ISSN: 0973-7510
E-ISSN: 2581-690X
This study investigated the prevalence, risk factors and antimicrobial resistance of Staphylococci and E. coli isolated from diabetic urinary tract infection patients. Urine samples collected from 694 cases admitted to the General Health Insurance Authority Hospital in Port Said City, Egypt. A total of 302 bacterial strains were isolated from 292 urine samples with glucosuria and infection positive. The prevalence of the S. saprophyticus, S. aureus, E. coli and other species isolates from the urine samples are 54.7%, 34%, 7.6% and 3.7%, respectively. The antibacterial resistance profile of the isolated bacteria was investigated against different antibiotics. Most bacterial isolates were more susceptible to oflaxacin antibiotic while resistance to ampicillin. Multiple antibiotic resistance (MAR) was found highest for bacterial isolates obtained from urine samples. S. aureus showed high resistance percentage than other bacterial strains to studied antibiotics. Vancomycin resistance was detected in 23% of all S. aureus isolates. Out of 165 isolates S. saprophyticus, 71% were ג-lactamase producers, while all S. aureus and E. coli isolates were ג-lactamase positive. Also the purpose of this study was to study any relative between the recognized antibiotic resistance of bacteria and the occurrence of plasmids. Molecular sizes of the detected plasmids were 39.306 kbp in S. aureus and 44.640 kbp in E. coli. Plasmid curing in combination with MIC determination revealed that resistance of staphylococci and E. coli isolates was plasmid-mediated linked. The high MAR recognized makes it essential for antibiotic resistance testing to be accompanied prior to antibiotics remedy for diabetic urinary tract infection patients.
Antibiotics Resistance, Staphylococci, E. coli, Diabetic Urinary Tract Infection.
Diabetes is very public health difficulties that evidently rise in occurrence and frequency with progressing age. Besides organ complications, diabetic patients also hurt extra recurrently after problematical contagions equated with non-diabetic patients (Carten et al., 1992; Foxman, 2002; Geerlings et al., 2002; Hoepelman et al., 2003; Brown et al., 2004; Brown et al., 2005). To complicate substances further, diabetes also may be a risk factor for single and multi-drug resistance. However, all investigators have found increased uropathogen resistance to antibiotics in diabetic patients (Stapleton and stamm, 1997; Fedele et al., 2000; Geerlings et al., 2001; Wullt et al., 2003).
Insufficient articles have observed endemic antimicrobial resistance in Egypt, espically UTI in diabetic patients (Haberberger et al. 1994; Oyofo et al. 1995; Ostroff et al. 1996; Hilal et al. 1997; El-Teheawy et al., 1988; El Kholy and Nassar, 1996; Samuel et al., 1996; El Kholy et al., 2003). This study was therefore supposed to study the occurrence, risk factors and antibiotic resistance of staphylococci and E. coli isolated from diabetic urinary tract infection patients in Egypt.
Sample collection and identification of bacteria
Urine samples were collected as described by Collins et al. 1998 from 694 cases admitted to the General Health Insurance Authority Hospital in Port Said city, Egypt. A total of 388 DM patient’s urine samples with glucosuria positive concerned for further bacterial examination. One ml aliquots from urine suspensions were dispensed into CLED and MacConkey media. All plates were incubated face down and the bacteria were allowed to grow at 37°C for 24-48 hours prior to enumeration and further identification. Pure well-isolated colonies were preceded for their biochemical tests according to Bergeys manual of determinative bacteriology (Holt et al., 1994).
Antibiotic resistance testing
All trials were piloted using the inventive typical cultures to escape the unstructured hurt of antibiotic resistance. Antibiotic resistance was tested using a modified Kirby-Bauer disc diffusion method (Robert et al., 2003). ג-lactamase detection and curing test was carried out as described by Odugbemi et al., (1977).
Plasmid Analysis
The plasmid isolated by mini-prep alkaline extraction method (Sambrook et al., 1989). Concentrations and purity of DNA were assessed spectrophotometrically using Spectro 22, Labo Med, Inc., USA. Gels were arranged by adding 1% agarose and 5 µL ethidium bromide (10 mg/mL) to the TBE buffer. Pure DNA sample (3µL) was added to 12µL deionized water and 1µL endonuclease (EcoR I, Hind III (Sigma Production), BamH I (Roche Diagnostics GmbH). Sequential dilutions of acridine orange were used for curing of isolated plasmids.
A full of 292 urine trials were found infection positive from all different ages and cases admitted to the General Health Insurance Authority Hospital (Table 1). The percentage of infection raised by increase the concentration of glucosuria. We detected 100% a bacterial infection in urine samples of ++++ glucosuria level on CLED medium. Also percentage of counts on MaCckonky medium increased from 35.21% on + glucosuria level to 66.67% on ++++ glucosuria level.
Table (1):
Showing distribution of collected urine samples during study
| Patients | Age | No. Cases | Urine samples with glucosuria positive | Urine samples with glucosuria and infection positive |
|---|---|---|---|---|
| Male patients | Less 30 | 1 | 1 | 1 |
| 30- | 17 | 7 | 7 | |
| 40- | 96 | 52 | 34 | |
| 50- | 104 | 54 | 44 | |
| 60- | 118 | 59 | 48 | |
| 70- | 39 | 20 | 11 | |
| 80 – more | 15 | 10 | 9 | |
| Total | 390 | 203 | 154 | |
| Female patients | Less 30 | 1 | 0 | 0 |
| 30- | 16 | 10 | 7 | |
| 40- | 95 | 54 | 41 | |
| 50- | 69 | 44 | 34 | |
| 60- | 81 | 52 | 39 | |
| 70- | 35 | 21 | 15 | |
| 80 – more | 7 | 4 | 2 | |
| Total | 304 | 185 | 138 | |
| Total | 694 | 388 | 292 | |
A whole of 302 bacterial isolates were isolated from urine samples. The distribution of staphylococci and E. coli is shown in Table 2. The incidence of the S. saprophyticus, S. aureus, E. coli and other species isolates from the urine samples are 54.7%, 34%, 7.6% and 3.7%, respectively (Table 2). Bacterial counts had flocculation on + and ++ glucosuria concentration but other concentration in female patients recorded high bacterial counts than male patients (Table 2).
Table (2):
Showing distribution of identified bacterial species in male and female patients accordance to glucosuria level
| Glucosuria level | + | ++ | +++ | ++++ | Total (%) | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Bacterial isolates | M | F | M | F | M | F | M | F | ||
| S. saprophyticus | 15 | 8 | 34 | 19 | 40 | 46 | 1 | 2 | 165 (54.7) | |
| S. aureus | 8 | 11 | 16 | 13 | 21 | 34 | 0 | 0 | 103 (34) | |
| E. coli | 4 | 1 | 3 | 4 | 5 | 6 | 0 | 0 | 23 (7.6) | |
| Other Species* | 2 | 1 | 1 | 3 | 1 | 3 | 0 | 0 | 11 (3.7) | |
| Total | 50 | 93 | 156 | 3 | 302 | |||||
Other species include: Pseudomonas aeruginosa and Streptococcus sp.
The antibiotic resistance profile of staphylococci and E. coli isolates from urine samples is shown in Table 3. Most bacterial isolates were more susceptible to oflaxacin antibiotic while resistance to ampicillin. MAR was main for bacterial isolates gained from urine samples. Greater degrees of resistance were confirmed for S. aureus as compared with S. Saprophyticus versus most studied antibiotics. S. aureus showed high resistance percentage than other bacterial strains. Alls of staphylococci isolates were susceptible to vancomycin, except 23% of S. aureus (Table 3). E. coli isolates showed low resistance pattern to studied antibiotics. Susceptibility to imipenem and septazole (trimethoprim and sulfamoxazole) antibiotics for E. coli was 100% (Table 3). b-lactamase enzyme manufacture was confirmed in all staphylococci and E. coli isolates. Out of 165 isolates S. saprophyticus, 71% were b-lactamase producers, while all S. aureus and E. coli isolates were ג-lactamase positive.
Table (3):
Antibiotic resistance profiles of isolated bacteria from urine samples
| Bacterial isolates resistance to antibiotics (%) | |||
|---|---|---|---|
| Antibiotics / Isolates | S. saprophyticus (no.= 165) | S. aureus (no.= 103) | E. coli (no.= 23) |
| Nitroforantain (300 µg) | 70 | 79 | 95 |
| Imipenem (10 µg) | 40 | 19 | 100 |
| Aztreonam (30 µg) | 100 | 100 | 76 |
| OFLAXACIN (5μg) | 0 | 20 | 0 |
| Amoxicillin (25 µg) | 50 | 56 | 60 |
| Erythromycin (5μg) | 79 | 100 | 40 |
| Trimethoprim (125μg)+Sulfamoxazole(23µg) | 60 | 80 | 100 |
| Amikacin (30 µg) | 36 | 20 | 60 |
| Cefotaxime (30 µg) | 60 | 100 | 40 |
| Amoxicillin+Clavulanic acid (30 µg) | 60 | 100 | 80 |
| Ampicillin (10 µg) | 71 | 100 | 100 |
| Tobramycin (10 µg) | 53 | 17 | 39 |
| Norfloxacin (10 µg) | 40 | 54 | 29 |
| Vancomycin (30 µg) | 0 | 23 | ND |
N: Number of studied isolates.
ND: Not Determined.
Plasmid outlines of the three bacterial isolates below study were strongminded (Table 4). E. coli and S. aureus contain one plasmid, though S. saprophyticus were establish to three plasmids (Fig. 1A). Breakdown of isolated plasmids of E. coli and S. aureus isolates presented in fig. 1B. Quantity of recognition sites, amount of fragments and the estimated molecular size of restricted fragments were as shown in Table 4.
Table (4):
The restriction patterns of plasmids from E. coli and S. aureus isolates
Plasmids |
Restriction Enzymes |
No. of Recognition Sites |
No. of Fragments |
Size of Fragments (Kbp) |
Plasmid Size (Kbp ) |
|---|---|---|---|---|---|
E. coli |
Hind III EcoR I BamH I |
6 6 5 |
5 5 4 |
23000-9416-5148-4361 and 3530 23100-9416-5148-3530 and 3410 23000-9416-5804 and 5643 |
44.640 |
S. aureus |
Hind III EcoR I BamH I |
6 6 8 |
5 5 7 |
23000-6557-5148-4973 and 3530 22000-5804-4973-3530 and 2037 9416-7421-5148-5100-3430-3530 and 2322 |
39.306 |
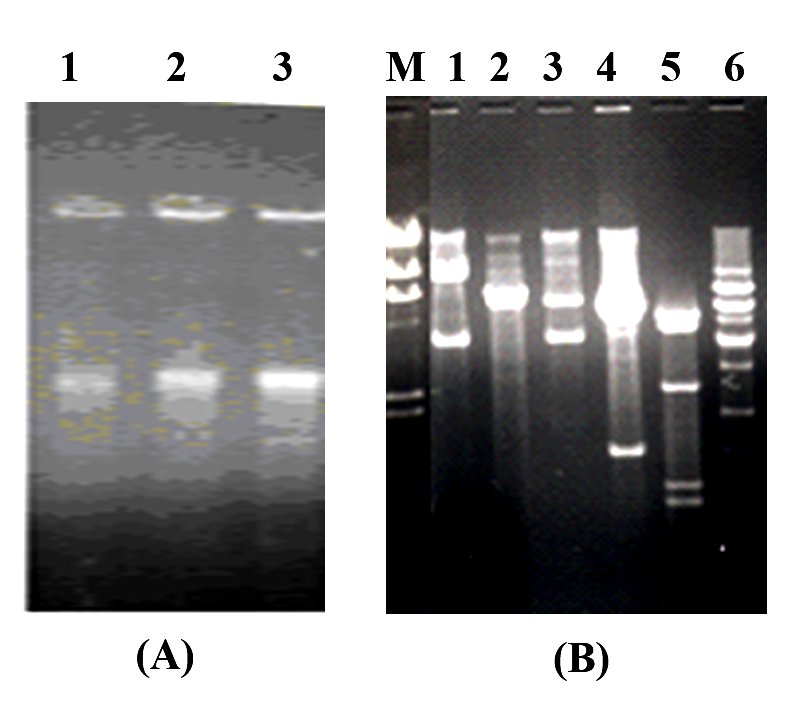 Fig. 1. Agarose gel electrophoresis of pure plasmids preparation from isolated strains. (A) lanes 1-3 pure plasmids of E. coli, S. aureus and S. saprophyticus respectively. (B) The restriction patterns of plasmids isolated from E. coli and S. aureus bacterial strains (contain only one plasmid) digested with Hind III, EcoR I and BamH I respectively. Where: Lane M: Lambda (λ) DNA + Hind III + EcoR I, lanes 1- 3: E. coli and lanes 4- 6: S. aureus
Fig. 1. Agarose gel electrophoresis of pure plasmids preparation from isolated strains. (A) lanes 1-3 pure plasmids of E. coli, S. aureus and S. saprophyticus respectively. (B) The restriction patterns of plasmids isolated from E. coli and S. aureus bacterial strains (contain only one plasmid) digested with Hind III, EcoR I and BamH I respectively. Where: Lane M: Lambda (λ) DNA + Hind III + EcoR I, lanes 1- 3: E. coli and lanes 4- 6: S. aureusPatients with diabetes frequently have enlarged difficulties of UTI, harshness and unfamiliar expressions (Stepleton, 2002). The risk for UTI complications is higher in people with DM. The foremost reasons of severe and straightforward UTI in ambulatory patients have been informed to be due to E. coli, S. aureus and Pseud. Aeruginosa (Foxman et al., 2000; Hoepelman et al., 2003). E. coli origins the common of UTIs between utmost clusters of patients deprived of diabetes and is likewise the maximum joint cause of UTI between men or women with diabetes (Parker et al., 2004). In our studies find similar results.
From the above data it can be mostly settled that staphylococci and E. coli isolates are relatively resistant to commonly used antibiotics. Resistance rates between staphylococci have been testified from other domestic geographical areas with results similar to ours (HEl-Kholy et al., 2003H). Amongst isolates of E. coli, 100% were resistance to ampicillin. Completely isolates were susceptible to imipenem and septazole. E. coli isolates in our study were have resistant outline to the antibiotics like to those from the El-Kholy et al. 2003. The occurrence of MAR organisms has attracted the attention of many workers and increasing antibiotic resistance is a factual problem in UTI (Foxman, 2002). It occurs from the infected patients who respond poorly to treatment and in a hospital, which may perform infection control and prevention programs. By definition, patients with complex UTI have a superior possibility of infection with an antimicrobial-resistant organism and/or an uncommon organism and also have a larger hazard of treatment disappointment (Patterson and Andriole, 1995; Ronald and Harding, 1997).
Most antibiotic- resistant microbes develop as a consequence of genetic change and following assortment courses by antibiotics. The resistance factor may be chromosomal, that developed as a result of spontaneous mutations and extrachromosomal resistance (Bekowitz, 1995; Selim, 2003). Plasmid- mediated antibiotic resistance in S. aurous has been described by many authors (Archer et al., 1986; Lyon and Skurray, 1987; Mansouri and Khaleghi, 1997; Shakibaie et al., 1999; Selim, 2003). The plasmid-linked resistance outlines of three bacterial ampicillin-resistance isolates were examined. For them plasmids were experimental and plasimd-linked resistance to ampicillin were established with relatively high MIC(S). The frequency of penicillin and cephalosporin- resistant clinical isolates of S. aureus has enlarged quickly and most of the isolates are -lactamase producers. In greatest strains, the enzyme is determined by plasmid but can also be establish on the chromosome (Shakibaie et al., 1999; Ako-Nai et al., 2005).
In, our data propose that antimicrobial resistance amongst staphylococci and E. coli is joint and important in diabetic patients in Egypt. The results obtained here, we confidence, will improve clinicians’ ability to improved achieve these diseases and decrease probable development. Our outcomes request for additional molecular biology studies to describe genes involved in antimicrobial resistance patterns in these isolates.
UTI, urinary tract infection; M, male Patients; F, Female Patients; +, Glucosuria concentration 180-200 mg/dl; ++, Glucosuria concentration 200-250 mg/dl; +++, Glucosuria concentration 250-350 mg/dl; ++++, Glucosuria concentration over 350 mg/dl.
Acknowledgements
None.
Conflicts of Interest
The authors declare that there is no conflict of interest.
Authors’ Contribution
All authors have made substantial, direct and intellectual contribution to the work and approved it for publication.
Funding
None.
Data Availability
All datasets generated or analyzed during this study are included in the manuscript.
Ethics Statement
The study was approved from the Universities Ethical Committee (ENREC).
- Ako-Nai, A.K., Adeyemi, F.M., Aboderin, O.A., Kassim, O.O. Antibiotic resistance profile of staphylococci from Clinical sources recovered from infants. African Journal of Biotechnology, 2005; 4: 816-822.
- Archer, G., Coughter, J.P., Johnston, J.L. Plasmid encoded trimethoprime resistance in Staphylococcus aureus. Antimicrob. Agent Chemother., 1986; 29: 733-40.
Crossref - Bekowitz, F.E. Antibiotic resistance in bacteria. South Med. J. 1995; 88: 797-804.
Crossref - Brown, J.S., Barrett-Connor, E., Nyberg, L.M., JW, K., TJ, O., Ma, Y., Vittinghoff, E. Incontinence in women with impaired glucose tolerance: results of the Diabetes Prevention Program. J. Urol., 2004; 171: 325–326 (2004).
Crossref - Brown, J.S., Wessells, H., Chanceller, M.B., Howards, S.S., Stamm, W.E., Staleton, A.E., Steers, W.D., Van Den Edden, S.K., McVary, K.T. Urologic complications of diabetes. Dia. Care. 2005; 28: 177-185.
Crossref - Carton, J.A., Maradona, J.A., Nuno, F.J. Diabetes mellitus and bacteraemia: a comparative study between diabetic and nondiabetic patients. Eur. J. Med. 1992; 5: 281-287.
- Collins, C.H., Lyne, P.M., Grange, J.M. 1998. Microbiological methods, pp178-205. Seventh edition, Butterworth Heinemaa.
- El Kholy, A.A., Nassar, Y.H. Phenotyping versus plasmid pattern analysis of Enterobacter cloacae in an outbreak in a pediatric intensive care unit. Egyptian Journal of Laboratory Medicine. 1996; 8: 467–83.
- El Kholy, A., Baseem, H., Hall, G.S., Procop, G.W., Longworth, D.L. Antimicrobial resistance in Cairo, Egypt 1999-2000: a survey of five hospitals. J. of Antimicro. Chemoth. 2003; 51: 625-630.
Crossref - El-Teheawy, M.M., El-Bokl, M.A., Abd el-Fattah, S.A., Sabbour, M.S. The pattern of antimicrobial use in general hospitals in Egypt. Chemioterapia. 1988; 7: 387–92.
- Fedele, D., Bortolotti, A., Coscelli, C., Santeusanio, F., Chatenoud, L., Colli, E., Lavezzari, M., Landoni, M., Parazzini, F. Erectile dysfunction in type 1 and type 2 diabetics in Italy. Int. J. Epidemiol. 2000; 29: 524-531.
Crossref - Foxman, B., Gillespie, B., Koopman, J. Risk factors for second urinary tract infection among college women. Am. J. Epidemiol. 2002; 151: 1194- 1999.
Crossref - Foxman, B. Epidemiology of urinary tract infections: incidence, morbidity, and economic costs. Am. J. Med. 2002; 113: 5S.
Crossref - Haberberger, R.L., Lissner, C.R., Podgore, J.K., Mikhail, I.A., Mansour, N.S., Kemp, L. Etiology of acute diarrhea among United States embassy personnel and dependants in Cairo, Egypt. American Journal of Tropical Medicine and Hygiene. 1994; 51: 870–874.
Crossref - Geerlings, S.E., Stolk, R.P., Camps, M.J. Consequences of asyymptomatic bacteriuria in women with diabetes mellitus. Arch. Itern. Med. 2001; 161: 1421-1427.
Crossref - Geerlings, S.E., Meiland, R., Hoepelman, A.I.M. Pathogenesis of bacteriuria in women with diabetes mellitus. Int. J. of Antimicro. Agents. 2002; 19: S39-S45.
Crossref - Hilal, S., El Kholy, A., El Ansary, M., El Rachidi, N. Comparison of a polymerase chain reaction (PCR) assay and standard susceptibility testing methods for detection of methicillin resistant Staphylococcus aureus. Journal of Cairo University. 1997; 65: 67–77.
- Hoepelman, A.I.M., Meiland, R., Geerlings, S.E. Pathogenesis and Management of bacterial urinary tract infections in adult with diabetes mellitus. Int. J. of Antimicro. Agents. 2003; 22: S35-S43.
Crossref - Holt, J.G., Krieh, N.R., Sneath, P.A., Staley, J., Williams, S.T. Bergey,s manual of determinative bacteriology. Ninth edition, Williams and wilkins Co., Baltimore, MD 1994.
- Lyon, B.R., Skurray, R. Antibiotic resistance of Staphylococcus aureus. Genetic bais. Microbiol. Rev. 1987; 51: 88-92.
- Mansouri, S., Khaleghi, M. Antibacterial resistance pattern and frequency of methicillin resistant Staphylococcus aureus isolated from different sources in southeastern Iran. Irn. J. Med. Sc. 1997; 22: 89-93.
- Patterson, J.E., Andriole, V.T. Bacterial urinary tract infections in diabetes. Infect. Dis. Clin. North Am. 1995; 9: 25–51.
- Odugbemi, T.O., Hafiz, S., McEntegart, M.D. Penicillinase-producing Neisseria gonorrhea detection by Starch-paper technique. Br. Med. J. 1977; 2: 500.
Crossref - Ostroff, S.M., Harrison, L.H., Khallaf, N., Assaad, M.T., Guirguis, N.I., Harrington, S. Resistance patterns of Streptococcus pneumoniae and Haemophilus influenzae isolates recovered in Egypt from children with pneumonia. The antimicrobial surveillance study group. Clinical Infectious Diseases. 1996; 23: 1069–1074.
Crossref - Oyofo, B.A., El Gendy, A., Wasfy, M.O., El Etr, S.H., Churilla, A., Murphy, J. A survey of enteropathogens among United States military personnel during operation bright star 94, in Cairo, Egypt. Military Medicine. 1995; 160: 331–334.
Crossref - Parker, A.S., Cerhan, J.R., Lynch, C.F. History of urinary tract infection and risk of renal cell carcinoma. Am. J. Epidemiol. 2004; 159: 42-48.
Crossref - Robert, S., Anders, R.L., Niels, F., Frank, E. Evaluation of different disk diffusion/media for detectionof methicillin resistance in Staphylococcus aureus and coagulase-negative staphylococci. APMIS. 2003; 111: 905-914.
Crossref - Ronald, A., Harding, G. Complicated urinary tract infections. Infect. Dis. Clin. North Am. 1997; 11: 583–592.
Crossref - Sambrook, J., Fritsch, E.F., Maniatis, T. Molecular cloning: A laboratory manual. Secound edition, Cold Spring Harbor Laboratory, Cold Spring Harbor, New York 1989.
- Samuel, S., Haroun, M., El Kholy, A., Mahmoud, S. Epidemiologic aspects of community-acquired pneumonia in infancy and childhood in summer versus wintertime. Journal of Arab Child. 1996; 7: 1–10.
- Selim, S. A. Plasmids and Resistance to Antimicrobial Agents in Pseudomonas aeruginosa and Staphylococcus aureus strains isolated from Burns. The Eleventh Conference of microbiology, Cairo, Egypt, 2003; 139-153.
- Shakibaie, M.R., Mansouri, S., Hakak, S. Plasmid Pattern of Antibiotic Resistance in Beta-Lactamase Producing Staphylococcus aureus Strains Isolated from Hospitals in Kerman, Iran. Archives of Iranian Medicine. 1999; 2: 4-7.
- Stapleton, A., Stamm, W.E. Prevention of urinary tract infection. Infect. Dis. Clin. of North America. 1997; 11: 719-733.
Crossref - Stepleton, A. Urinary tract infections in patients with Diabetes. The Am. J. of Med. 2002; 113: 80S-84S.
Crossref - Wullt, B., Bergsten, G., Fischer, H., Godaly, G., Karpman, D., Leijonhufvud, I., Lundstedt, A. C., Samuelsson, P., Samuelsson, M., Svensson, M. L., Svanborg, C. The host response to urinary tract infection. Infect. Dis. Clin. North America. 2003; 17: 279-301.
Crossref
© The Author(s) 2019. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


