergence of antibiotic resistance is due to the traditional antibacterial therapy much before availability of susceptibility and urine culture reports. The resistance occurs as a result of b-lactamase enzyme production, drug modification and target site alteration.20 A. baumannii exhibited similar sensitivity for ciprofloxacin and piperacillin i.e. 75.9% and high rate was shown by gentamicin (62%) and imipenem (86.2%), whereas <50% was seen for ceftazidime and ceftriaxone, these findings were higher than other study.23,25 The susceptibility of A. baumannii for cotrimoxazole in our study is higher compared to the result shown by the study31,32 in which it is only 3.7%, also no susceptibility was seen for nitrofurantoin in which similar pattern is observed concerning other studies.23,25 The least susceptibility is due to the resistance to the above-mentioned antibiotics which occurs by the early exposure of the isolates. K. pneumoniae exhibited high sensitivity for ciprofloxacin (83.6%) followed by amoxicillin/clavulanic acid (81.7%), imipenem (73.8%), these susceptibility rates deviate from other studies18,26,31 in which least rate of susceptibility was observed. The sensitivity to ceftriaxone and ceftazidime goes in similar pattern consistently below 60%.30,31 In our study high resistance was observed for ampicillin (>70%) this may be the result of the non-judicious use of antibiotics and their easier accessibility without a physician’s prescription. The overall result of the present study has shown that the susceptibility rate for fluoroquinolones, imipenem, amoxicillin/clavulanic acid, piperacillin and aztreonam is high whereas it is least for third-generation cephalosporins, ampicillin and nitrofurantoin. The low resistance drugs may be considered as highly active agents for uropathogens in the current study setting.
In the present study, E. coli followed by K. pneumoniae and E. fergusonii were the most occurring uropathogens isolated from female subjects with UTI. Catheter use, menopause, place of residence, and history of UTI are the main risk factors. Third-generation cephalosporins depicted low susceptibility for all isolates. Fluoroquinolones, aminoglycosides and carbapenems may be considered as effective for treatment. These findings will promote/create awareness for physicians about medication for UTIs. Based on the antibiogram of the isolate empirical or specific treatment can be determined for better therapeutic outcomes.
ACKNOWLEDGMENTS
The authors are thankful to CSIR, UGC for providing financial support as JRF-KK10000775 and are thankful to other laboratories for providing clinical samples.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
NRB and MBH conceptualized and designed the study. SD and RV performed data acquisition. NRB and MBH performed statistical analysis and data interpretation. NRB wrote the manuscript. MBH reviewed the manuscript and supervised the study. All authors read and approved the final manuscript for publication.
FUNDING
None.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
This study was approved by KLES Kidney Foundation Institutional Ethics Committee, Belagavi, with approval Number – KLESKF/IEC/23/017.
INFORMED CONSENT
Written informed consent was obtained from the participants before enrolling in the study.
- Medina M, Castillo-Pino E. An introduction to the epidemiology and burden of urinary tract infections. Ther Adv Urol. 2019:1756287219832172.
Crossref - Behzadi P, Behzadi E, Pawlak-Adamska EA. Urinary tract infections (UTIs) or genital tract infections (GTIs)? It’s the diagnostics that count. GMS Hyg Infect Control. 2019;14:Doc14.
Crossref - Zeng Z, Zhan J, Zhang K, Chen H, Cheng S. Global, regional, and national burden of urinary tract infections from 1990 to 2019: an analysis of the global burden of disease study 2019. World J Urol. 2022;40(3):755-63.
Crossref - Guglietta A. Recurrent urinary tract infections in women: risk factors, etiology, pathogenesis and prophylaxis. Future Microbiol. 2017;12(3):239-46.
Crossref - Kaur R, Kaur R. Symptoms, risk factors, diagnosis and treatment of urinary tract infections. Postgrad Med J. 2021;97(1154):803-812.
Crossref - Davenport M, Mach KE, Shortliffe LM, Banaei N, Wang TH, Liao JC. New and developing diagnostic technologies for urinary tract infections. Nat Rev Urol. 2017;14(5):296-310.
Crossref - Khoshnood S, Heidary M, Mirnejad R, Bahramian A, Sedighi M, Mirzaei H. Drug-resistant gram-negative uropathogens: A review. Biomed Pharmacother. 2017;94:982-994.
Crossref - Karam MR, Habibi M, Bouzari S. Urinary tract infection: Pathogenicity, antibiotic resistance and development of effective vaccines against Uropathogenic Escherichia coli. Mol Immunol. 2019;108:56-67.
Crossref - Fatima H, Goel N, Sinha R, Khare SK. Recent strategies for inhibiting multidrug-resistant and b-lactamase producing bacteria: A review. Colloids Surf B Biointerfaces. 2021;205:111901.
Crossref - Bauer AW, Kirby WM, Sherris JC, Turck M. Antibiotic susceptibility testing by a standardized single disk method. Am J Clin Pathol. 1966;45(4):493-496.
Crossref - Humphries RM, Ambler J, Mitchell SL, et al. CLSI methods development and standardization working group best practices for evaluation of antimicrobial susceptibility tests. J Clin Microbiol. 2018;56(4):10-128.
Crossref - Seifu WD, Gebissa AD. Prevalence and antibiotic susceptibility of Uropathogens from cases of urinary tract infections (UTI) in Shashemene referral hospital, Ethiopia. BMC Infect Dis. 2018;18:1-9.
Crossref - Kumar MA, Majmundar A, Patel S, et al. Antibiotic Resistance Pattern amongst Uropathogens Extracted from Patients Presenting with Urinary Tract Infection to Tertiary Care Teaching Hospital: A Cross Sectional Study. 2023.
- Athinarayanan G, Mariselvam R, Dhasarathan P, Ranjitsingh AJ. Epidemiology of urinary tract infection in South India. World J Biol Pharm Health Sci. 2020;1(1):025-32.
Crossref - Chandra H, Singh C, Kumari P, et al. Promising roles of alternative medicine and plant-based nanotechnology as remedies for urinary tract infections. Molecules. 2020;25(23):5593.
Crossref - Gupta K, Scholes D, Stamm WE. Increasing prevalence of antimicrobial resistance among uropathogens causing acute uncomplicated cystitis in women. Jama. 1999;281(8):736-738.
Crossref - Neumann I, Moore P. Pyelonephritis (acute) in non-pregnant women. BMJ Clin Evid. 2014;2014
- Akhtar SM, Sattar A, Hayat K, Cheema NA, Channa AA, Sarwer A. Uropathogens spectrum and antibiotic susceptibility in sexually active females of age group 20 to 40 years. Rawal Medical Journal. 2021;46(1):14.
- Seid M, Markos M, Aklilu A, et al. Community-Acquired Urinary Tract Infection Among Sexually Active Women: Risk Factors, Bacterial Profile and Their Antimicrobial Susceptibility Patterns, Arba Minch, Southern Ethiopia. Infect Drug Resist. 2023;16:2297-2310.
Crossref - Ali SB, Perdawood D, Abdulrahman R, Al Farraj DA, Alkubaisi NA. Vitamin D deficiency as a risk factor for urinary tract infection in women at reproductive age. Saudi J Biol Sci. 2020;27(11):2942-2947.
Crossref - Caretto M, Giannini A, Russo E, Simoncini T. Preventing urinary tract infections after menopause without antibiotics. Maturitas. 2017;99:43-46.
Crossref - Muhammad A, Khan SN, Ali N, Rehman MU, Ali I. Prevalence and antibiotic susceptibility pattern of uropathogens in outpatients at a tertiary care hospital. New Microbes New Infect. 2020;36:100716.
Crossref - Ali SA, Mandal S, Georgalas A, Gilani SA. A Pattern of Antibiotic Resistance in Gram-Negative Rods Causing Urinary Tract Infection in Adults. Cureus. 2021;13(1):12977.
Crossref - Mohamed AH, Omar NMS, Osman MM, Mohamud HA, Eraslan A, Gur M. Antimicrobial resistance and predisposing factors associated with catheter-associated UTI caused by uropathogens exhibiting multidrug-resistant patterns: a 3-year retrospective study at a tertiary Hospital in Mogadishu, Somalia. Trop Med Infect Dis. 2022;7(3):42.
Crossref - Mohapatra S, Panigrahy R, Tak V,et al. Prevalence and resistance pattern of uropathogens from community settings of different regions: an experience from India. Access Microbiol. 2022;4(2):00032.
Crossref - Pandey B, Pandit M, Jaiswal S, Sah AK, Chand RS, Shrestha R. Antimicrobial susceptibility pattern of pathogenic bacteria causing urinary tract infection in tertiary care hospital in Kathmandu, Nepal. IJPSR. 2020;11:6448-55.
Crossref - Ravishankar U, Sathyamurthy P, Thayanidhi P. Antimicrobial Resistance Among Uropathogens: Surveillance Report From South India. Cureus. 2021;13(1):e1213.
Crossref - Zang YM, Liu JF, Li G, et al. The first case of Escherichia fergusonii with biofilm in China and literature review. BMC Infect Dis. 2023;23(1):1-7.
Crossref - Malik S, Rana JS, Nehra K. Prevalence and antibiotic susceptibility pattern of uropathogenic Escherichia coli strains in Sonipat region of Haryana in India. Biomed Biotechnol Res J. 2021;5(1):80-87.
Crossref - Ahmed N, Khalid H, Mushtaq M, et al. The molecular characterization of virulence determinants and antibiotic resistance patterns in human bacterial uropathogens. Antibiotics. 2022;11(4):516.
Crossref - Bhargava K, Nath G, Bhargava A, Kumari R, Aseri GK, Jain N. Bacterial profile and antibiotic susceptibility pattern of uropathogens causing urinary tract infection in the eastern part of Northern India. Front Microbiol. 2022;13:965053.
Crossref - Bahrami S, Shafiee F, Hakamifard A, Fazeli H, Soltani R. Antimicrobial susceptibility pattern of carbapenemase-producing Gram-negative nosocomial bacteria at Al Zahra hospital, Isfahan, Iran. Iran J Microbiol. 2021;13(1):50.
Crossref
© The Author(s) 2024. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.